Abstract
Background
Myasthenia gravis is an autoimmune disease which can impact pregnancy.
Methods
Six databases were systematically searched for studies with at least five subjects reporting pregnancy outcomes for women with myasthenia gravis in pregnancy. Assessment of bias was performed for all included studies. Forty-eight cases from our own centre were also included in the analysis.
Results
In total, 32 publications met inclusion criteria for systematic review, for a total of 33 unique data sets including 48 cases from our institution. Outcome data was available for 824 pregnancies. Spontaneous vaginal delivery occurred in 56.3% of pregnancies. Overall risk of myasthenia gravis exacerbation was 33.8% with a 6.4% risk of myasthenic crisis in pregnancy and 8.2% postpartum. The incidence risk of transient neonatal myasthenia gravis was 13.0%.
Conclusions
The current systematic review provides the best estimates of risk currently available to aid in counselling women with myasthenia gravis in pregnancy.
Introduction
Myasthenia gravis (MG) is an antibody-mediated disorder affecting the neuromuscular junction. 1 The majority of affected individuals will have antibodies against acetylcholine receptor (AChR) or muscle specific kinase. Patients experience fluctuating weakness in the skeletal muscles, primarily affecting the ocular, bulbar, limb and respiratory muscles. Though uncommon, with a reported prevalence in the general population of 0.3–7.7 per 100,000,2, 3 MG incidence spikes for women during the second and third decades of life and therefore can impact pregnancy. Recent guidelines make recommendations about the optimal management of MG in pregnancy4, 5 though high-quality data is lacking.
MG is a disease with a variable course, with episodic exacerbations requiring more intensive treatment. During an exacerbation, individuals experience worsening muscle weakness or other related symptoms and may require increased dosages or additional types of medication for symptom control. Myasthenic crisis on the other hand, is a life-threatening complication involving weakness of the respiratory and/or bulbar muscles leading to respiratory compromise requiring ventilatory support. 6
MG treatment primarily involves symptomatic management with anticholinesterase agents, with steroids and other immunosuppressive agents used in more severe disease. For refractory cases or myasthenic crises, intravenous immunoglobulin (IVIG) and/or plasmapheresis can be employed. Thymectomy has been demonstrated to improve symptoms for individuals with a thymoma or those with thymic hyperplasia in young-onset AChR antibody cases. 7
MG has a variable course in pregnancy. Though it may happen at any time, exacerbation seems most likely to occur in the first trimester and during the postpartum period. 8 Previous studies report overall pregnancy exacerbation rates on the order of 30%,9, 10 though higher11, 12 and lower 13 rates have been reported. The literature is similarly discordant with regard to pregnancy outcome, with some studies reporting increased rates of preterm prelabour rupture of membranes (PPROM), preterm birth (PTB), small for gestational age (SGA) babies and caesarean delivery,14, 15 and others reporting rates similar to that of the general population.16, 17 Transient neonatal myasthenia gravis (TNMG) is a complication whereby the autoantibodies pass through the placenta causing a self-limited myasthenic syndrome in the neonate. Risk of TNMG is generally quoted to be 10–15%, with individual series demonstrating rates ranging between 3–4%15, 18 and 33%. 19
The present study sought to systematically review all reported case series (5 or more cases) of MG during pregnancy and to include a large case series from our institution in an effort to summarize the present literature and better understand the risks associated with MG in pregnancy.
Materials and methods
The review was registered in the PROSPERO database (2019; registration number CRD42019143072) prior to study initiation. The methods were performed in accordance with the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines (http://www.prisma-statement.org).
Search strategy
The following electronic bibliographic databases were searched using a comprehensive search strategy developed by an information specialist (M.R.): (1) Ovid MEDLINE, (2) Ovid EMBASE, (3) Ovid EBM Reviews – Cochrane Central Register of Controlled Trials, (4) PubMed (Non-Medline records), (5) Web of Science and (6) LILACS (Latin American & Caribbean Health Science Literature). Additionally, all registered controlled trials were screened in ClinicalTrials.gov, the WHO International Clinical Trials Registry Platform and the International Standard Randomised Controlled Trial Number Registry. The search strategy was structured according to the 2015 Peer Reviewed Electronic Search Strategies Guidelines. Subject headings and free text terms related to ‘myasthenia gravis’, ‘pregnancy’ and ‘obstetric delivery’ were included. No restrictions were applied to publication year, language or age. All databases were searched for relevant references published from their inception up to 31 July 2020. All references and duplicate records were managed and screened using EndNote citation management software and Covidence systematic review software (Covidence systematic review software, Veritas Health Innovation, Melbourne, Australia; www.covidence.org).
Inclusion criteria included randomized controlled trials, prospective or retrospective cohort studies and case series of at least five subjects with MG in pregnancy. Exclusion criteria included reviews without individual patient-level data, publication of data reported in previous publications and abstract presentations.
Data extraction and assessment of risk of bias
Two authors (HB and KMN) independently screened all titles and abstracts. Relevant studies were then pulled for full text review. These two authors independently reviewed, extracted data and assessed risk of bias, using “Quality Assessment Tool for Case Series” (NIH.gov), for each full text article. Disagreements were settled via consensus or if necessary, with discussion involving a third author (KEM).
Data was extracted from included studies using a standardized data extraction form. Extracted data included disease information, including MG treatment, proportion of pregnancies with exacerbation, stable disease and asymptomatic/symptomatic improvement in pregnancy, and the rate of myasthenic crisis during pregnancy and up to 6 weeks postpartum. Data was also extracted regarding pregnancy outcomes, including mode of delivery as well as the rate of pregnancy complications, including: PTB less than 34 weeks and less than 37 weeks gestational age, PPROM, SGA less than 10th centile (based on birthweight for given gestational age) and the proportion of neonates experiencing TNMG. Studies were screened to exclude data sets which included previously published patient data.
Institutional case series
After local Research Ethics Board approval (MSH REB #19-0183-C, 26 July 2019), a retrospective chart review of all pregnant women with myasthenia gravis cared for at Mount Sinai Hospital, Toronto, was conducted from the Medical Disorders of Pregnancy clinic database from 1 January 2000 to 31 August 2019.
Cases were included if there was a documented diagnosis of myasthenia gravis made prior to or during the pregnancy based on clinical or electrophysiological criteria. Cases of congenital myasthenic syndromes were excluded as this was felt to represent a separate disease entity. Only cases which were followed for the duration of pregnancy at our institution with available birth outcomes were included.
For the purposes of this study, each pregnancy was treated as a separate case for women having more than one pregnancy. Although multiple pregnancies in the same individual will have correlated outcomes, the previously published literature on this topic included subsequent pregnancies in the same woman and it was not possible to assign pregnancies to individuals in these studies. Therefore, this convention was followed in our case series to maintain consistency. This was felt to be reasonable as comparative statistics were not planned.
For each case, data regarding demographics, diagnosis, MG treatment, disease course during pregnancy and up to 6 weeks postpartum, mode of delivery and maternal-fetal-neonatal outcomes were collected. Disease course was defined as “asymptomatic/symptomatic improvement” if the woman had an improvement in their symptoms or remained asymptomatic throughout the pregnancy, “unchanged” if symptoms were stable throughout pregnancy and “exacerbation” if the woman experienced a documented increase in the frequency or severity of symptoms or required an increase in medication dose or an additional treatment modality in pregnancy. Neonates were considered to be SGA if their birthweight was less than the 10th percentile for gestational age based on the growth curves created by Kramer et al. 20
Statistical methods
Descriptive statistics were performed using SPSS (IBM SPSS Statistics, Version 23.0, Armonk, NY: IBM Corp).
Results
Our literature search yielded 980 citations. After removing 334 duplicates, 646 titles and abstracts were screened for inclusion. Of these, 227 were retrieved for full-text review and 32 ultimately were included in our review (Figure 1). Our case series data was included in the final review for a total of 33 unique data sets involving MG in pregnancy.
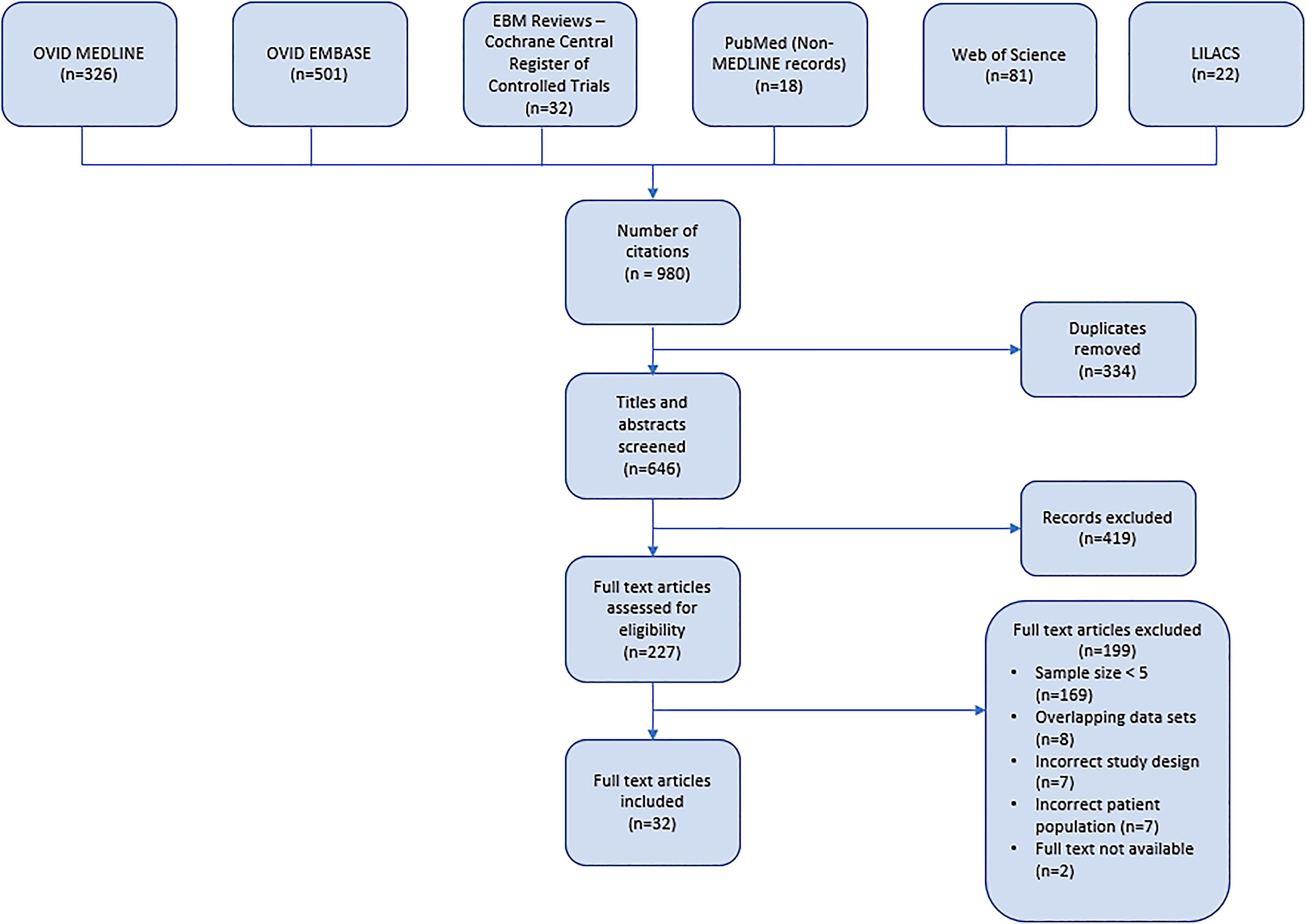
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of systematic literature search for studies related to myasthenia gravis in pregnancy.
In our institutional case series, 39 women with MG had 48 pregnancies. The demographic information, MG treatment and pregnancy outcomes for women at our centre are summarized in Table 1. Individual patient level data is available as a Supplemental Table.
Demographic information, disease course and pregnancy outcomes of women with myasthenia gravis in case series.

IVIG : intravenous immunoglobulin; PLEX: plasmapharesis; SVD: spontaneous vaginal delivery; PPROM: preterm prelabour rupture of membranes; TNMG: transient neonatal myasthenia gravis.
The data collected from the 32 studies8, 11, 16, 21–46 included in the systematic review as well as our own case series are summarized in Table 2. The included studies represent a heterogeneous group from 22 different countries with case data collected from 1947 up until 2020. Twenty-eight of the included studies were case series, four used a retrospective cohort design and one case-control study was included. Sample sizes ranged from 5 to 163 subjects with MG in pregnancy. There was significant variability in the type of data collected between studies. The significant heterogeneity in study design and reporting precluded meaningful comparative analysis of the study data.
Description of individual studies retrieved in the systematic review of myasthenia gravis in pregnancy.
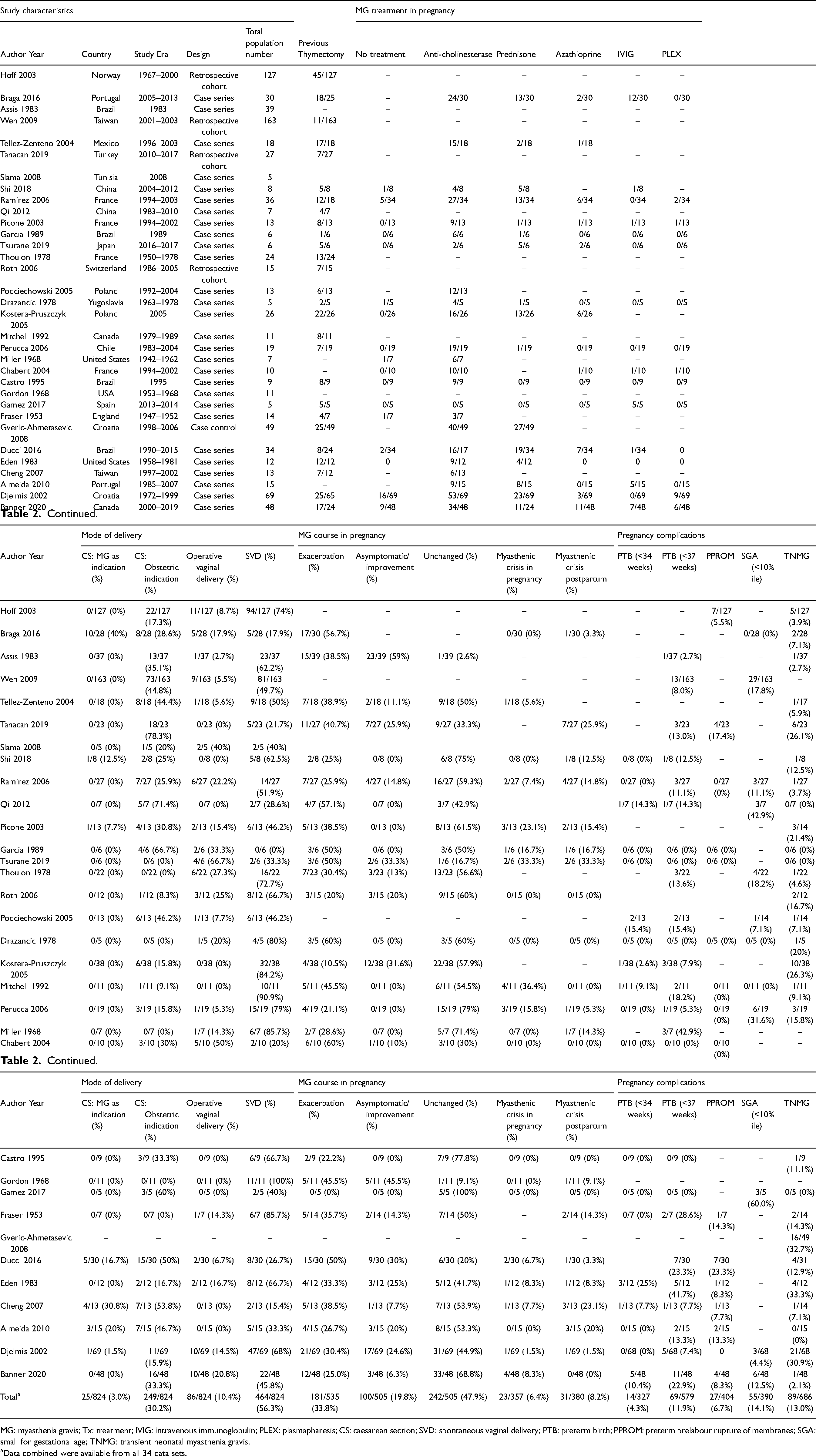
MG: myasthenia gravis; Tx: treatment; IVIG: intravenous immunoglobulin; PLEX: plasmapharesis; CS: caesarean section; SVD: spontaneous vaginal delivery; PTB: preterm birth; PPROM: preterm prelabour rupture of membranes; SGA: small for gestational age; TNMG: transient neonatal myasthenia gravis.
Data combined were available from all 34 data sets.
Risk of bias was assessed and this is summarized in Figure 2. Due to the nature of methodology used for these studies, the overall risk of bias is quite high.

A) Summary of risks of bias with the use of the Quality Assessment Tool for Case Series (NIH.gov). Green represents low risk of bias. B) Risk of bias items from the Quality Assessment Tool for Case Series represented by percentages.
MG treatment in pregnancy
The proportion of women with a thymectomy prior to pregnancy ranged from 16.7% 36 to 100%.19, 37 Across studies, the majority of women were treated with anti-cholinesterase medications during pregnancy, though a significant number also required prednisone.34, 39, 46 More recent studies were more likely to include women treated with Azathioprine, 39 IVIG 11 or plasmapheresis. 41 Management changes over time reflect practice changes as new medications and evidence for treatment has become available.
MG course in pregnancy
Across all studies reporting this outcome, 181/535 (33.8%) women experienced an exacerbation of their MG during pregnancy. While not all studies differentiated between stable disease and improvement, in those that did, 242/505 (47.9%) had stable disease and 100/505 (19.8%) had an improvement in their symptoms from baseline.
In studies reporting this outcome, 23/357 (6.4%) women experienced an episode of myasthenic crisis in pregnancy, and 31/380 (8.2%) experienced a myasthenic crisis during the postpartum period.
Mode of delivery
The overall rate of caesarean delivery done for MG was 25/824 (3.0%), with an additional 249/824 (30.2%) women having a caesarean delivery for an obstetric indication. The rate of operative vaginal delivery was 86/824 (10.4%), with 464/824 (56.3%) of women having a spontaneous vaginal birth.
Pregnancy complications
PTB less than 34 and 37 weeks’ gestation occurred in 14/327 (4.3%) and 69/579 (11.9%) pregnancies, respectively. The overall risk of PPROM was 27/404 (6.7%).
In studies providing information on birthweight for gestational age, 55/390 (14.1%) neonates were smaller than the 10th percentile. The risk of TNMG was 89/686 (13.0%).
Discussion
Myasthenia gravis is an uncommon neuromuscular condition which can have a significant impact on pregnancy. Previous publications have described a wide range of risk of MG exacerbation during pregnancy, ranging in individual series from 0% 37 to 60%40, 47 depending on the study. In our systematic review, 33.8% of women with MG overall experienced an exacerbation of their symptoms during pregnancy, a finding consistent with the generally quoted risk of 30–45%.9, 10 Previous authors have stated that exacerbation is most likely to occur in the first trimester and in the postpartum period, 8 though the current review did not examine the timing of exacerbation within pregnancy since the timing of exacerbation was only reported in a small minority of studies.
Myasthenic crisis, an important and potentially life-threatening complication of MG, occurred in 6.4% of women during pregnancy and in 8.2% of women during the postpartum period.
The method by which women with MG give birth shows significant regional and temporal variation in the previously published literature, with the proportion of women having a vaginal birth ranging from 0% 36 to 100% 35 in some small series. Overall, 56.3% of births across all included studies occurred by SVD and an additional 10.4% were operative vaginal deliveries. In total, 33.3% of deliveries were via caesarean section, with 30.2% done for an obstetric indication. Only 3.0% of women had a caesarean delivery with MG as the indication.
For women with symptomatic MG at the time of birth, there is some concern that the voluntary striated muscles used during active pushing may be weakened by MG exacerbation and further compromised by excessive maternal effort, precipitating myasthenic crisis. 48 For women with exacerbation symptoms at the time of delivery, an assisted second stage of labour may decrease the need for maternal effort and minimize this risk. This has not been universally adopted as standard practice, and indeed current guidelines note that spontaneous vaginal delivery is the objective and should be encouraged. 4
Women with MG seem to be at increased risk of PPROM compared to the 3% 49 risk in the general population, with this complication occurring in 6.7% of all pregnancies in women with MG. This finding has been previously reported,8, 39 though a pathophysiologic explanation remains elusive.
Whether or not MG increases the risk of preterm labour and birth has been the subject of previous controversy in the literature, with some populations, our own included, appearing to demonstrate an elevated risk. 33 Our systematic review did not find an elevated risk of PTB in women with MG, with overall risks of PTB of 11.9% prior to 37 weeks and 4.3% prior to 34 weeks, which is similar to the population risk. 50 Tanacan et al. 8 have previously suggested that an elevated risk of PTB may exist in the population who experience exacerbation in pregnancy 8 though it was not possible to assess this in the current study.
Previous studies have demonstrated conflicting findings regarding the association between MG and fetal growth, with high rates of SGA babies reported in some series12, 16, 17 but not others.11, 17, 41 Our review found that 14.1% of babies born to mothers with MG had an infant with a birthweight less than the 10th percentile for gestational age overall.
Finally, the occurrence of TNMG in individual studies has ranged from 0%12, 21, 36, 37, 46 to 33.3%.19, 34, 41 In our large sample, the overall rate was 89/686 (13.0%). Previous studies have demonstrated a lack of correlation between anti-AChR antibody titre, 42 duration of illness and treatment modalities 34 and the risk of TNMG. The current understanding of risk factors for this complication remains limited. Women should be counselled about this risk and parents and care providers should be aware of the signs of this condition and the fact that there is no known association between this outcome and any predictive risk factor.
The current systematic review represents an accumulation of data, which to our knowledge has not previously been synthesized systematically. Previous estimates of risk and recommendations for management in the literature have been based on individual single-centre case series, the results of which differ considerably.
We used a search strategy which encompassed multiple databases and did not eliminate studies based on publication language which allowed us to capture a diverse group of data sets. By excluding studies with sample sizes less than 5, some of the bias inherent in the publication of extreme case studies was minimized. This study extracted data on a large range of factors related to the treatment and outcomes of MG in pregnancy, and due to the large total sample represents the best estimate of the true risk presently available.
The heterogeneous nature of the included studies means that data is represented from many different geographical regions. The fact that we did not use time or geographical exclusions increases the generalizability of our data, but does mean that our findings include regional and temporal variations in practice.
The main limitation of a study of this nature is the inclusion of low-quality evidence, as the majority of included studies were small and did not use a comparative design. Case series are particularly vulnerable to convenience sampling and selection bias, and may not appropriately represent the wider population of women with MG embarking on pregnancy. The lack of a comparator group in the majority of included studies compromises internal validity and means that results are presented in a descriptive manner rather than employing meaningful comparative statistical analysis. Finally, most studies reported on pregnancies rather than by individual patients. It is possible that women with less severe disease and fewer complications may have contributed more pregnancies to the overall number. Risk estimates may have been under- or over-estimated as a result of the nature and quality of the studies included in the review, however the large cumulative number of included women allows the risk estimates to be as accurate as possible.
For women with MG entering pregnancy, the current study allows the current best risk estimates for use in counselling regarding disease behaviour as well as the risk of myasthenic and pregnancy complications.
Conclusion
Myasthenia gravis has important implications and considerations for pregnancy which should be discussed with women. A counselling summary based on the findings of our study is provided in Figure 3. Women with MG are at increased risk of requiring assisted vaginal delivery or caesarean delivery compared to the general population, though more than half of women with MG will be able to have a spontaneous vaginal birth. The overall risk of exacerbation in pregnancy is about 33%, with a small but significant subset experiencing potentially life-threatening myasthenic crisis in pregnancy or postpartum. MG does not appear to significantly increase the rate of other pregnancy complications, with the exception of PPROM, which occurs at a rate roughly double that of the general population. Neonates should be observed for signs of TNMG, regardless of antenatal risk factors or maternal status.

Clinical Summary for the clinician counselling about myasthenia gravis in pregnancy.
Supplemental Material
sj-xlsx-1-obm-10.1177_1753495X211041899 - Supplemental material for Myasthenia gravis in pregnancy: Systematic review and case series
Supplemental material, sj-xlsx-1-obm-10.1177_1753495X211041899 for Myasthenia gravis in pregnancy: Systematic review and case series by Harrison Banner, Kirsten M Niles, Michelle Ryu, Mathew Sermer, Vera Bril and Kellie E Murphy in Obstetric Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The Research Ethics Board of Mount Sinai Hospital approved this study (#19-0183-C, July 26, 2019).
Trial registration
The review was registered in the PROSPERO database (2019; registration number CRD42019143072) prior to study initiation.
Guarantor
KEM is the guarantor of the present work.
Contributorship
HB and KEM conceived, designed and oversaw the conduct of the study. HB wrote the initial draft of the manuscript, conducted abstract and full-text screening, conducted risk of bias assessment and synthesized the data. KMN conducted screening of abstracts and full texts and conducted risk of bias assessment. MR developed and carried out the search strategies. MS, VB and KEM provided clinical and methodological input. All authors critically revised the article and approved the final version for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
