Abstract
We present the case of a woman with a history of biliopancreatic diversion and duodenal switch procedure who developed severe malnourishment requiring total parenteral nutrition during three pregnancies. The widespread use of bariatric surgery, particularly among those of reproductive age, has led to an increase in the number of women who become pregnant following bariatric surgery. There is a paucity of evidence to guide nutritional recommendations for women during pregnancy post bariatric surgery. We review this literature and summarize key published evidence and provide comprehensive recommendations concerning the common challenges in the management of nutrition status during pregnancy. The focus is on the impact of malabsorptive bariatric surgeries on pregnancy outcomes, nutrient deficiencies, recommendations for micro- and macronutrient monitoring and supplementation, and altered glucose metabolism and implications for diabetes screening. Optimizing pregnancy outcomes for individuals following bariatric surgery requires multidisciplinary team management including obstetrical providers, obstetric medicine specialists, and dietitians.
Introduction
Obesity has become one of the most important public health concerns in many resource-rich countries. Obesity is defined by the World Health Organization as a body mass index (BMI) greater than or equal to 30 kg/m2, which may reflect a state of excess body fat that increases the risk of detrimental health effects. 1 The number of adults with obesity tripled from 1975 to 2016, with obesity becoming one of the most common chronic health conditions in people of reproductive age and a contributor to infertility. Risks during pregnancy include early pregnancy loss, medical complications during pregnancy and postpartum weight retention.2,3
Bariatric surgery has emerged as the most effective long-term treatment for obesity. 4 More than 80% of bariatric surgeries are performed for women and approximately half of these are performed for reproductive-aged women. 5 Sleeve gastrectomy (SG), a restrictive bariatric surgery, is the primary surgery performed worldwide with North America performing more of this type of surgery compared to Europe. The reverse is true when comparing the percentage of roux-en-Y gastric bypass (RYGB) procedures, both a restrictive and malabsorptive surgery, between North America and Europe. 6
Bariatric surgery has been shown to reduce the overall risk of maternal and fetal complications associated with obesity. And yet, paradoxically, it is associated with an increased risk of small for gestational age (SGA) infants and potentially an increased risk of stillbirth. 7 Furthermore, malabsorptive surgeries are associated with a significantly increased risk of intrauterine growth restriction (IUGR) when compared to restrictive surgeries. 8
We present a case and a review of the evidence on the management challenges during pregnancy following bariatric surgery, as well as provide recommendations for pre-pregnancy counselling and the antenatal and postnatal management of this patient population.
Case
A 34-year-old primigravid woman was referred to the Obstetric Medicine clinic at 22 weeks of gestation for management of malnutrition. She had a biliopancreatic diversion and duodenal switch procedure nine years previously with a subsequent weight loss of 80 kg. Her pre-pregnancy weight was 93 kg with a BMI of 27.1 kg/m2. Her medications included esomeprazole 40 mg daily, prenatal vitamins one tablet daily, vitamin D3 5000 international units (IU) daily, calcium gluconate 500 mg three times per day, magnesium 300 mg daily and vitamin A 10,000 IU three times per week.
Her first trimester was complicated by significant morning sickness. She noted night blindness (nyctalopia) and progressive peripheral swelling. On examination, her weight was 83 kg, blood pressure was 92/55 mmHg with heart rate 80 beats/min. She had peripheral lower limb oedema. Her investigations demonstrated a hemoglobin of 99 g/L, albumin of 26 g/L, ferritin 131 mcg/L, international normalized ratio (INR) was 1.1, vitamin B12 level 1190 pmol/L, 25-OH vitamin D level 8 nmol/L and vitamin A level that was undetectable.
She presented at 25 weeks with decreased fetal movements and was found to have an intrauterine fetal demise (IUFD) and gave birth to a stillborn male infant weighing 510 g. Prenatal screening and anatomical survey were normal. The fetus was appropriately grown for gestational age.
Autopsy and genetic testing were declined. The placental pathology was unremarkable. She was readmitted 2 weeks postpartum with progressive weight loss, anasarca, nyctalopia and steatorrhoea. Her serum albumin was 16 g/L and faecal fat testing demonstrated significant fat malabsorption of 85%. Computerized tomography (CT) abdomen showed extensive bowel wall thickening with mucosal oedema, bilateral pleural effusions and moderate volume ascites. Her common surgical anastomosis was patent. Gastroscopy was normal. Given the suspicion of micro- and macro-malnutrition, she was started on total parenteral nutrition (TPN) with good effect and was subsequently weaned after one month.
She presented the following year with her second pregnancy. This pregnancy was uncomplicated up until 26 weeks of gestation when she developed nyctalopia and peripheral oedema. By 28 weeks, she had lost 5 kg, developed steatorrhoea, and worsening nyctalopia despite changing her vitamin A from the oral route to intramuscular injections. The fetus was on the 40th percentile for estimated fetal weight (EFW). She was admitted at 34 weeks with worsening anasarca and EFW on the 8th centile. She was commenced on TPN. She had an induction at 36 weeks for IUGR and fetal well-being. She delivered a healthy girl weighing 2020 g. TPN was ceased immediately postpartum and her symptoms resolved.
She returned for care the following year with her third pregnancy. At 25 weeks, she developed steatorrhoea and peripheral swelling. She denied nyctalopia. The fetus was on the 46th growth percentile. It was noted that her albumin had reduced from 31 g/L to 26 g/L. She was commenced on TPN electively at 27 weeks and was transitioned to home TPN with improvement in symptoms. This was continued for the remainder of the pregnancy. She had a home birth, as was her wish, via a spontaneous vaginal delivery at 39 weeks and had a healthy baby girl who weighed 2977 g. TPN was ceased postpartum.
This case illustrates severe malnourishment during pregnancy following bariatric surgery. The woman’s first pregnancy was complicated by IUFD, and then iatrogenic late preterm birth secondary to poor fetal growth and well-being. With prompt initiation of TPN, she had a successful third pregnancy and term birth. In retrospect, it is possible that earlier initiation of TPN may have prevented severe malnutrition and complications in her earlier two pregnancies.
Overview of bariatric surgery and physiological impact
Types of bariatric surgeries and anatomical changes
Bariatric surgeries are classified as restrictive, malabsorptive, or a combination of the two. Common bariatric surgeries include: (1) gastric banding, in which a band is placed around the upper portion of the stomach to restrict intake; (2) sleeve gastrectomy, in which the greater curvature of the stomach is resected leading to restriction; (3) roux-en-Y gastric bypass, in which a small gastric pouch is created in addition to a gastrojejunostomy that bypasses the duodenum and early jejunum leading to both restriction and malabsorption; (4) biliopancreatic diversion (BPD), in which a subtotal gastrectomy is performed along with diversion of the biliopancreatic limb that carries bile and pancreatic juice away from the alimentary limb that transports food. A modification of the latter is the biliopancreatic diversion/duodenal switch (BPD/DS), in which a sleeve gastrectomy is performed, the pylorus is preserved, and an ileoduodenostomy is formed distal to the pylorus.9,10
Physiological changes and impacts
Metabolic and nutritional derangements are more common after malabsorptive surgeries than restrictive surgeries. Screening and monitoring of bloodwork is important to ensure serum vitamin and mineral levels are within normal limits. 11
The cause for nutrient deficiencies after these surgeries is multifactorial and relates to how absorption is impacted. More specifically, partial stomach resection removes the cells that secrete intrinsic factor necessary for vitamin B12 absorption; gastric acid secretions are altered, which influence absorption of iron, calcium, selenium, copper, and zinc; the duodenum and jejunum are bypassed with the RYGB and BPD/DS, and these are the primary sites of absorption for many nutrients; and vitamins A, D, E, and K are poorly absorbed due to inadequate mixing of bile and pancreatic enzymes in addition to reduced small bowel absorption. These factors may be coupled with gastrointestinal and dietary pattern changes in pregnancy that minimize food intake such as food aversions, changes in taste and smell, hyperemesis gravidarum, and constipation. 12
The malabsorptive effects of surgery are further worsened by changes to maternal physiology during pregnancy, including the effects of hemodilution due to increased maternal blood volume, decrease in concentration of nutrient-binding proteins, increase in renal perfusion leading to increased excretion of water-soluble vitamins, and effects on fat storage. Later in pregnancy there are increasing demands from the growing fetus.2,3 Any combination of these factors could lead to consequences such as a fetus who is SGA, preterm birth, congenital abnormalities, and perinatal mortality.
Inadequate supplementation has been linked to adverse fetal and maternal outcomes. A 2014 prospective cohort study found that the mean birthweight of babies to individuals who had a malabsorptive surgery was significantly lower compared to those who had a restrictive surgery (3.06 ± 0.56 kg versus 3.43 ± 0.47 kg, respectively; p = 0.024). 13 A 2015 systematic review demonstrated that the most common adverse neonatal outcomes related to maternal micronutrient deficiencies were visual complications (vitamin A), intracranial hemorrhage (vitamin K), neurological and developmental impairment (vitamin B12), and neural tube defects (folate). 14 Furthermore, a 2018 systematic review that looked at the maternal blood concentrations of 2056 women with pregnancies after bariatric surgery identified deficiencies in vitamins A, B1, B6, B12, C, D, and K, and in the minerals iron, calcium, selenium, and phosphorous. Documented maternal symptoms for these deficiencies include anemia (vitamin B12 and iron), night blindness (vitamin A), and urinary tract infections (vitamin A and D). 15 Despite these findings, both reviews conclude that the evidence on the association between micronutrient deficiencies in pregnant and postpartum women after bariatric surgery and adverse neonatal outcomes are inconclusive.
Pre-pregnancy counselling
A short interval period from the time of surgery to before weight has stabilized may be associated with potentially a higher risk for prematurity, SGA fetus, and neonatal intensive care unit admission. 8 The American Society for Metabolic and Bariatric Surgery (ASMBS) recommends waiting until weight has stabilized, which is generally 12–18 months after surgery. As fertility may resume rapidly following bariatric surgery, care providers should discuss pregnancy timing and use of contraception. 16 The use of non-oral contraceptives is recommended over oral contraceptives following malabsorptive surgeries such as RYGB, BPD, and BPD/DS due to the limited absorption of oral contraceptives. 17
Antenatal Care
Recommended macronutrient intake levels
There is a lack of evidenced-based specific dietary recommendations for pregnant individuals post bariatric surgery. 18 The recommendations used in practice at this time are limited to what is known about the diet advice for the non-pregnant post-surgical population combined with what is known about the diet advice for the pregnant non-surgical population.
Energy requirements are individualized with considerations to pre-pregnancy BMI, gestational weight gain (GWG), and physical activity level. GWG is the preferred indicator of energy adequacy. Energy requirements are typically less for individuals with a pre-pregnancy BMI of 30kg/m2 and above, excessive GWG, and lower physical activity level, and vice versa. 19
Daily protein requirements in the pregnant non-surgical patient ranges from 0.8 g/kg to 1.1 g/kg of pre-pregnancy body weight. However, this is highly dependent on the individual. In the non-pregnant post-surgical patient, protein requirements may be up to 1.5 g/kg of ideal body weight. 18 The general recommendation is a minimum intake of 60 grams of protein per day.18,20,21
Enteral nutrition should be considered in women who are unable to meet their nutritional needs orally. Parenteral nutrition should be considered when enteral nutrition is not indicated.
When parenteral nutrition is required, the American Society for Parenteral and Enteral Nutrition recommend hypocaloric (50–70% of estimated calorie needs) high nitrogen (protein provision of 1.2 g/kg actual body weight or 2–2.5 g/kg ideal body weight) feeding. This minimizes the risk of overfeeding in the acute phase of illness and refeeding syndrome while providing adequate micronutrients.
Recommended micronutrient intake levels for the prevention and repletion of micronutrient deficiencies, and maintenance of normal serum levels
Our recommendations for optimum micronutrient supplementation during pregnancy have been extrapolated from data from bariatric and obstetric literature (Table 1). The Institute of Medicine's Recommended Dietary Allowance (RDA) values for select micronutrients are listed in Table 2. Individuals should continue their pre-conception (a time period of at least 3–6 months prior to conception) micronutrient regimen during pregnancy while replacing the multivitamin with a prenatal multivitamin. Daily prenatal multivitamin is recommended before conception, during pregnancy, and postpartum for individuals who choose to breastfeed. Prenatal multivitamin should contain the minimum amounts of the following micronutrients and additional single micronutrients as needed to maintain serum concentrations of these nutrients within normal limits.
ASMBS: American Society for Metabolic and Bariatric Surgery; DS: duodenal switch; RYGB: roux-en-Y gastric bypass; BPD/DS: biliopancreatic diversion/duodenal switch; RDA: recommended dietary allowance.
Regular screening of micronutrient deficiencies will allow for individualized therapy and adjustment of dosages as required. Supplementation and screening should continue postpartum in those who breastfeed.
For women who are refractory to, unable to tolerate, or have difficulty complying with oral micronutrient supplementation, then intravenous (IV) or intramuscular (IM) injections are recommended. In practice, this has been observed to be most often the case for iron, vitamin D, and vitamin B12.
Iron
Iron deficiency and iron deficiency anemia are more common with malabsorptive surgeries than restrictive surgeries. 20 Explanations for this include decreased meat tolerance, decreased gastric acid secretion, and exclusion of the duodenum where the bioavailable form of iron is absorbed. Menstruating individuals are at risk of iron deficiency owing to blood loss. Therefore, they are often advised to commence oral iron supplementation prior to surgery to maximize iron stores. During pregnancy, iron requirements increase due to expanding blood volume. Regular monitoring is recommended to enable timely correction if necessary.
Symptoms and signs include fatigue, pica, pale conjunctiva, and koilonychia. Maternal iron deficiency is associated with adverse neonatal outcomes such as delayed growth and development. 23 Laboratory findings for iron deficiency include microcytic anemia (low red blood cell count, hemoglobin, hematocrit, and mean corpuscular volume), low serum iron, high total iron binding capacity, and/or low serum ferritin. Hemoglobin level below 105 g/L should be evaluated further for iron deficiency; ferritin levels below 30ug/L are diagnostic of deficiency.
Ferritin values between 30ug/L and 140ug/L do not rule out iron deficiency and should be evaluated further with a transferrin saturation, whereby levels below 15% also indicate iron deficiency. 24
Options to treat iron deficiency include oral and IV supplementation. Oral iron may be available in three main forms: non-heme iron, polysaccharide-iron complex, or heme iron. Non-heme ferrous salts such as ferrous sulphate are less well absorbed. They should be taken on an empty stomach and with vitamin C to aid absorption. They should not be taken with calcium and phytate-containing foods such as tea, coffee, and bran as these inhibit absorption. Side effects may include nausea and constipation. Polysaccharide-iron complex such as Feramax® are less likely to cause nausea and constipation. Heme iron polypeptide such as Optifer Alpha® is best absorbed and a good choice for those who experience constipation or stomach upset. Individuals may need 1–2 tablets daily or to try various types.
IV iron is indicated for women who are symptomatic, refractory to and/or unable to tolerate oral iron supplements, and have hemoglobin levels less than 100 g/L. IV iron can be administered as iron dextran (50 mg elemental iron/mL), iron sucrose complex (Venofer®, 20 mg elemental iron/mL), or iron sodium gluconate complex (Ferrlecit®, 12.5 mg elemental iron/mL). The dose is calculated based on body weight, hemoglobin level, and the amount of elemental iron/mL of the product. In practice, 1000 mg is generally sufficient to treat anemia. Doses above 1000 mg have not been shown to be clinically useful. This could be administered either as a single infusion or through multiple infusions depending on the product. Hemoglobin level takes approximately six to eight weeks to normalize. Other measures of iron stores also improve. 25 The duration of therapy is dependent on improvement in iron status and stores as determined by monitoring complete blood counts, ferritin, and iron studies.
Vitamin D
Among vitamin and mineral deficiencies, low vitamin D has been identified in people with obesity before and after surgery despite supplementation. Vitamin D deficiency is reported to occur in up to 100% of women. 20 Multiple clinical practice guidelines exist for vitamin D repletion recommendations in the non-pregnant post bariatric surgery population (Table 3). 26
Recommended dietary allowances and tolerable upper intake levels for select nutrients during pregnancy.
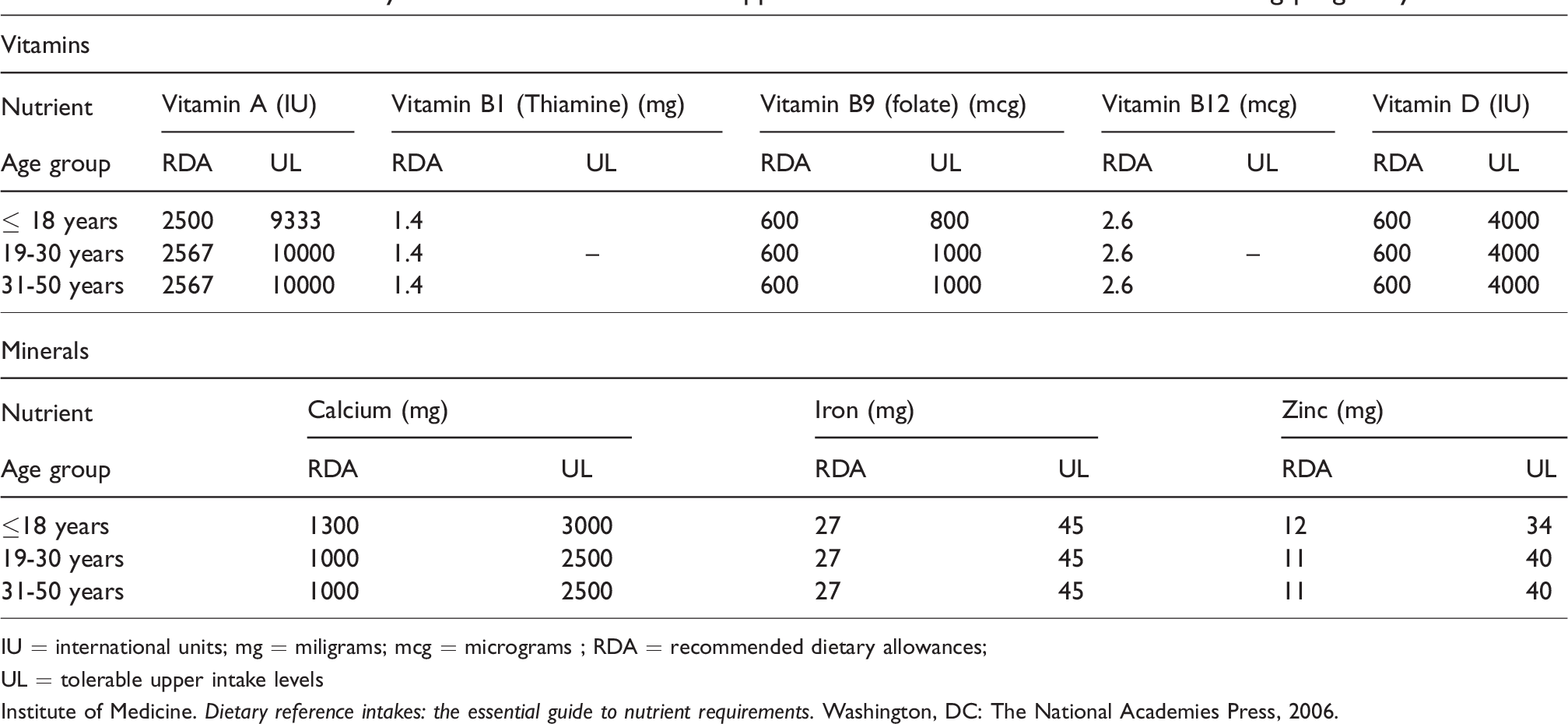
IU = international units; mg = miligrams; mcg = micrograms ; RDA = recommended dietary allowances;
UL = tolerable upper intake levels
Institute of Medicine. Dietary reference intakes: the essential guide to nutrient requirements. Washington, DC: The National Academies Press, 2006.
In a 2019 retrospective matched cohort study, a group of 50 women were provided with 1000 mg calcium carbonate and 800 IU oral vitamin D3 while a second group of 50 women were provided with the same supplements plus an additional 50,000 IU liquid vitamin D3 monthly.
Within each group, there were a greater number of people who underwent SG than RYGB, but this was not a statistically significant difference. Both groups saw an increase in serum 25-(OH) D levels compared to baseline, with the second group that received an additional 50,000 IU vitamin D3 each month seeing a higher level compared to the first group. 27
We recommend a baseline oral vitamin D3 supplement of at least 3000 IU (in addition to vitamin D that may be available in a prenatal multivitamin and calcium supplement). If serum 25-(OH) D levels do not improve and are below 75 nmol/L, we recommend increasing supplementation by 1000 IU, up to 6000 IU, until target serum 25-(OH) D level of at least 75 nmol/L is achieved.
For women who do not respond to the up titration of vitamin D3, we recommend a stronger dose of either 50,000 IU vitamin D2 or 20,000 IU vitamin D3 one to three times per week. The frequency and duration would be assessed on a case-by-case scenario depending on laboratory trends.
Vitamin B12
Vitamin B12 absorption is impaired after malabsorptive surgery because stomach size and therefore gastric acid production is reduced. The protein intrinsic factor that is required to bind to and absorb vitamin B12 is also isolated from the food stream. Deficiency, which is defined by the ASMBS as a serum vitamin B12 concentration below 200 pmol/L, occurs in less than 20% of patients with RYGB and 4–20% of patients with SG two to five years after surgery. 20
In a study that compared the effect of oral supplementation versus IM injection on serum vitamin B12 concentration, there was no significant difference in serum vitamin B12 and methylmalonic acid concentrations between the two groups after six months of therapy. Both treatments normalized previously low serum vitamin B12 concentrations. Oral treatment was provided in the dose of 1000 mcg daily and IM injection was provided at a one-time loading dose of 2000 mcg at baseline and then 1000 mcg bi-monthly thereafter. 28 The choice between the two should depend on which is most practical and effective for the patient.
Recommended micro- and macronutrient laboratory screening and monitoring
Individuals who have had prior malabsorptive surgeries are at greatest risk for micronutrient deficiencies. 13 Micronutrient screening has been recommended by the American College of Obstetrics and Gynecology, a consortium of the American Association for Clinical Endocrinologists, The Obesity Society, and the American Society for Metabolic and Bariatric Surgery. Similarly, the Society of Obstetricians and Gynecologists of Canada recommend that during pregnancy, women with obesity have their levels of hemoglobin, mean corpuscular volume, ferritin, and vitamin B12 assessed. Our proposed recommendations for nutrient screening and monitoring are detailed in Table 4 and laboratory reference ranges for key biochemical indices are provided in Table 5.
Comparison of vitamin D supplementation guidelines for non-pregnant individuals after malabsorptive bariatric surgery. 26
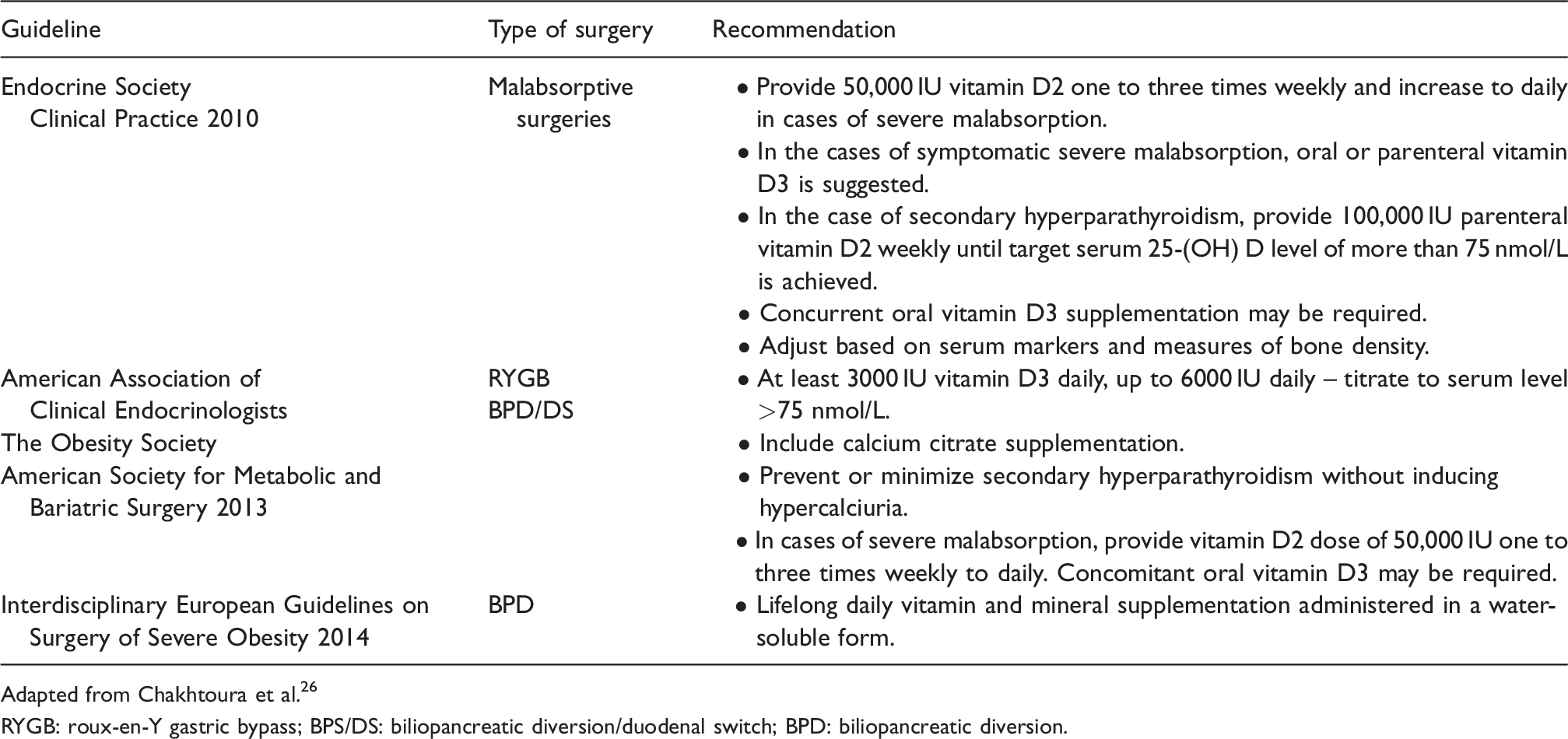
Adapted from Chakhtoura et al. 26
RYGB: roux-en-Y gastric bypass; BPS/DS: biliopancreatic diversion/duodenal switch; BPD: biliopancreatic diversion.
Proposed tests (fasting) for each stage of the peri-conception, pregnancy, and postpartum/breastfeeding periods post bariatric surgery. 18
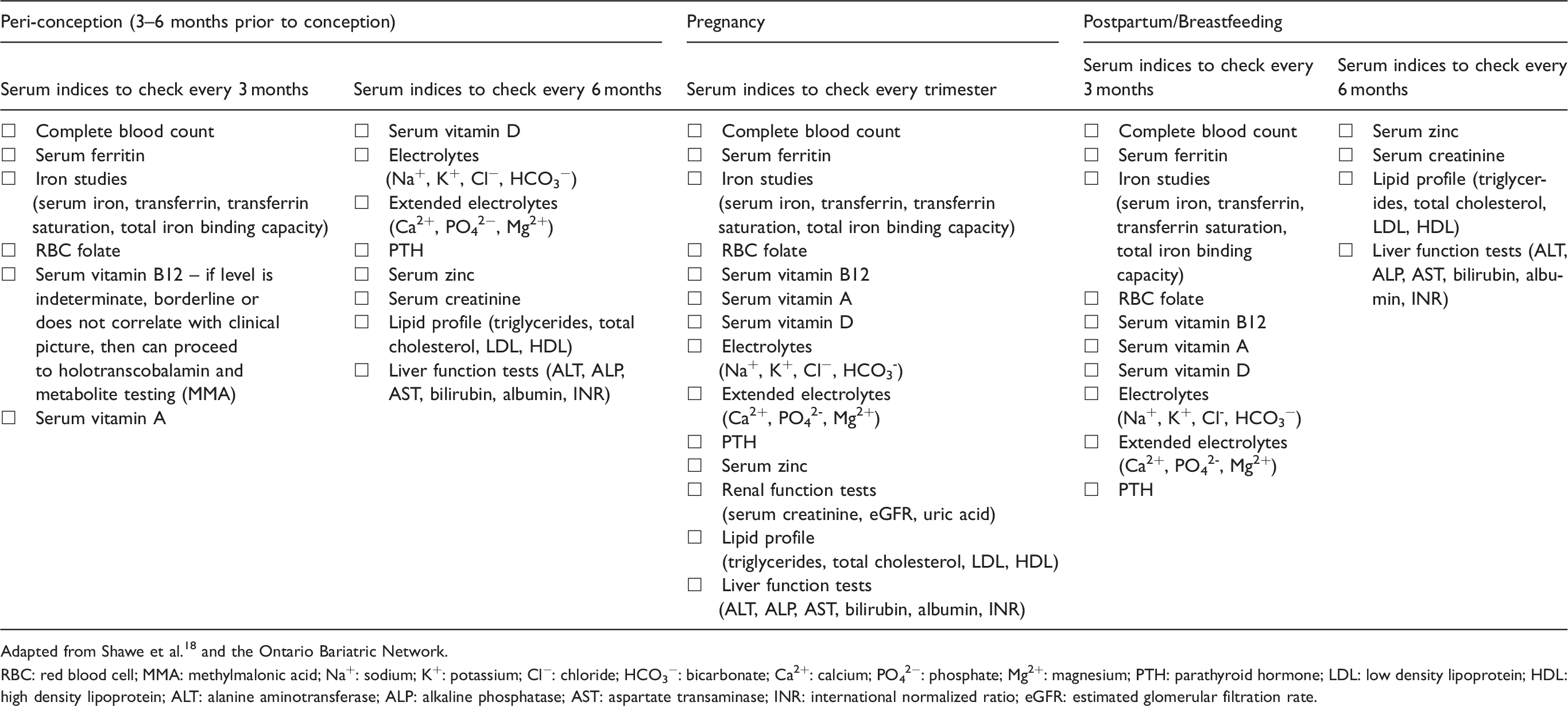
Adapted from Shawe et al. 18 and the Ontario Bariatric Network.
RBC: red blood cell; MMA: methylmalonic acid; Na+: sodium; K+: potassium; Cl−: chloride; HCO3−: bicarbonate; Ca2+: calcium; PO42−: phosphate; Mg2+: magnesium; PTH: parathyroid hormone; LDL: low density lipoprotein; HDL: high density lipoprotein; ALT: alanine aminotransferase; ALP: alkaline phosphatase; AST: aspartate transaminase; INR: international normalized ratio; eGFR: estimated glomerular filtration rate.
Gestational diabetes and diabetes screening
Individuals with obesity are at an increased risk of type 2 diabetes mellitus and gestational diabetes mellitus (GDM). First trimester screening should be considered for individuals who are at risk for diabetes such as those with a history of previous GDM, and may facilitate the diagnosis of pre-gestational diabetes. The International Federation of Gynecology and Obstetrics recommend screening and diagnosis of GDM between 24 and 28 weeks of gestation. The classic method of screening for individuals with no history of bariatric surgery is an oral glucose tolerance test (OGTT) in which 50 g or 75 g of liquid glucose is consumed. 30
For individuals who have had a malabsorptive bariatric surgery, the OGTT is inaccurate and can cause significant symptoms. This is due to the diversion of solid and liquid food contents away from the proximal small intestine and pyloric sphincter, which causes food to more rapidly exit the small gastric pouch and into the small intestine. Consumption of food and beverages high in glycemic index carbohydrates could cause dumping. Early dumping syndrome occurs 30–60 minutes after a meal in response to the body’s release of gut peptides after eating, which affects intestinal transit and causes symptoms such as low blood pressure, rapid heart rate, skin flushing and may be accompanied by diarrhea. Late dumping syndrome occurs 60–180 minutes after a meal and occurs in response to the body’s release of insulin after eating a large glucose load and causes symptoms related to reactive hypoglycemia including pre-syncope, diaphoresis, confusion, fatigue, heart palpitations, and tremors.
The proposed method of diabetes screening for pregnant individuals who have had a malabsorptive bariatric surgery is to test for fasting blood glucose and HbA1c in place of the glucose challenge and/or OGTT (Table 6) in early pregnancy, and with seven-point capillary blood glucose or one week of continuous glucose monitoring.
Reference laboratory ranges during pregnancy. 29
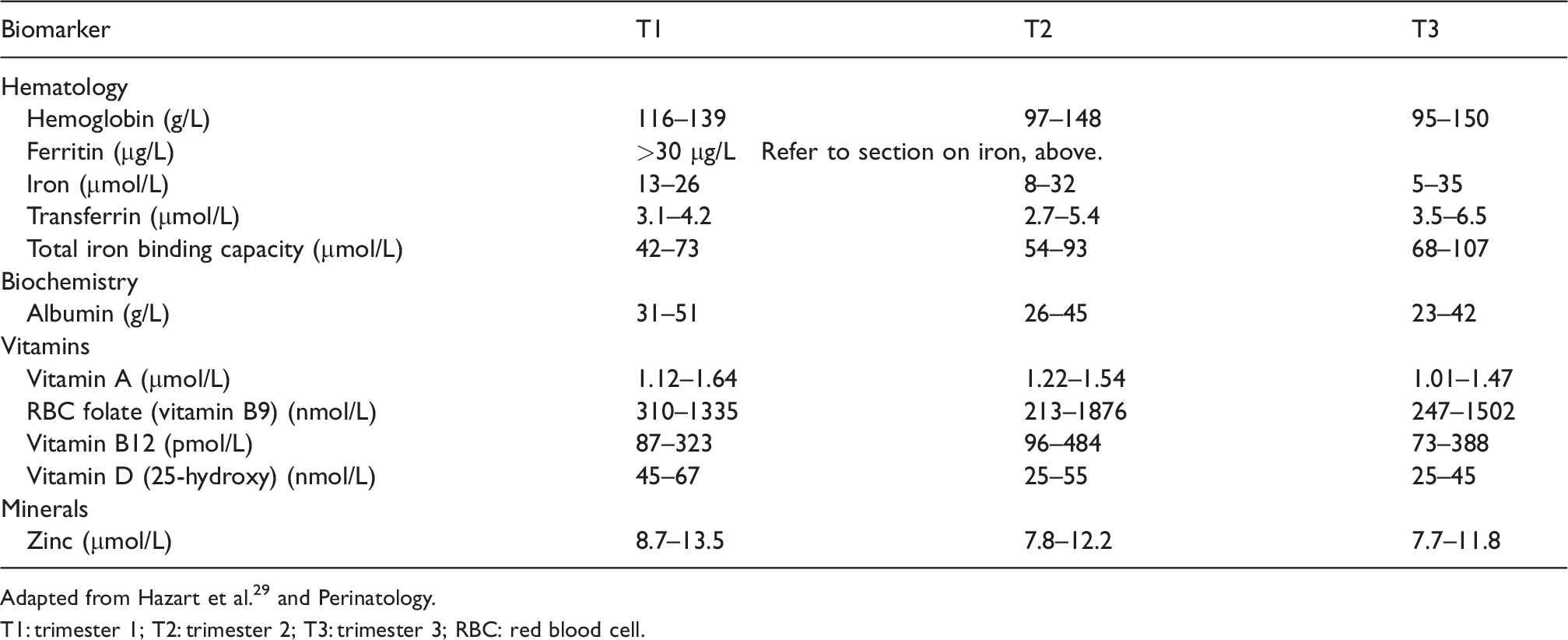
Adapted from Hazart et al. 29 and Perinatology.
T1: trimester 1; T2: trimester 2; T3: trimester 3; RBC: red blood cell.
Proposed tests for screening and monitoring gestational diabetes. 18

Adapted from Shawe et al. 18 and the Ontario Bariatric Network.
HbA1C: hemoglobin A1C; FBG: fasting blood glucose; T1: trimester 1; T2: trimester 2; T3: trimester 3; T2DM: type 2 diabetes mellitus; CBG: capillary blood glucose; CGM: continuous glucose monitoring; GDM: gestational diabetes mellitus.
Dietary interventions for the post-surgical patient remain the same as for the non-post surgical patient. They include small frequent meals, protein and low glycemic index carbohydrates at each meal and snack, and replacing sugar with sweeteners that can be safely consumed during pregnancy such as aspartame, sucralose, and stevia.
Gestational weight gain (GWG)
Weight gain during pregnancy can be a sensitive topic. After bariatric surgery, about 11% of patients still have obesity. This is concerning for increased risk of anaesthetic complications during labour and delivery, increased risk of thromboembolism, difficult delivery, and difficulty initiating and maintaining breastfeeding in the postpartum period. 17 Despite these concerns, pregnant individuals should avoid weight loss and instead focus on ensuring adequate nutritional intake and preventing excessive weight gain.18,19 While there are no specific recommendations for weight gain in the pregnant post-surgical population, interpretation and individualization of the recommended GWG for the non-surgical population could be applied. The Institute of Medicine recommends that for a singleton pregnancy, individuals with a pre-pregnancy or first trimester BMI of 30kg/m2 and above should gain about 5–9 kg. 31 Furthermore, minimizing GWG during the first half of pregnancy and in general for people with class III obesity (BMI of 40kg/m2 and above) may be beneficial. The interpretation and recommendations around GWG should be made in consort with fetal growth monitoring. 19
Fetal growth
As pregnancy following bariatric surgery can have effects on fetal growth, fetal monitoring in the form of ultrasound for growth and well-being during the third trimester is recommended. 19 Discussions regarding mode of birth are beyond the scope of this review.
Conclusion
Obesity is a growing concern worldwide and bariatric surgery is the most effective treatment. Most bariatric surgeries are performed for individuals in their reproductive years. Pregnant women who have had malabsorptive bariatric surgeries are at a high risk of protein energy malnutrition and micronutrient deficiencies. Therefore, an individualized approach for pre-pregnancy and antenatal counseling with patients on recommended dietary and supplemental intakes, laboratory screening and monitoring, and fetal monitoring with a team of obstetrical care providers, obstetric medicine specialists, and dietitians, among others, is recommended.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors did not receive financial support for the research, writing, and publication of this article.
Ethical approval
Ethical approval was not required for this article due to the nature of the research being a review of published literature.
Informed consent
The patient has given written consent for inclusion in the case presentation.
Guarantor
CM is the guarantor of the present work.
Contributorship
BH researched the literature and wrote the first draft of the manuscript. JY, SH, and CM provided direction in the conceptualization of this article. CM conceived the idea for this article and its collaborations. All authors reviewed and edited the manuscript, and were in agreement prior to final submission.

