Abstract
Anomalous systemic arterial supply to the basal segment of the lung (ABLL) is a rare cause of haemoptysis. ABLL may be complicated by massive haemoptysis, heart failure due to left-to-right shunt and infection. We describe a case of this condition presenting in the third trimester of pregnancy. Computed tomography chest angiogram confirmed the diagnosis. A multidisciplinary approach was necessary to determine treatment, which ultimately consisted of elective caesarean section at 36 weeks and 4 days’ gestation, followed by transarterial embolisation 9 days postpartum.
Introduction
Anomalous systemic arterial supply to the basal segment of the lung (ABLL) is a rare congenital malformation characterised by aberrant systemic arterial supply to the basal segment of the lung, with normal venous drainage to the pulmonary veins and normal bronchial branching. 1 It was previously classified as one form of pulmonary sequestration (PS) according to Pryce's terminology. 2 It has been proposed that these malformations exist on a spectrum of congenital pulmonary abnormalities, however ABLL is currently distinguished as an independent clinical entity from PS. 3 In contrast to PS, the lung segment in ABLL has normal connections with the tracheobronchial tree. 4
Most cases are recognised in the fourth decade of life, with the most common symptoms being haemoptysis, exertional dyspnoea and cough. Asymptomatic cases have been discovered incidentally through abnormal imaging studies or detection of a cardiac murmur. 5 ABLL confers a risk of complications such as massive haemoptysis, heart failure due to left-to-right shunt and infection.6, 7 Therefore, patients with ABLL require treatment even in the absence of symptoms. Traditionally management involved surgical resection of the aberrant artery and lung resection, however transarterial embolisation has recently been utilised for certain cases.8–10
There has been a single case report of ABLL 11 and few reports of the related condition of PS presenting for the first time in pregnancy.12, 13 We describe a case of ABLL presenting with haemoptysis in the third trimester. To our knowledge this is the first report of ABLL presenting in pregnancy managed with postpartum transarterial embolisation. The management challenges in this case included decision-making regarding the timing and modality of delivery, as well as the choice of definitive management modality in the postpartum setting.
Case report
A 36-year-old woman in her first pregnancy presented at 35 weeks’ gestation to the emergency department of a tertiary obstetric centre with a 4-day history of low-volume frank haemoptysis. She was expectorating one to two teaspoons of frank red blood and blood clots, once to twice daily. There was no coughing between haemoptysis episodes, and no dyspnoea, chest pain, or fever. She had never had similar symptoms before and the only history of respiratory disease was mild childhood asthma. The pregnancy had been uncomplicated to date.
Her respiratory rate was 18 breaths/min, peripheral oxygen saturations were 99% on room air, heart rate was 82 beats/min and blood pressure 129/87 mmHg. Breath sounds were normal. Calves were soft and non-tender.
Routine haematology and biochemistry were normal. Haemoglobin was 123 g/L. Prothrombin time and activated partial thromboplastin clotting time were normal. Chest radiograph was normal. Fetal assessment was normal for cardiotocography, and ultrasound showed satisfactory interval growth, with heart activity present, normal amniotic fluid, and normal placental blood flow.
Computed tomography (CT) abdomen scan from 9 months prior (performed during a gastroenterological work-up for altered bowel habit) was reviewed. This incidentally demonstrated a systemic arterial supply arising from the thoracic aorta at the level of the diaphragmatic crus, supplying the posterior basal segment of the right lower lobe. This abnormality was commented upon in the report, but the patient was not aware of the finding and did not undergo further investigation or management at that time.
CT pulmonary angiogram was performed and demonstrated a wedge-shaped focus of consolidation in the posteromedial right lower lobe suggestive of pulmonary infarction (Figure 1). This pulmonary segment was supplied by the systemic artery arising from the aorta. CT chest angiogram confirmed a focal area of air space opacity with adjacent bronchial luminal filling in the posterior medial right lower lobe with arterial systemic feeder 5.9 mm in diameter, and pulmonary venous drainage, consistent with ABLL. There was a small rounded arterially enhancing focus in the right lower lobe possibly representing a pseudoaneurysm in the systemic feeder artery (Figure 2). Transthoracic echocardiogram was normal.
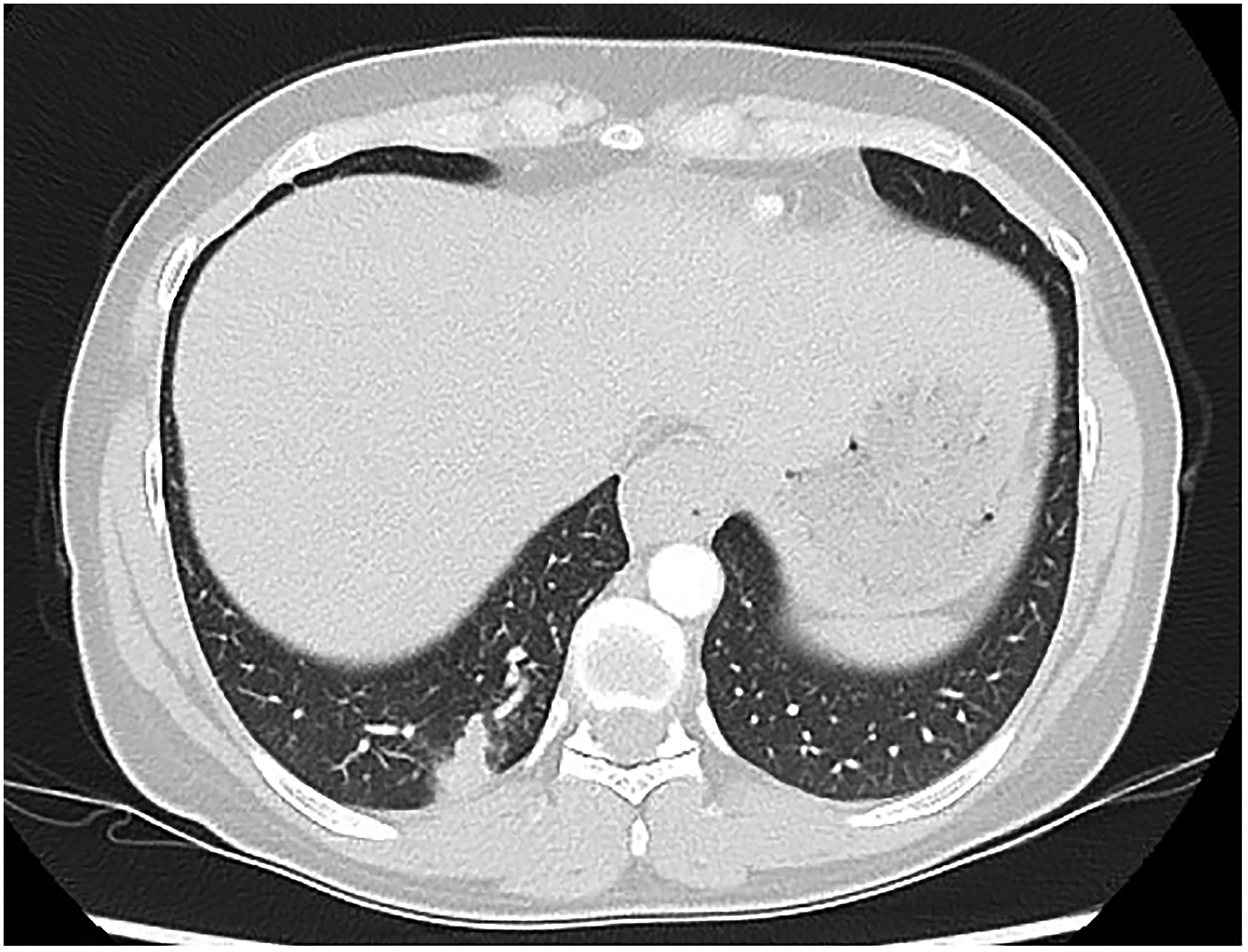
Computed tomography pulmonary angiogram demonstrating a wedge-shaped focus of consolidation in the posteromedial right lower lobe suggestive of pulmonary infarction.
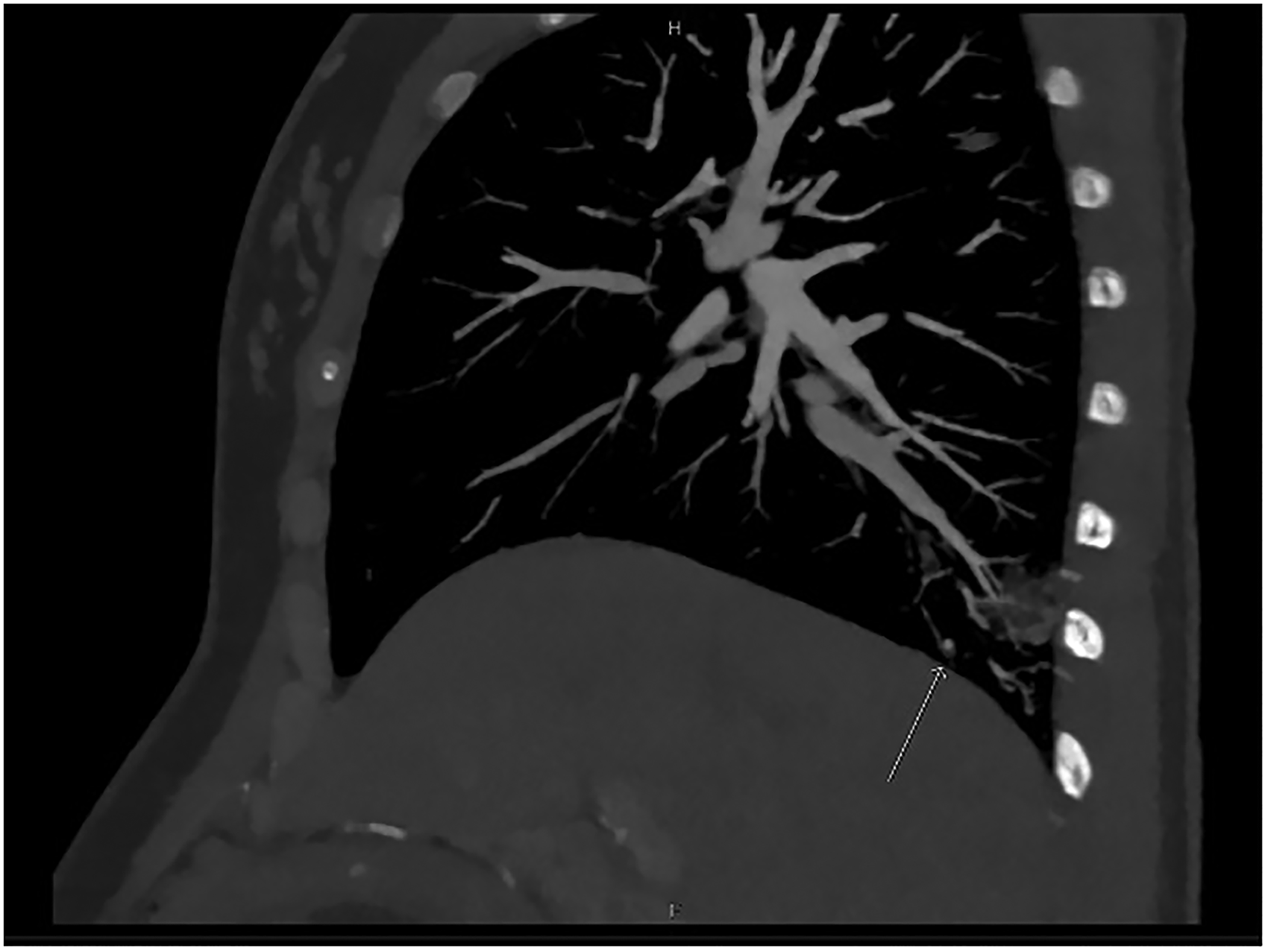
Computed tomography chest angiogram demonstrating (arrow) a small rounded arterially enhancing focus in the right lower lobe possibly representing a pseudoaneurysm.
Multidisciplinary input was sought from obstetrics, respiratory medicine, thoracic surgery, interventional radiology and anaesthetic teams. Due to ongoing daily haemoptysis during the hospital admission, and the gestation of 36 weeks and 4 days, the decision was made to deliver. Caesarean section under spinal anaesthesia was performed on day 6 of admission with no complications and a live male infant was born in good condition. Haemoptysis, which had been gradually decreasing in volume and frequency, ceased completely after delivery.
Arterial embolisation was performed 9 days postpartum with Medtronic MVP™ microvascular plug system (Figure 3). The embolisation was technically successful with no complications, with discharge 2 days post-procedure.
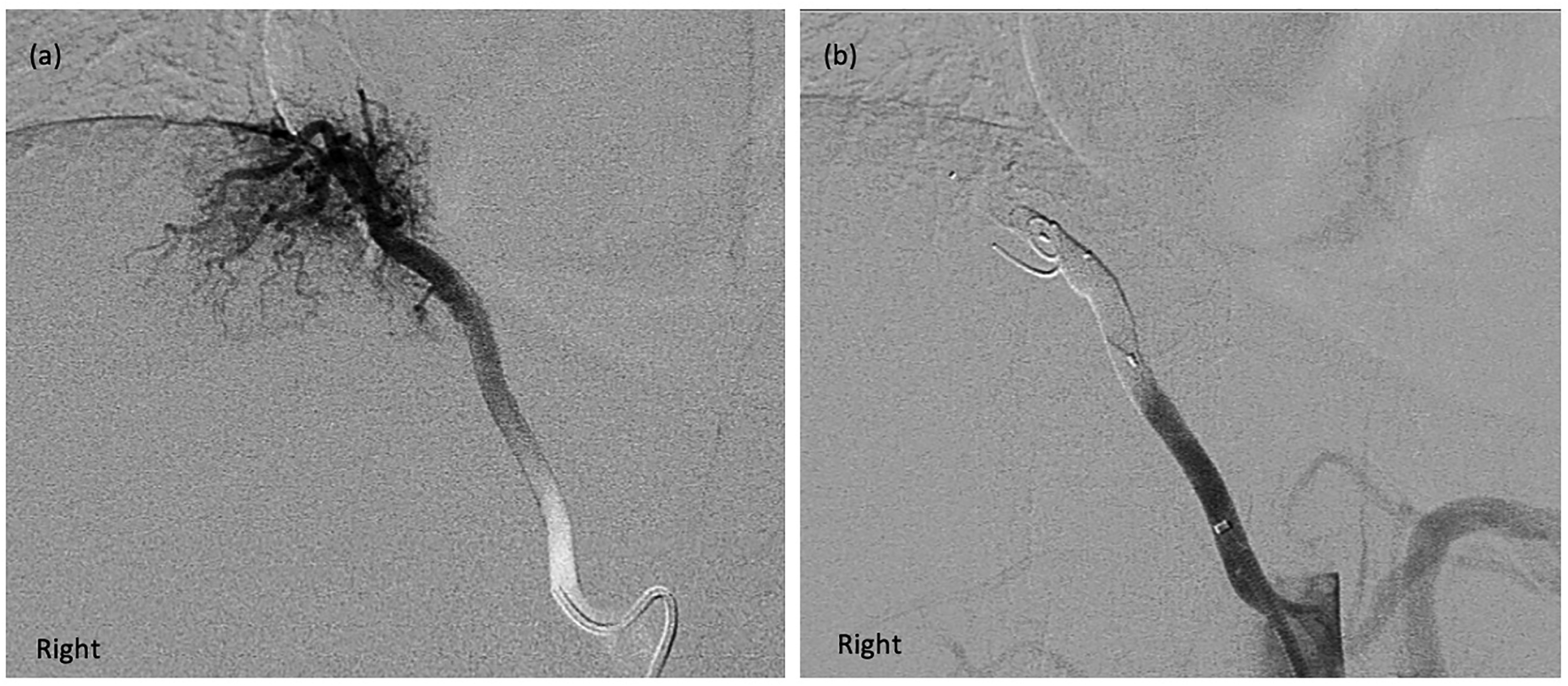
(a) Chest angiogram prior to embolisation, demonstrating an arterial systemic feeder into the posterior medial right lower lobe of lung. (b) Chest angiogram after embolisation, with occlusion of the vessel just proximal to branch point with two 7 mm Medtronic MVP™ and an 8 mm coil in between the two plugs.
When seen for outpatient evaluation 1 month later, the woman remained well, with no recurrent haemoptysis and no evidence of complications.
Discussion
The differential diagnosis of new onset haemoptysis in pregnant women includes thromboembolism, pulmonary infection, vasculitis, neoplasm, arteriovenous malformation, haemostatic disorders and ‘idiopathic haemoptysis of pregnancy’, which has been attributed to hormone-induced bronchial arterial dilatation and tortuosity. 14 Congenital malformations such as ABLL and PS are rare causes.
Cases of ABLL and PS in pregnancy and postpartum
The three prior case reports of ABLL and PS presenting for the first time in pregnancy all described onset of symptoms in the second trimester.11–13 In the single published case to date of ABLL presenting in pregnancy, Shaikh et al. 11 described that a woman at 20 weeks’ gestation with haemoptysis and a radiological diagnosis of medial right lower lobe PS, managed conservatively until caesarean section. Histopathology from the postpartum thoracoscopic surgery in fact confirmed ABLL with evidence of dissection of the abnormal artery, with pulmonary haemorrhage.
There have been two reports of PS presenting in pregnancy, though neither represent the classic congenital PS with aortic-source aberrant arterial supply. In the first, Freeman and Maxwell attributed a case of PS presenting with haemoptysis in the second trimester to an acquired mechanism; that of chronic inflammation from pulmonary tuberculosis altering the arterial supply of the lung. 12 This relates to a long-standing theory that some PS cases may be due to progressive obliteration of subsegmental pulmonary arteries, producing parasitisation and hypertrophy of the abdominal and thoracic aortic branches. The patient underwent thoracotomy and tuberculosis treatment, and delivered a healthy term infant. Secondly, Sayegh et al. reported a case of an enlarged, abnormal bronchial arterial supply, in a woman who presented with haemoptysis at 26 weeks’ gestation. She underwent arterial embolisation for right lower lobe PS, had no further episodes of haemoptysis during her pregnancy, and delivered a healthy infant. 13
Fatal massive haemoptysis has occurred in the postpartum setting with these conditions. 15 A woman with known PS diagnosed in infancy had never been treated. Following an uncomplicated pregnancy, 3 weeks postpartum she developed massive haemoptysis culminating in cardiac arrest. 15 Conversely, a seemingly uncomplicated pregnancy and postpartum period occurred in a woman identified 2 years later to have ABLL with recurrent haemoptysis. 16
Physiology of ABLL and PS presenting in pregnancy
Changes to cardiac output, systemic arterial dilatation and elevation of the diaphragm may be relevant to the presentation of this condition in pregnancy. The new onset of haemoptysis during pregnancy in the above three prior cases of ABLL or PS coincided with development of peak cardiac output in the second trimester, likely causing increased blood flow via the aberrant artery.11–13 In our patient's case there was no haemoptysis until the 35th week, making this mechanism unlikely the sole factor.
Physiological changes in arterial structure and function are greatest in the third trimester and peripartum. Arterial dilatation occurs from early pregnancy, with the greatest arterial diameter, greatest pulse-wave velocity and lowest intima-media thickness shown in the third trimester. 17 This may increase the likelihood of haemoptysis in the third trimester from abnormally dilated arteries with hyperaemia, 14 and also the risk of arterial rupture. 18
Progressive uterine distension results in elevation of the diaphragm and altered thoracic configuration. The enlarging uterus increases the end-expiratory abdominal pressure, thereby displacing the diaphragm upwards. In our patient the systemic arterial supply to the right lower lobe arose from the thoracic aorta at the level of the diaphragmatic crus. This may have resulted in mechanical compression of the arterial supply to the involved pulmonary tissue, leading to infarction and haemoptysis.
Timing and mode of delivery
Given the persistence of haemoptysis in our patient, and the potential for massive haemoptysis, intervention was deemed warranted. Both thoracic surgical and endovascular interventions for PS at this gestation were considered technically difficult and of relatively high risk. Therefore, the decision for delivery at 36 weeks and 4 days’ gestation was taken.
Regarding the mode of delivery, vaginal delivery with a symptomatic unsecured aberrant arterial supply to the lungs has not been described in the literature. In our patient's case the anomalous artery arose from the aorta at the level of the diaphragmatic crus, and there was concern for a pseudoaneurysm shown on Computed Tomography Chest Angiogram (CTCA). We were concerned that repeated expulsive contractions with Valsalva during the second stage of labour may increase the risk of arterial rupture, with subsequent catastrophic haemorrhage. Furthermore, nulliparous women who are induced prior to 37 weeks have lower rates of vaginal delivery than parous women, 19 with a vaginal delivery rate of 52.8% in one study. 20 In our patient, the risks of labour, and the significant potential for an unsuccessful vaginal delivery requiring emergency caesarean section, prompted a decision to proceed with elective caesarean section.
Choice of definitive management modality in the postpartum setting
Surgical resection with ligation of the anomalous arteries has been the treatment of choice historically, for both symptomatic and asymptomatic cases of ABLL and PS.9, 21 An alternative treatment is exclusion of the aberrant arterial supply via an endovascular approach, utilising various occlusion devices, resulting in the embolised lesion undergoing ischaemia, infarction and even disappearance. 22 No head-to-head studies have been performed comparing these approaches. For cases identified incidentally and with no symptoms, a surveillance approach has been adopted in some centres. Among nine unresected cases with mean follow-up of 19 months available, no complications related to PS were reported. 23
Transarterial embolisation for PS was first described in the 1980s and 1990s, with its initial role to control massive haemoptysis prior to surgical management. 24 It is now an accepted stand-alone therapy with a good efficacy and safety profile in selected cases. 22 It has also been used in multiple cases for ABLL since 2000. 25 The risks include embolisation of non-target arteries, recanalisation, incomplete embolisation, and subsequent pulmonary infection because the abnormal parenchyma has not been resected. 22
There is a paucity of data available regarding the optimal management of this condition in pregnancy, with the options being conservative management until delivery, thoracic surgery or transarterial embolisation. The key considerations are the severity and trajectory of haemoptysis and other symptoms, the size and number of pseudoaneurysms on imaging, and gestation. Where there is massive haemoptysis, increasing volume or frequency of haemoptysis, or other worsening symptoms, emergent or urgent embolisation or surgery may be indicated. The presence of large or multiple pseudoaneurysms on imaging may also prompt intervention during pregnancy due to the risk of rupture. An earlier gestation at presentation would prompt greater consideration of intervention during pregnancy, due to the more prolonged risk period for massive haemoptysis associated with conservative management in this instance. Although thoracic embolisation would likely result in below-threshold dose during the second or third trimester (e.g. <50 mGy), 26 in the semi-urgent setting, consultation/informed consent with the patient and medical physicist and performing interventionalist would be ideal to discuss the potential radiation-related risks to the fetus.
In our patient's case, given the presentation with haemoptysis alone, and the absence of prior recurrent infections of the affected segment, a transarterial embolisation approach was chosen. The procedure was deferred until 9 days postpartum, to reduce the impact of pregnancy-related haematological changes including gestational thrombocytopaenia. 27
Given that our patient underwent uncomplicated caesarean section and successful treatment of ABLL, vaginal birth after caesarean section would be an option in a future pregnancy.
Conclusion
To our knowledge this is the first case of a pregnant woman presenting with haemoptysis due to ABLL, managed postpartum with transarterial embolisation. A multidisciplinary approach was necessary to determine treatment options.
Footnotes
Acknowledgements
The authors would like to thank the patient for allowing us to discuss her case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This project has been reviewed by the Chair's Delegate of the Gold Coast Hospital and Health Service Human Research Ethics Committee (on behalf of Metro North Hospital and Health Service HREC) and deemed not requiring HREC review on the basis that it is a Case Study. The exemption reference is EX/2022/QRBW/87264.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
Guarantor
The guarantor is Dr Kate Hawke.
Contributorship
All authors contributed to the patient care. KH and KJ drafted the manuscript. The article was reviewed and edited by all authors and all approved the final version of the manuscript.
