Abstract
Background:
The escalating morbidity and mortality of chronic obstructive pulmonary disease (COPD) necessitates improved diagnostic approaches for comorbid infections. COPD patients exhibit heightened susceptibility to opportunistic pathogens like Nocardia species due to compromised airway defenses and frequent glucocorticoid/immuno-suppressant use. Despite its clinical significance, Nocardia infection remains diagnostically challenging due to nonspecific presentations and imaging features.
Objectives:
To develop and validate a diagnostic model integrating clinical characteristics and risk factors for COPD complicated by Nocardia infection.
Design:
A retrospective analysis was conducted on clinical data from 586 patients diagnosed with COPD and Nocardia infection, including clinical symptoms, laboratory tests, imaging findings, and treatment outcomes. Patients were screened according to inclusion and exclusion criteria and divided into two groups: COPD with Nocardia infection group (infection group) and COPD-only group (control group).
Methods:
This retrospective study analyzed 586 COPD patients (2019–2024), stratified into Nocardia-infected (n = 289) and noninfected (n = 297) cohorts. Demographic, laboratory, pulmonary function, and imaging data were collected. Multivariate logistic regression identified independent predictors, which informed a nomogram model. Model performance was assessed via concordance index (C-index), calibration curves, and ROC analysis.
Results:
Independent risk factors included hemoptysis (OR = 1.99, 95% CI: 0.76–5.26), lymphocyte count (OR = 6.81, 95% CI: 4.06–11.42), hemoglobin (OR = 1.01, 95% CI: 0.99–1.03), and pulmonary function parameters (FEV₁/FVC ratio OR = 12.47, 95% CI: 1.25–124.16). The model demonstrated excellent discrimination (C-index: 0.895 infected, 0.829 noninfected) and calibration (mean absolute error: 0.127–0.170). ROC analysis revealed AUCs of 0.896 (95% CI: 0.90–0.97) and 0.830 (95% CI: 0.77–0.89) for infected and noninfected groups, respectively.
Conclusion:
This validated nomogram provides a clinically actionable tool for early Nocardia detection in COPD patients, addressing a critical diagnostic gap. External validation is warranted to confirm generalizability.
Plain language summary
This study developed a tool to predict Nocardia infection in COPD patients using key risk factors like coughing blood and poor lung function, aiding early diagnosis. Further validation is needed. Why was the study done? COPD patients have weakened immune systems and often use medications like steroids, making them more likely to get infections. One rare but dangerous infection is caused by Nocardia, which is hard to diagnose because its symptoms (like cough and fever) look like other lung problems. Doctors often miss it or treat it too late, which can be life-threatening. This study aimed to create a simple tool to help doctors spot Nocardia infection early in COPD patients, so they can start treatment faster. What did the researchers do? The researchers studied COPD patients to identify factors that increase the risk of a rare but serious infection called Nocardia. They developed a tool to help doctors diagnose this infection earlier. What did the researchers find? They found that symptoms like coughing up blood, low immune cell counts, poor lung function, and certain blood test results were strong indicators of Nocardia infection. A model based on these factors accurately predicted the infection risk. What do the findings mean? This tool can help doctors detect Nocardia infection sooner in COPD patients, leading to faster treatment and better outcomes. More testing in different hospitals is needed to ensure the tool works for all patients.
Introduction
COPD represents a global health burden characterized by progressive airflow limitation and chronic inflammation. 1 Immuno-compromised states secondary to corticosteroid use and structural lung damage predispose patients to opportunistic infections, including nocardiosis. 2 Among these, Nocardia infection is relatively rare but associated with high mortality, presenting significant challenges in clinical treatment. 3 Early and accurate diagnosis of COPD combined with Nocardia infection is crucial for improving patient prognosis. Prompt diagnosis enables timely targeted treatment, effective infection control, and reduced disease severity, thereby enhancing patients’ quality of life and survival rates. However, current clinical practice lacks efficient and accurate diagnostic methods. The clinical manifestations of Nocardia infection are nonspecific, resembling those of other common pathogens, such as fever, cough, expectoration, and dyspnea. 4 Additionally, its imaging features are atypical and easily confused with other lung diseases, leading to frequent misdiagnosis and missed diagnosis. 5 This diagnostic dilemma often delays effective treatment, exacerbating disease progression and increasing patient suffering and medical burdens. 6 To address this issue, this study aimed to comprehensively analyze the clinical characteristics and risk factors of COPD patients and construct a validated diagnostic model for COPD combined with Nocardia infection. By identifying key diagnostic indicators, we hope to provide clinicians with a reliable tool for early diagnosis and treatment, ultimately improving patient outcomes.
Objects and methods
Subjects
A total of 586 COPD patients who were hospitalized in the Department of Respiratory Medicine of our hospital from January 2019 to December 2024 were selected as the research subjects. Inclusion criteria: (1) Patients met the diagnostic criteria for COPD established by the Global Initiative on Chronic Obstructive Pulmonary Disease (GOLD) 7 ; (2) Age ⩾60 years old; (4) The clinical data are complete, including medical history, symptoms, signs, laboratory tests, and imaging tests. Exclusion criteria: ① those combined with other lung malignant tumors; (2) Complicated with severe heart, liver, kidney, and other important organ failure; ③ Combined with human immuno-deficiency virus (HIV) infection; (4) Use of immuno-modulators in the past 3 months; (5) Qualified sputum or bronchoalveolar lavage fluid samples could not be obtained for pathogen detection.
Diagnostic criteria
The diagnostic basis of COPD combined with Nocardia infection: (1) The patient has a history of COPD; (2) The nocardia is found in sputum or bronchoalveolar lavage fluid smear, or cultured; (3) The clinical manifestations include increased fever, cough, expectoration, dyspnea, or accompanied by chest pain and coughing up blood, and lung lesions caused by infection of other pathogens were excluded.
Data collection
Collect general data of patients, including age, gender, cough, coughing up blood, and smoking; Laboratory indicators such as routine blood tests (white blood cell count, neutrophils, immune cells, and hemoglobin), C-reactive protein (CRP), procalcitonin (PCT), erythrocyte sedimentation rate (ESR), albumin; Pulmonary function indicators, such as forced expiratory volume in the first second (FEV), percentage of FEV to estimated value (FEV₁% pred), and FEV₁/FVC ratio. At the same time, collect imaging examination data such as chest X-rays and lung CT scans from patients, and record in detail the location, morphology, density, and other characteristics of lung lesions, such as the presence of nodules, cavities, consolidation shadows, and streaks. The indicators derived from whole blood cell count include mean platelet volume (MPV), lymphocyte to monocyte ratio (LMR); Tumor necrosis factor alpha (TNF alpha) and interleukin-6 (IL-6), among others.
Grouping method
According to the diagnosis results, the patients were divided into COPD combined with nocardial infection group (infection group) and simple COPD group (control group).
Statistical methods
The reporting of this study conforms to the STARD (Standards for Reporting Diagnostic Accuracy Studies) statement. 8 Data on general patient information, laboratory test indicators, pulmonary function parameters, and imaging findings were collected. SPSS 26.0 software (IBM Corporation, Armonk, NY, USA) and R software (version 4.0.3; R Foundation for Statistical Computing, Vienna, Austria) were used for data analysis. When the measurement data conforms to the normal distribution, it is expressed by the mean standard deviation (S), and the comparison between the two groups is made by independent sample t-test. When it does not conform to the normal distribution, it is expressed by the median (interquartile interval) (m (q, q)), and Mann–Whitney U test is used for comparison between groups. The counting data were expressed by the number of cases (percentage) (n(%)), and the comparison between groups was made by χ test or Fisher exact probability method. Single factor logistic regression analysis was used to screen the factors related to COPD complicated with Nocardia infection, and the factors of p < 0.05 in single factor analysis were included in multivariate logistic regression analysis to determine the independent risk factors. Based on the independent risk factors, a Nomogram diagnosis model is constructed and realized by “rms” package in R software. The bootstrap method is used for internal verification, sampling is repeated for 1000 times, C-index is calculated to evaluate the discrimination of the model, calibration curve is drawn to evaluate the calibration degree of the model, receiver operating characteristic curve (ROC) is drawn to evaluate the prediction accuracy of the model, and the area under the curve (AUC) and its 95% confidence interval (CI) are calculated. Inspection level α = 0.05.
Results
Comparison of general data between the two groups
A total of 586 patients with COPD were included, including 289 cases in the infected group and 297 cases in the noninfected group. There was no significant difference in age, gender, cough, smoking, white blood cell, neutrophil, CRP, PCT, ESR, and albumin between the two groups (p > 0.05). The infection group showed higher levels of coughing up blood, MPV, TNF alpha, and IL-6 compared to the control group, but lower levels of immune cells, hemoglobin, FVC, FEV₁, FEV₁% pred, FEV₁/FVC ratio, and LMR compared to the control group, and the differences were statistically significant (p < 0.05), as shown in Table 1.
Comparison of baseline data of patients in infected and noninfected groups.

Results of logistic regression analysis on factors of COPD combined with Nocardia infection
The results of univariate analysis showed that there were statistically significant differences in coughing up blood, immune cells, hemoglobin, FVC, FEV₁, FEV₁% pred, FEV₁/FVC ratio, MPV, LMR, TNF alpha, and IL-6 between the infected and uninfected groups (p < 0.05). The infection status was taken as the dependent variable (0 = No, 1 = Yes); the variable assignment method is shown in Table 2, and the results are shown in Table 3. Further multi-factor logistic regression analysis was performed with the factor p < 0.05 in single factor analysis as the covariate. The results showed that coughing up blood, immune cells, hemoglobin, FVC, FEV₁, FEV₁% pred, FEV₁/FVC ratio, MPV, LMR, TNF alpha, and IL-6 were related to COPD combined with Nocardia infection (p < 0.05). In the regression model, the tolerance of each variable was >0.1, VIF was <10, condition index was <30, and the proportion of variances of multiple covariates was >50% without the same feature value. Hence, there was no collinearity of each covariate, as shown in Table 4.
Variable assignment method.

Univariate logistic regression analysis of factors for chronic obstructive pulmonary disease combined with Nocardia infection.

Multi-factor logistic regression analysis of factors of COPD combined with Nocardia infection.
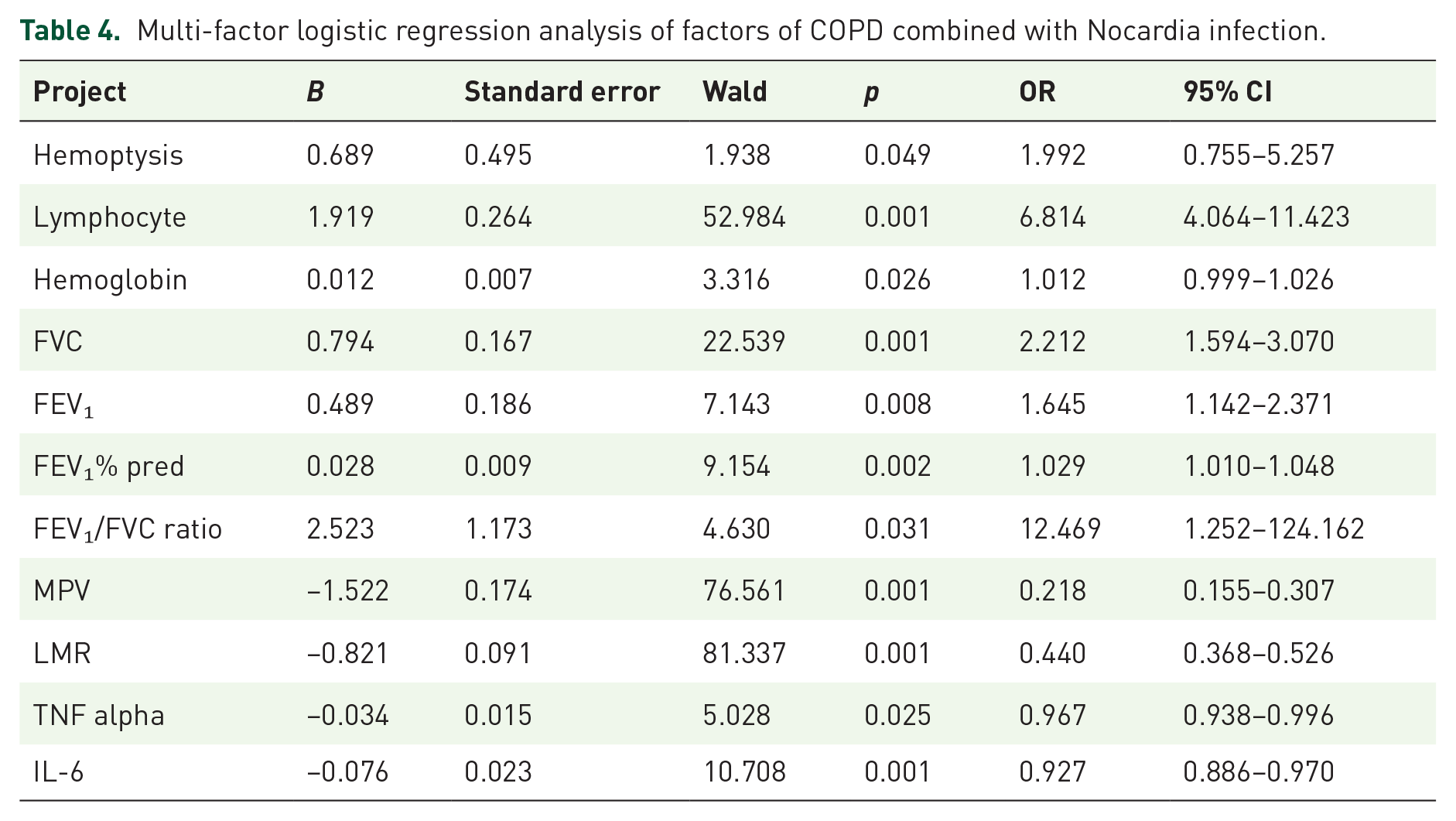
The establishment of COPD with Nocardia infection diagnosis model
The establishment of a diagnostic model for COPD combined with Nocardia infection was based on multivariate logistic regression analysis identifying independent risk factors. A prediction model of a diagnosis model of COPD combined with nocardia infection is constructed, each independent risk factor in the model is assigned, and the total score for predicting COPD combined with nocardia infection is calculated and reflected by the probability for predicting COPD combined with nocardia infection. The higher the total score is, the higher the accuracy for predicting the acceptance of COPD combined with nocardia infection is. See Figure 1.

Nomogram of a prediction model for chronic obstructive pulmonary disease complicated with nocardia infection.
Validation and evaluation of diagnostic model of COPD combined with Nocardia infection
Evaluation and validation of a predictive model for COPD combined with Nocardia infection in the infected group and the uninfected group, the C-index of the nomogram model was 0.895 and 0.829, respectively, and the mean absolute errors of the predicted value and the true value in the calibration curve were 0.127 and 0.170, respectively. The results of Hosmer–Lemeshow test were χ2 = 19.034, p = 0.014 and χ2 = 12.150, p = 0.144, respectively. The ROC curve was shown in the infected group and the uninfected group. The AUC of COPD combined with nocardial infection predicted by the nomogram model was 0.896 (95% CI: 0.866–0.927) and 0.830 (95% CI: 0.770–0.891), respectively. The sensitivity and specificity were 0.932, 0.737, and 0.904, 0.609, respectively. See Figure 2 for the calibration curve and Figure 3 for the ROC analysis.
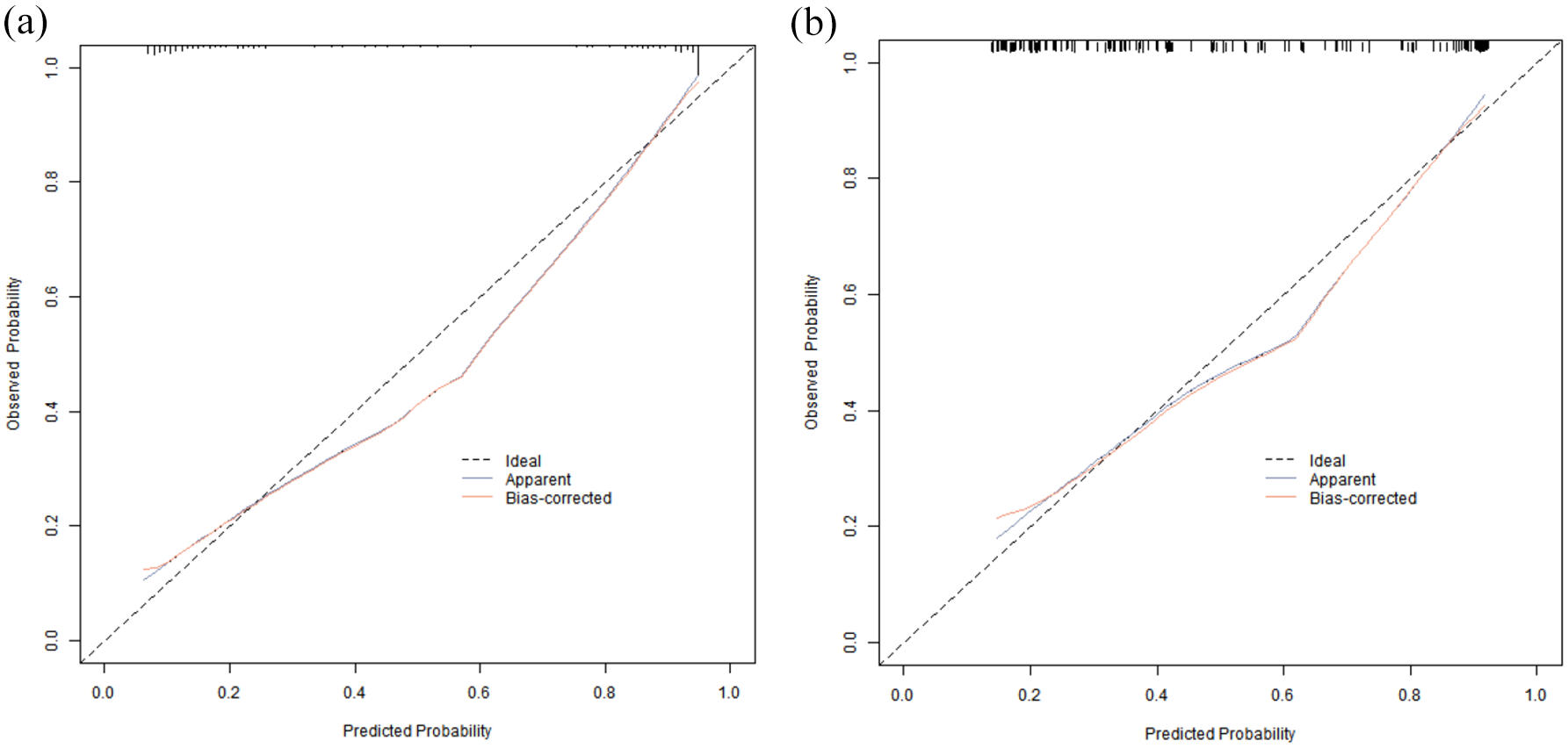
Calibration curve in the training set (a) and the verification set (b).
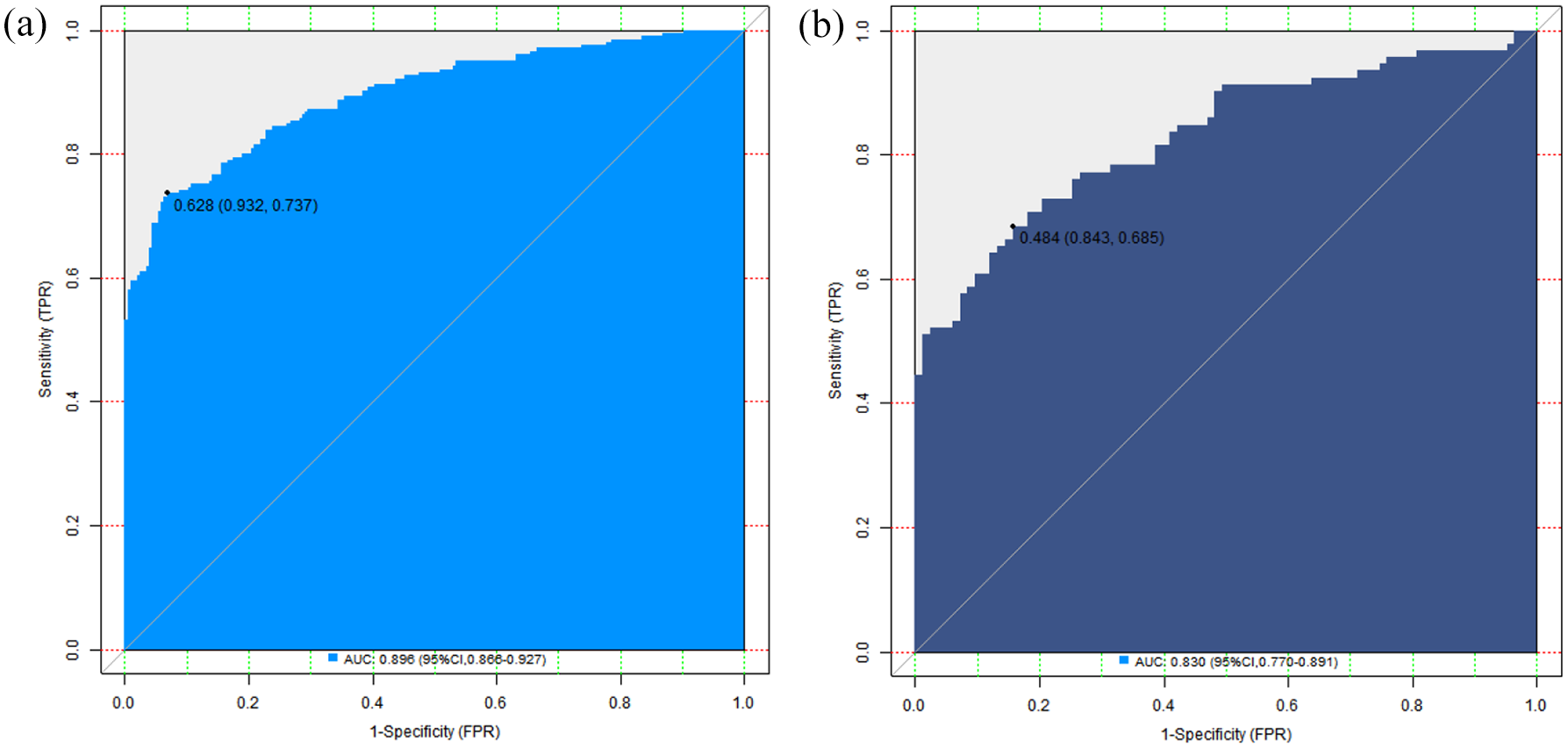
ROC curve in the training set (a) and the verification set (b).
Analysis of decision curve of nomogram prediction model
The decision curve analysis of the prediction model of COPD combined with Nocardia infection showed that when the threshold probability was about 0.05–0.92, the application of the nomogram model constructed in this study had more clinical benefits than the preoperative decision that all patients had COPD combined with Nocardia infection or none had COPD combined with Nocardia infection. See Figure 4.

Decision curve in the training set (a) and the verification set (b).
Discussion
The coexistence of chronic obstructive pulmonary disease (COPD) and Nocardia infection presents a formidable clinical challenge, with mortality rates exceeding 30% when diagnosis is delayed. The purpose of this study was to construct and verify a diagnostic model based on clinical characteristics and risk factors, to provide a scientific basis for early clinical diagnosis. The results have important clinical significance and value, and also provide a direction for future research. 9 Multivariate analysis identified coughing up blood, immune cells, hemoglobin, FVC, FEV, FEV₁% pred, and FEV₁/FVC ratio as independent risk factors, which was of great significance and worthy of further discussion.
Coughing up blood, as an independent risk factor, has special significance in COPD combined with Nocardia infection. Nocardial infection can cause damage to lung tissue, invade blood vessels, and destroy the normal structure of lung tissue, thus causing coughing up blood. 10 Compared with patients with simple COPD, combined with nocardial infection, pulmonary inflammation is more serious and complex, and the continuous stimulation of inflammation increases the fragility of the vascular wall and is more likely to rupture and bleed. 11 Hemoptysis often reflects a more advanced disease state; in severe cases, it may lead to reduced hemoglobin levels and worsening pulmonary function. When patients with COPD have coughing up blood, clinicians should be highly alert to the possibility of nocardia infection and timely conduct further examinations, such as pathogen detection in sputum or bronchoalveolar lavage fluid, in order to make an early diagnosis and take targeted treatment measures. 12 Immune cells play a key role in the body’s immune defense, and their quantitative changes are closely related to COPD combined with Nocardia infection. 13 The role of lymphocyte levels in predicting Nocardia infection among COPD patients warrants further investigation. This may be because patients with COPD often experience recurrent infections, chronic airway inflammation, and prolonged use of corticosteroids, all of which can compromise immune function. 14 When the number of immune cells is reduced, the immune defense ability of the body against pathogens such as Nocardia sp. is significantly reduced. As an opportunistic pathogen, Nocardia sp. is more likely to invade and proliferate when the body’s immune function is low, leading to the occurrence and aggravation of infection. 15 This suggests that the lymphocyte count is an important reference index for clinicians to assess the risk of infection in patients with COPD. For patients with decreased immune cells, infection monitoring and preventive measures should be strengthened, such as strengthening ward environmental management and improving the nutritional support of patients, in order to reduce the risk of nocardial infection. 16 Changes in hemoglobin levels were also associated with COPD with nocardial infection. 17 In this study, we found that decreased hemoglobin was one of the risk factors for infection. In the infected state, the body is under stress, which can interfere with iron metabolism and inhibit red blood cell production. 18 At the same time, nocardial infection may lead to chronic blood loss that further aggravates anemia. Anemia will make the body’s oxygen-carrying capacity decreased, and tissues and organs cannot get enough oxygen supply, thus affecting the body’s immune function and repair ability, making the infection more difficult to control. 19 In patients with COPD complicated with nocardial infection, correction of anemia not only helps to improve the general condition of the patient, but may also enhance the body’s anti-infection ability. Clinicians can correct anemia and increase the hemoglobin level of patients by supplementing iron, erythropoietin, and other means, thereby improving the prognosis of patients. The FVC, FEV, FEV₁% pred, and FEV₁/FVC ratios reflected the patient’s pulmonary function, and abnormalities in these indicators were strongly associated with COPD with nocardial infection. 20 Patients with COPD have limited airflow and gradually decreased lung function. 21 Nocardial infection further aggravates lung inflammation, leading to airway stenosis, obstruction, and damage to lung tissue, further exacerbating lung function. For example, FEV is an important indicator of the severity of COPD, and a lower FEV means a more serious patient and a higher risk of nocardial infection. At the same time, the infection in turn will affect the recovery of lung function, forming a vicious circle. Therefore, in clinical treatment, in addition to active anti-infection treatment, attention should be paid to the maintenance and improvement of lung function, such as through rehabilitation training, the rational use of bronchodilators, to break the vicious circle, and improve the quality of life and survival rate of patients. 22 In addition to the factors mentioned earlier, such as MPV, LMR, TNF alpha, and interleukin-6, these indicators are also related to the coexistence of COPD and Nocardia infection. MPV can reflect changes in platelet volume. When COPD is complicated with Nocardia infection, inflammation in the body stimulates platelets, causing changes in MPV levels. If MPV shows abnormalities, it may indicate an increased risk of infection with Nocardia in patients. LMR comprehensively reflects the immune status of the body. A decrease in LMR indicates that the body’s immune balance has been disrupted and immune function has deteriorated, making it easier for Nocardia to invade the body and cause infection. Tumor necrosis factor alpha and interleukin-6 are both inflammatory markers in the body. When patients with COPD are complicated with Nocardia infection, the body’s inflammatory response will be enhanced, and the levels of these two indicators will also increase. By monitoring them, doctors can have a clearer understanding of the inflammation in patients’ bodies, determine whether the infection is caused by Nocardia, and assess the severity of the condition. So, these indicators can help doctors better determine whether patients with COPD are infected with Nocardia, which is very helpful for diagnosis and treatment.
The Nomogram model constructed in this study performed well in both the infected and uninfected groups, with high discrimination and calibration, providing a powerful tool for early clinical diagnosis. Chest X-rays and lung CT scans are widely used in the diagnosis and treatment of patients with chronic lung diseases. Exploring the predictive value of specific pulmonary imaging patterns is of great significance in the diagnosis of COPD combined with Nocardia infection. Nocardia infection may present unique imaging features, such as nodules with irregular edges and inflammatory exudate around them. The formation of voids may have certain characteristics, such as wall thickness and smoothness, which may differ from voids caused by other pathogen infections. The distribution and density characteristics of pulmonary consolidation shadows may also provide clues for diagnosis. By conducting in-depth analysis of these imaging features and incorporating them into the diagnostic model, it is expected to further improve the predictive accuracy of the model. Future research can focus on this aspect by collecting a large amount of imaging data from patients with COPD complicated with Nocardia infection, combining clinical features and other examination indicators to construct more comprehensive prediction models, and providing stronger support for early clinical diagnosis. The model comprehensively considered multiple independent risk factors, and could accurately predict the risk of COPD patients combined with Nocardia infection. According to the specific situation of patients, clinicians can calculate the probability of infection through the model, so as to develop a more reasonable diagnosis and treatment plan. For high-risk patients, monitoring can be strengthened and anti-infection treatment can be carried out as soon as possible. For low-risk patients, unnecessary examinations and treatments can be appropriately reduced to avoid the waste of medical resources. Nonetheless, due to the absence of large-scale, multicenter studies, the clinical utility of this model requires further validation. The lack of external validation poses a serious obstacle to the widespread application and promotion of the model. There are significant differences in patient populations across different regions and hospitals, with variations in disease spectrum, treatment methods, testing methods, and other aspects. To conduct external verification, it is necessary to collaborate deeply with multiple medical institutions and collect a large amount of external data. However, in the actual operation process, there are many challenges that are difficult to overcome. On the one hand, the data formats and quality of different medical institutions vary greatly, requiring complex data standardization processing to ensure data consistency and comparability; On the other hand, coordinating and communicating with multiple medical institutions is not an easy task, as it involves the interests of all parties, differences in work processes, and other issues, requiring a lot of time and energy. In addition, the limited research time and resources also greatly limit the development of external verification. Although the internal validation results are relatively satisfactory, external validation is crucial for evaluating the applicability and accuracy of the model in different clinical environments, and is an indispensable link for the model to be widely applied in clinical settings.
Based on the above limitations, future research can adopt the following strategies to further validate and improve the model. In terms of data collection, actively carry out multicenter data collection work. Collaborate with respiratory departments from hospitals of different regions and levels to establish unified data collection standards and standardized processes, ensuring that the collected data has broad representativeness. Through multicenter collaboration, diverse patient data can be obtained to more comprehensively evaluate the performance of the model in different clinical environments. Participate in or conduct collaborative registration research on research collaboration models. Establish a specialized registration platform for COPD patients with concurrent Nocardia infection, and all participating medical institutions will include eligible patient information into the platform for long-term follow-up and data updates. This approach not only expands the sample size, but also enables dynamic observation of patients, which helps to more accurately validate the predictive value of the model, improve its universality, and provide more reliable basis for early diagnosis and treatment of COPD complicated with Nocardia infection. The diagnostic model established in this study provides an important reference for the early diagnosis of COPD combined with Nocardia infection, and the identified independent risk factors have important guiding significance for clinical practice. Based on this, in clinical practice, the column chart model can be applied to patients with COPD, especially hospitalized patients aged 60 and above, during the initial assessment. Medical staff collect data on patients’ coughing up blood, lymphocyte count, hemoglobin level, FVC, FEV₁, FEV₁% pred, FEV₁/FVC ratio, etc., and input them into the model to calculate the probability of infection. If the calculated probability of infection is high, high vigilance should be exercised against Nocardia infection, and pathogen testing of sputum or bronchoalveolar lavage fluid should be carried out in a timely manner to facilitate early diagnosis and targeted treatment. If the probability of infection is low, diagnosis and treatment can be carried out according to conventional procedures, but attention should still be paid to changes in the condition. By integrating this column chart into the evaluation process for patients with COPD, it is expected to improve the early identification ability of COPD complicated with Nocardia infection and improve patient prognosis.
Conclusion
This study successfully established and validated a diagnostic model for COPD complicated by Nocardia infection based on clinical characteristics and risk factors. Multivariate logistic analysis identified coughing up blood, lymphocyte count, hemoglobin, FVC, FEV₁, FEV₁% pred, FEV₁/FVC ratio, MPV, LMR, TNF α, and IL-6 as independent risk factors. The constructed nomogram model demonstrated excellent performance in both the infected and noninfected groups, exhibiting high discriminative ability, calibration, and predictive accuracy, thereby providing a robust tool for the early clinical diagnosis of COPD complicated by Nocardia infection. However, this study only conducted internal validation and has not yet performed external validation, which limits the broader application and generalizability of the model. Future efforts should focus on further validating the model through multicenter data collection and collaborative registry studies to provide more reliable evidence for the early diagnosis and treatment of COPD complicated by Nocardia infection. In clinical practice, for eligible COPD patients, this nomogram model can be applied to assess infection risk, guide diagnostic and therapeutic decisions, enhance early identification of COPD complicated by Nocardia infection, and improve patient prognosis.
