Abstract
Background:
Therapeutic management of idiopathic pulmonary fibrosis (IPF) remains challenging. IPF patients frequently exhibit a hypercoagulable state, and anticoagulant therapy has emerged as a potential strategy; however, its clinical utility remains controversial.
Objectives:
To systematically evaluate the efficacy and safety of anticoagulant therapy in IPF patients.
Design:
Systematic review and meta-analysis of randomised controlled trials.
Data sources and methods:
Comprehensive searches were conducted in PubMed, Cochrane Library, EMBASE, Web of Science, CNKI, Wanfang Data, VIP, and CBM databases for randomised controlled trials (RCTs) investigating anticoagulant therapy in IPF, from database inception to February 2025. Two investigators independently screened the literature, extracted data, and assessed risk of bias. Meta-analysis was performed using RevMan 5.3.
Results:
Seven RCTs involving 504 patients with IPF were included. Compared with control groups not receiving anticoagulants, anticoagulant therapy significantly improved PaO2 (MD = 11.64, 95% CI 7.09–16.18, p < 0.00001), SaO2(MD = 4.44, 95% CI 2.42–6.47, p < 0.0001), and HGF levels (MD = 108.74, 95% CI 88.58 to 128.90, p < 0.00001) but significantly reduced the mMRC score (MD = −0.38, 95% CI −0.62 to −0.14, p = 0.002) and D-dimer levels (MD = -0.08, 95% CI −0.11 to −0.05, p < 0.00001). No significant difference was observed in PaCO2 levels (MD = −4.26, 95% CI −9.09 to 0.56, p = 0.08). Anticoagulant therapy did not demonstrate benefit in reducing all-cause mortality (RR = 3.46, 95% CI 1.57–7.61, p = 0.002) or adverse reactions (MD = 1.44, 95% CI 1.15–1.81, p = 0.002).
Conclusion:
Anticoagulant therapy may offer clinical benefits in IPF management. However, its lack of mortality benefit and safety concerns warrant cautious interpretation. Clinicians should carefully assess individual bleeding risks before initiating treatment. Due to the limited number of included studies and data constraints, further large-scale, high-quality, multicenter, and long-term RCTs are needed. Future research should prioritise risk stratification, optimised anticoagulation protocols and identification of biomarkers predictive of bleeding risk to inform clinical decision-making.
Trial registration:
The study protocol was registered with PROSPERO (Registration number: CRD42022349940).
Plain language summary
Study combining results from multiple clinical trials to evaluate blood-thinning drugs for idiopathic pulmonary fibrosis (IPF) patients Why was the study done? Idiopathic pulmonary fibrosis (IPF) is a serious lung disease with limited treatment options. Some studies suggest that blood-thinning drugs (anticoagulants) might help IPF patients because their blood tends to clot more easily. However, whether these drugs truly benefit patients or cause more harm remains highly debated. This study aimed to clarify the benefits and risks of anticoagulants for IPF. What did the researchers do? Researchers gathered and analysed data from all available high-quality clinical trials (published up to February 2025) comparing anticoagulants with no anticoagulants (or placebo) in IPF patients. They combined results from 7 trials involving 504 patients to assess effects on breathing (oxygen levels, shortness of breath), blood markers, survival, and side effects. What did the researchers find? Potential benefits: Anticoagulants improved blood oxygen levels (PaO2 and SaO2), reduced shortness of breath (mMRC score), and lowered blood clotting markers (D-dimer) and a disease-related factor (HGF). No clear change: Blood carbon dioxide levels (PaCO2) were unaffected. Important risks: Anticoagulant therapy did not demonstrate benefit in reducing all-cause mortality or adverse reactions. What do the findings mean? While anticoagulants may improve some symptoms in IPF patients, these potential benefits are outweighed by significantly higher risks of death and adverse effects. Anticoagulants are not recommended as routine treatment for IPF. If considered for individual patients, doctors must carefully weigh bleeding risks. More large, long-term studies are needed to confirm these findings. Future research should focus on: Identifying which patients might benefit safely, Developing safer anticoagulant strategies, Finding biomarkers to predict bleeding risk.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, severely debilitating, and ultimately fatal disease that poses a significant threat to public health. Its incidence rate is 17.4/100,000 1 and continues to rise annually. 2 The median survival after diagnosis is approximately 4–5 years, 3 with mortality during hospitalisation for acute exacerbations reaching 50%–80%.4–6 The aetiology and pathogenesis of IPF remain poorly understood, and there is currently no universally accepted optimal treatment. Clinical studies have shown elevated concentrations of tissue factor and thrombin in the bronchoalveolar lavage fluid of patients with IPF compared to healthy people, 7 and the risk of developing a prothrombotic state in IPF is estimated to be four times higher than in the general population. 8 Autopsy studies have also identified blood clots in alveolar capillaries and fibrin deposition in the alveolar space. 9 Based on these findings, it is hypothesised that early anticoagulant therapy may slow disease progression by limiting fibrin deposition. In recent years, numerous international and domestic studies have explored the use of anticoagulant drugs for the treatment of IPF, aiming to improve clinical outcomes and identify novel therapeutic strategies. While some findings suggest that anticoagulation may improve survival, pulmonary function, and oxygenation indices in patients,10–12 significant controversy remains due to inconsistent study results, concerns about bleeding risks, and inconsistent guideline recommendations.13,14 Furthermore, the efficacy and safety profiles of clinically available anticoagulants (e.g. heparins, warfarin, and direct oral anticoagulants) may differ substantially, 15 further complicating clinical decision-making. Given the conflicting evidence and lack of consensus, this study conducted a systematic review and meta-analysis of randomised controlled trials (RCTs) involving anticoagulant therapy for IPF, aiming to comprehensively evaluate its impact on clinical effectiveness and safety in IPF patients. The selection of outcome measures for this study was guided by the PRISMA framework, with emphasis on their mechanistic relevance, clinical decisiveness, and safety, with the goal of providing evidence to inform clinical practice and guide future research.
Methods
The reporting of this study conforms to the PRISMA checklist. 16
Inclusion criteria
Studies were included if they met the following criteria, based on PICOS principles:
(1)
(2)
(3)
(4)
(5)
Exclusion criteria
Studies were excluded if they met any of the following criteria: (1) Participants were not diagnosed with IPF. (2) Non-randomised designs (e.g., retrospective studies, cohort studies), or other non-RCT literature. (3) Interventions based on traditional Chinese medicine, including herbal prescriptions and injections. (4) Animal studies, review articles, conference abstracts, case reports, and other literatures. (5) Studies for which data could not be extracted or full texts were unavailable.
Search strategy
Eight electronic databases were searched from inception to February 2025: The Cochrane Library, PubMed, Embase, Web of Science, China National Knowledge Infrastructure (CNKI), WanFang Data, Chinese Science and Technology Periodicals Database (VIP), and SinoMed. RCTs investigating anticoagulant therapy in patients with IPF were screened. Searches were limited to Chinese and English publications, using a combination of subject headings and free words. An example of the PubMed search strategy is shown in e-Table 1 in Supplemental Materials.
Data extraction
All search results were imported into EndNote X8 for management, and two researchers independently screened the literature according to the inclusion and exclusion criteria, and then cross-checked. First, after both systematic and manual duplicate checking, duplicate records are removed. Then, the titles and abstracts are screened to preliminarily exclude documents that do not meet the inclusion criteria. Finally, full texts are reviewed for documents that are uncertain or potentially eligible, and those not meeting the criteria are excluded. In cases of disagreement, inclusion decisions are resolved through discussion or consultation with a third party. Excel 2019 was used to extract the following data from eligible studies: title, name, and year of the first author, study type, sample size, randomisation method, blinding status, case dropout information, intervention measures in the experimental and control groups, and outcome indicators. The extracted outcomes included: all-cause mortality; arterial blood gas (ABG) parameters: arterial partial pressure of oxygen (PaO2), arterial partial pressure of carbon dioxide (PaCO2), and blood oxygen saturation (SaO2); the modified Medical Research Council Dyspnea Scale (mMRC score); D-dimer; hepatocyte growth factor (HGF); and adverse events. Adverse event types and frequencies were extracted exactly as reported in the original studies, with focused analysis on haemorrhagic events (major and minor bleeding), respiratory system events (such as haemoptysis or pulmonary haemorrhage), and thromboembolic events (including deep vein thrombosis and pulmonary embolism), among others.
The selection of these outcome measures was primarily guided by the PRISMA-P framework and further informed by the three endpoint selection principles outlined in the 2018 IPF Diagnostic Guidelines 2 : ① Mechanistic relevance (e.g. D-dimer as a marker of fibrin-driven pathology); ② Clinical decisiveness (mortality and ABG parameters as predictors of respiratory failure); and ③ Safety requirements (anticoagulant-specific bleeding events).
Risk of bias assessment
The Cochrane Risk of Bias Assessment Tool was used to evaluate the methodological quality of the included studies. The assessment covered seven domains: (1) the method of random sequence generation; (2) whether allocation concealment was performed; (3) whether blinding was implemented for participants and personnel; (4) whether outcome assessors were blinded; (5) completeness of outcome data; (6) selective outcome reporting; and (7) the presence of other potential sources of bias. 22 Based on these criteria, each included study was classified as having a “low risk of bias,” “high risk of bias,” or “unclear risk of bias.”
Statistical analysis
Statistical analysis was performed using RevMan 5.3. The I² statistic was used to assess heterogeneity. 23 When I2<50% and p > 0.05, heterogeneity was considered low, and a fixed-effects model was applied. If I2⩾50% or p ⩽ 0.05, indicating substantial heterogeneity, a random-effects model was used, and sensitivity analysis was performed to explore potential sources of heterogeneity. For continuous variables, mean difference (MD) and 95% confidence intervals (CIs) were used as summary statistics; when different measurement units were involved, the standardised MD (SMD) with 95% CI was used. For dichotomous variables, the relative risk (RR) and corresponding 95% CI were calculated. Funnel plots were used to assess publication bias when the number of included studies for a given outcome was four or more.
Results
Search results
A total of 1550 records were retrieved. After systematic and manual deduplication, 995 duplicates were excluded. An additional 514 records were excluded after screening titles and abstracts. Full-text screening of the remaining studies resulted in the exclusion of 41 articles that did not meet the inclusion criteria. Ultimately, seven studies were included in the final analysis.9,24–29 These included four studies published in Chinese and three in English. The process of literature selection is shown in Figure 1.

PRISMA flowchart for the study selection process.
Study characteristics
All seven included studies were RCTs investigating the use of anticoagulant therapy in patients with IPF, involving a total of 504 patients 193 in the experimental group and 311 in the control group. Detailed characteristics of the included studies are presented in Table 1.
Details of the characteristics of the studies.

(1) All-cause mortality; (2) PaO2; (3) PaCO2; (4) SaO2; (5) mMRC score; (6) D-dimer; (7) HGF; (8) Adverse Events.
C, control group; CWM, Western medicine conventional treatment; E, Experimental group; NCT, ClinicalTrials.gov identifier; NR, Not mentioned.
Risk of bias within studies
Randomisation: Three studies mentioned randomisation without specifying the method (unclear risk), while the remaining studies provided detailed methods (low risk). Allocation concealment: Only one study explicitly reported allocation concealment (low risk); others were unclear. Blinding: Two studies employed double-blinding (low risk); the others were rated as high or unclear risk. Incomplete outcome data: One study had participant dropouts (high risk); other studies reported complete data (low risk). Selective reporting: All studies fully reported prespecified outcomes (low risk). Other biases: Risk was unclear across all studies. The overall risk of bias assessment and individual evaluations are shown in Figure 2.
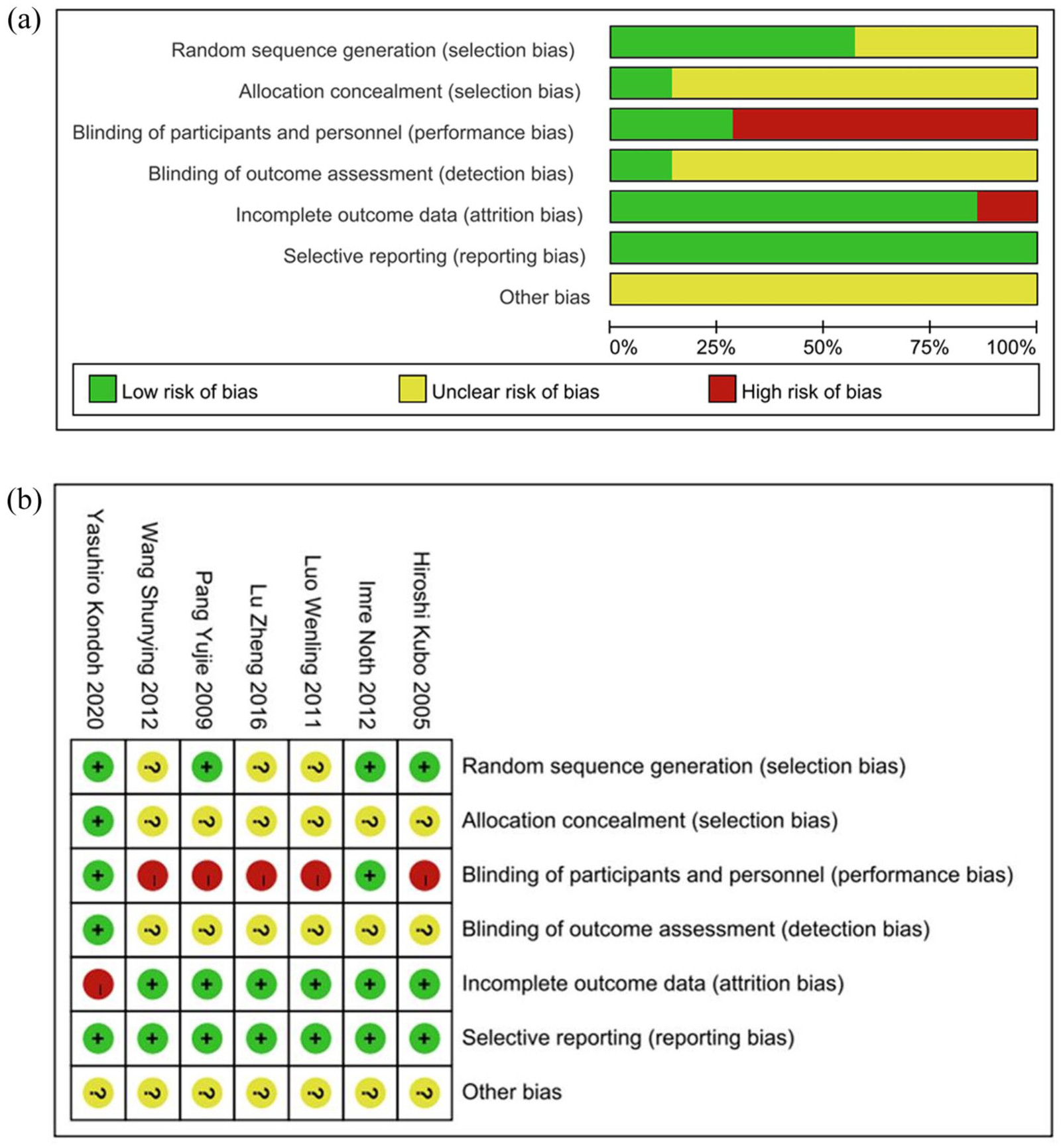
(a) Risk of bias summary. (b) Risk of bias graph.
Meta-analysis results
All-cause mortality
Three studies, with a total of 278 patients (135 in the experimental group, 143 in the control group) were included. Heterogeneity testing indicated substantial heterogeneity (p = 0.0005, I2 = 87%; Figure 3(a)), and a random-effects model was used for meta-analysis. Sensitivity analysis to identify the source of heterogeneity revealed that exclusion of the study by Hiroshi Kubo et al.’s study eliminated the observed heterogeneity. After this exclusion, the results indicated that anticoagulant therapy may not provide a significant advantage in reducing all-cause mortality in patients with IPF compared with the non-anticoagulant group (RR = 3.46, 95% CI 1.57–7.61, p = 0.002; Figure 3(b)). However, due to the presence of residual heterogeneity and other confounding factors, these findings should be considered exploratory and do not offer conclusive evidence regarding the impact of anticoagulant therapy on all-cause mortality in IPF.
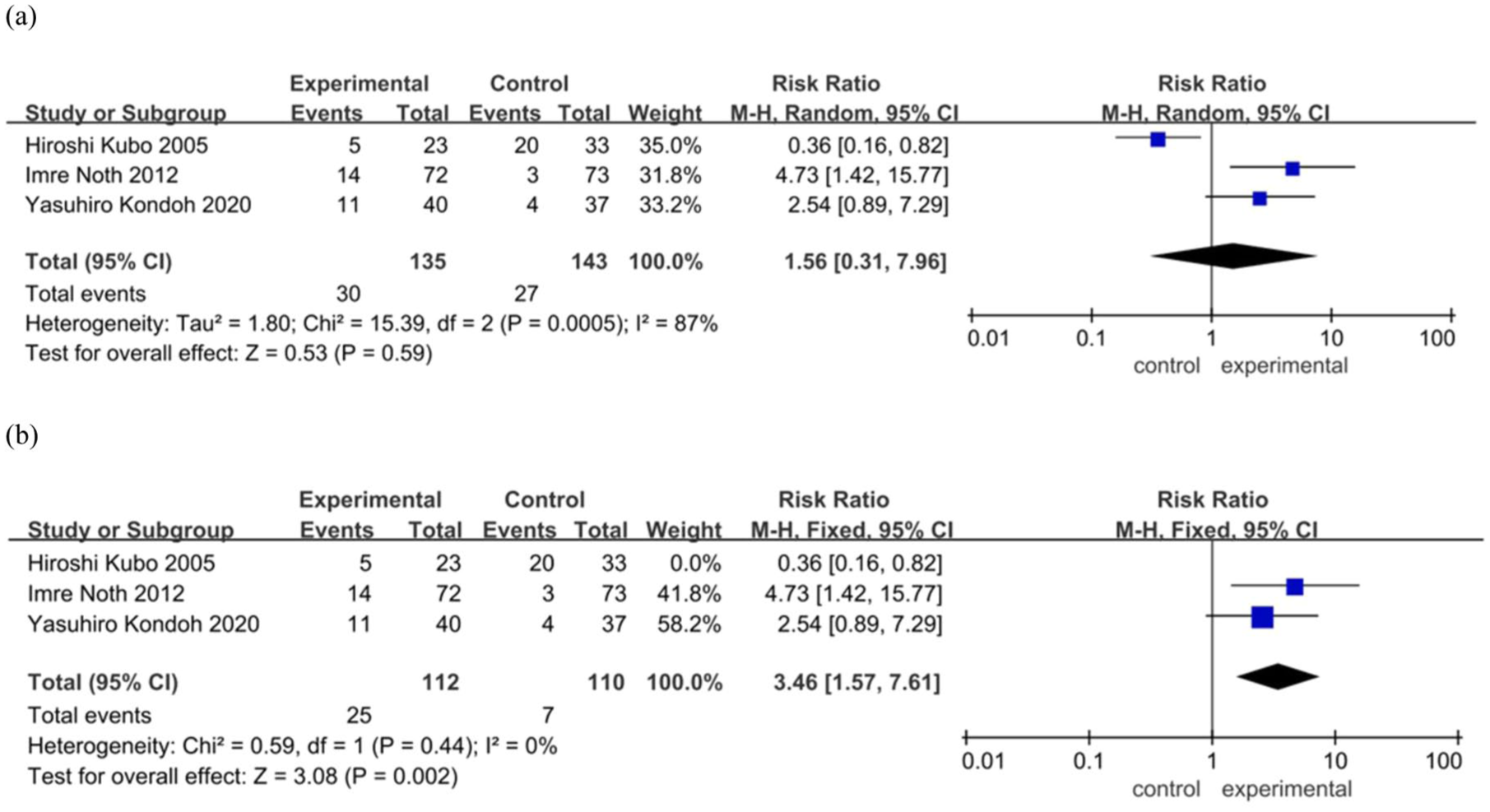
Forest plot of all-cause mortality between experimental group and control group. (a) Before eliminating the sources of heterogeneity. (b) After eliminating the sources of heterogeneity.
Arterial blood gas analysis
PaO2. Four studies were included, comprising a total of 99 patients (51 in the experimental group and 48 in the control group). The heterogeneity test yielded p = 0.07, I2 = 58% (Figure 4(a)), indicating moderate heterogeneity; thus, a random-effects model was applied in the meta-analysis. Sensitivity analysis identified the study by Lu Zheng et al. as the primary source of heterogeneity. After excluding this study, the heterogeneity was resolved, and the results demonstrated that anticoagulant therapy significantly increased PaO2 levels in patients with IPF (MD = 11.64, 95% CI 7.09–16.18, p < 0.00001; Figure 4(b)).
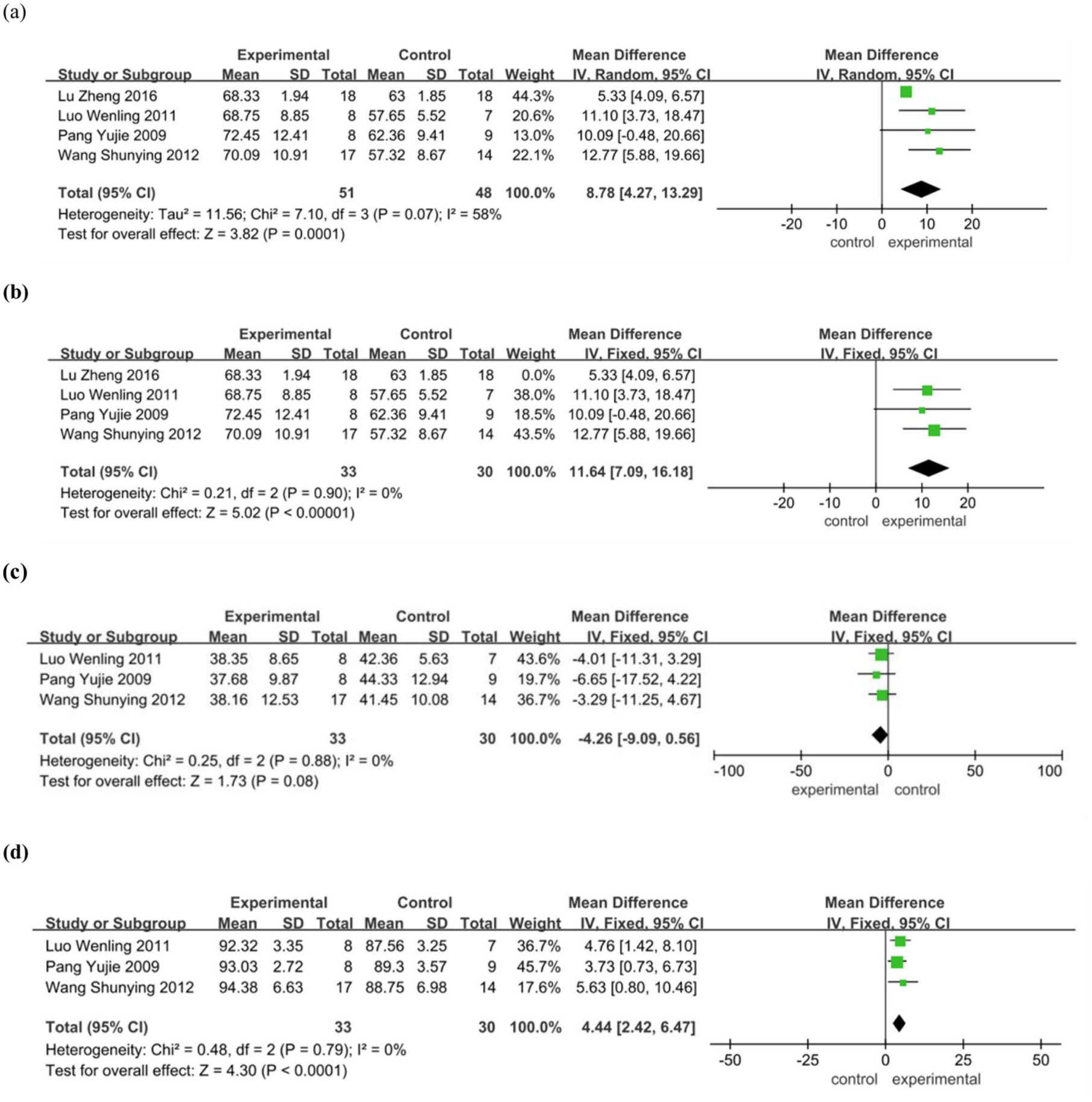
Forest plot of arterial blood gas analysis between experimental group and control group. (a) PaO2. Before eliminating the sources of heterogeneity. (b) PaO2. After eliminating the sources of heterogeneity. (c) PaCO2. (d) SaO2.
PaCO2. Three studies were included, involving a total of 63 patients (33 in the experimental group and 30 in the control group). The heterogeneity test showed p = 0.88, I2 = 0%, indicating no significant heterogeneity. A fixed-effects model was used for the meta-analysis. The results showed no statistically significant difference in PaCO2 levels between the anticoagulant and control groups (MD = −4.26, 95% CI −9.09 to 0.56, p = 0.08 > 0.05; Figure 4(c)).
SaO2. Three studies were included, with a total of 63 patients (33 in the experimental group and 30 in the control group). Heterogeneity was low (p = 0.79, I2 = 0%), and a fixed-effects model was applied. The meta-analysis showed that anticoagulant therapy significantly increased SaO2 levels in patients with IPF (MD = 4.44, 95% CI 2.42–6.47, p < 0.0001; Figure 4(d)).
MMRC score
Two studies were included, involving a total of 48 patients (25 in the experimental group and 23 in the control group) were included. The heterogeneity among the studies was low (p = 0.29, I2 = 12%), and a fixed-effects model was applied. The meta-analysis demonstrated that anticoagulant therapy significantly reduced the mMRC dyspnea score in patients with IPF (MD = −0.38, 95% CI −0.62 to −0.14, p = 0.002) (e-Figure 1 in Supplementary Materials).
D-dimer
Four studies were included, comprising a total of 117 patients (56 in the experimental group and 61 in the control group). The heterogeneity test showed significant heterogeneity among the studies (p = 0.001, I2 = 81%), prompting the use of a random-effects model. Sensitivity analysis identified the study by Hiroshi Kubo et al. as the primary source of heterogeneity. Upon exclusion of this study, heterogeneity was resolved. The updated analysis showed that anticoagulant therapy significantly reduced D-dimer levels in patients with IPF. (MD = −0.08, 95% CI −0.11 to −0.05, p < 0.00001; Figure 5(b)).

Forest plot of D-dimer between experimental group and control group. (a) Before eliminating the sources of heterogeneity. (b) After eliminating the sources of heterogeneity.
Hepatocyte growth factor
Two studies were included, involving 48 patients (25 in the experimental group and 23 in the control group). Heterogeneity was low (p = 0.25, I2 = 24%), and a fixed-effects model was used. The results indicated that anticoagulant therapy significantly increased HGF levels in patients with IPF (MD = 108.74, 95% CI 88.58–128.90, p < 0.00001) (e-Figure 2 in Supplemental Materials).
Adverse events
Adverse events were reported in four studies, involving a total of 278 patients (140 in the experimental group and 138 in the control group). Of these, two studies explicitly stated that no significant adverse reactions were observed, while the other two studies provided detailed accounts of specific adverse events (Table 2). The reported events were predominantly haemorrhagic, with additional occurrences of insomnia, delirium, and other reactions. Haemorrhagic events primarily involved the respiratory system, as well as skin and subcutaneous tissue disorders. Across all studies, the anticoagulant-treated group consistently exhibited a higher incidence of haemorrhagic events compared to the control group. The heterogeneity among the studies was low (p = 0.32, I2 = 0%), and a fixed-effects model was applied. Meta-analysis indicated that anticoagulant therapy may increase the incidence of adverse events (MD = 1.44, 95% CI 1.15–1.81, p = 0.002; Figure 6).
Details of the adverse events of the studies.


Forest plot of adverse events between experimental group and control group.
Publication bias
Funnel plot analysis was conducted for outcomes with ⩾4 included studies (PaO2 and D-dimer) to assess publication bias. As shown in e-Figures 3 and 4 (Supplemental Materials), both funnel plots exhibited asymmetry, suggesting potential publication bias. Contributing factors may include the small number of included studies, heterogeneity in anticoagulant regimens, and differences in disease severity among patient populations.
Discussion
This study included seven domestic and international RCTs on anticoagulant therapy in IPF and conducted a meta-analysis of the following outcomes: all-cause mortality, PaO2, PaCO2, SaO2, mMRC score, D-dimer, HGF levels, and the incidence of adverse events. The results indicated that, compared with the non-anticoagulant group, anticoagulant therapy may not offer a significant advantage in reducing all-cause mortality among IPF patients. In IPF clinical trials, all-cause mortality is widely regarded as the gold-standard endpoint for evaluating the net effect of therapy; it directly captures survival benefit and is one of the most scientifically robust and clinically meaningful ultimate outcomes. 30 Our finding that anticoagulant therapy failed to significantly reduce all-cause mortality may represent a fundamental constraint on its clinical utility. Several factors may account for the observed heterogeneity in all-cause mortality outcomes across studies: (1) differences in baseline disease severity; (2) inconsistent inclusion and exclusion criteria; (3) variability in anticoagulant type and dosage. Recent real-world evidence emphasises the imperative need for a nuanced interpretation of the effects of different anticoagulant classes. As demonstrated by King et al. 15 in their analysis of the Pulmonary Fibrosis Foundation Registry, warfarin significantly increased mortality risk in IPF patients (adjusted HR 2.566; 95% CI 1.095–6.016), whereas direct oral anticoagulants (DOACs) did not (adjusted HR 1.368; 95% CI 0.500–3.737). This distinction highlights warfarin’s potential to exacerbate fibrosis via protein C inhibition, in contrast to DOACs’ capacity to attenuate fibrotic signalling through direct thrombin or factor Xa blockade, suggesting a potential safety advantage for the latter; (4) variation in therapeutic regimens, specifically, the transition from warfarin to low-molecular-weight heparin during rehospitalisation for acute exacerbations, which may influence mortality outcomes. Among the three studies included in the mortality analysis, only Hiroshi Kubo et al. 9 reported that switching from warfarin to low-molecular-weight heparin during rehospitalisation significantly improved survival and reduced mortality without increasing adverse events. This finding may have contributed to the overall heterogeneity. In contrast, Imre Noth et al. 27 reported a higher mortality rate with warfarin than with the control group, and Yasuhiro Kondoh et al. 29 observed increased mortality with thrombomodulin-α than compared with the control group, leading to the recommendation against its clinical use. Notably, warfarin was not recommended for the treatment of IPF as early as 2011 in the IPF Clinical Diagnosis and Treatment Guidelines. 31 However, this does not mean that anticoagulant drugs are prohibited for patients with IPF in a hypercoagulable state. 13 An increasing number of studies have shown that heparin-based anticoagulants and certain new anticoagulants may offer new therapeutic pathways for IPF.11,32,33 These newer anticoagulants do not appear to increase mortality in patients with IPF. 34 Therefore, whether they are effective in reducing mortality in IPF remains to be further studied.
In terms of improving PaO2 and SaO2 levels and reducing mMRC scores in patients with IPF, the results of this study indicated that the anticoagulant group demonstrated certain advantages over the non-anticoagulant group. The improvements in these functional parameters hold distinct clinical value. ABG analysis (PaO2, SaO2) provides direct insight into the efficiency of gas exchange, the impairment of which is a core pathophysiological alteration closely linked to IPF severity and a key determinant of functional status and prognosis. 35 Concurrently, the reduction in the mMRC score is an important patient-centred finding, as it directly reflects a patient-perceived alleviation of dyspnea symptoms and an improvement in daily activity tolerance, suggesting that anticoagulant therapy may enhance the quality of life for individuals with IPF. 36 These benefits are likely mediated through multiple mechanisms of anticoagulant drugs, particularly low-molecular-weight heparin (LMWH). Research by Tsushima et al. 37 suggested that anticoagulants might mitigate fibrosis progression through direct or indirect inhibition of thrombin. LMWH has multiple biological activities, including anticoagulant, anti-fibrotic, and anti-inflammatory effects. It improves pulmonary microcirculation and reduces blood viscosity, thereby preventing pulmonary thrombosis. In addition, LMWH inhibits platelet release of mediators such as 5-HT and bradykinin, which can help relieve bronchospasm-induced ventilation dysfunction.24,38,39 These mechanisms collectively contribute to elevated PaO2 and SaO2 levels and a reduction in mMRC scores in IPF patients. However, no significant difference was observed between the groups in reducing PaCO2 levels. Regarding PaO2 outcomes, sensitivity analysis identified that exclusion of studies such as Lu Zheng et al. 28 resolved the observed heterogeneity, suggesting that this study was a primary source of variation. A plausible explanation is that, unlike the other three studies, the intervention in the Lu Zheng trial did not include concomitant glucocorticoid therapy. This therapeutic inconsistency may have led to differential treatment responses, thereby contributing to heterogeneity. D-dimer and HGF serve as mechanistic biomarkers. D-dimer levels reflect the extent of fibrin deposition in the alveoli, offering insight into the fibrotic pathology and risk of acute exacerbation in IPF. 40 HGF contributes to alveolar epithelial and endothelial repair following lung injury by inducing anti-inflammatory and antioxidant gene expression, thereby mitigating tissue fibrosis. 41 In terms of D-dimer levels, sensitivity analysis demonstrated that heterogeneity resolved upon exclusion of studies such as Hiroshi Kubo et al. 9 suggesting that this study was the primary contributor to the observed heterogeneity. This divergence likely stems from discrepancies in anticoagulation regimens. In the Kubo study, a larger proportion of patients were rehospitalised and received LMWH instead of the initial oral warfarin therapy. This therapeutic switch may have influenced D-dimer levels, thereby contributing to substantial heterogeneity. LMWH was also used as anticoagulant in all the other included studies, which consistently reported reduced D-dimer levels in patients with IPF; this reduction is likely related to LMWH’s ability to improve hypercoagulability through the mechanisms mentioned above. In terms of HGF levels, the results of this study indicate that LMWH treatment may enhance HGF expression in patients with IPF. Previous studies have shown that activation of the fibrinolytic system can upregulate HGF, which promotes alveolar epithelial cell proliferation and repair, thereby exerting anti-fibrotic effects.42,43 In vitro experiments further support this mechanism, as serum levels of active HGF were shown to increase following LMWH administration. 38 These findings provide mechanistic support for the observed improvement in HGF levels with LMWH therapy in this study.
Regarding safety, only two studies27,29 in this analysis reported specific adverse events. The meta-analysis indicated that, compared with the non-anticoagulant group, anticoagulant therapy in IPF patients may be associated with certain adverse effects, particularly haemorrhagic events. In the study by Imre Noth et al, 27 both major and minor bleeding events occurred more frequently in the experimental group than in the control group. Similarly, Yasuhiro Kondoh et al.’s 29 study reported a higher incidence of adverse events, both in terms of event types and number of affected participants, in the anticoagulant group. The probability of haemorrhagic events was also higher in the experimental group compared to the control. Haemorrhagic adverse events primarily involved respiratory, thoracic, and mediastinal conditions, such as epistaxis, haemoptysis, and pulmonary haemorrhage, as well as skin and subcutaneous tissue disorders, including subcutaneous haemorrhage and purpura. In severe cases, additional adverse effects such as insomnia and delirium were reported. These findings collectively suggest that anticoagulant use in IPF patients may increase the risk of bleeding-related complications. Therefore, when anticoagulation is required for comorbid conditions (e.g., atrial fibrillation or venous thromboembolism) in IPF patients, it is recommended that clinicians refer to the Padua Prediction Score 44 to assess venous thromboembolism (VTE) risk and carefully evaluate patient comorbidities (e.g., atrial fibrillation, deep vein thrombosis) before initiating therapy. Throughout the treatment course, patient symptoms and coagulation parameters should be closely monitored. Special attention should be paid to early signs of respiratory bleeding, such as epistaxis or haemoptysis, as well as dermatologic signs, such as subcutaneous haemorrhage or purpura. Clinicians should also exercise caution in selecting appropriate anticoagulant agents in this patient population. A multidisciplinary approach is recommended to rigorously assess indications for anticoagulation, ensuring that therapeutic benefits are achieved while minimising the risk of adverse events.
Limitations and future research
This study has several limitations that must be acknowledged. (1) First, due to the presence of heterogeneity and other methodological constraints, the findings, particularly regarding all-cause mortality, should be considered exploratory. The current data do not allow for definitive conclusions on mortality outcomes. Sensitivity analyses investigating sources of heterogeneity may not fully account for observed heterogeneity. Therefore, interpretation of pooled results for outcomes with high heterogeneity (mortality, PaO2, and D-dimer) requires particular caution, as these may reflect true variations in treatment effects across diverse patient populations or study contexts rather than a single universal effect size. (2) Second, the analysis is limited by the small number of included publications (n = 7), which reduces statistical power and increases the influence of individual studies on the pooled results. Furthermore, most of the included trials were small-scale studies conducted in China, which limits the generalizability of the findings. The paucity of reported outcome measures also precluded more detailed subgroup analyses (e.g., stratification by specific anticoagulant classes). As a result, the pooled effect estimate reflects an average across different anticoagulant agents and may not reliably guide the clinical selection of specific therapeutics. (3) Third, many of the included studies inadequately reported key aspects of study design, such as randomisation methods, allocation concealment, and blinding. Lack of proper allocation concealment could introduce selection bias, while the absence of blinding for outcome assessors may lead to detection bias, both of which may result in overestimation of the true intervention effect. (4) Fourth, this study did not control for concomitant treatment measures aside from anticoagulants across the included trials. Differences in patient condition severity and variations in experimental design may have contributed to additional heterogeneity. Moreover, the limited number of studies precluded separate analyses of the efficacy and safety of individual anticoagulants. This aggregated approach may have introduced variability and affected the interpretability of the results. (5) Fifth, pulmonary function tests (PFTs), such as forced vital capacity (FVC) and maximum voluntary ventilation, are widely recognised as key clinical outcome measures in IPF. However, only one of the included studies reported PFT-related outcomes, while the remaining six did not. Due to this lack of data, a meta-analysis of pulmonary function outcomes could not be performed, which represents an important limitation of this study.
Recommendations: (1) The mortality rate of IPF is as high as 80%, 45 Therefore, there is a pressing need for future clinical research to address this highly unmet medical need. To truly capture the clinical course of IPF, these studies should align their outcome indicators closely with the disease characteristics and prioritise patient-relevant endpoints, such as time to disease progression or all-cause mortality. 46 Further research is equally needed to explore and validate the value of other important indicators, including the incidence of acute exacerbations, progression-free survival, and all-cause hospitalisation rates. 47 Furthermore, establishing a scientific and standardised core outcome set for IPF, utilising both qualitative and quantitative research methodologies, represents another critical objective for future research, aiming to ensure consistency and comparability across different studies. (2) Future studies should ensure the consistent reporting of core PFT parameters (such as FVC % predicted) to enable more comprehensive and meaningful evidence synthesis in this field. (3) Anticoagulant therapy for IPF remains in an exploratory stage. While several novel anticoagulants, such as thrombin inhibitors, selective factor Xa inhibitors, and dabigatran etexilate, may offer potential advantages over traditional agents, including lower bleeding risk and fewer drug, drug interactions,14,33 there is a current lack of clinical RCTs investigating these newer agents. It is therefore recommended that future research include large-scale, multicenter, high-quality RCTs to provide robust evidence-based guidance for anticoagulant use in IPF. These studies should strictly adhere to CONSORT (Consolidated Standards of Reporting Trials) guidelines and include detailed methodological reporting. (4) Future studies should also prioritise prospective trials evaluating DOACs in fibrotic ILD, especially given their neutral effect on survival as reported by King et al. 15 and their mechanistic potential to attenuate fibrosis via direct thrombin/factor Xa inhibition. There is an urgent need for RCTs comparing DOACs with warfarin for the management of venous thromboembolism in IPF cohorts, with an emphasis on clarifying class-specific effects rather than attributing outcomes broadly to anticoagulation.
Conclusion
In summary, anticoagulant therapy appears to improve PaO2, SaO2, and HGF levels, while reducing mMRC scores and D-dimer levels in patients with IPF. However, a core therapeutic paradox remains: these physiological improvements do not translate into survival benefits. Due to high heterogeneity among studies, anticoagulants demonstrated no significant advantage in reducing all-cause mortality. Moreover, reported adverse events, particularly bleeding, highlight safety concerns. This delicate risk–benefit balance necessitates that clinicians carefully assess individual bleeding risk before initiating anticoagulant therapy in patients with IPF. Subgroup analyses to determine the advantages of specific classes of anticoagulants could not be performed due to data limitations. Therefore, the conclusions drawn here require validation through large-scale, high-quality, multicenter, and long-term RCTs. Clinicians should rigorously evaluate the indications for anticoagulation and closely monitor relevant parameters to minimise the risk of adverse events during treatment. As evidence supporting novel anticoagulants continues to evolve, future research must address the current contradictions through three strategic pathways: (1) develop predictive models incorporating VEGF genotypes and fibrosis risk stratification; (2) conduct dose-finding studies and RCTs to determine the optimal treatment duration of DOACs; and (3) identify bleeding-risk biomarkers and design multicenter, adaptive phase III trials, such as randomised embedded multifactorial adaptive platform trials, to establish precision medicine-based anticoagulation protocols in IPF.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251393078 – Supplemental material for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tar-10.1177_17534666251393078 for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis by Jiamin Liu, Lu Wang, Guixiang Zhao and Hailong Zhang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251393078 – Supplemental material for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tar-10.1177_17534666251393078 for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis by Jiamin Liu, Lu Wang, Guixiang Zhao and Hailong Zhang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666251393078 – Supplemental material for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis
Supplemental material, sj-pdf-3-tar-10.1177_17534666251393078 for Clinical efficacy and safety of anticoagulants in the treatment of idiopathic pulmonary fibrosis: a systematic review and meta-analysis by Jiamin Liu, Lu Wang, Guixiang Zhao and Hailong Zhang in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
