Abstract
Background
Hypertensive disorders of pregnancy (HDP) are common obstetric medical problems. Compliance with clinical guidelines and evidence from major trials has the potential to translate to significantly improve maternal and perinatal outcomes. The aims of this study were to prospectively review management of HDP in an Australian cohort in the context of the Society of Obstetric Medicine of Australian and New Zealand (SOMANZ) guidelines and current evidence in published literature regarding management controversies.
Methods
The management of 100 pregnant women with HDP and prescription for antihypertensive medication at two tertiary obstetric centres was prospectively reviewed in 2013. Compliance with SOMANZ guidelines, uptake of findings from the HYPITAT trial and the Control of Hypertension In Pregnancy Study (CHIPS) trial were assessed.
Results
Sixty-eight women had chronic hypertension, while 32 had gestational hypertension. Management of HDP was mostly consistent with current SOMANZ guidelines and evidence from CHIPS and HYPITAT.
Conclusion
Clinicians were practising according to the current SOMANZ guidelines, indicating vigilance on behalf of the treating team.
Keywords
Background
Hypertensive disorders of pregnancy (HDP) affect about 10% of pregnancies in Australia 1 and are a leading cause of maternal mortality and perinatal death worldwide. 2 It is estimated that 500,000 perinatal deaths and 30,000 maternal deaths are attributable to HDP annually worldwide. 3 Optimising antenatal management of HDP is core to reducing maternal and fetal risks, and a key objective of clinical practice guidelines.
The Society of Obstetric Medicine of Australian and New Zealand (SOMANZ) guidelines on management of HDP,1,4 form the basis for the management of HDP at the studied institutions. Both 2008 and 2014 SOMANZ guidelines recommend labetalol, methyldopa and oxprenolol as first-line antihypertensive medications and hydralazine, modified-release nifedipine and prazosin as second line.1,4 Moreover, both guidelines advise that angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) are contraindicated in pregnancy.1,4 Unless deemed essential due to comorbidities, they also do not recommend atenolol and other highly-selective beta blocker drugs as they are associated with fetal growth restriction (FGR), or thiazide diuretics, which may restrict the natural plasma volume expansion of pregnancy.1,4
In Australia, there are three main models of care available for pregnant women, namely general practitioner (GP)-led model known as ‘shared care’, midwife-led-midwifery group practice model and an obstetrician-led obstetric model.1,4 The GP shared care model allows the woman to see a trained GP of her choice for the majority of the antenatal appointments, with the remainder with a midwife at the nominated hospital. 1 The midwifery group practice model 1 allows for one-to-one maternal care, often with the same midwife throughout the term of pregnancy, with little involvement by an obstetrician. The obstetric model 1 involves antenatal appointments with an obstetrician with the addition of an obstetric physician if needed, especially for the close monitoring of blood pressure (BP) and prescription of antihypertensive medications.
The Control of Hypertension In Pregnancy Study (CHIPS) 5 found no significant differences in the risk of pregnancy loss, high-level neonatal care or overall maternal complications between less-tight (target diastolic BP (DBP) 100 mmHg) and tight (target DBP 85 mmHg) control of hypertension. There was, however, an increase in the incidence of severe maternal hypertension in the less-tight group. 5 Further published analysis demonstrated that this was associated with poorer maternal outcomes, even when corrected for the development of pre-eclampsia. 6 Despite this, the BP threshold to initiate antihypertensive treatment in pregnant women with mild to moderate hypertension remains in contention, leaving the decision up to the treating clinician's judgement. The 2014 SOMANZ guidelines have also added a recommendation regarding the timely delivery of women with mild to moderate hypertensive disease, in light of findings from the ‘Induction of labour (IOL) versus expectant monitoring for gestational hypertension (GH) or mild pre-eclampsia after 36 weeks' gestation’ (HYPITAT-I) trial. 7
There is a paucity of studies investigating the prospective management of HDP in the context of clinical guidelines in Australia and internationally. Few studies that have assessed the uptake of HDP guidelines recommendations have been retrospective.8,9 Similarly, studies investigating the uptake of recommendations from either CHIPS or HYPITAT-I have also been scarce and have not been undertaken in an Australian context.10,11
The aim of this study was to prospectively review management of HDP in an Australian cohort, in the context of clinical guidelines and current evidence in the published literature regarding management controversies. The specific objectives were:
To evaluate compliance with clinical guidelines
4
for management of HDP during pregnancy, specifically:
appropriateness of antihypertensives used and monitoring of blood pressure To review the extent of use of IOL at term for women with GH or pre-eclampsia as per HYPITAT-I
7
and To evaluate uptake of findings from the CHIPS
5
trial regarding less-tight versus tight DBP control.
Methods
Study design
Prospective cohort study.
Study setting
Antenatal clinics of two large tertiary maternity hospitals in Melbourne, Australia. Together, these hospitals provide antenatal care to approximately 13,000 women annually.
Inclusion criteria were:
≥18 years of age; Able to understand English; Diagnosis of a HDP (either chronic or gestational); Current prescription of an antihypertensive medication.
Eligible participants were identified by author AH after reviewing the medical records of pregnant women scheduled to attend out-patient antenatal clinics (February to December 2013). Potential participants were approached individually and provided with written information about the study and a questionnaire. Those who consented to the study signed a written consent form. In-depth interviews were carried out with a subset. The medical records of all participants were prospectively reviewed until delivery.
Participants’ medical records were reviewed manually by AH after each appointment, and relevant data were extracted. Maternal data included demographics, medical and obstetric history, progression of the HDP, including development of moderate to severe hypertension (systolic BP (SBP) ≥ 150–169 mmHg and/or DBP ≥ 100–109 mmHg), 4 severe hypertension (SBP ≥ 170 mmHg and/or DBP ≥ 110 mmHg) 4 and pre-eclampsia. 4 Management of the HDP, including use of antihypertensive medication, admission to the Pregnancy Day Assessment Centre (PDAC) as well as antenatal hospital admission were recorded. Antihypertensive agents were classified as first line and second line according to the SOMANZ guidelines that were in use at the time of the study. 4
All participants were followed up until delivery and neonatal data were also recorded, including gestational age at delivery, birthweight and need for special or neonatal intensive care admission. Customised centiles were calculated and FGR recorded, if birthweight was less than the 10th customised centile. Centiles were calculated using the calculator devised by Mongelli et al. specifically for the Australian population, allowing adjustment for maternal characteristics such as height and weight, ethnicity, gestational age and fetal gender. 12 One participant had a intrauterine death in utero and maternal data extraction ceased at that point. One other participant was transferred to a different maternity hospital during their pregnancy and provided additional consent to have the relevant data, including that of the baby, extracted from her medical records at the new hospital.
Target DBP among women who had non-proteinuric chronic or GH between 14 weeks 0 days to 33 weeks 6 days of gestation, were analysed according to the CHIPS 5 description of less-tight control (target DBP, 100 mm Hg) versus tight control (target DBP, 85–99 mm Hg).
Among women who reached 36 weeks of gestation with mild GH or pre-eclampsia and a single fetus in a cephalic presentation (‘HYPITAT eligible’), outcomes were compared between those managed expectantly and those managed with immediate/as soon as practicable IOL, as described in the HYPITAT-I trial. 7 Outcomes compared between groups were those reported in HYPITAT-I, namely maternal mortality, maternal morbidity (eclampsia; haemolysis, elevated liver enzymes and low platelets (HELLP) syndrome; pulmonary oedema; thromboembolic disease; and placental abruption), progression to severe hypertension or proteinuria, and major postpartum haemorrhage (>1000 mL blood loss). 7
Data were analysed using descriptive statistics and appropriate univariate analysis (Pearson chi-square and Student's t-tests) and multivariate analysis (logistic regression) using SPSS (version 26.0, IBM, New York, NY, USA). The p < 0.05 was considered statistically significant.
Results
A total of 100 pregnant women were recruited. Among these participants, 68 had chronic hypertension (CH), while 32 had GH; these groups are compared in Table 1. Thirteen participants with CH were diagnosed during pregnancy, 10 of whom were diagnosed by their local GP at 4 to 9 weeks of gestation.
Baseline maternal characteristics and obstetric history (N = 100).
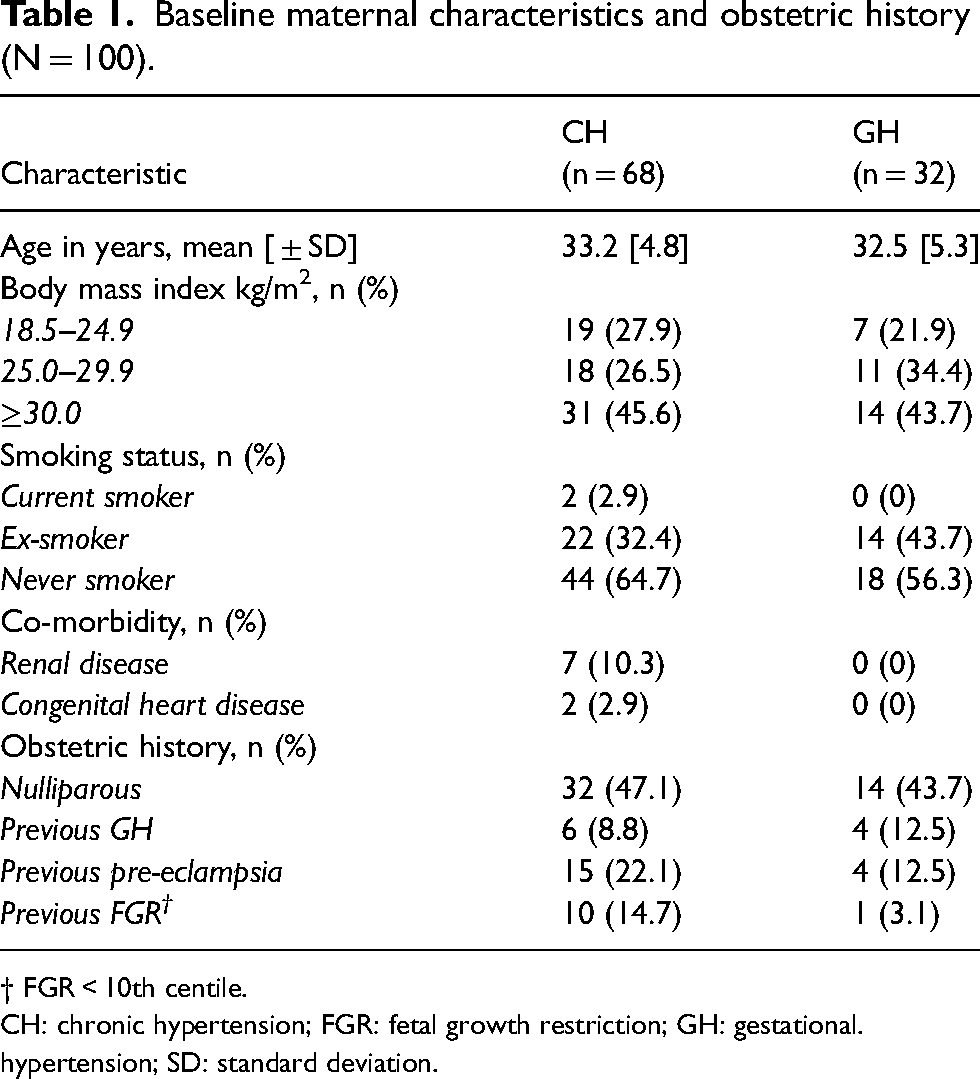
† FGR < 10th centile.
CH: chronic hypertension; FGR: fetal growth restriction; GH: gestational. hypertension; SD: standard deviation.
The specific antihypertensives prescribed in this population are summarised in Table 2. Labetalol was the most common agent prescribed in CH, whereas methyldopa was the preferred agent in GH.
Summary of antihypertensive medication (N = 100).
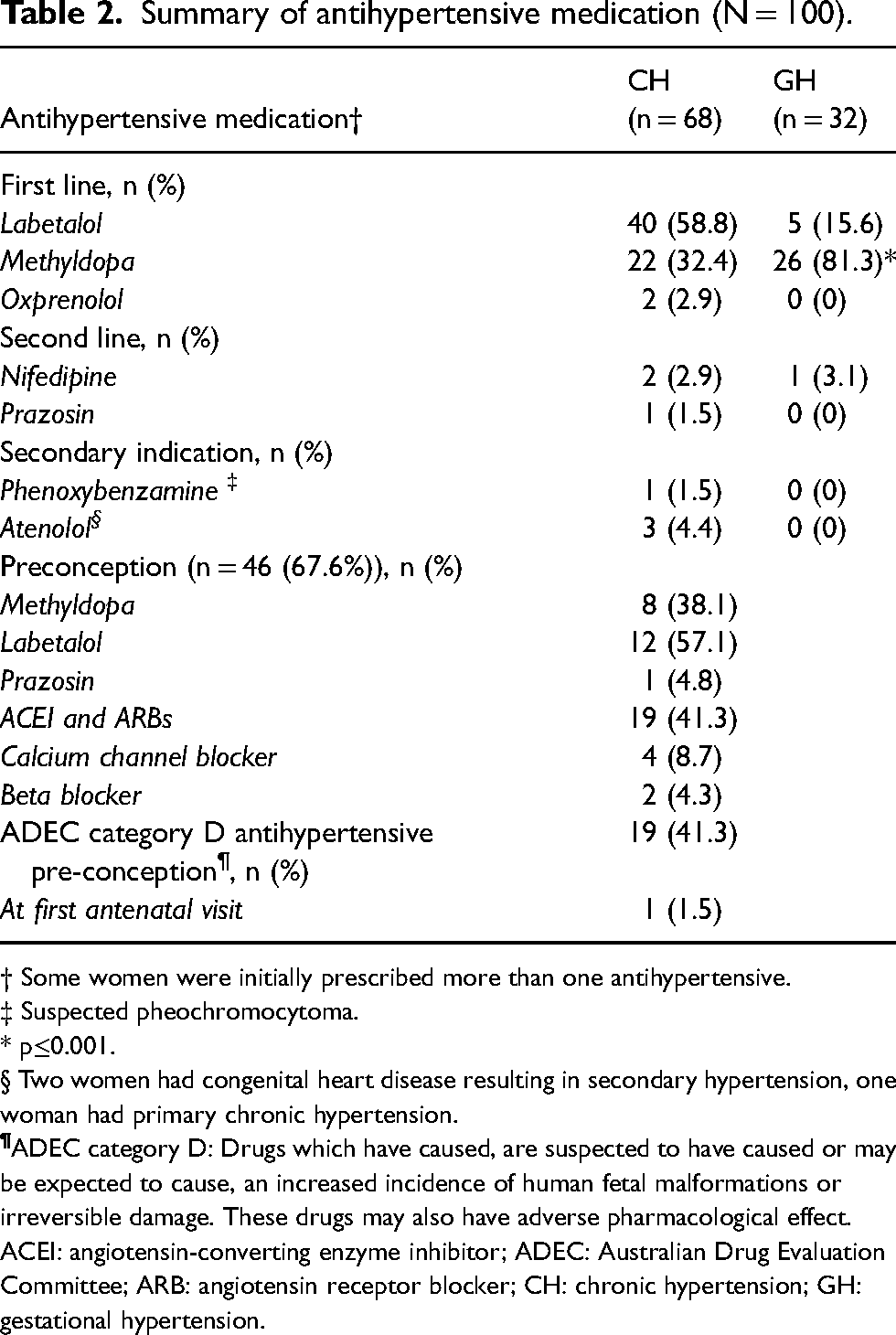
† Some women were initially prescribed more than one antihypertensive.
‡ Suspected pheochromocytoma.
* p≤0.001.
§ Two women had congenital heart disease resulting in secondary hypertension, one woman had primary chronic hypertension.
ACEI: angiotensin-converting enzyme inhibitor; ADEC: Australian Drug Evaluation Committee; ARB: angiotensin receptor blocker; CH: chronic hypertension; GH: gestational hypertension.
Table 3 describes the type of monitoring that the women in the cohort received during the pregnancy as well as the rates of pre-eclampsia development, episodes of severe hypertension and the need for emergency caesarean delivery.
Antenatal monitoring and hospital admission for management of HDP (N = 100).

‡ Some women had > 1 indication for hospital admission.
§ Some women had an initial diagnosis for pre-eclampsia but had hypertension that was gestational in nature.
BP: blood pressure; CH: chronic hypertension; GH: gestational hypertension; HDP: hypertensive disorders of pregnancy; IQR: interquartile range; PDAC: Pregnancy Day Assessment Centre; SD: standard deviation.
In the only significant result found by logistic regression was that a well-timed first antenatal visit (by 12 weeks of gestation) was less likely to be associated with severe hypertension—odds ratio 0.160 (95% confidence interval 0.037–0.683).
Of the 68 women with mild to moderate hypertension, 59 had non-proteinuric mild to moderate gestational or CH DBP of 85–99 mmHg; the remaining nine had DBP reading ≥ 100 mmHg at a stage prior to 33 weeks 6 days of gestation. Their outcomes are compared in Table 4.
Outcomes of ‘tight’ vs ‘less-tight’ BP control (N = 68).
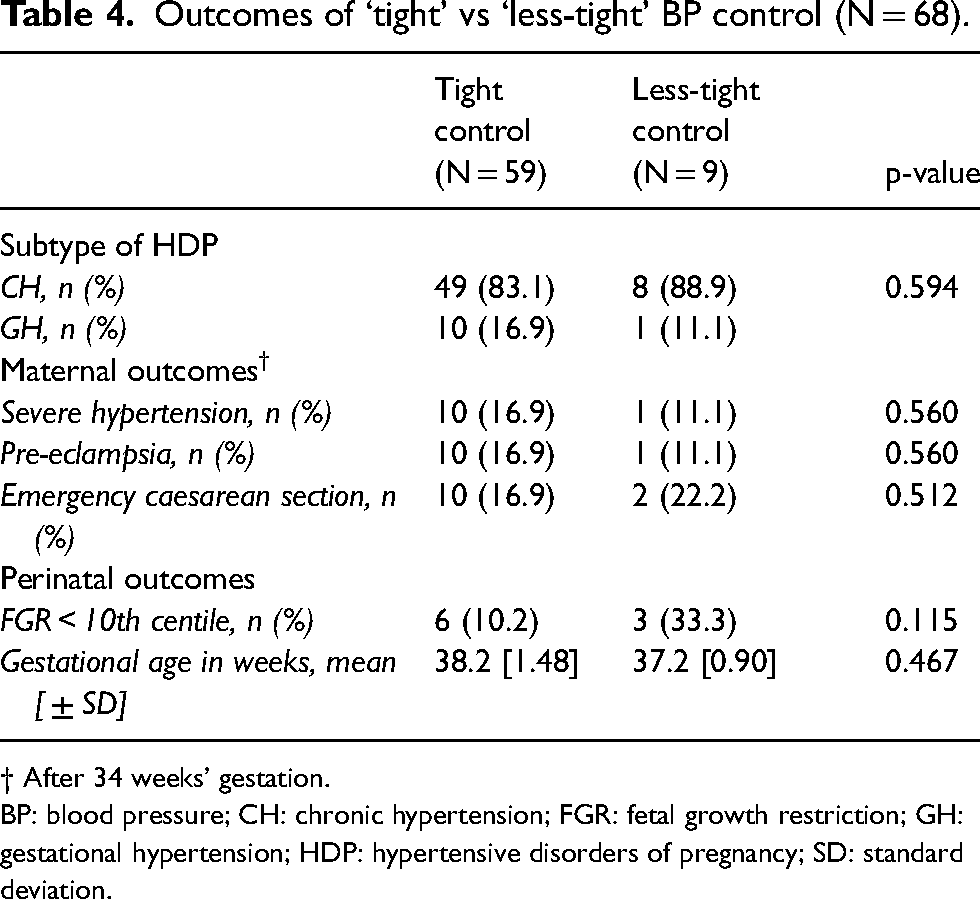
† After 34 weeks' gestation.
BP: blood pressure; CH: chronic hypertension; FGR: fetal growth restriction; GH: gestational hypertension; HDP: hypertensive disorders of pregnancy; SD: standard deviation.
Of the 21 ‘HYPITAT-I eligible’ women with mild GH or mild pre-eclampsia, 16 (76.2%) had not been delivered at 36 weeks and had no contraindication for IOL, such as breech or planned caesarean. Of these, 4 (25%) appeared to have been managed with immediate/as soon as practicable IOL, while 12 (75.0%) were considered safe for expectant management and were delivered between 37 (+2 days) and 39 weeks (+6 days). Of these, one woman progressed to severe hypertension and another progressed to severe pre-eclampsia; however, neither developed a composite severe adverse outcome, as defined by HYPITAT-I. 7 There was no significant difference in the rate of caesarean section between these groups. The FGR (<10th centile) rate was 50% in the expectant management group and 50% in the immediate management group.
Discussion
This study showed that management of HDP was mostly consistent with current guidelines (SOMANZ1,4) and evidence (CHIPS, 5 HYPITAT-I 7 ) at the time. This study has also highlighted that pre-pregnancy or early pregnancy consultation can ensure that women with CH are on appropriate antihypertensive medications, thus helping to reduce the potential for birth defects following inadvertent exposure in early pregnancy. 13
In our study, more than half of the women in the CH group were using an antihypertensive medication prior to pregnancy and a little over half of these were not on a pregnancy-safe medication. Nevertheless, the majority of women with CH had their antihypertensive switched to a pregnancy-safe agent by the time of their first hospital antenatal appointment, attesting to the value of pre-pregnancy consultation. Since the time this study was undertaken, there has been a stronger recommendation from several clinical guidelines to cease antihypertensives that are not classified as safe in the pre-conception period.1,14 The 2016 Australian guidelines for the diagnosis and management of hypertension in adults mentions pregnancy and the potential for pregnancy in women of reproductive age as a contraindication for ACEI and ARBs. 15
The majority of women in both CH and GH groups were prescribed a first-line antihypertensive, in compliance with clinical guidelines. Methyldopa was used significantly more often in the GH than the CH group. Although this may have been acceptable at the time, 4 current clinical recommendations are to change methyldopa to an alternative antihypertensive if treatment should continue in the immediate postpartum period, due to its potential to exacerbate post-natal depression. 14
The timing of the first antenatal appointment was significantly later for the women in the CH group compared to the women in the GH group. This is despite that prescription of an antihypertensive would have been valid upon conception in the majority of women with CH. In our study, a later initial booking visit was more likely to be associated with an incidence of severe hypertension. A study in the United Kingdom that explored the reasons for delayed access to antenatal care, found that although there were patient-related factors such as knowledge of pregnancy, there were also administrative failures such as letters not being sent and/or received in a timely manner. 16 There is, however, a dearth of studies into why pregnant women do not access timely, free and locally available antenatal care both in the United Kingdom 17 and Australia.
The rate of admission to PDAC was as expected across both groups 18 and may have contributed to the low incidence of severe hypertensive disease in this cohort. There were, however, some women who required hospitalisation to reduce the risks of severe hypertension and/or PE for both the mother and the baby, indicating close monitoring.
The majority of women with mild to moderate non-proteinuric hypertension had their BP tightly controlled as defined by CHIPS. 5 This may have contributed to the low incidence of severe hypertension in the cohort. It also reinforces that pregnant women who require antihypertensive treatment should be under an obstetrician model of care as recommended in the current SOMANZ guidelines. 1
Only one-quarter of ‘HYPITAT eligible’ patients were delivered at 37 weeks, yet we observed a lower rate of adverse maternal outcomes in this group than what would have been anticipated by HYPITAT-I. This rate was similar to that seen in a retrospective cohort study in Australia, 8 but contrasts with results from an earlier study in the United States. 10 The SOMANZ guidelines advise considering delivery after 37 weeks but prior to 41 weeks, particularly if maternal or fetal complications have developed. 1 Many patients in this study were managed expectantly until 37 to 41 weeks, which may explain some of the observed differences in less major maternal and neonatal adverse events, such as progression to severe hypertension and neonatal respiratory distress in this cohort compared to HYPITAT-I. 7 This suggests that clinicians actively weighed up the potential maternal benefit of immediate delivery versus neonatal harm, including the long-term consequences of late preterm and early term birth.
It is expected that routine introduction of soluble fms-like tyrosine kinase 1/placental growth factor (sFlt/PlGF) ratio into clinical practice will be an important addition to the current diagnostic algorithm for pre-eclampsia in pregnant women with either CH or GH. The sFlt/PlGF ratio has recently been found to differentiate between the diagnosis of CH with superimposed pre-eclampsia and uncontrolled CH. 19 Similarly, the Preeclampsia Triage by Rapid Assay (PETRA) group recently undertook a prospective observational study of pregnant women with signs or symptoms of PE between 20 and 35 weeks of gestation at 24 centres in the United States. They found that sFlt/PlGF ratio can help to predict the potential for adverse pregnancy outcomes better than clinical markers, and thus inform timing of delivery. 20
Strengths and limitations
To the best of our knowledge, this is the first study to prospectively evaluate the Australian management of HDP according to the SOMANZ guidelines. Although the division between CH and GH groups was not equal, the ratio is consistent with that observed in the CHIPS trial. 5 Although recruitment was from two major tertiary maternity hospitals in Melbourne, the study was not powered to detect differences in the clinical management between groups. The low sample size also compromised the adequacy for multivariate analyses. Moreover, the women were followed up only until delivery, so if they developed postpartum pre-eclampsia or their hypertension persisted after 6 weeks postpartum the data were not captured. Further research involving larger numbers of participants is warranted, and would require recruitment from a larger and broader range of tertiary maternity centres.
Conclusions
In conclusion, the clinical management guidelines for HDP were followed well. Pre-pregnancy or early pregnancy counselling for women with CH helped to ensure that antihypertensive medication was switched in a timely manner to a pregnancy-safe agent. Recommendations from the CHIPS trial were generally followed in this cohort. Although HYPITAT-I was available at the time of this cohort study, the clinicians were already practising according to the current SOMANZ guidelines indicating vigilance on behalf of the treating team and providing reassurance to pregnant women.
Footnotes
Acknowledgements
Our gratitude goes to the pregnant women who participated in this research. The authors would also like to thank the staff at the Mercy Hospital for Women and the Royal Women's Hospital for accommodating this research. We would also like to thank Professor Susan Walker for her guidance and comments on the manuscript, Dr Fiona Cullinane for her assistance at the Royal Women's Hospital, and Dr Ian Hunt for statistical support.
Contributorship
AH carried out patient recruitment, data collection and writing of first draft. AH and JG were involved with data analysis. All authors were involved in the concept and design of the study as well as ethical approval. All authors contributed to data analysis and interpretation; and reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained from participating institutions and the University. Mercy Health Human Research Ethics Committee (R12/62) 08/01/2013, The Royal Women's Hospital Research and Human Research Ethics Committee (R13/18) 12/07/2013, and Monash University Human Research Ethics Committee (CF13/117) 18/01/2013.
Informed consent
Potential participants were approached individually and provided with written information about the study and a questionnaire. Those who consented to the study signed a written consent form. Written informed consent was obtained from the patients for their anonymised information to be published in this article.
Guarantor
JG.
