Abstract
Background:
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease that affects motor neurons, resulting in muscle weakness, loss of function, and ultimately death due to respiratory failure. Due to the lethal prognosis of ALS, respiratory muscle training has been proposed as a potentially beneficial intervention.
Objectives:
To systematically review the efficacy of respiratory muscle training on lung function and respiratory muscle strength in ALS patients.
Design:
A systematic review and meta-analysis of randomized controlled trials.
Data sources and methods:
Articles published in PubMed, PEDro, Scopus, and Web of Science databases up to July 2024. The Preferred Reporting Items for Systematic reviews and Meta-Analyses 2020 statement guideline was followed. Included studies had (1) ALS patients, (2) respiratory muscle training, (3) physical exercise, usual care or no intervention were provided as a comparison group, (4) assessments of lung function, respiratory muscle strength, quality of life, survival, fatigue, and functional capacity outcome measures, and (5) a randomized controlled trial design. Methodological quality was analyzed using the PEDro scale, and risk of bias with the Cochrane Collaboration Risk of Bias Tool. Meta-analyses were performed with Review Manager software.
Results:
Five randomized controlled trials with 170 participants were included. The results showed that respiratory muscle training improved muscle strength, particularly maximum expiratory and inspiratory pressures. One study suggested inspiratory muscle training as a survival predictor in ALS patients. No significant effects were observed in forced vital capacity or quality of life. No adverse effects were reported.
Conclusion:
Respiratory muscle training improves ventilatory function, particularly respiratory muscle strength, in people with ALS. While evidence is limited, it shows promise as an adjuvant therapy to enhance quality of life and survival. It has been registered in the PROSPERO (CRD42024568235).
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a progressive and disabling neurodegenerative disease that involves the loss of cortical, brainstem, and spinal motor neurons.1 –3 It is characterized by rapid muscle weakness affecting axial muscles and limbs, leading to loss of ambulation, dysphagia, dysarthria, respiratory dysfunction, and death due to respiratory failure, mainly from diaphragm weakness.1,3,4 The mean survival time from symptom manifestation is 3–5 years, although some patients with slower disease progression may live longer.2,5,6
ALS is clinically classified according to symptom onset: spinal, initially affecting the upper or lower limbs; and bulbar, initially affecting head and neck regions. Spinal onset is more prevalent than bulbar onset (58% vs 82%).3,7,8 From the first stage of the disease, progressive muscle weakness may appear asymmetrically in the limbs, reducing functional performance and significantly impacting patients’ quality of life.3,9
As the disease progresses, respiratory muscle weakness becomes a significant complication, reducing lung volume, causing ineffective cough, and leading to secretion accumulation and recurrent respiratory infections.1,2,10 Respiratory insufficiency primarily arises from weakened inspiratory muscles due to motor neuron loss in the intercostal and axial musculature, leading to a restrictive pattern. This may also reduce functional capacity in these patients, so respiratory muscle training (RMT) may improve their quality of life.2,10,11
Respiratory dysfunction becomes a terminal event for most patients, 2 with a decrease in total lung capacity (TLC), vital capacity, and functional residual capacity.2,12 Diaphragmatic weakness due to phrenic nerve degeneration leads to additional complications, impairing inspiratory capacity and reducing the peak cough flow required to clear secretions. If glottic insufficiency is present, adequate pressures are not achieved to develop an effective cough, while weakened expiratory muscles also limit secretion clearance.2,13 –15 Therefore, physiotherapeutic interventions targeting inspiratory and expiratory muscle weakness and secretion management are essential components of ALS treatment.2,15,16
Due to the prognosis and lethal nature of ALS, treatment is based on multidisciplinary, personalized care.3,17 Physiotherapy plays a key role, aimed at symptom management, maximizing function, and enhancing quality of life.3,10,18,19 Breathing exercises in physical therapy aim to maintain lung function and prevent complications. Studies suggest potential benefits for both neurological and cardiopulmonary conditions.10,20,21 Though few studies have examined the efficacy of RMT in neurodegenerative diseases, existing research indicates that RMT may increase respiratory muscle strength, reduce complications, and lower hospitalization rates in these patients.10,22,23 Reyes et al. 24 conducted a systematic review of RMT in patients with multiple sclerosis and Parkinson’s disease, concluding that this training can improve respiratory strength, although functional capacity was not evaluated.10,24
Several systematic reviews have investigated RMT and found increases in respiratory muscle strength; however, these studies were either not conducted on patients with ALS, 25 did not include randomized controlled trials (RCTs),25,26 or did not focus exclusively on this population. 10 This highlights the need for further research to conclusively assess the efficacy of RMT as a supportive intervention for respiratory management in this disease.
Therefore, the aim of this review is to evaluate the effect of RMT on lung function and respiratory muscle strength in ALS patients.
Methods
Design
A systematic review and meta-analysis was conducted taking into account the recommendations of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement. 27 It has been registered in the PROSPERO International Prospective Registry of Systematic Reviews (CRD42024568235).
Data sources and searches
An electronic search for articles was carried out using PubMed, PEDro, Scopus, and Web of Science databases between June and July 2024, with no restrictions on language or publication state. The descriptors used were: “amyotrophic lateral sclerosis,” “respiratory training,” “inspiratory muscle training,” and “breathing exercises,” joined by the Boolean operator “AND” (see Table 1).
Search strategy and article selection.

Source: Own elaboration.
Data collection and analysis
Inclusion criteria: studies assessing the effect of respiratory physiotherapy were included according to the following criteria:
Participants: patients with ALS, without limitation by age or nationality.
Intervention: use of RMT, without combination with physical exercise.
Comparison: control groups (CGs) in which physical exercise, usual care or no intervention was performed.
Outcomes: assessments of lung function, inspiratory and expiratory muscle strength, fatigue, quality of life, functional capacity, survival, and adverse events.
Study design: RCTs.
Exclusion criteria: quasi-randomized clinical trials, quasi-experimental trials, observational studies, pilot studies, preliminary studies, or study protocols. Articles with only one intervention arm or performing acute interventions (single session). Interventions related to Jacobson’s relaxation exercises, high-intensity exercise, aquatic exercise, or martial arts.
Study selection, data extraction, and assessment of methodological quality and risk of bias
Two independent authors (MJBI and PRP) reviewed the titles and abstracts of the studies included in the searches, removed duplicates, and identified potentially relevant articles. The authors selected the titles and abstracts of references from the searches that met the inclusion and exclusion criteria. They obtained the full texts of studies deemed relevant to complete the review. In cases of doubt, they consulted a third researcher (RLL).
Author MJBI extracted the following characteristics from each included study whenever possible: number of participants, participant characteristics, interventions, duration of intervention, and outcomes. All extracted characteristics were presented in tables.
The methodological quality of the studies was assessed using the PEDro scale, 33 with low quality considered below 6 points, and the Cochrane Collaboration Risk of Bias Tool was used to assess the risk of bias.34,35
Meta-analysis
Finally, three meta-analyses of the main outcome measures were conducted with the Review Manager software (RevMan version 5.4.1), including three to four of the five RCTs according to outcome. The I2 statistic was used to determine the degree of heterogeneity: 25% = low, 50% = medium, and 75% = high heterogeneity. All the results included were the data collected on maximum expiratory pressure (MEP), maximum inspiratory pressure (MIP), and forced vital capacity (FVC). The effect size was calculated using the mean difference between post-intervention and pre-intervention values for each study, with a confidence interval of 95%. Effects plots (forest plots) were generated to illustrate the overall effect of the interventions on each outcome. To calculate the standard deviation of the difference (SDdifference), we used the following formula:

In this analysis, we assumed a correlation coefficient R = 0.5 to reflect a moderate correlation between pre- and post-intervention values, as respiratory function parameters are expected to exhibit some variability over time. All calculations were performed using data reported in the included studies, and the results were synthesized using a random-effects model to account for variability among studies, considering potential differences in populations, interventions, and study designs.
Results
The database searches provided 178 records. After removing duplicates, 81 records were analyzed, excluding 76 according to the inclusion and exclusion criteria. Finally, five articles were selected (Figure 1).

Flow diagram of electronic database searches. Methodology of article selection.
Five RCTs were included, with a total of 170 participants. Table 2 shows the characteristics of each included RCT.
Summary of the characteristics of the selected studies (results table).

Source: Own elaboration.
6MWT, 6-minute walk test; ALS, amyotrophic lateral sclerosis; ALSFRS-R, Amyotrophic Lateral Sclerosis Functional Rating Scale-Revised; CG, control group; DIGEST, Dynamic Imaging Grade of Swallowing Toxicity; EG, experimental group; EMT, expiratory muscle training; FVC, forced vital capacity; IMT, inspiratory muscle training; MEP, maximum expiratory pressure; MIP, maximum inspiratory pressure; MVV, maximal voluntary ventilation; NIV, noninvasive mechanical ventilation; PIF, peak inspiratory flow; SNIP, sniff nasal inspiratory pressure; VC, vital capacity.
Participants
The age of participants ranged from 38 to 75 years, with a mean age of 53 to 63 years.28 –32 Regarding sex, all studies had a higher percentage of males (58.8%–69.2%) than females (30.8%–41.2%).28 –32
All five included trials reported on disease onset, with the majority of participants having spinal onset (72.9%–84.6%), while the remainder had bulbar onset (15.4%–24%).28 –32 Only the study by Plowman et al. 28 reported that 4.2% of participants had mixed disease onset.
Disease duration was quite variable, ranging from 11.4 months on average in the Pinto et al. 31 study, to 34 months on average in the Pinto and de Carvalho 30 study. Only the study by Plowman et al. 32 did not report on the duration of illness of their participants.
Three of the five studies reported on the use of tracheostomy or noninvasive ventilation before the start of the trial.28,29,32 In two of these studies, participants did not use tracheostomy or noninvasive ventilation,28,32 but in the study by Cheah et al., 29 one experimental group (EG) participant used noninvasive ventilation.
Type of intervention
All trials had a single comparison group that performed the same intervention as the CG, but in a simulated manner, with no or minimal loading.28,29,31,32 An exception was the study by Pinto and de Carvalho, 30 in which the comparison CG consisted of historical controls who did not perform the intervention (inspiratory muscle training). In the study by Pinto et al., 31 the CG was a delayed-start parallel group that performed sham inspiratory muscle training (IMT) for the first 4 months and then received the same intervention as the EG for the subsequent 4 months.
Three of the five trials performed IMT.29 –31 In the study by Plowman et al., 28 expiratory muscle training was performed, while the study by Plowman et al. 32 included IMT and expiratory muscle training. The training load with the device varied: two studies used a load of 30%–40% of the MIP,30,31 Plowman et al. 28 used 50% of the MEP, and Plowman et al. (2023) 32 applied a load of 30% of both MIP and MEP. In the study by Cheah et al., 29 the load progressed from 15% of MIP in the first week to 60% of MIP in the fourth week.
Only two studies reported the number of repetitions, sets, and rests to be performed by the participants. In both studies, the protocol consisted of five sets of five repetitions each, with rests every five breaths.28,32 Pinto et al. 31 specified that participants performed 14–16 breaths per minute.
Three studies conducted 10-min sessions,29 –31 Plowman et al. 28 conducted 20-minute sessions, and Plowman et al. 32 conducted 30-min sessions.
Two studies conducted a single daily session, 5 days a week,28,32 while two other studies conducted two daily sessions, every day of the week.30,31 Cheah et al. 29 conducted three daily sessions, every day of the week.
In terms of the duration of the interventions, the shortest study was that of Plowman et al., 28 which lasted 8 weeks (2 months). Two studies had a duration of 12 weeks (3 months),29,32 although the study by Cheah et al. 29 included an additional 8-week (2-month) post-intervention period to observe the effects of ceasing training. The study by Pinto et al. 31 had a duration of 32 weeks (8 months), and the study by Pinto and de Carvalho 30 study had a duration ranging from 8 to 32 months, due to the use of historical controls.
Outcome measures
Regarding lung function, four of the five included studies that assessed FVC28,29,31,32; three studies assessed peak expiratory flow (PEF),28,31,32 and two assessed peak inspiratory flow (PIF).28,32 Additionally, two studies assessed sniff nasal inspiratory pressure29,31 Other measures, such as TLC and vital capacity, were assessed in the study by Cheah et al., 29 and maximum voluntary ventilation was assessed in the study by Pinto et al. 31
Plowman et al.28,32 assessed PEF and PIF through cough spirometry using an oral pneumotachograph.
Four studies assessed respiratory muscle strength, including both inspiratory using MIP and expiratory using MEP. These studies also evaluated the effect of the intervention on participation and survival using the ALS Functional Rating Scale-Revised (ALSFRS-R).28,29,31,32
Two studies assessed health-related quality of life.29,31 The study by Cheah et al. 29 used the Short Form-36, while Pinto et al. 31 used the EuroQol-5D questionnaire.
Two studies assessed the neurophysiological index of the ulnar nerve.29,31
Two studies assessed swallowing in participants.28,32 In the study by Plowman et al., 28 three measures of swallowing were used: global swallowing function assessed using the validated Dynamic Imaging Grade of Swallowing Toxicity (DIGEST) and its efficiency subscale from a videofluoroscopic swallowing study, airway safety measured using the validated Penetration-Aspiration Scale (PAS), and daily oral intake evaluated using the Functional Oral Intake Scale (FOIS). Additionally, swallowing function was reported by patients using the Eating Assessment Tool-10 (EAT-10). In the study by Plowman et al., 32 swallowing was assessed using the Oral Ingestion Status, categorizing it as null if no food or liquid was consumed by mouth, and oral feeding if any food or liquid was consumed.
The study by Cheah et al. 29 also included measures of functional exercise capacity, which was assessed through the 6-Minute Walk Test (6-MWT), and hand grip strength.
Pinto et al. 31 also collected the following outcome measures: arterial blood gas, Nocturnal Pulse Oximetry, dyspnoea via a Visual Analogue Scale, fatigue via the Fatigue Severity Scale and the Borg scale during the intervention, depression via the Hamilton Rating Scale for Depression, daytime sleepiness via the Epworth Daytime Sleepiness Scale, and the patient’s level of independence via the Functional Independence Measurement scale.
Pinto and de Carvalho 30 assessed participant survival time and factors influencing survival, comparing EG with CG according to gender, age at presentation, bulbar or spinal onset, delay in diagnosis, ALSFRS-R score, and respiratory tests such as FVC and mean phrenic nerve response amplitude.
Effect of interventions
Regarding lung function, four studies assessed FVC28,29,31,32 and none found significant differences.
Likewise, three studies assessed PEF28,31,32 without finding significant differences either.
Two studies assessed PIF.28,32 The study by Plowman et al. 28 found no significant differences, but Plowman et al. 32 reported an improvement in the EG and a decrease in the CG, resulting in a significant difference between groups in favor of EG (p = 0.02).
Two studies assessed sniff nasal inspiratory pressure.29,31 Neither study found significant differences during the intervention. However, Cheah et al. 29 found a significant decrease in both intervention groups when the intervention was withdrawn (p < 0.05).
Cheah et al. 29 also assessed other measures of lung function, such as TLC and vital capacity, which revealed a significant decrease at the cessation of the intervention (p < 0.05). Pinto et al. 31 assessed maximum voluntary ventilation and found a significant improvement in the EG in both the sitting (p = 0.017) and lying (p = 0.042) positions at the end of the intervention (first 4 months).
Four studies assessed expiratory muscle strength using MEP.28,29,31,32 The results of the studies by Plowman et al.28,32 concurred, showing a significant improvement in the EG pre- to post-intervention (p < 0.05). Additionally, the Plowman et al. 32 study revealed a between-group difference in favor of the EG (p < 0.05). The study by Cheah et al. 29 observed a significant decrease in both intervention groups at the cessation of the intervention (p < 0.05), while the study by Pinto et al. 31 found no significant difference.
Three studies assessed inspiratory muscle strength using MIP.29,31,32 The study by Cheah et al. 29 reported a significant decrease at the cessation of the intervention in both groups (p < 0.05). The studies by Pinto et al. 31 and Plowman et al. 32 found no significant differences.
Four studies assessed the effect of the intervention on participation and survival using the ALSFRS-R.28,29,31,32 The study by Plowman et al. 32 was the only one to show a significantly lower score for the EG compared to the CG on the bulbar subscale (p = 0.02).
Two studies assessed health-related quality of life,29,31 and neither showed significant differences.
Two studies assessed the neurophysiological index of the ulnar nerve.29,31 In the study by Cheah et al., 29 there were no significant differences, but the neurophysiological index in the EG was higher than in the CG throughout the study. Pinto et al. 31 reported a decrease in neurophysiological index in both intervention groups throughout the study.
Two studies measured swallowing function in participants.28,32 In the study by Plowman et al., 28 significant differences were observed between treatment groups in global swallowing function and Dynamic Imaging Grade of Swallowing Toxicity efficiency score (p = 0.02), with EG patients maintaining their function while CG patients worsened. There was also an improvement, although not significant, in functional oral intake (Functional Oral Intake Scale scores), with the EG showing a 14.4% improvement and the CG experiencing an 11.8% worsening over the 2-month period. In the study by Plowman et al., 32 3 individuals in the EG (13%) and 8 in the CG (36%) stopped eating by mouth within 12 months (p = 0.07).
In the study by Cheah et al., 29 a significant reduction in the 6-MWT was observed after the cessation of the intervention in both groups (p = 0.01). This study also assessed manual grip strength, finding no significant differences; however, the EG scores were higher than those of the CG throughout the study, and there was a significant decrease in grip strength at the cessation of the intervention period (p < 0.01).
In terms of Nocturnal Pulse Oximetry, Pinto et al. 31 found a smaller decrease in oxygen saturation in the EG compared to the CG during the first four months of the study, which was the intervention period for the EG.
Pinto et al. 31 reported no significant differences in dyspnoea across the Visual Analogue Scale, Fatigue Severity Scale, Hamilton Rating Scale for Depression, Epworth Daytime Sleepiness Scale, and Functional Independence Measurement scales. However, they did find more favorable progress in the EG in the nonintervention period (4–8 months) on the Borg scale for dyspnoea.
Pinto and de Carvalho 30 found that survival time was longer in the EG compared to the CG (p < 0.001) and identified that IMT performance (p = 0.032), phrenic nerve response amplitude (p = 0.015), and male gender (p = 0.004) were significant predictors of mortality at disease onset.
Three studies collected on adverse effects,29,31,32 and none reported the appearance of serious adverse effects in participants.
Methodological quality and risk of bias of the included studies
A summary of the methodological quality of the five included studies, assessed using the PEDro scale, is presented in Table 3. The study by Pinto and de Carvalho (2013) scored 7 points on the PEDro scale, 30 two studies scored 8 points,28,31 and two studies scored 10 points.29,32 Overall, the trials demonstrated high methodological quality, with three studies rated as moderate quality (7–8 points) and two studies rated as high quality (10 points), indicating a reduced risk of bias.
Evaluation of the methodological quality of the articles included.

Source: Own elaboration.
The first item (Specified selection criteria) does not participate in the total score count.
1, meets PEDro scoring criteria; 0, does not meet PEDro scoring criteria.
Figure 2 shows the risk of bias of the studies included in the quantitative analysis, assessed using the Cochrane Risk of Bias tool. All included studies had low bias arising from the randomization process, due to deviations from intended interventions and in selection of the reported result.28,29,31,32 In 75% (three studies) of the articles, the bias due to missing outcome data were well described, with a low risk of bias in this analysis.28,29,31 The bias in measurement of the outcomes was well reported in 50% (two studies) of the articles29,32 and the other 50% presented some concerns in this bias.28,31

Meta-analysis
A meta-analysis was performed on respiratory muscle strength in three RCTs,28,31,32 measured via MEP and comparing RMT with CG (Figure 3). The results were statistically significant in favor of the EG (RMT intervention; p = 0.02). That is, the MEP in the group that performed RMT was superior to the CG in patients with ALS (SMD: 19.53; IC95%: 2.58–36.48; I2: 0%). Regarding heterogeneity, the I2 statistic was 0%, suggesting no apparent heterogeneity among the studies. However, the 95% confidence interval (CI) for I2 was wide [0%–95.96%], indicating that substantial heterogeneity cannot be ruled out.
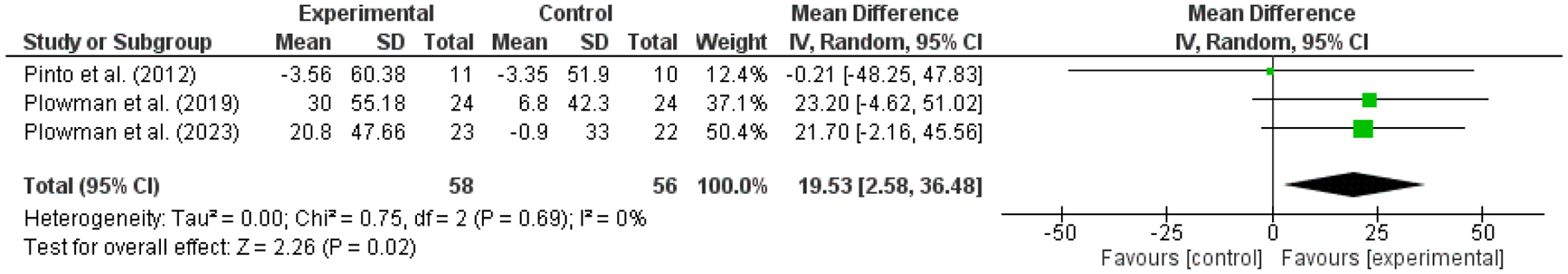
Also, three of the included studies29,31,32 compared respiratory muscle strength in RMT group versus CG, measured via MIP (Figure 4). The results of this meta-analysis were statistically significant (p = 0.010), so there is a difference in favor of RMT intervention (SMD: 13.96; IC95%: 3.35–24.56; I2: 30%). The level of heterogeneity across the studies was moderate (I2 = 30%). However, the 95% CI for I2 ranged from 0% to 98.22%, indicating substantial uncertainty. This wide range suggests that while moderate heterogeneity was observed, it is also possible that there was little to no heterogeneity, or conversely, substantial heterogeneity.

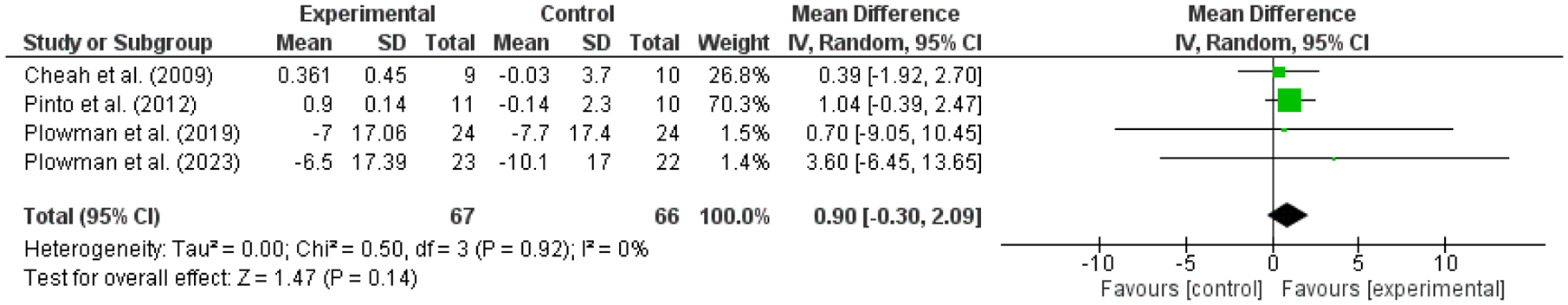
Another meta-analysis was conducted on FVC in four of the included articles 28, 29, 31, 32, comparing RMT group with CG (Figure 5). The results of this meta-analysis do not indicate a statistically significant differences between the EG and CG (p = 0.14) (SMD: 0.90; IC95%: −0.30–2.09; I2: 0%). The included samples appeared to be similar across studies, with no observed heterogeneity (I2 = 0%). However, the 95% CI for I2 was [0%–57.02%], indicating that although no heterogeneity was detected, the possibility of low to moderate heterogeneity cannot be excluded.
Discussion
This systematic review and meta-analysis of RCTs evaluated the efficacy of RMT in ALS patients and showed significant improvements in respiratory muscle strength, particularly in measures such as MEP and MIP. These findings align with the review’s objective to explore the impact of RMT on pulmonary function and muscle strength, as well as with previous studies.10,22 –24 However, no significant improvements were observed in FVC, consistent with prior research indicating that increased muscle strength does not necessarily translate to improved lung volumes.10,20,36
The improvement in respiratory muscle strength suggests that RMT may help ALS patients achieve better respiratory conditions, enabling them to generate sufficient strength to expel secretions and clear their airways. Improved expiratory muscle strength, as evidenced by increased MEP, is associated with better PEF and enhanced forced expiratory volume in the first second.10,37 These improvements are particularly relevant in ALS, where progressive respiratory muscle weakness leads to reduced lung volumes, ineffective cough, secretion accumulation, and recurrent respiratory infections.1,2,10 By improving expiratory muscle strength, RMT may enhance cough efficiency and secretion clearance airway,2,13,38 reducing the risk of complications such as upper airway infections and bronchoaspiration.1,10,39 Studies in healthy, elderly people have shown that RMT improves peak expiratory pressure and, as a consequence, cough function.24,40
Qualitative analysis revealed that while the intervention yielded benefits in some outcome measures, the overall effects were not significant across all studies. Subgroup findings included increases in MEP,28,32 PIF, 32 and maximum voluntary ventilation, 31 which reflect strengthened respiratory muscles and support the potential for clinical benefits of RMT. Notably, improvements in maximum voluntary ventilation suggest enhanced respiratory capacity that may contribute to a better quality of life for ALS patients. These benefits are critical in managing a condition where respiratory dysfunction is the terminal event for most patients.2,31
RMT aims to improve respiratory muscle strength, leading to increased chest strength and stability, and preventing respiratory fatigue.10,41 This has been proved even in patients with dyspnoea and previous muscle weakness,10,42 and our findings suggest that similar benefits may be achieved in neurodegenerative diseases such as ALS. Although ALS is a progressive neurodegenerative disease, and patients’ conditions worsen over time, the observed improvements underscore the importance of RMT as a therapeutic intervention.
One of the most important findings of the included studies was the worsening on several measures after cessation of interventions found in the study by Cheah et al. 29 These findings are consistent with previous studies suggesting that, while benefits of RMT may not always reach statistical significance, they may still yield clinically relevant improvements in respiratory function10,22,26 and, the most important thing, slow the progression of the restrictive respiratory impairment in patients with ALS. 29 Due to the chronic and degenerative condition of the disease, we suggest patients to continue treatment in order to maintain the benefits obtained and delay the deterioration of the disease as long as possible.
Regarding quality of life and survival, most studies did not report significant improvements in health-related quality of life. However, Pinto and de Carvalho 30 highlighted that IMT could independently predict survival, aligning with research suggesting that RMT could positively impact survival and functionality in these patients. 10 Conversely, studies such as Pinto et al. 31 did not observe a protective effect on the ALSFRS-R, but reported that early respiratory training could help prevent respiratory symptoms, potentially enhancing quality of life over time, which has also been described by other studies.24,26
Additional outcomes related to swallowing and cough function were highlighted in studies by Plowman et al.,28,32 which found improvements in global swallowing function and cough efficiency. These findings are consistent with existing literature suggesting that RMT can maintain critical functions like swallowing and coughing, essential for respiratory health in patients with progressive neuromuscular disorders.10,22,24 Studies has shown that impaired expiratory muscle contraction also reduces the ability to cough and clear secretions,2,14,15 so the improvement in MEP could be related to the improvement in cough efficiency generating enough powerful compression.
Importantly, no studies reported serious adverse effects, supporting RMT as a safe intervention for patients with ALS. This further emphasizes its potential as a valuable component of multidisciplinary ALS care, targeting inspiratory and expiratory muscle weakness and secretion management to improve respiratory function and potentially prolong survival.
Limitations and future research
Despite these encouraging findings, limitations in the current literature include the small sample sizes and the inclusion of only five RCTs, totaling 170 participants, which limits the generalizability of the results. Furthermore, it should be noted that the meta-analyses conducted involve only three to four studies, and while the methodological quality is high and the risk of bias is low, the results should be interpreted with caution. The limited number of studies may impact the robustness of the findings, restrict the ability to perform sensitivity analyses, and hinder the exploration of potential sources of heterogeneity, particularly in the presence of clinical or methodological differences between the included studies. While some meta-analyses showed no heterogeneity (I2 = 0%), the wide CIs around these values indicate that substantial heterogeneity cannot be ruled out. This is particularly relevant in analyses where the 95% CI for I2 extended up to 96% or even 98%, highlighting the imprecision inherent in small meta-analyses. Therefore, results should be interpreted with caution, considering this potential variability among studies even when heterogeneity is not statistically detected.
However, it is important to consider that ALS is a rare disease and with low prevalence, so the number of participants may still be significant. Additional limitations include heterogeneity across interventions and variability in follow-up duration, both of which contribute to the inconsistency in outcomes and hinder the evaluation of long-term effects. Although the included studies are of high methodological quality, risk of bias remains due to factors such as the lack of blinding and reliance on historical controls in study by Pinto and de Carvalho. 30
Future research should aim to standardize respiratory training protocols to enable better comparison across studies and extend post-intervention follow-up to capture potential long-term benefits of respiratory training. Following the findings of this review, we believe that it is important to continue to explore this field further, conducting more studies involving only patients with ALS in order to find results that allow us to state with certainty that respiratory muscle strength training can improve the quality of life of these patients and increase their life expectancy. Moreover, exploring RMT effects in specific subgroups, such as those with bulbar versus spinal onset, could reveal distinct benefits. Consideration should also be given to including, among the outcome measures, forced expiratory volume in the first second, as well as measures of fatigue, sleep quality, and functional capacity, as these would provide a more comprehensive understanding of how respiratory training affects various aspects of patient well-being.
Conclusion
In relation to the treatment of people with ALS, we found moderate evidence that RMT is more effective than the no-intervention CG (or sham training) in improving ventilatory function in these patients, particularly in respiratory muscle strength. RMT shows potential as an adjuvant intervention for patients with ALS. However, the evidence is limited, and results should be interpreted with caution. Despite these limitations, RMT represents a promising area of research that could contribute significantly to improving the quality of life and survival of people with ALS.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251346095 – Supplemental material for Efficacy of respiratory muscle training in improving pulmonary function and survival in patients with amyotrophic lateral sclerosis: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tar-10.1177_17534666251346095 for Efficacy of respiratory muscle training in improving pulmonary function and survival in patients with amyotrophic lateral sclerosis: a systematic review and meta-analysis by María Jesús Benzo-Iglesias, Patricia Rocamora-Pérez, María de los Ángeles Valverde-Martínez, Amelia Victoria García-Luengo, Pedro Miguel Benzo-Iglesias and Remedios López-Liria in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251346095 – Supplemental material for Efficacy of respiratory muscle training in improving pulmonary function and survival in patients with amyotrophic lateral sclerosis: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tar-10.1177_17534666251346095 for Efficacy of respiratory muscle training in improving pulmonary function and survival in patients with amyotrophic lateral sclerosis: a systematic review and meta-analysis by María Jesús Benzo-Iglesias, Patricia Rocamora-Pérez, María de los Ángeles Valverde-Martínez, Amelia Victoria García-Luengo, Pedro Miguel Benzo-Iglesias and Remedios López-Liria in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
