Abstract
Background:
Some novel anticancer agents are associated with drug-induced interstitial lung disease (ILD), a critical and potentially fatal adverse event. Lung cancer patients appear particularly susceptible, yet the risk and clinical characteristics remain incompletely analyzed.
Objectives:
To comprehensively evaluate ILD risk and characteristics induced by novel anticancer agents in non-small cell lung cancer (NSCLC) using large-scale real-world data.
Design:
A retrospective pharmacovigilance study based on spontaneous adverse event reports.
Methods:
Data from 2014 to 2024 were extracted from the Food and Drug Administration Adverse Event Reporting System (FAERS). Reports of NSCLC patients who developed ILD during treatment with FDA-approved novel anticancer agents (immune checkpoint inhibitors, targeted therapies, antibody-drug conjugates). The reporting odds ratio (ROR) was used to assess the disproportionate reporting signals for each drug. Statistical significance was defined when the lower 95% confidence interval (CI) exceeded 1 with at least three reports.
Results:
A total of 4712 NSCLC cases were analyzed. Eight agents were identified with positive signals for ILD: ROR and 95% CI for nivolumab was 1.28 (1.20–1.38), pembrolizumab 1.47 (1.36–1.59), durvalumab 7.38 (6.90–7.89), atezolizumab 1.25 (1.12–1.39), ipilimumab 1.96 (1.74–2.21), tremelimumab 3.58 (1.97–6.50), trastuzumab-deruxtecan 3.14 (2.29–4.30), osimertinib 1.12 (1.03–1.23). The median onset time was 33 days, with 48.59% of ILD events occurring within the first month. The fatal cases experienced a significantly shorter onset time than non-fatal cases. Older age, male sex, and lower body weight were identified as factors affecting ILD, whereas lower body weight, male sex, and a higher number of concomitant drugs were linked to increased mortality.
Conclusion:
Our study identifies positive signals for ILD with eight novel antineoplastic agents in NSCLC, including nivolumab, pembrolizumab, durvalumab, atezolizumab, ipilimumab, tremelimumab, trastuzumab-deruxtecan, and osimertinib, highlights the importance of monitoring during the first month of therapy, and identifies older male patients with lower body weight as a high-risk group.
Plain language summary
Novel anticancer agents, including targeted therapies, immune checkpoint inhibitors (ICIs), and antibody-drug conjugates (ADCs), have improved outcomes for patients with non-small cell lung cancer (NSCLC). However, these drugs can sometimes cause interstitial lung disease (ILD), a potentially serious lung condition. Previous studies have been limited by small patient numbers and strict inclusion criteria, leaving gaps in knowledge about the risk and characteristics of ILD in real-world settings.
We analyzed data from the FDA Adverse Event Reporting System (FAERS), a large-scale, real-world database, to assess ILD risk associated with novel anticancer agents in NSCLC patients. We examined which drugs were more likely to induce ILD, when the ILD started, and what patient characteristics might increase the risk.
Eight drugs showed positive signals for ILD: nivolumab, pembrolizumab, durvalumab, atezolizumab, ipilimumab, tremelimumab, trastuzumab-deruxtecan, and osimertinib. Durvalumab had the strongest signal. Nearly half of ILD cases occurred within the first month of treatment, highlighting the importance of early monitoring. Male sex, older age, and lower body weight were associated with higher ILD risk.
This study provides real-world evidence to guide safer drug selection and highlights the need for personalized monitoring in NSCLC patients receiving novel anticancer therapies. Clinicians should closely monitor patients during early treatment and consider individual risk factors to reduce the likelihood of severe ILD and improve patient safety.
Keywords
Introduction
Lung cancer is the leading cause of cancer incidence and mortality, accounting for 18.7% of cancer-related deaths. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer cases. 2 Chemotherapy remains standard treatment across all stages, but response rates are limited to 20%–30% because of drug resistance. 3 Recent advances in novel anticancer agents, including molecular targeted therapies, immune checkpoint inhibitors (ICIs), and antibody-drug conjugates (ADCs), have radically improved treatment for NSCLC. 4
Despite their efficacy, these agents are associated with drug-induced interstitial lung disease (ILD), which may affect treatment continuity and patients’ quality of life. Drug-induced ILD is characterized by pulmonary interstitial inflammation and fibrosis caused by drug exposure.5,6 It lacks specific clinical manifestations, and its severity varies from mild to life-threatening. 6 The incidence of drug-induced ILD has shown a rising trend in recent years, with anticancer agents representing the leading cause (23%–51%). 7 ICI-related ILD has been reported between 3% and 5% in clinical trial settings, while reaching 19% in real-world settings, accounting for 35% of ICIs-associated deaths.8 –10 In addition, 10.5% of patients receiving trastuzumab-deruxtecan (T-DXd) developed ILD. 11 Lung cancer patients appear particularly susceptible to drug-induced ILD, exhibiting higher rates of severe events and mortality compared with other cancers.12,13 Real-world data showed a severe drug-induced ILD incidence of 2.52% in lung cancer compared to 0.62% across all tumors. 12 Onset time varies from days to years, with median onset at 3 months for ICIs (<0.5–19), 5.5 months for T-DXd (<0.1–46.8), and 4 weeks for epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs).7,14,15
The heterogeneity of ILD poses substantial challenges for both clinical practice and research. The majority of previous studies were clinical trials focused on a single drug or a particular drug class. However, discrepancies may exist between clinical trial results and real-world outcomes, partly due to cohort selection. Real-world studies often include older patients and individuals with poorer performance status, both of which are recognized risk factors for drug-related ILD.5,16 These findings highlight the importance of characterizing the drug-induced ILD in NSCLC patients treated with novel anticancer agents in real-world settings.
The U.S. Food and Drug Administration Adverse Event Reporting System (FAERS) is a publicly accessible pharmacovigilance database that includes spontaneous reports of adverse drug events from patients, healthcare professionals, and pharmaceutical companies. Updated quarterly, FAERS enables the analysis of long-term adverse event trends and the identification of emerging safety signals for marketed products and drugs.
This study aims to conduct a comprehensive real-world analysis of ILD associated with different classes of anticancer agents in NSCLC based on the data from the FAERS. It will provide real-world insights and identify potential safety signals to enhance clinical awareness, support early detection, and guide appropriate management.
Methods
Data sources
The analysis was conducted using data derived from the FAERS database. The extracted datasets included DEMO (demographic and administrative information), DRUG (drug information), REAC (coded adverse events), OUTC (patient outcomes), RPSR (report sources), THER (therapy start and end dates for reported drugs), and INDI (indications for drug administration). These datasets were linked via the unique PRIMARYID. FAERS is updated quarterly and is publicly accessible: https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html.
In FAERS, all recorded adverse events are coded using preferred terms (PTs) from the Medical Dictionary for Regulatory Activities (MedDRA).
Procedures
This study is a retrospective pharmacovigilance analysis focusing on ILD induced by novel anticancer agents used in the treatment of NSCLC.
Data were extracted from the first quarter of 2014 to the fourth quarter of 2024, and duplicate reports were removed following Food and Drug Administration (FDA) recommendations by retaining the latest FDA_DT and the higher PRIMARYID for the same CASEID. In this study, novel anticancer agents included targeted therapies, immune checkpoint inhibitors, and ADCs approved by the FDA for NSCLC indications as of December 2024. Both generic and brand names were used for the search (Table S1). ILD cases were identified in the FAERS database using the narrow “ILD” Standardized MedDRA Query (SMQ) (SMQ code: 20000042). Details for PTs are listed in Table S2. To ensure relevance to NSCLC, we further restricted the indication to “NSCLC”. In FAERS, reported drugs are categorized as primary suspect (PS), secondary suspect (SS), concomitant (C), or interacting (I). For disproportionality analyses of individual agents, only PS drugs were included to improve accuracy and reduce confounding effects from concomitant therapies.
In summary, we included ILD adverse event reports from the FAERS database (from the first quarter of 2014 to the fourth quarter of 2024) associated with novel anticancer agents approved by the FDA for NSCLC indications, focusing on PS cases. Reports that did not meet these criteria or contained duplicate submissions were excluded.
The reporting of this study conforms to the Reporting of Observational Studies in Epidemiology (STROBE) statement (Table S3). 17 The research procedure was detailed in Figure 1.

The flow chart of the study.
Statistical analysis
This study was a retrospective pharmacovigilance analysis based on the publicly available database. The sample size was determined by the number of reports meeting the study criteria.
Disproportionality analysis is widely used to evaluate potential associations between drugs and adverse events by comparing the proportion of adverse event reports for the drug of interest with that for all other drugs. In this study, we used the reporting odds ratio (ROR) to evaluate the potential association between novel antineoplastic agents used in NSCLC treatment and the occurrence of ILD. 18 ROR is widely used by various reporting agencies and the World Health Organization to assess disproportionality between cases and non-cases. In addition, compared to the proportional reporting ratio, ROR is compatible with logistic regression modeling and allows for the estimation of adjusted RORs when relevant covariates are available. 19 A significant signal was defined as the lower limit of the 95% confidence interval (CI) for the ROR exceeded 1, with at least three records in PT. Additional details about disproportionality analysis are reported in Table S4.
Time to onset (TTO) was defined as the duration from the start of treatment with novel antineoplastic agents to the occurrence of ILD events. Reports with missing onset times were excluded. To illustrate the TTO of ILD events, we reported the median number of days along with the interquartile range (IQR), which extends from the first to the third quartile. We additionally applied the Weibull distribution to characterize the distribution of TTO. 20 This distribution is characterized by two parameters: the scale parameter (α), which reflects the time scale of event occurrence, and the shape parameter (β), which characterizes the hazard pattern over time (β < 1: decreasing risk; β ≈ 1: constant risk; β > 1: increasing risk).
To mitigate potential biases and ensure the robustness of the results, multivariate logistic regression was conducted to adjust the RORs for potential confounding related to patient characteristics and concomitant therapies, thereby improving the robustness and validity of the results. Age, sex, and the number of concomitant medications were included as covariates, and concomitant drugs potentially affecting ILD were included as additional confounding factors in the model.
The chi-square test was used to compare patient characteristics between patients with and without drug-induced ILD, as well as between ILD cases with and without fatal outcomes. A two-sided p < 0.05 was considered statistically significant.
Data mining and statistical analyses were performed using the R software (version 4.3.0; R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Results
Descriptive analysis
From the first quarter of 2014 to the fourth quarter of 2024, a total of 14,695,638 reports were retrieved from the FAERS database. After restricting the analysis to novel antineoplastic agents with an indication for NSCLC and removing duplicates as recommended by the FDA, 4712 cases and 4968 reports of ILD were included in the study. These comprised 3499 cases associated with ICIs, 1171 cases with targeted therapies, and 42 cases with ADCs. Table 1 summarizes the clinical characteristics of the included patients. Most reports originated from Japan (59.06%) and the United States (16.17%). The median age was 69 years (IQR: 62–75), with the highest proportion in the 70–79 age group (1260, 26.74%). The proportion of males was notably higher than that of females (59.95% vs 27.70%). We also summarized and compared the clinical characteristics of patients with and without ILD, as shown in Table S5.
Characteristics of 4712 reported cases of ILD in NSCLC patients from the FAERS.

ADC, antibody-drug conjugate; FAERS, Food and Drug Administration Adverse Event Reporting System; ICI, immune checkpoint inhibitor; ILD, interstitial lung disease; IQR, interquartile range; NSCLC, non-small cell lung cancer.
Figure 2 displays the number of ILD cases and deaths caused by ILD over time. The proportion of fatal outcomes was highest in the ADCs group (45.24%), exceeding that observed with immunotherapy (29.10%) and targeted therapy (34.50%). However, fewer ILD cases were reported with ADCs, probably because of their later approval for NSCLC compared with ICIs and targeted therapies.
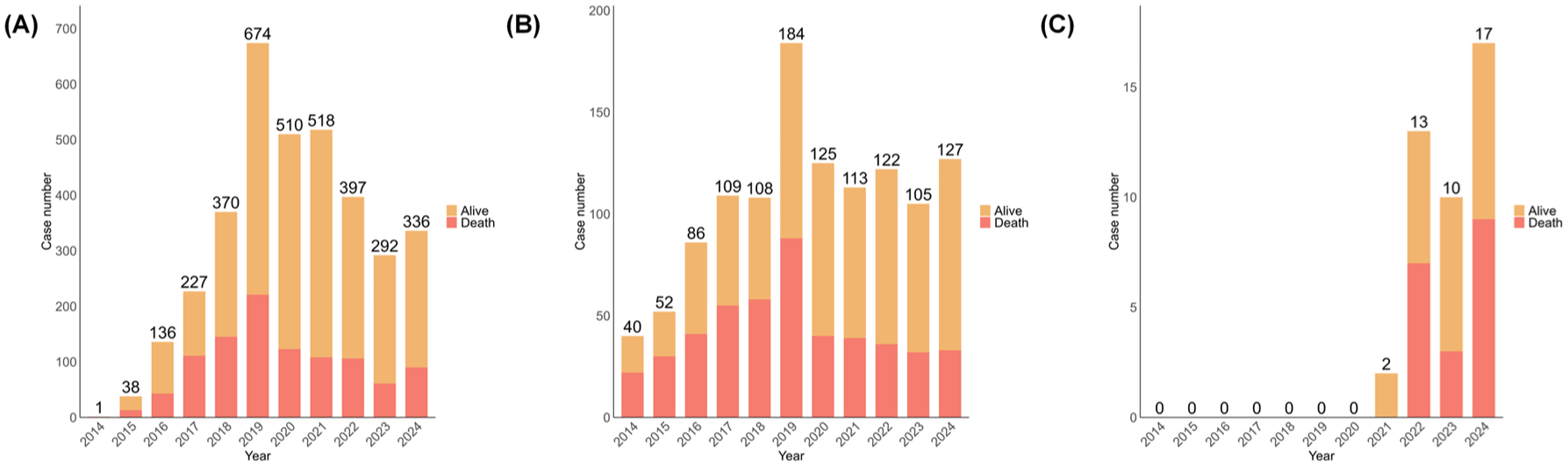
The number of interstitial lung disease cases and deaths associated with different classes of anticancer drugs from 2014 to 2024. Orange represents alive cases, and red represents death cases. (A) Immune checkpoint inhibitors; (B) Targeted therapy; (C) Antibody-drug conjugates.
Disproportionality analysis
We calculated the RORs for adverse event reports of ILD associated with each included drug (Figure 3(a)). In this study, eight agents were identified with positive signals associated with ILD, including nivolumab (ROR 1.28; 95% CI 1.20–1.38), pembrolizumab (ROR 1.47; 95% CI 1.36–1.59), durvalumab (ROR 7.38; 95% CI 6.90–7.89), atezolizumab (ROR 1.25; 95% CI 1.12–1.39), ipilimumab (ROR 1.96; 95% CI 1.74–2.21), tremelimumab (ROR 3.58; 95% CI 1.97-6.50), trastuzumab-deruxtecan (ROR 3.14; 95% CI 2.29–4.30), and osimertinib (ROR 1.12; 95% CI 1.03–1.23). Among these, durvalumab, a PD-L1 inhibitor, had the highest number of reported ILD cases (N = 1130) and showed the highest ROR signal value (ROR 7.38; 95% CI 6.90–7.89). It was followed by trastuzumab-deruxtecan (ROR 3.14; 95% CI 2.29–4.30), with only 42 reported cases. Among the 26 targeted agents included in this study, only osimertinib showed a positive signal (ROR 1.12; 95% CI 1.03–1.23).

The signal of ILD associated with novel antineoplastic agents in the treatment of NSCLC. (A) The forest plots of ROR values of novel antineoplastic agents associated with ILD in the treatment of NSCLC. (B) Heatmap showing the associations between novel antineoplastic agents and ILD. Red indicates a positive signal, defined as the lower limit of the 95% CI of ROR > 1 with at least three cases reported. Orange indicates a negative signal, defined as the lower limit of the 95% CI of ROR ⩽ 1 or fewer than three cases reported. Gray indicates the absence of a signal for the PT.
At the PT level, a total of 47 positive signals were identified. Durvalumab and nivolumab were associated with more PT signals, with 11 and 6 signals, respectively (Figure 3(b)). In total, ILD (N = 1737), pneumonitis (N = 1523), radiation pneumonitis (N = 865), pulmonary toxicity (N = 309), immune-mediated lung disease (N = 214), pulmonary fibrosis (N = 85), lung opacity (N = 54), lung infiltration (N = 52), hypersensitivity pneumonitis (N = 25), and eosinophilic pneumonia (N = 23) were the top 10 most frequently reported PTs in class-specific ILD.
Time to onset
After excluding records with missing onset time, 2245 ILD cases associated with novel antineoplastic agents in NSCLC treatment were included. The median onset time of ILD was 33 days overall (IQR: 13–81). Fatal cases developed ILD significantly earlier than non-fatal cases (p < 0.001), with a median TTO of 26 days (IQR: 10–68.5) versus 37 days (IQR: 14–85), respectively. The majority of ILD reports occurred within the first 30 days after drug initiation (48.59%, N = 1084), particularly for ICIs and targeted therapies (Figure 4(a)). Within 3 months, the cumulative proportion of ILD adverse event records was 77.86% (1748/2245; Figure 4(a)).

TTO of ILD adverse events associated with novel anticancer agents. (A) Proportion of ILD adverse event records at different time points among the three classes of novel anticancer drugs. (B) Proportion of ILD adverse event records at different time points across the top 10 PTs.
The onset times and Weibull shape parameters for each drug are presented in Table S6. To ensure reliable parameter estimation and valid confidence intervals, Weibull analysis was only performed for medications with at least 10 ILD cases. Weibull analysis indicated that all included ICIs and targeted therapies (except capmatinib) exhibited β < 1, characteristic of early failure type, with a progressively lower reporting of ILD over time. For trastuzumab-deruxtecan and capmatinib, the 95% CI of the Weibull parameters encompassed 1, indicating that ILD could occur at any time during treatment, without a time dependency. Among all agents, brigatinib had the shortest median onset time (8 days, IQR: 2–61, N = 34), while binimetinib had the longest (167 days, IQR: 110–204, N = 3; Table S6).
The onset times for the top 10 most frequently reported PTs among ILD cases associated with novel antineoplastic agents are shown in Figure 4(b). ILD had the shortest median onset (27 days, IQR: 10–67.5), followed by radiation pneumonitis (28 days, IQR: 14–55.5) and lung opacity (28 days, IQR: 10–70), whereas hypersensitivity pneumonitis (81 days, IQR: 34.5–237.8) and pulmonary fibrosis (74 days, IQR: 38.5–176.8) had longer median onset times.
Sensitivity analysis
To mitigate potential confounding effects, we performed multivariate logistic regression to adjust for age, sex, and the number of concomitant drugs, and calculated adjusted RORs (aROR). In addition, to further reduce the influence of other concomitant medications that potentially contribute to ILD, we presented the ILD risk of the top 30 most frequently used concomitant agents based on FDA label information (Table S7). Pemetrexed, paclitaxel, and cisplatin were identified as potentially associated with ILD. These three agents, together with other potential confounders, were included as covariates in the multivariate logistic regression model. To mitigate indication bias, only patients with NSCLC who received novel anticancer agents were included in the analysis. After adjusting for confounders, five agents were identified with positive signals for ILD: pembrolizumab (aROR 1.28; 95% CI 1.16–1.41), durvalumab (aROR 6.35; 95% CI 5.72–7.04), ipilimumab (aROR 1.76; 95% CI 1.52–2.03), trastuzumab-deruxtecan (aROR 5.72; 95% CI 3.60–8.90), and osimertinib (aROR 1.30; 95% CI 1.13–1.48). Notably, these five agents also showed positive signals before adjustment, indicating the robustness of the findings.
Risk factors associated with ILD
We conducted univariate and multivariate logistic regression analyses associated with novel antineoplastic agents in the treatment of NSCLC (Table 2). Univariate logistic regression analysis indicated that older age, male sex, a higher number of concomitant medications, and lower body weight were associated with ILD reporting. In the multivariable logistic regression model, older age, male sex, and lower body weight remained associated with ILD reporting, whereas the number of concomitant medications was no longer statistically significant.
Univariate and multivariate logistic regression analyses of factors associated with drug-induced ILD in NSCLC patients receiving novel antineoplastic agents.

CI, Confidence interval; ILD: interstitial lung disease; NSCLC, non-small cell lung cancer; OR, odds ratio.
Fatal and non-fatal analysis
Overall, the mortality rate among patients with NSCLC who developed ILD associated with novel antineoplastic agents was 30.60%. ADCs had the highest mortality rate (45.24%), followed by targeted therapies (34.50%) and ICIs (29.10%). At the PT level, ILD and pneumonitis accounted for the majority of fatal cases, with 761 (50.36%) and 460 (30.44%) deaths, respectively.
We further performed univariate and multivariate logistic regression analyses to identify factors associated with mortality in patients with ILD (Table 3). The results showed that a higher number of concomitant medications was significantly associated with increased risk of death (multivariate OR = 1.06, 95% CI 1.04–1.08, p < 0.001). Male patients had a higher risk of death compared with females (multivariate OR = 1.37, 95% CI 1.07–1.76, p = 0.014). In addition, body weight was found to exert a protective effect against mortality (multivariate OR = 0.99, 95% CI 0.98–0.99, p = 0.001). Age was associated with increased risk of death in univariate analysis (univariate OR = 1.01, 95% CI 1.01–1.02, p < 0.001), but this association was no longer significant after multivariate adjustment (multivariate OR = 1, 95% CI 0.99–1.01, p = 0.559).
Factors associated with mortality in ILD patients: univariate and multivariate logistic regression.

CI, confidence interval; ILD: interstitial lung disease; OR: odds ratio.
Comparison with conventional chemotherapy
We also evaluated ILD signals for conventional chemotherapies in NSCLC (Figure 5). Overall, novel anticancer therapies exhibited more positive signals than traditional chemotherapeutic agents.

The signal of ILD associated with conventional chemotherapy agents in the treatment of NSCLC.
Discussion
In recent years, novel anticancer agents have significantly improved the treatment landscape for NSCLC. However, some agents can cause drug-induced ILD, a potentially fatal adverse event that is often difficult to detect due to its nonspecific clinical manifestations. Early recognition and management are critical to avoid treatment interruption or mortality. 21
Based on the FAERS database, we conducted the first large-scale, real-world study to systematically evaluate the reporting characteristics, signal strength, and factors associated with ILD among different novel anticancer drugs in NSCLC, thereby providing real-world evidence to enhance drug safety and optimize treatment outcomes.
In this study, the highest proportion of reports of drug-induced ILD associated with novel anticancer agents in NSCLC patients came from Japan (59.06%). This is consistent with previous studies observing a higher incidence of ILD associated with novel anticancer agents in Japan compared to other regions.22 –24 There are several possible contributing factors that still need further verification, including genetic susceptibility, greater clinical focus, as well as the country’s higher access to lung cancer treatment, which possibly leads to more ILD-related adverse reports.25 –29 Despite the higher number of reports from Japan, the FAERS database, as a global platform, reflects ILD risk across different regions, but may limit the generalizability of the findings to regions with lower reporting rates.
Disproportionality analyses were conducted to identify reporting signals. A positive signal indicates a higher-than-expected reporting frequency for a specific drug compared with other agents in the database, which can be influenced by factors such as reporting bias, drug utilization patterns, and levels of clinical awareness. The absence of a significant signal should not be simply interpreted as indicating lower risk.
In this study, among ICIs, all agents except cemiplimab demonstrated positive signals for ILD, with durvalumab showing the strongest signal and reporting the highest number of ILD cases before and after adjustment. At the PT level, durvalumab showed significantly stronger reporting associations with radiation pneumonitis compared to other agents, potentially related to its clinical application settings. Based on the PACIFIC study results, durvalumab has become the standard consolidative treatment following chemoradiotherapy for patients with unresectable, stage III NSCLC.30,31 In clinical practice, durvalumab is frequently prescribed to patients with prior thoracic radiotherapy or combination chemotherapy, both of which are known risk factors.5,6,32 This could lead to channeling bias, as these patients may already have a higher risk of developing ILD, potentially overestimating the drug’s associated risk.
From a mechanistic perspective, radiotherapy and ICIs may have synergistic effects, potentially increasing the risk of pneumonitis. Radiation directly damages vascular endothelial and alveolar epithelial cells, triggering cytokine release and inflammatory cell recruitment. 33 In addition, radiotherapy induces immunomodulatory changes, including upregulation of PD-L1 and the release of tumor antigenic peptides, which enhance antitumor T-cell recognition and activity.31,34,35 ICIs further relieve inhibitory immune signaling, thereby enhancing cellular immune activation. 36 In the context of radiation-induced lung injury and a preexisting inflammatory microenvironment, such immune activation may amplify local inflammatory responses and increase susceptibility to immune-mediated lung injury. Therefore, closer monitoring and management of pulmonary toxicity during combined ICI and radiation therapy is essential, especially with durvalumab, and be prepared for early intervention if drug-induced ILD is in doubt.
T-DXd, an anti-human epidermal growth factor receptor 2 (HER2) agent, was approved by the FDA for the treatment of NSCLC in 2022. 37 The DESTINY-Lung 01 trial reported an objective response rate (ORR) of 55% in HER2-mutated NSCLC, but 26% of patients developed drug-related ILD. 38 In a pooled analysis of nine T-DXd monotherapy studies, 25 of 1150 patients (2.2%) experienced a death outcome due to ILD. 23 We also identified a significant ILD signal associated with T-DXd, reinforcing concerns about its pulmonary toxicity in real-world settings. The mortality rate of ADC-related ILD was also significantly higher than that associated with targeted therapies and ICIs. The mechanism of T-DXd–related ILD has not been fully elucidated. These toxicities may be explained by off-target effects, either through premature release of the cytotoxic payload in the circulation or tumor microenvironment, or by binding the ADC to noncancerous cells expressing HER2.39,40 A Recent study indicated that uptake and redistribution of T-DXd by alveolar macrophages may be involved. 41 The majority of T-DXd studies have focused on breast or gastric cancers, with limited studies in lung cancer. Our findings suggest that T-DXd is associated with a notable risk of ILD in patients with NSCLC, and its use in clinical practice should be carefully monitored.
For the 26 targeted drugs included in the study, only osimertinib reached a positive signal for ILD, suggesting a potential association with ILD occurrence. This finding is consistent with previous studies that have reported an increased risk of ILD with osimertinib, particularly in Japanese populations.42,43 It should be noted that although other targeted agents did not reach the positive threshold in this study, they are not without risk in clinical practice. These results should be interpreted as differences in signal strength rather than an absence of risk. For first-generation EGFR-TKIs, such as gefitinib, although previous studies have suggested an increased risk of ILD, no positive signal was detected in this study, which could be related to lower usage and the limited number of case reports in recent years. 44 This result should not be directly interpreted as indicating a safer clinical profile. Similarly, no ILD positive signals were detected for other novel targeted drugs (e.g., ALK, MET, RET, and KRAS inhibitors), partly due to their shorter periods of clinical application and limited real-world data. Furthermore, reporting bias is inherent to the FAERS database, as ILD events are more likely to be reported for drugs with higher usage or more attention.
It is challenging to predict drug-induced ILD, but individual factors such as advanced age, prior history of ILD, male sex, and concomitant use of pulmonary toxic drugs are associated with an increased risk.5,42 After adjusting for age, sex, and concomitant medications in multivariate logistic regression, we identified five agent signals that are still associated with ILD: pembrolizumab, durvalumab, ipilimumab, trastuzumab-deruxtecan, and osimertinib. These findings suggest that when using these drugs, especially in high-risk populations, enhanced early monitoring and timely intervention are recommended to reduce severe or fatal adverse events.
Nearly half of the cases occurred within the first month of treatment, with a median onset of approximately 33 days. Weibull shape parameter analysis revealed an early-onset pattern. Notably, fatal cases developed ILD significantly earlier than non-fatal cases, highlighting the need for close monitoring of pulmonary symptoms, imaging, and functional parameters, particularly during the early phase of therapy, to enable timely identification and intervention of severe events.
We also explored characteristics associated with ILD reporting in FAERS. Older age, male sex, and lower body weight were associated with a higher frequency of ILD reporting, with male sex and lower body weight remaining at a higher risk in multivariate analysis, consistent with previous reports.5,45 Moreover, male sex and the number of concomitant drugs were linked to higher reporting of fatal outcomes, whereas higher body weight appeared protective. Compared with traditional chemotherapy, novel anticancer agents demonstrated stronger ILD reporting signals.
There are several limitations in this study. First, the FAERS database is inherently subject to incomplete information, reporting bias, and heterogeneity in report quality, which may lead to overestimation or underestimation of the association between drugs and the target events. Second, some key clinical characteristics were unavailable, such as baseline health status, tumor stage, smoking history, and prior treatments. Although we attempted to control for potential confounders, residual confounding cannot be excluded, and these missing variables also limited the feasibility of more detailed subgroup analyses. Third, information on concomitant medications in FAERS is often incomplete and lacks details on treatment sequence and dosing, preventing us from performing analyses of drug combinations. Finally, the disproportionality analysis used in this study reflects the strength of reporting signals rather than causal inference. In the future, further validation through multicenter, prospective, or international studies is needed to enhance external validity and confirm these findings. Despite these limitations, the large sample size and systematic evaluation provide valuable real-world evidence, complementing clinical trial data and highlighting the need for early detection and monitoring of ILD in NSCLC patients on novel anticancer treatments.
Conclusion
This real-world analysis systematically evaluated disproportionality signals of ILD across novel anticancer agents used in NSCLC. Eight agents were identified with positive signals for ILD (nivolumab, pembrolizumab, durvalumab, atezolizumab, ipilimumab, tremelimumab, trastuzumab-deruxtecan, osimertinib). Nearly half of ILD cases occurred within the first month, and fatal cases developed earlier. Older age, male sex, and lower body weight were factors that affected ILD. Compared with conventional chemotherapy, novel anticancer agents showed more positive signals for ILD. This study provides real-world evidence to support early detection and prevention of ILD related to novel anticancer agents, thereby enhancing patient safety. In addition, these findings complement existing clinical trial data and offer further insights into the features of ILD induced by these therapies.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666261444082 – Supplemental material for Interstitial lung disease associated with novel anticancer agents in non-small cell lung cancer: a pharmacovigilance analysis using the FAERS database
Supplemental material, sj-pdf-1-tar-10.1177_17534666261444082 for Interstitial lung disease associated with novel anticancer agents in non-small cell lung cancer: a pharmacovigilance analysis using the FAERS database by Yunfei Zhu, Jingjing Sun and Shuyun Xu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
