Abstract
Background:
Social determinants of health (SDOH) and health-related social needs (HRSN) drive disparities in lung function, nutrition, and survival in People with Cystic Fibrosis (PwCF). Addressing HRSN can improve access to care, yet standardized screening and intervention methods remain underutilized.
Objectives:
The aim of this project was to develop, test, and refine a remote HRSN screening and intervention model across multiple cystic fibrosis (CF) centers.
Design:
A multicenter, prospective Quality Improvement (QI) initiative
Methods:
Four CF centers, serving both pediatric and adult populations, piloted an electronic HRSN screening tool and a remote social need intervention strategy. Developed collaboratively with CF clinicians and patient and family partners (PFPs), the tool assesses nine HRSN domains. Multidisciplinary teams, including PwCF, held regular meetings to tailor implementation to each site’s existing clinical workflow and staff structure. Over 1 year, each site conducted iterative Plan-Do-Study-Act (PDSA) cycles every 2 weeks to refine the screening process, sharing adaptations across centers.
Results:
All four CF centers successfully implemented the remote HRSN screening and intervention workflows, completing 26 iterative PDSA cycles to refine site-specific processes. Study site meetings were held with multidisciplinary attendance at 100% of meetings. The screening tool was integrated into pre-visit planning and telehealth workflows, allowing for social worker follow-up of identified needs. Multidisciplinary collaboration from all sites resulted in the generation of a comprehensive library of local and regional resources to support unmet needs identified during screening. Narrative patient testimonial highlighted the screening tool’s effectiveness in facilitating discussions about social needs and connecting individuals to available resources from the perspective of PwCF.
Conclusion:
Our study has shown that HRSN screening and intervention are feasible, adaptable and acceptable to PwCF. Next steps include gathering comprehensive data on screening and intervention rates, domains of unmet social needs across regions, and sustainability interventions. Expanding HRSN screening and intervention to other CF Centers can provide data to support public policy and advocacy initiatives for reducing health disparities driven by SDOH.
Plain language summary
Social determinants of health (SDOH) are an individual’s personal circumstances that impact their overall health and well-being. SDOH can lead to health-related social needs (HRSN) that include limited access to healthy food, affordable housing, transportation, education, and access to health insurance. These factors have been shown to impact lung health, nutrition status, and survival in people with cystic fibrosis (CF). While the use of telehealth has increased access to care for many people with CF, it has also created some challenges in how we can help people get access to the social resources that they might need. The goal of our project is to test an HRSN screening tool and resource packet that can be used with telehealth in CF. We hope to increase identification of unmet needs and be able to provide remote interventions to connect people with resources and services that can help. Four different CF Programs tested a virtual HRSN screener and intervention packet. These teams meet regularly and work together to find the best way to use the screener and provide resources to patients who are in need, and patients and families at their center are helping to guide the process. Our goal is to improve access to care through picking up on areas of need and linking patients with resources without patients having to come into the clinic in-person. What we learn from this project can be used in other CF programs and can be used to develop resources for the national CF community.
Keywords
Introduction
Cystic fibrosis (CF) is the most common life-limiting genetic multi-organ disease in the United States and affects 100,000 people worldwide, though the true global population is likely higher due to underdiagnosis in low- and middle-income countries. 1 Recent therapeutic advancements have significantly improved clinical outcomes and survival for the majority of people with CF (PwCF).2,3 However, these advancements in care have also brought about new challenges and emphasized known health disparities in this population driven by social determinants of health (SDOH). These disparities include difficulty obtaining stable employment, housing, transportation, education, food, experiences of racial biases, and access to healthcare. 4 While SDOH are systematic policies and practices that have long-term impact on a community level, health-related social needs (HRSN) are “social and economic needs that individuals experience that affect their ability to maintain their health and wellbeing.” 4 SDOH and HRSN can both play a factor in health disparities for certain groups and have been shown to impact pulmonary function, nutrition status, and survival in PwCF.5–12 Screening for HRSN and providing interventions is a recommended approach within the healthcare model. 13 At present, methods for screening and connecting PwCF to resources for unmet social needs have not been broadly tested and implemented in the CF care model. 14
Broad adoption of providing care to PwCF through telehealth (TH) has occurred in response to the COVID-19 pandemic, with multiple studies indicating a high level of patient satisfaction and positive experiences.15,16 Post-pandemic, the TH utilization in the CF community declined to 20% of all clinical encounters, 17 but remained utilized regularly by most CF centers based on PwCF preferences and will likely play a significant role in the future model of CF chronic care. Remote clinical monitoring in PwCF during TH visits (i.e., home-based spirometry and sputum cultures) has allowed widespread utilization of TH, but there has been limited work on how to remotely assess HRSN and intervene with resources in a virtual environment. Highlighting the need for screening for social needs, a recent cross-sectional analysis from a single CF program demonstrated that 30% of PwCF report an undesired change in at least one SDOH domain during the pandemic; 18 however, there has been limited focus on how to remotely assess and intervene on HRSN in the setting of a virtual environment. Further research is necessary to understand the landscape of unmet social needs, available resources, and their impact on PwCF, and specifically optimizing methods of screening for social needs and intervention delivery through a hybrid TH and in-person model of care has become an opportunity for improving care. In this report, we describe the use of the Model for Healthcare Improvement to address HRSN, design a multicenter Quality Improvement (QI) intervention for screening and intervening on unmet social needs in PwCF. The global aim of this project is to develop and test the feasibility and sustainability of a remotely performed HRSN screening tool and intervention package in outpatient adult and pediatric CF clinics representing different regions of the U.S. Specific aims of the project include to (1) increase the percentage of eligible PwCF screened for social risk factors with a remote social needs screening tool at each site to 90% and to (2) increase the amount of eligible PwCF who receive resource packets and Social Work TH follow-up for identified social need(s) to 85% by December 31, 2024.
Design and methods
This multicenter QI initiative began in July 2022 and is still ongoing across four U.S.-based CF care programs. The project aimed to codesign and test a HRSN screening and intervention process for the CF population, tailored to each site. Methods are presented according to the staged improvement framework shown in Figure 1: Scoping, Design, Planning, and Testing. While the study framework includes future phases on Sustainability and Scale/Spread, the current report focuses on the Scoping–Testing phases. This quality improvement project has been reported in accordance with the SQUIRE 2.0 (Standards for Quality Improvement Reporting Excellence) guidelines. 19
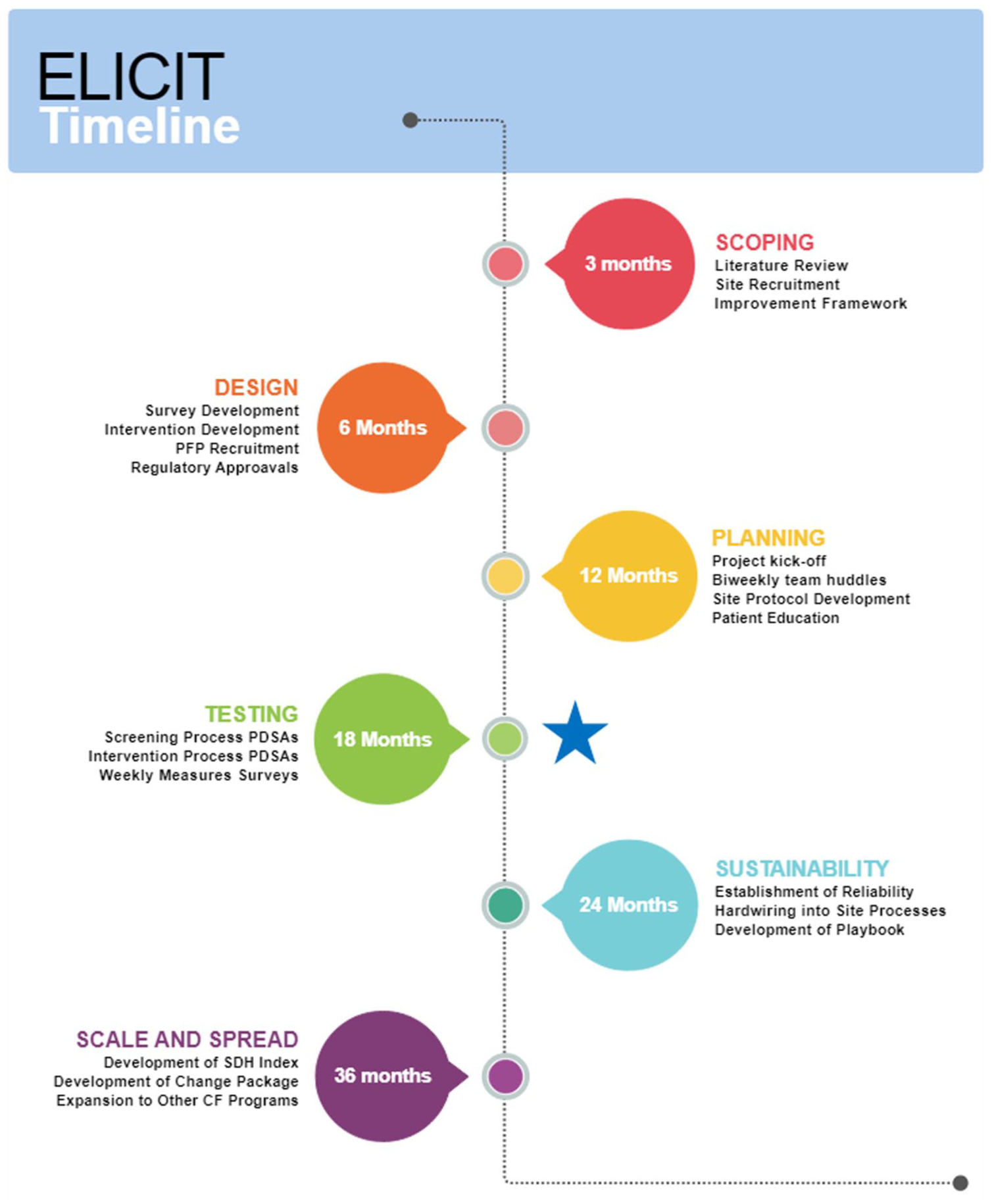
Timeline and overview of project phases and activities.
Scoping phase
Site recruitment
Four CF programs located at large academic programs in the United States collaborated to pilot a social needs screening tool and intervention process. Sites include Boston Children’s Hospital Pediatric CF Center (BCH), Oregon Health and Sciences University Pediatric CF Center (OHSU), University of Alabama at Birmingham Adult CF Center (UAB), and University of Virginia Adult CF Center (UVA). Each site had preexisting experience with QI methodology. Each site also had a history of screening for HRSN, although previously each team used different screening tools, methods, and frequencies of screening. Sites were chosen based on additional factors of relatively large patient populations served, and included both pediatric and adult care centers, across a diversity of geographic spread to optimize a broader understanding of regional variation in unmet social needs and available resources.
Participants
Eligible participants were all individuals with a confirmed diagnosis of CF, followed clinically at one of the four participating CF care centers, and willing to provide informed consent. Participants were excluded if they were unwilling to provide informed consent for the HRSN survey/screening at a routine clinic visit annually.
Improvement framework
In this multisite QI project, participating teams used the Model for Health Care Improvement20,21 to design and test iterative Plan-Do-Study-Act (PDSA) cycles of fielding the HRSN screening and intervention processes detailed herein. This framework was selected to support structured development and iterative testing of a social needs screening and intervention process within routine CF care. The six-phase approach allowed for a balance of centralized coordination and site-level customization, with embedded feedback loops at each stage. It reflects a learning health system model in which coproduction with patients and continuous improvement are foundational elements in each stage.
Design phase
Survey development and preliminary testing
After a comprehensive literature review of screeners, the HRSN screening tool was developed collaboratively by a participating site in conjunction with PwCF through rapid, iterative testing cycles and has been detailed in previously published work. 22 An existing HRSN screener was not utilized, as no tool specific to the CF population was available, and the voice of PwCF was crucial in creating a screener that addressed the unique needs of this community. The tool screens for HRSN in nine domains: housing, food, transportation, utilities, access to TH care/high-speed internet, healthcare access, medication access, income/employment, and education. The screening tool is 21 questions and takes approximately 10–15 min to complete. In addition to the screening tool, demographic information about age, race, ethnicity, sexual orientation, and gender identity was gathered from respondents. Input from PwCF was gathered at each stage of the testing process and drove changes made to the screening tool and the screening and intervention delivery process. The screener was built out in REDCap with a unique QR code for each participating site.
Remote screening infrastructure
Once the screener was finalized, it was adapted to an electronic format so that it could be administered to patients remotely. The survey was administered, and survey data were collected using the REDCap electronic data capture tools hosted at UAB Department of Medicine IT (DOM IT).23,24 The screening tool was adapted for each site to tailor implementation needs to their center and their center’s population, and to comply with each site’s regulatory board (ethics) approvals.
Survey refinements
Multiple rounds of adaptations to the screener occurred in the project design phase prior to implementation. Iterative testing with PFP feedback was performed to adapt the HRSN screener for an electronic format, tailoring to pediatric populations, adjusting for specific site needs, and strategies to reduce survey fatigue. Global adjustments made to tailor the screener to an electronic version include creation of site specific QR codes for use ahead of clinic and during clinic, addition of a field for respondents to enter their email address if they wanted to be sent a links to resources, and changes made to the visual appearance of the screener to make it easier to fill out on a mobile device. The screening tool was further tailored to meet the unique needs of pediatric patients across different age ranges in collaboration with PFPs from participating pediatric sites. Adjustments made to the pediatric population screener include additional logic for multiple children with CF in the same household, identification of which questions applied to all children in the household versus individual children, adjustments to remove pieces of the screener that did not apply to children (i.e., help finding work), and age-related logic to ensure that only age-appropriate demographic questions would present.
Site-specific adaptation to ensure that electronic screening fit into existing clinic processes and workflow included providing the screener using a tablet in the clinic for individuals who did not complete the survey remotely prior to clinic, an email alert system for positive screens, and the generation of site reports that provide feedback regarding positive responses that requested assistance. To mitigate survey fatigue, PFP feedback was incorporated in early piloting of the electronic screener. Additional adjustments included separating demographic questions from HRSN screening questions to reduce scrolling, grouping questions onto pages rather than separating each question to reduce clicking fatigue, and PFP-led improvements in the visual appearance of the screener. Both the adult and pediatric HRSN screeners are included in the Supplemental Materials.
Intervention design
Interventions to address HRSN are offered alongside the screening process from the beginning of testing, given that it is unethical to screen for social needs without providing intervention when needs are identified by the screening and desired by the patient/family. Interventions consist of providing respondents who screen positive with electronic resources for the domain(s) of reported need and SW TH follow-up to discuss and coproduce a plan to support patients with unmet needs. Electronic intervention packets with appropriate resources are deployed to patients asynchronously via secure email or the EMR portal and can also be sent synchronously via the chat and screen-sharing features during TH visits. Paper resources are also available for patients who do not have internet access or prefer paper resources during in-person clinic appointments. Electronic resource templates are available for each of the nine domains on the screener, and each program can customize resources based on regional and local program variation.
A library of resources was created by teams that are updated regularly. Additionally, each team contributed to a HRSN Resource Library that includes national, regional, and local resources in each of the nine domains of social risk. The Resource Library is a living document, with teams adding resources as they are created or discovered. SWs from each site met separately for huddles to review, compare, and research available resources for each of the nine domains assessed in the screening, and PFPs also provided feedback on the resource library. In addition, multiple resources are available through the Cystic Fibrosis Foundation (CFF) Compass Program site, and as necessary, the teams and PwCF are encouraged to contact the CFF Compass program (https://www.cff.org/support/get-help-cf-foundation-compass). A Voice of the Consumer tool and targeted patient interviews were used at each participating site to elicit broad feedback on the screening and intervention processes and identify opportunities for improvement.
Planning phase
Project team and leadership structure
Each site assembled its project team comprised of the CF Care Center Medical Director, CF Care Team Social Workers, Quality Improvement Coordinators, and Patient/Family Partners (PFPs). Additionally, a research assistant built all REDCap databases, and a statistician assisted with the development of data collection tools for process measurement, provided input on survey design, and created data displays throughout the planning process. Both individuals participated in site huddles and will continue to do so throughout the duration of the project. PFPs with lived experience were recruited at each site. This collaborative was modeled using the Cystic Fibrosis Learning Network (CFLN) framework 16 and has previously been successful across a variety of QI portfolio projects at the participating sites.
Coproduction with PFP
In addition to each team’s dedicated PFP, site Patient Family Advisory Committees have been utilized for feedback at each participating program on an ad hoc basis throughout the project when additional or broader perspective was needed. PFP Teams were selected based on QI experience and to provide a balance between age ranges and regional location. PFPs were provided with QI training and onboarding at the beginning of the project. Teams utilized materials developed by the CFLN in addition to site-specific onboarding materials. Time commitment was estimated to be 2 h per week with some electronic communication between meetings as needed in the PFP preferred format (secure messaging, email, phone calls). PFPs met initially in their specific site teams during onboarding to the project, and continued participation at the local level in site QI meetings with a goal to begin joining large project huddle meetings across sites in the next phase of the project. PFPs will also be involved in any sub-projects that occur, such as the creation of additional surveys or interview scripts to further explore findings generated from this work.
Planning huddles
Project planning huddles began in November 2022 with project teams including PFPs, site principal investigators, and project staff meet biweekly for brief QI huddles to ensure effective collaboration and shared learning. Team huddles utilize timed agendas and assign rotating roles for effective meetings. A formal kickoff meeting occurred in January 2023 with clinical team members from all sites to provide orientation to the project, timeline, and global data tracking plans for outcome and process measures. Huddles were framed on discussing ways to screen and intervene on social needs in an “all teach all learn” environment. Individual site teams also meet every other week to discuss implementation of social needs screening and intervention processes and to tailor social needs intervention packets to fit the needs of their site. While awaiting regulatory approvals to begin fielding the screener and intervention packets, teams used their weekly site-specific meetings to develop simplified Failure Modes and Effects Analysis (sFMEA), Process Maps, and Project Key Driver Diagram (Figure 2). Patient education materials were also developed in collaboration with PFPs for educational campaigns at each site with the goal of improving acceptability and reducing the stigma of social needs screening. Teams continued to meet biweekly from June–December 2023 to plan for implementing the screening and intervention processes, and to refine screening tools.

Global Project Key Driver Diagram, adapted from Cincinnati Children’s Medical Center Template.
HRSN screening and intervention process overview
Teams developed site-specific processes for implementing the screening and intervention. Broadly, the patient/family receives and completes the screener, with results followed up by team social workers who provide interventions intended to mitigate unmet needs, often through referral(s) to appropriate resources. A high-level process map is provided in Figure 3. Social work intervention for positive screens occurs in tandem with screening, with a TH contact from an SW occurring in the event of a positive screen. In cases where the PwCF or their family cannot be reached by phone or virtual visit, the SW follows up at the next in-person clinic visit. Examples of additional interventions that may occur during SW TH follow-up on positive are presented in Figure 4. Each site developed a tailored implementation protocol for the specific needs of their programs, staffing, and workflow.

Example high-level process map of remote health-related social needs screening and intervention from participating site.

Potential SW Interventions in response to positive social needs screen.
Measures
During the planning phase, a family of measures was developed. Planning includes team engagement measured by frequency of huddles, huddle attendance, PFP participation in huddles, frequency of data submission by teams, and number of PDSA cycles completed. Coproduction was measured through narrative feedback elicited from PFPs and patient family advisory boards in site QI meetings and between meetings. Project testing measures were identified in the planning phase to guide ongoing monitoring and improvement. These include SMART aims for screening completion and intervention follow-up, as well as process and balancing measures to capture feasibility, patient experience, and impact on staff workload (Table 1).
Project measures for screening and intervention SMART Aims.

Potential sources of bias and mitigation strategies
As a multisite quality improvement initiative, several potential sources of bias were identified when planning. These include selection bias due to variability in patient/family participation in the screening and measurement bias, given the reliance on self-reported data or inconsistent documentation. To address these, all sites utilized a standardized screening tool built into REDCap with common guidance for administration, but it can be tailored to each site’s workflow. Project teams received shared orientation and ongoing support through collaborative huddles, allowing for real-time adjustments and knowledge sharing. Observed improvements may be influenced by attribution bias, whereby changes could reflect external factors rather than the intervention itself; therefore, run charts and statistical process control methods are planned to monitor trends over time and distinguish real change from natural variation. Additionally, social desirability bias may influence responses related to sensitive domains of need. Engagement of PFPs at each site throughout design and testing phases also served to reduce bias by ensuring that processes and materials reflected lived experience and diverse perspectives. Ongoing education with patients and families to reduce stigma around HRSN may help to mitigate social desirability bias in screening disclosure and utilization of SW intervention.
Patient education campaigns
Prior to testing the screener, each site conducted a comprehensive patient education campaign in 2023 to improve patient awareness, reduce stigma associated with screening, and promote informed decision-making about reporting unmet social needs and receiving resources and SW intervention. Channels of communication at each site included face-to-face education by allied health team members during clinic encounters, flyers, center-wide emails, and CF Center newsletter articles. Educational materials were codesigned with PFPs from each center and pilot-tested with multiple PwCF in an iterative process prior to distribution. Examples of patient education materials on the resource library are included in the Supplemental Materials.
Testing phase
Screening and intervention implementation
Screening and intervention processes began in January 2024. The HRSN screener is sent both asynchronously to PwCF as part of pre-visit planning forms, and synchronously during both TH and in-person visits, if the PwCF did not complete the survey prior to the visit. The screening tool was designed to be deployed in multiple ways to fit within each program’s existing workflow processes, including synchronous to clinic visits or asynchronous prior to clinic encounters. Individuals with HRSN identified in at least one domain and who answered “yes” to wanting to receive assistance were defined as a positive screen, received electronic resource packets, and had SW follow-up within 1 week via TH to address the identified domain(s) of need.
Testing huddles
During the testing phase, huddle focus shifted to iterative PDSA cycle ramps and the opportunity for teams to share key learning on results, barriers, and adaptations of site-specific delivery of the screening tool and intervention resource packet. During this time, project huddles are held bimonthly. Testing began in January 2024 and is ongoing, with teams continuing to share learnings at project huddles in a round-robin format from PDSA ramps, review of measures data, and PFP feedback. Additionally, team huddles focus on plans to spread learning throughout the CF Care Center Network, re-scoping test plans, and manuscript development.
Data analysis plan
A data team from UVA supports the project and develops time series and control charts for screening rate and intervention rate; data are collected weekly and reviewed twice monthly during site meetings. A research coordinator reviewed the de-identified Excel tracking tools, which are uploaded directly to REDCap to gather information on response rate and intervention rate at each site as PDSAs progress. Process measures are then pulled from the tracking tool and compiled into reports by the UVA data core to examine key process measures—screening response rate, timely intervention rate, screening decline rate. Weekly data submissions are compiled and reviewed by the UVA data team. Process control charts will be created once sufficient data points are available. This analysis will inform adaptations to the intervention and planning for future sustainability and scale-up.
Results
Participants
A total of four CF Programs, serving 916 PwCF, are currently participating in feasibility testing of the screening tool and electronic intervention package. Site-level demographic and clinical characteristics were obtained from the most recent Cystic Fibrosis Foundation Patient Registry site-specific reports and are presented in Table 2. These data reflect the total care populations for each site and are intended to contextualize the study population, rather than represent the subset of individuals who will ultimately complete the screening tool when testing is complete. The four teams include Boston Children’s Hospital (BCH), Oregon Health Science University (OHSU), University of Alabama at Birmingham (UAB), and University of Virginia (UVA). Project team members include 1 QI Coordinator, 7 PFPs, 6 SWs, 4 physicians, and broader multidisciplinary QI teams from each CF program site, with representation from Allied Health Disciplines, Nurses, and Data Coordinators.
Clinical and demographic characteristics of project site cohorts.

Note that pediatric sites are broken into age categories to report on lung function (FEV1 pp), consistent with the CF Foundation Patient Registry site-specific reports. Children younger than age 6 cannot reliably perform spirometry and are therefore not included in the median FEV1 pp.
CFRD, cystic fibrosis-related diabetes.
Feasibility, acceptability, and participant feedback
A total of 26 multidisciplinary team meetings were held in 2023 to develop QI tools and share learning from PDSA cycles. Initial PDSA cycles were carried out at each site in the first quarter of 2024. Small-scale testing began in January of 2024, and each site has conducted a total of 26 PDSA cycles, expanding and refining the site-specific screening processes and adapting SW follow-up for interventions within one week of positive screens. The multisite project team met biweekly in 2024 (n = 10), and 100% of meetings included multidisciplinary representation from each project team, indicating that multidisciplinary participation across sites is feasible and sustainable. SWs from each site also met separately as a discipline group every second week (n = 10) in 2024 for cross-sharing, troubleshooting, building a resource library, and profession-specific discussions related to intervening on unmet social needs in different subsets of their cohorts. Site multidisciplinary QI teams met weekly throughout 2023 and 2024 for project planning and to progress the interventions tailored to their unique site needs. Each team had existing QI meetings at their CF Center to integrate this intervention into their existing structures. Testing is ongoing in 2025 and all four sites are offering social needs screening and intervention to their entire program cohort while continuously refining the process at each site.
PFPs serve as champions of social needs screening and intervention and provide leadership around tool and education material development, process refinement, and feedback during each project phase. Testimonials describing the work, impact of the project, and lived experience from project PFPs are shown below in Figure 5.

Lived experience testimonial from project PFP.
Discussion
HRSN contributes to health disparities in the CF population. Routinely detecting and responding to unmet social needs can improve clinical outcomes, health equity, and quality of life in the CF population. The ability to remotely screen for and address HRSN in a virtual environment will allow CF teams to provide high-quality and holistic multidisciplinary care in a clinical environment that has become more reliant on TH and distance health monitoring. There is also evidence to suggest that electronic screening for unmet basic needs, rather than face-to-face or paper-based, improves response rate. 26 In addition to creating a CF-specific HRSN screener, our work has adapted to an electronic format, which may promote uptake. ELICIT demonstrates a multicenter quality improvement initiative for screening and intervening on unmet social needs in PwCF, with the potential for tailoring and expanding to more CF programs in a rapidly changing CF model of care.
A major strength of this project is the multicenter, multidisciplinary collaboration and PFP participation in the coproduction of tools and processes. PwCF coproduced all phases of the project, from tool design and process refinement to implementation. All sites in this project participated in the CFLN,27,28 a learning health network focused on improving clinical and quality-of-life outcomes through multisite, data-driven QI projects. In addition, including both pediatric and adult teams, each representing a different region of the United States in this multicenter project allowed shared learning in subpopulations of PwCF that would not have been possible otherwise. Potential for variability in survey completion rates underscores the importance of incorporating flexible delivery methods. An additional strength of this methodology described is the customizability and adaptability of the processes to meet the needs of each site’s existing workflow and cohorts while collecting the same measures across sites allowing for improved understanding of how the processes positively and negatively affect barriers to implementation.
This project has several limitations. First, there is potential for increased burden to teams with time required to perform screening, intervention, and data tracking. The pilot work conducted by this group addresses this limitation by establishing a robust framework that enables the expansion of tested processes to additional care centers. Rigorous testing in QI-proficient teams has allowed refining, optimizing, and simplifying the processes and tools so that they may be implemented more easily in CF programs with less robust QI experience and resources. Further, implementation of this screening tool has the potential to highlight a broader range of social and environmental challenges faced by people with CF (PwCF) and their families, which raises questions about the social work resources needed to respond effectively. The Cystic Fibrosis Foundation has recognized that PwCF must navigate HRSN alongside complex disease-specific psychosocial factors that directly influence health and quality of life. 29 Social workers are identified as required members of the multidisciplinary CF care model and play a pivotal role in proactively identifying and addressing these concerns, 29 suggesting that use of the tool would not necessarily require adding new team members. Insights from implementation could help clinics better map unmet social needs, connect patients with available resources, and evaluate intervention outcomes and, in settings where demand surpasses capacity, support a case for additional social work staffing to sustain comprehensive care. Other difficulties include survey fatigue and that HRSN questions could be considered intrusive by some PwCF, given the stigma associated with unmet social needs and accepting assistance. To overcome these limitations, our group has extensively piloted and adapted the screener with input from PFP. Codesigned educational campaigns were also utilized to make patients aware of the rationale for the project and reduce potential stigma associated with seeking assistance for unmet social needs.30,31 We believe that explaining the reasons behind the screening and providing education about resources has improved feasibility to care teams and acceptability to PwCF as part of routine clinical care. Further, incorporating these processes into routine clinical SW assessment as part of the standard of care can offset survey fatigue through personalization, one-on-one education about the purpose and potential benefits of the screening. The screener may also be paired with other routine screenings administered by the CF team including pre-visit planning and mental health screening to ease staff burden. Another key limitation is that our sites have small populations of ethnic/racial minorities, consistent with the broader CF population. However, it is important to identify trends using the demographic data collected to inform targeted interventions if disparities are discovered. Additionally, while the pediatric screening tool was tailored with input from PFPs at each of the pediatric sites, it was not formally pilot-tested in the pediatric CF population prior to this work; subsequent results from the testing phase should evaluate its feasibility, acceptability, and validity in younger age groups. Finally, this study did not include formal power calculations, as it was implemented as a multicenter QI initiative embedded in routine care. All eligible participants were included at each site to maximize real-world applicability and system-level learning. While this approach enhances generalizability within the included settings, it limits the ability to determine the statistical power to detect small or site-specific effects. This is a recognized trade-off in pragmatic QI designs, and future studies may benefit from incorporating power calculations when feasible.
Next steps in ELICIT will involve completing the testing phase and reporting on outcome measures to assess project impact and assess differences between subpopulations. While the pediatric screening tool was tailored with input from parent partners at each of the pediatric sites, it was not formally pilot-tested in the pediatric CF. The current testing phase will evaluate its performance and applicability in younger age groups and provide valuable insight into further adaptations required in pediatric populations. Following the testing phase, the project will progress into the sustainability phase, focusing on re-screening protocols and embedding practices into routine care. Lessons learned in this phase will then inform scale and spread, with attention to maintaining fidelity while adapting to local context. Initial planning is underway to adapt the screening and intervention model for broader dissemination across additional CF programs. This includes developing standardized tools such as a social determinants of health (SDH) index and a change package for additional sites.
The data obtained from this project regarding identified needs and resource availability can be used to inform targeted interventions in the broader CF care center network with the aim of improving access to care by intervening in subsets of the CF population that are especially vulnerable due to unmitigated HRSN. TH and remote methods to identify and address HRSN can serve as powerful tools in improving access to care and reducing the rate of patients and families lost to follow-up. This work also provides an opportunity for enhanced partnership in care, coproduced development of resources, and shared decision-making tools that can be utilized throughout the broader CF Community. Data on the prevalence of HRSN and gaps in individual-level resources may also be used to advocate for systems-level changes to affect SDOH more broadly in the CF Community including data-driven advocacy for resource allocation with Medicaid/Medicare, private insurance, and health system networks.
Conclusion
ELICIT designed and tested a multicenter QI project for screening and intervening on HRSN in PwCF. Intentional design and planning, including partnership with PFPs and the identification and compilation of locally available resources, is essential so that support can be offered promptly to PwCF who screen positive for unmet needs. Learning, tools, and resources generated from this project may be spread to other centers in the CF Care Center Network and other multidisciplinary teams involved in screening and mitigation of HRSN. This work may improve access to care by assessing and addressing unmet needs in the most vulnerable subsets of the CF Community, including subgroups who are typically marginalized and minorities, so that all people with CF can lead not only long, but fulfilling lives, regardless of social factors.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251393088 – Supplemental material for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project
Supplemental material, sj-docx-1-tar-10.1177_17534666251393088 for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project by Julianna Bailey, Deirdre Jennings, Melissa Alao, Justin D. Anderson, Bryan Garcia, Rachel Gordon, Katie Gravley, Moira Harrison, Michael Powers, Stacie Prettiman, Nathan Salter, Gregory S. Sawicki and Dana Albon in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666251393088 – Supplemental material for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project
Supplemental material, sj-pdf-2-tar-10.1177_17534666251393088 for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project by Julianna Bailey, Deirdre Jennings, Melissa Alao, Justin D. Anderson, Bryan Garcia, Rachel Gordon, Katie Gravley, Moira Harrison, Michael Powers, Stacie Prettiman, Nathan Salter, Gregory S. Sawicki and Dana Albon in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666251393088 – Supplemental material for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project
Supplemental material, sj-pdf-3-tar-10.1177_17534666251393088 for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project by Julianna Bailey, Deirdre Jennings, Melissa Alao, Justin D. Anderson, Bryan Garcia, Rachel Gordon, Katie Gravley, Moira Harrison, Michael Powers, Stacie Prettiman, Nathan Salter, Gregory S. Sawicki and Dana Albon in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-4-tar-10.1177_17534666251393088 – Supplemental material for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project
Supplemental material, sj-pdf-4-tar-10.1177_17534666251393088 for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project by Julianna Bailey, Deirdre Jennings, Melissa Alao, Justin D. Anderson, Bryan Garcia, Rachel Gordon, Katie Gravley, Moira Harrison, Michael Powers, Stacie Prettiman, Nathan Salter, Gregory S. Sawicki and Dana Albon in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-5-tar-10.1177_17534666251393088 – Supplemental material for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project
Supplemental material, sj-pdf-5-tar-10.1177_17534666251393088 for Remotely addressing health-related social needs (ELICIT) in cystic fibrosis: design and implementation of a multicenter screening and intervention quality improvement project by Julianna Bailey, Deirdre Jennings, Melissa Alao, Justin D. Anderson, Bryan Garcia, Rachel Gordon, Katie Gravley, Moira Harrison, Michael Powers, Stacie Prettiman, Nathan Salter, Gregory S. Sawicki and Dana Albon in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
