Abstract
Background:
Radial probe endobronchial ultrasound–transbronchial lung biopsy (rEBUS-TBLB) enables accurate and safe sampling of small peripheral pulmonary lesions (PPLs) inaccessible to conventional flexible bronchoscopy. Although widely adopted and effective, the standard approach using fluoroscopic guidance and a guide sheath (GS) has limitations, including reduced instrument flexibility, greater procedural complexity, and higher demands for equipment and radiation safety.
Objective:
This study aimed to evaluate the diagnostic efficacy and safety of rEBUS-TBLB technique performed without the use of a GS or fluoroscopy in patients with PPLs.
Design:
This was a prospective, interventional, non-controlled study conducted in 67 patients with PPLs of unknown etiology that could not be accessed by flexible bronchoscopy for diagnostic sampling. All patients were admitted to the Department of Respiratory Medicine, Military Hospital 175, between February 2024 and March 2025.
Methods:
rEBUS-TBLB was performed by two experienced pulmonologists. Primary outcomes included the specimen acquisition rate, number, and quality, while secondary outcomes comprised diagnostic yield, complication rates, and the association of chest CT and rEBUS lesion characteristics with diagnostic performance. Data were collected at admission and throughout hospitalization using a standardized medical record format.
Results:
The mean age of the patients was 62.03 ± 9.8 years, with a male-to-female ratio of 2.7:1. Solid nodular lung lesions accounted for 73.1% of cases. The mean lesion size was 42 ± 14 mm, and the mean distance from the lesion to the pleura was 8.5 ± 12 mm (range, 0–45 mm). All lesions were sampled using rEBUS-TBLB, with ⩾3 specimens obtained in 90% of cases. Histopathological diagnoses were achieved in 74.6% of cases, of which 84% were lung cancer and 16% were tuberculosis. The most common complication observed was mild localized bleeding.
Conclusion:
The rEBUS-TBLB technique, even without GS and fluoroscopy support, remains a highly effective and safe diagnostic approach for PPLs, particularly for malignant lesions and those ⩾20 mm in size. This technique may be appropriate for wider implementation in hospitals with limited equipment and financial resources, provided that flexible bronchoscopy systems, rEBUS equipment, and adequately trained personnel are available.
Trial registration:
The study was approved by an independent ethics committee (Ethics Committee of Military Hospital 175, No. 2637/QĐ-BV) and conducted in accordance with the Declaration of Helsinki and Guidelines for Good Clinical Practice.
Introduction
The technique of radial probe endobronchial ultrasound (rEBUS) combined with transbronchial lung biopsy (TBLB) has been widely implemented and has demonstrated both efficacy and safety in diagnostic applications. rEBUS enables the guidance of biopsies for small peripheral pulmonary lesions (PPLs) that cannot be reached by conventional flexible bronchoscopy.1,2 This technique is typically performed with fluoroscopic guidance to enhance biopsy accuracy.
rEBUS can accurately localize lesions and the bronchi leading to them; however, a major limitation is the inability to observe the biopsy process in real time. Once a lesion is localized by rEBUS, the probe must be withdrawn from the bronchoscope’s working channel before the biopsy instrument is introduced. As a result, it is not possible to determine whether the biopsy instrument has been accurately advanced into the bronchial pathway leading to the target lesion. To address this limitation, Kurimoto et al. developed a guide sheath (GS) that extends from the bronchoscope tip to the lung lesion, serving as an extended working channel. The GS allows repeated biopsies of the same lesion, improving procedural consistency. The rEBUS-GS technique has proven to be a valuable method for sampling PPLs, even those too small to be visualized fluoroscopically. 3 Subsequent studies have demonstrated that rEBUS-TBLB with GS and fluoroscopy yields high diagnostic efficacy, and this approach is now widely recognized as a standard procedure.4–7
Nevertheless, the use of GS and fluoroscopy is not without limitations. First, biopsy forceps compatible with the GS are smaller in size, resulting in smaller tissue specimens that may compromise the quality of histopathological evaluation. Additionally, the GS may restrict access to multiple sites within a lesion, potentially reducing diagnostic yield. Technical challenges such as stabilizing the GS tip during patient coughing or deep respiration, as well as displacement caused by the force applied when inserting biopsy instruments, may also hinder procedural success. 8
Furthermore, performing the procedure under fluoroscopic guidance exposes both patients and healthcare staff to radiation, increases demands on space and personnel, and raises costs. In many settings, particularly in resource-limited countries or healthcare facilities, the lack of adequate equipment can be a major barrier to implementing the technique with GS and fluoroscopy according to standard protocols. 7
In a retrospective Korean study of 184 patients who underwent rEBUS-guided biopsy without a GS or fluoroscopy, initial pathological assessment identified malignancy in 109 cases (59%). Among the remaining 75 patients (41%) with non-malignant results, additional diagnostic evaluation detected malignancy in 34 more patients. Based on the final diagnoses, the overall diagnostic accuracy was 80% (136/170), with a sensitivity of 76% (109/143) and a specificity of 100% (27/27) for malignancy. Molecular and genetic analyses were successfully completed in all but one case, in which the specimen was inadequate. 9
In light of these challenges, we conducted this study to evaluate the diagnostic efficacy and safety of the rEBUS-TBLB technique when performed without GS and fluoroscopy support in patients with PPLs.
Methods
Study design and subjects
This was a prospective, non-randomized interventional study that included 67 patients with PPLs identified on chest computed tomography (CT). All patients were admitted for treatment at the Department of Respiratory Medicine, Military Hospital 175, between February 2024 and March 2025.
The inclusion criteria were: (I) patients with PPLs defined as abnormal growths detected on CT scans and (II) age ⩾18 years. The exclusion criteria were: (I) age < 18 years, (II) pure ground-glass opacities, (III) lesion diameter < 10 mm, and (IV) contraindications to bronchoscopy, including hemodynamic instability, coagulation disorders, respiratory failure, severe heart failure, or angina within the preceding month. Informed consent was obtained from all participants.
Procedures of radial probe endobronchial ultrasound–transbronchial lung biopsy
All procedures were performed by two experienced pulmonologists using a BF-H190 flexible bronchoscope (Olympus Ltd., Tokyo, Japan) equipped with a 2.0 mm working channel and a 4.0 mm outer diameter, in conjunction with an endoscopic ultrasound system (Olympus Ltd., Tokyo, Japan). The radial probe EBUS (UM-S20-17S, Olympus Ltd., Tokyo, Japan) measured 1.4 mm in diameter, and disposable biopsy forceps with a 1.8-mm outer diameter were utilized during rEBUS-TBLB. Pre-procedural chest CT images were reviewed for each patient, with lesion size measured at the largest diameter on axial lung window settings, and the location and distance of each PPL from the visceral pleura were recorded.
Based on chest CT, the location of each lung lesion was determined, and a bronchial map was constructed to identify the optimal entry route and bronchus signs within the lesion (Figure 1). 10

Bronchus sign images: (a) schematic illustration of common patterns and (b) representative chest CT image showing the responsible bronchus directly reaching the target lesion with evidence of obstruction and narrowing. 1
Procedures were performed under conscious sedation with superficial intravenous anesthesia using midazolam at a starting dose of 0.04–0.06 mg/kg. Standard bronchoscopy techniques were followed to approach the lesion, which was subsequently assessed using rEBUS imaging (Figure 2).

Peripheral lung lesions observed by rEBUS: (a) schematic illustration of the ultrasound probe positioned within the lesion and (b) representative real-time rEBUS image of the lesion.
The bronchoscope tip was fixed at the location of the lesion as determined by rEBUS. The length of the biopsy forceps was matched to that of the rEBUS probe and marked with a fixed button. Forceps were inserted into the working channel of the bronchoscope to obtain multiple biopsy specimens from different areas of the lesion. Lesion localization was rechecked using rEBUS during the biopsy process. A chest radiograph was obtained within 2–4 h after the procedure to detect any pneumothorax.
Characteristics of rEBUS imaging: Position was classified as within when the radial probe was completely surrounded by the lesion and adjacent when the probe was located at the lesion margin (Figure 3) 11 ; size was measured from the center of the ultrasound probe to the lesion border; echogenicity was defined as hyperechoic when the lesion demonstrated higher echogenicity than the surrounding structures and hypoechoic when it demonstrated lower echogenicity; echotexture was categorized as homogeneous when echogenicity was uniform throughout the lesion and heterogeneous when echogenicity varied within the lesion.7,12

EBUS image according to the relationship between the peripheral lung lesion and the bronchus. The radial probe EBUS was positioned inside the peripheral lung lesion (a, within), in the bronchus adjacent to the lung lesion (b, adjacent to) and outside the lung lesion (c, outside). 1
Bleeding complications were classified into four grades: grade 0 (no or minimal bleeding, no intervention required), grade 1 (mild bleeding requiring suction only, procedure continued), grade 2 (bleeding requiring local hemostatic measures such as adrenaline injection or cold saline, procedure terminated), and grade 3 (severe bleeding requiring embolization or surgery, endoscopic intervention not feasible). 6
A positive histological diagnosis was defined as confirmation of malignancy or identification of a specific benign pathology, such as tuberculosis or pneumonia. If the TBLB result was nondiagnostic (e.g., showing only chronic inflammation), the patient was either offered video-assisted thoracoscopic surgery (VATS) for further evaluation or monitored for radiological improvement following appropriate treatment.
Data collection
Epidemiological, demographic, clinical, and treatment-related data were collected at admission and during hospitalization using a standardized medical record format.
Outcome measures
The primary outcomes were the success rate of lesion specimen acquisition, the number of biopsy specimens obtained, and the adequacy and quality of specimens for histopathological evaluation, while the secondary outcomes included the diagnostic yield of histopathological and cytological analyses of specimens obtained by rEBUS-TBLB without fluoroscopy or a GS the incidence of procedure-related complications, and the relationship between lesion characteristics on chest CT and rEBUS imaging and the overall diagnostic performance of the technique. The results related to the diagnostic yield of the technique were evaluated according to the consensus standards of the American Thoracic Society. 13
Statistical analysis
Data were analyzed using SPSS for Windows, version 22.0 (SPSS Inc., Chicago, IL, USA). The Kolmogorov–Smirnov (K–S) test was used to assess data normality. Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables were expressed as percentages.
Correlations between technical effectiveness and other variables were analyzed using Spearman’s correlation coefficients. The Mann–Whitney
The reporting of this study conforms to the TREND statement. 14
Results
Baseline characteristics and chest CT findings in patients with PPLs
A total of 67 subjects with PPLs were enrolled. The study flowchart is shown in Figure 4. The mean age of the patients was 62.03 ± 9.8 years (range, 34–88 years), with males accounting for 73.1% and females 26.9% of the study population; 56.7% of patients (38/67) were smokers. On chest CT, the mean lesion size was 42 ± 14 mm, and all patients (100%) demonstrated a positive bronchus sign, with a mean distance from the lesion to the parietal pleura of 8.5 ± 1.2 mm. Lesions were located in the upper lobe in 47.7% of cases, the lower lobe in 44.8%, and the middle lobe in 7.5%; solid nodular lesions accounted for 73.1% of cases, and contrast enhancement was observed in 74.6%. Serum tumor marker concentrations were slightly elevated and variable, with carcinoembryonic antigen (CEA) levels of 27.8 ± 68.9 ng/ml (range, 0.9–332.6), neuron-specific enolase (NSE) levels of 21.8 ± 13.1 ng/ml (range, 7–56), and cytokeratin-19 fragment (Cyfra 21-1) levels of 11.9 ± 27.4 ng/ml (range, 1.39–166.5). Final diagnoses based on biopsy or surgery revealed that 74.6% of lesions were malignant, 20.9% were benign, and 4.5% remained of undetermined etiology (Table 1).

Study flowchart.
Baseline characteristics, chest CT, and histopathology findings in patients with peripheral lung lesions.
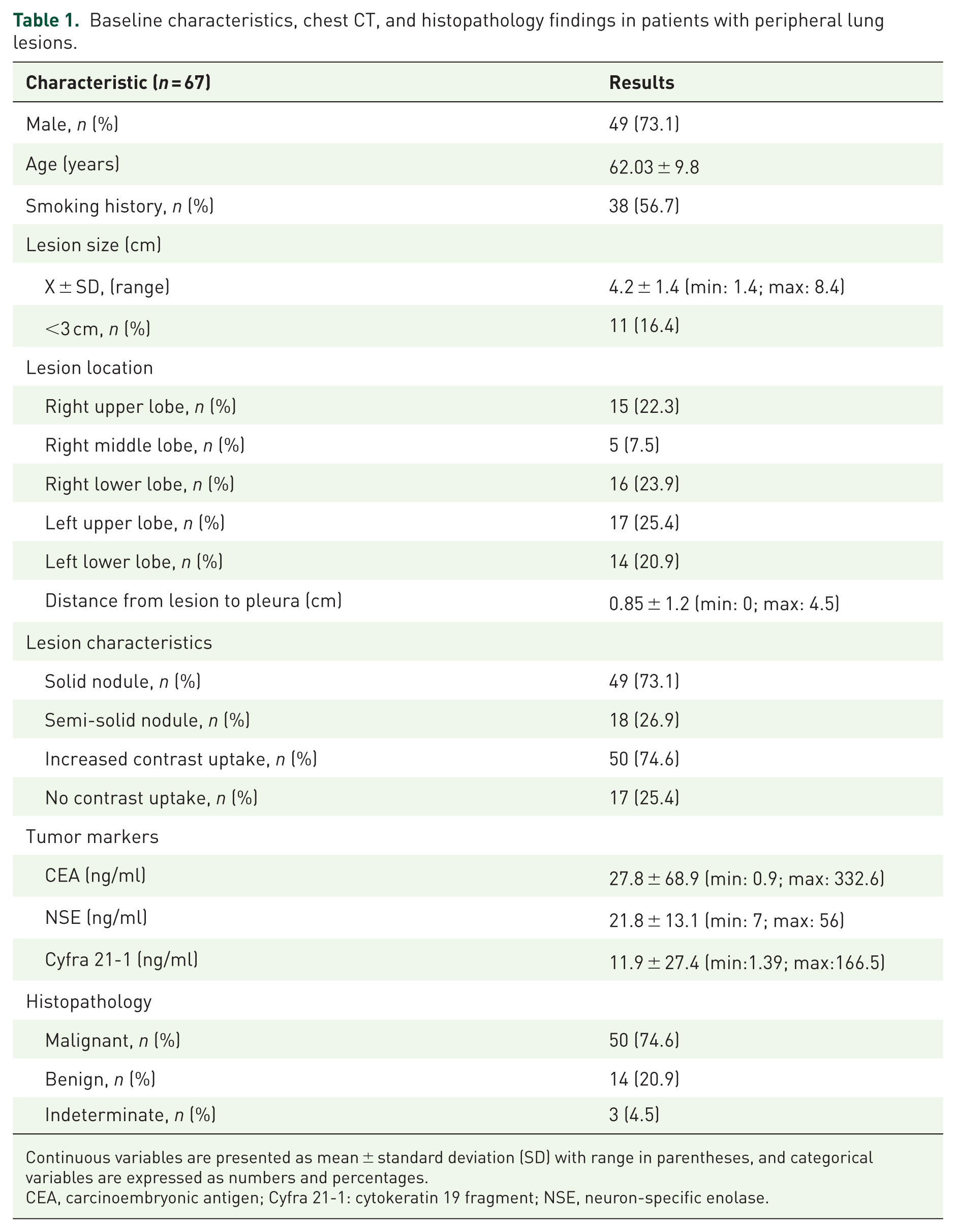
Continuous variables are presented as mean ± standard deviation (SD) with range in parentheses, and categorical variables are expressed as numbers and percentages.
CEA, carcinoembryonic antigen; Cyfra 21-1: cytokeratin 19 fragment; NSE, neuron-specific enolase.
Diagnostic efficacy and safety of rEBUS-TBLB without guide sheath or fluoroscopy
Lesion characteristics and sampling efficacy: All patients had lesions detected by rEBUS. Of these, 79.1% were categorized as
rEBUS imaging characteristics of lesions and outcomes of rEBUS-TBLB.

Lesion position was classified as within when the rEBUS probe was completely surrounded by the lesion and as adjacent when it was located at the lesion margin. Echogenicity and echotexture were defined according to the intensity and uniformity of ultrasound signals. Technical outcomes included the proportion of cases in which forceps biopsy was performed, the number of biopsy specimens obtained, and the incidence of procedure-related complications.
rEBUS, radial endobronchial ultrasound; TBLB, transbronchial lung biopsy.
Histopathological diagnostic yield: Definitive histopathological diagnoses were obtained in 50 patients (74.6%). Among these, 42 patients (84%) were diagnosed with lung cancer, and the remaining eight patients (16%) were diagnosed with tuberculosis. Seventeen patients (25.4%) did not receive a definitive diagnosis from rEBUS-TBLB and subsequently underwent surgical intervention with video-assisted thoracic surgery (VATS). Postoperative histopathology confirmed eight cases of lung cancer and six cases of benign inflammatory lesions. In three patients, the nature of the lesions could not be determined because they declined surgery and did not return for re-evaluation (Table 3). Illustrative cases of lung cancer (Figure 5) and pulmonary tuberculosis (Figure 6) confirmed by rEBUS-TBLB biopsy samples.
Histopathological diagnosis results from rEBUS-TBLB specimens.
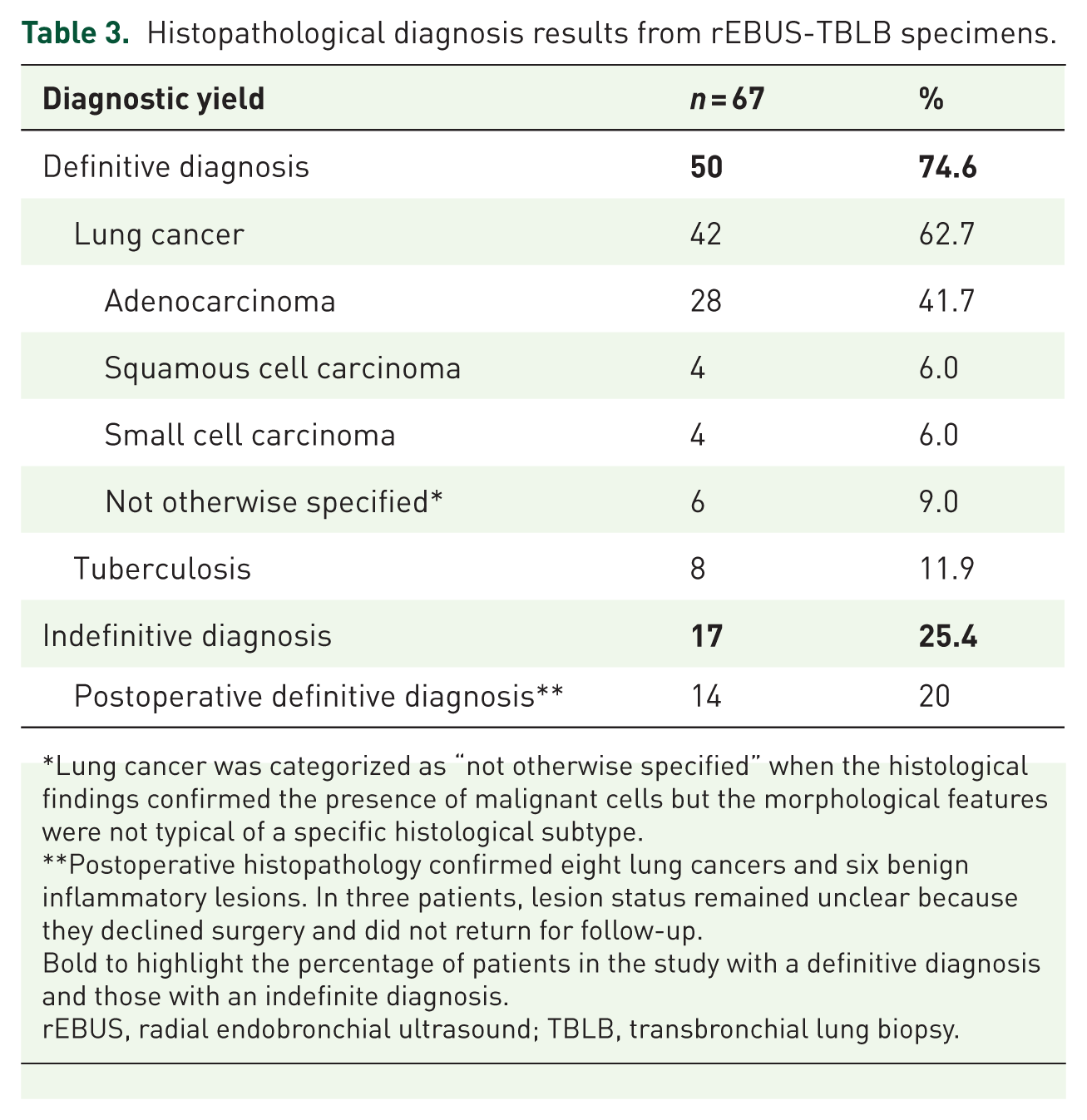
Lung cancer was categorized as “not otherwise specified” when the histological findings confirmed the presence of malignant cells but the morphological features were not typical of a specific histological subtype.
Postoperative histopathology confirmed eight lung cancers and six benign inflammatory lesions. In three patients, lesion status remained unclear because they declined surgery and did not return for follow-up.
Bold to highlight the percentage of patients in the study with a definitive diagnosis and those with an indefinite diagnosis.
rEBUS, radial endobronchial ultrasound; TBLB, transbronchial lung biopsy.

Lesion located in the S6 segment of the left lung adjacent to the thoracic aorta (a). rEBUS imaging demonstrated a hyperechoic, heterogeneous, adjacent lesion (b). rEBUS-TBLB obtained two biopsy specimens, and histopathological analysis confirmed adenocarcinoma (c).

Details of transbronchial lung biopsy guided by rEBUS: (a) Chest CT scan showing a semi-solid pulmonary nodule located in segment S1 of the right upper lobe; (b) flexible bronchoscopy showing no visible endobronchial lesions; (c) placement of the rEBUS probe into the target bronchus with rEBUS imaging demonstrating an “adjacent” lesion; (d) histopathological analysis confirming lung tuberculosis.
Factors associated with diagnostic efficacy: On lung CT imaging, solid nodules were significantly more likely to yield a definitive diagnosis than semisolid nodules (
Factors influencing the diagnostic results of rEBUS-TBLB.

Discussion
General characteristics of patients
Risk factors such as advanced age, male sex, smoking history, and family history are well recognized as contributing to the development of lung cancer, with smoking identified as the most significant factor. In this study, the mean age of patients was 62.03 ± 9.8 years (range, 34–88 years), the male-to-female ratio was 2.7:1, and more than 50% of patients had a history of smoking. These findings are consistent with the established epidemiological profile of lung cancer. Serum tumor markers have long been utilized as screening tools in high-risk populations and as monitoring tools for treatment response.
The development and widespread application of chest CT, particularly low-dose chest CT has significantly improved the early detection of lung lesions. Early detection, in turn, facilitates timely access to advanced diagnostic techniques such as rEBUS, which are particularly valuable for confirming the nature of lesions at an early stage, especially in cases of lung cancer. Increasingly, PPLs require histopathological confirmation for definitive diagnosis, allowing the initiation of individualized treatment strategies. 15
Key information obtained from CT imaging, such as lesion size, morphology, location, number (solitary or multiple), lymph node involvement, invasion of adjacent structures, and contrast enhancement patterns, can provide valuable clues regarding the nature of a lesion. In the present study, solid nodular lesions accounted for the majority of cases (73.1%), while semi-solid nodular lesions comprised only 26.9%. Most lesions were relatively large, with a mean diameter of 4.2 ± 1.4 cm, and 83.6% were larger than 3 cm. Furthermore, 74.6% of the lesions demonstrated contrast enhancement. Consistent with previous reports, larger lesion size and more pronounced, heterogeneous contrast enhancement were more commonly associated with malignancy compared with benign lesions.
The lesion sizes observed in this study were larger than those reported in international studies. For instance, Sojung Park et al. reported that lesions biopsied using the rEBUS-TBLB technique had a mean size of 28 mm (range, 17–79 mm), with lesions ⩽30 mm accounting for 58.3% of cases. 16 This discrepancy may be attributable to earlier implementation of lung cancer screening programs in other countries, which facilitates detection of smaller lesions.
Diagnostic efficacy and safety of rEBUS-TBLB without guide sheath or fluoroscopy in patients with PPLs
PPLs are commonly detected on chest radiography or CT, yet conventional bronchoscopy often cannot visualize lesions beyond the bronchial lumen. rEBUS, using a 20-MHz radial probe with a 4-cm penetration depth, improves localization of peripheral lesions by visualizing the bronchial wall and surrounding tissues. In our study, the rEBUS detection rate was 100% (79.1% within-lesion; 20.9% adjacent), comparable to Hayama et al., who reported an 88% detection rate and 52% within-lesion and 36% adjacent-lesion positioning. 17
The positional relationship between the probe and the lesion (within, adjacent, or invisible) strongly determines diagnostic yield; when instruments cannot be advanced into the lesion, yield drops to ~60% or lower. 18 Beyond localization, rEBUS imaging aids differential diagnosis: anechoic components are more common in proliferative lesions, whereas their absence favors non-neoplastic disease. 19 Additional imaging features associated with malignancy include larger lesion size, lobulated morphology, well-defined but non-sharp borders, absence of vascular structures, and lack of discrete linear air-bronchograms. 20
EBUS image analysis is a simple and time-efficient approach that can provide valuable information for real-time diagnostic decision making during rEBUS procedures. Recently, there has been increasing interest in the use of artificial intelligence to develop predictive models based on EBUS imaging patterns, which could further enhance the diagnostic accuracy for PPLs.18,21,22
Once rEBUS localizes the lesion, transbronchial forceps biopsies are obtained. The number of samples depends on lesion accessibility, patient cooperation, and procedural complications. In our study, the mean number of specimens was 3.75 ± 1.1 (range 2–6), with ⩾3 specimens collected in 90% of patients. Prior studies have shown that diagnostic yield increases—and complication risk may rise—with a greater number of biopsy samples, and several authors recommend obtaining at least five specimens. As expected, within-lesion sampling provides higher diagnostic yield than adjacent sampling.
Adjunctive techniques such as bronchial washing, brushing, and cytologic aspiration can further enhance diagnostic yield and should be applied selectively. 16 Transbronchial lung cryobiopsy (TBLC) is a promising alternative that yields larger, better-preserved tissue samples with fewer crush artifacts compared with conventional forceps biopsy, potentially overcoming limitations of current devices. Rapid on-site evaluation (ROSE) can also improve efficiency by assessing sample adequacy in real time, though its availability remains limited across medical centers. 15
In our study, the overall diagnostic efficiency of rEBUS-TBLB for PPLs was 74.6%, with 84% of definitive diagnoses being malignant (42/50) and 16% being benign (8/50). These findings were consistent with previously published studies that performed rEBUS-TBLB using standard protocols with GS and fluoroscopy,7,23,24 suggesting that the diagnostic yield of histopathology using rEBUS-TBLB without GS and fluoroscopy is not significantly inferior.
Both GS-assisted and non-GS rEBUS-TBLB techniques have distinct strengths and limitations, and their use should be tailored to lesion characteristics (size, location, and morphology), operator experience, and equipment availability. 5 A systematic review and meta-analysis of 57 studies (7872 lesions) reported an overall rEBUS diagnostic rate of 70.6% (95% CI: 68%–73.1%), with higher yields in lesions >20 mm, malignant lesions, those with a positive bronchus sign, and when the probe was positioned within rather than adjacent to the lesion. The overall complication rate was 2.8%. 2 In a study of 496 patients, Alexander Chen et al. found an overall diagnostic efficiency of 69% for rEBUS-TBLB using GS and fluoroscopy, with yields increasing by lesion size: 58% (1–2 cm), 72% (2.1–3 cm), 77% (3.1–4 cm), 87% (4.1–5 cm), and 88% (>5.1 cm). 25
The GS-rEBUS technique involves advancing the ultrasound probe within a GS to the lesion, then withdrawing the probe while keeping the GS in place for forceps sampling. This stabilizes the target and improves biopsy accuracy, particularly for small peripheral lesions or ground-glass opacities.3,26 Multiple studies have shown that GS-rEBUS achieves higher diagnostic accuracy than non-GS approaches, with reported accuracy rates up to 93% and specificity and PPV approaching 100%. GS-assisted procedures also demonstrate higher yields in lesions ⩾20 mm and lower complication rates, including pneumothorax (~2.2%).5,7 GS is especially beneficial for small lesions in the middle or lower lobes, reducing bleeding and pneumothorax risks. 27
However, GS use has limitations. It may restrict instrument size and reduce sampling flexibility, whereas non-GS rEBUS allows larger forceps and sampling from multiple areas within a lesion, improving the chance of obtaining a diagnostic specimen. 28 Technical challenges such as GS displacement from coughing or deep breathing, twisting, bending, or resistance during instrument insertion can also occur.29,30 Additionally, GS increases procedural time and cost, which may limit its routine application.
Interestingly, a controlled interventional study reported that the diagnostic yield of rEBUS-TBLB for small PPLs (⩽15 mm) was significantly higher when performed without GS compared to with GS (80.7% vs 50.0%), with no major complications observed in either group. 28 Other studies have also demonstrated that the addition of GS does not necessarily improve diagnostic yield for malignant PPLs.27,31 Overall, the choice to use GS should be individualized, based on lesion size and location, operator experience, and institutional resources.
The diagnostic yield of rEBUS generally ranges from 66% to 88%, with fluoroscopically assisted procedures often demonstrating higher or equivalent yields compared to non-fluoroscopy methods.4,32,33 Fluoroscopy combined with rEBUS without GS has been shown to be both safe and effective, with diagnostic yields comparable to those obtained using a GS. This approach also offers procedural simplicity, which may be particularly advantageous in resource-limited settings. 28
Although fluoroscopic guidance may enhance diagnostic yield in certain studies, its routine use remains controversial due to concerns regarding radiation exposure, additional equipment costs, and logistical constraints. 34 Eliminating fluoroscopy entirely avoids radiation exposure, thereby improving safety for both patients and healthcare staff. rEBUS-TBLB without fluoroscopy maintains high diagnostic accuracy and an acceptable complication rate in diagnosing PPLs, while also reducing procedural complexity and resource requirements. 7
A systematic review of 31 studies involving 6491 patients reported a pooled diagnostic yield of 70% (95% CI: 67%–74%) for rEBUS-TBLB without fluoroscopy, though there was considerable inter-study variability. Diagnostic yield was significantly higher in patients with a positive bronchus sign on CT, larger PPL size, probe position
A pooled analysis of 46 studies encompassing 7252 PPLs was conducted. The overall diagnostic yield of rEBUS was 73.4%. Further subgroup analysis showed that lesions located in the middle or lower lobes, those larger than 2 cm, malignant lesions, solid-appearing lesions on computed tomography (CT), lesions with a positive bronchus sign, a within-lesion probe position, and the use of rapid on-site evaluation (ROSE) were all associated with higher diagnostic yield. In contrast, the use of a GS, bronchoscopy type, and multimodality approaches—including combinations with virtual bronchoscopic navigation (VBN) or electromagnetic navigation bronchoscopy (ENB)—did not significantly influence diagnostic performance. The pooled incidence rates of overall complications, pneumothorax, and moderate to severe bleeding were 3.1% and 1.1%, respectively. 36
In our study, several factors were associated with improved diagnostic efficiency. Patients with solid nodular lesions on CT and homogeneous lesions on rEBUS had significantly higher diagnostic rates compared to those with semisolid or heterogeneous nodules. Additionally, obtaining a greater number of biopsy specimens was associated with improved diagnostic accuracy. There was no significant difference in diagnostic efficiency between
Histopathological diagnosis depends largely on the quality and adequacy of the biopsy specimen, making the operator’s sampling technique a critical determinant of success. In this study, GS was not used; therefore, the sampling location varied, and larger biopsy forceps were employed. These factors may have contributed to the improved histopathological diagnostic yield observed. Lesion size ⩾20 mm, the presence of a positive bronchial sign on chest CT, solid lesion characteristics, and positioning of the probe within the lesion were identified as important factors for achieving a successful diagnosis. 7 In malignant PPLs, echogenicity characteristics were more strongly associated with the diagnostic yield of TBLB than lesion diameter. A within-lesion probe position, discontinuous lesion borders, and heterogeneous echogenicity were identified as independent predictors of higher diagnostic yield for TBLB in peripheral lung lesions. 37
Prior controlled studies have demonstrated that, for lesions >20 mm with bronchus signs on CT and located in the middle lobe or lingula, the diagnostic yield of rEBUS-TBLB is comparable whether GS is used or not. Accurate pre-procedural identification of the bronchus leading to the lesion based on CT analysis is, therefore, essential for procedural success. Incorporating virtual bronchoscopy may further improve localization and procedural planning.38,39 Additionally, re-confirming the GS position or re-localizing the lesion with rEBUS during the biopsy procedure can further improve diagnostic efficiency.
40
Overall, lesion characteristics (diameter, location, and bronchus sign), probe position (
Recent studies have evaluated the application of the EBUS-TBNA technique for diagnosing PPLs and have reported promising diagnostic efficiency. The diagnostic yield of bronchoscopy ultrasound for PPLs may be limited because the radial EBUS probe is not always positioned
Combining rEBUS with adjunctive technologies, such as virtual bronchoscopy or electromagnetic navigation, may improve diagnostic efficiency compared with rEBUS alone. A meta-analysis of 363 studies found an overall diagnostic efficiency of 78.1%, highest with CT-guided transthoracic biopsy (CT-TBNA; 88.9%), followed by robot-assisted bronchoscopy (RAB; 84.8%), and lowest with rEBUS (72%). Although CT-TBNA achieved the highest yield, it had significantly higher complication rates, whereas RAB provided a comparable yield with substantially lower risk. 42
The rEBUS-TBLB technique itself remains highly safe, with an extremely low incidence of major complications. In our study, complications were limited to grade 1 (6%) and grade 2 (4.5%) localized bleeding. There were no cases of severe bleeding, respiratory failure, or pneumothorax. Published studies have consistently demonstrated the safety of rEBUS-TBLB. For instance, Hayama Manabu et al. reported an overall complication rate of only 1.3%, with pneumothorax (0.8%) and pulmonary infection (0.5%) being the most common adverse events. Ultrasound probe fracture or path rupture was exceedingly rare (0.4%). 17
Overall, existing evidence supports the high efficacy, favorable safety profile, and low complication rates of the rEBUS-TBLB technique, with or without GS support or fluoroscopy. rEBUS is particularly effective for lesions with a
Study limitations
This study has several limitations. First, it is a single-center study with a relatively small sample size, convenience sampling, and no control group using rEBUS-TBLB with GS or fluoroscopy. Second, most lesions were ⩾30 mm (83.6%), which limits the evaluation of diagnostic performance in smaller PPLs and the associated pneumothorax risk. Future multicenter studies with larger sample sizes and lesion-size stratification are needed to validate and expand upon these findings.
Conclusion
This study demonstrates that the rEBUS-TBLB technique without fluoroscopy or GS support is a highly effective and safe diagnostic approach for PPLs, particularly for malignant lesions and those ⩾30 mm in size. The method eliminates radiation exposure, reduces procedural complexity, and maintains high diagnostic efficiency and safety.
Given these advantages, this approach may be appropriate for widespread implementation in hospitals with limited equipment and financial resources, provided that facilities are equipped with a flexible bronchoscopy system, rEBUS equipment, and adequately trained personnel.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666261430508 – Supplemental material for Efficacy and safety of radial probe endobronchial ultrasound–transbronchial lung biopsy in peripheral pulmonary lesions without guide sheath and fluoroscopy
Supplemental material, sj-pdf-1-tar-10.1177_17534666261430508 for Efficacy and safety of radial probe endobronchial ultrasound–transbronchial lung biopsy in peripheral pulmonary lesions without guide sheath and fluoroscopy by Cong Nguyen Hai, The Nguyen Minh, Toan Hoang Thanh, Loi Trinh Duc and Cuong Hoang Xuan in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
