Abstract
Background:
Chronic glucocorticoid therapy is known to heighten the risk of secondary pulmonary infections. However, the impact of cumulative glucocorticoid dosage (CGD) on mortality risk in patients who develop community-acquired pneumonia (CAP) while undergoing glucocorticoid therapy remains inadequately explored.
Objectives:
This study aims to clarify the relationship between CGD and mortality outcomes in immunocompromised patients with CAP.
Design:
This study is a retrospective cohort analysis utilizing data from the DRYAD database.
Methods:
We examined data from 561 patients diagnosed with CAP who had received either oral or intravenous glucocorticoids prior to their CAP diagnosis. To evaluate the effect of CGD on mortality risk, we employed piecewise linear regression and Cox regression analyses, adjusting for relevant confounders.
Results:
Among the study population, the median CGD was 4 g of methylprednisolone (interquartile range 2.16–8.80 g). The 30-, 60-, and 90-day mortality rates were 22.28%, 25.13%, and 25.49%, respectively. Piecewise linear regression analysis revealed a nonlinear relationship between methylprednisolone dose and mortality risk, indicating a threshold effect at a methylprednisolone level of 20 g. In addition, Cox regression analysis showed a significantly higher mortality risk in patients with CGD exceeding 40 g of methylprednisolone compared to those with CGD between 20 and 40 g, after adjusting for potential confounding factors (adjusted HR 5.16, 95% CI: 1.16–22.99, p < 0.05).
Conclusion:
CAP occurring in close proximity to recent high doses of steroids is associated with pathogens typically seen in immunocompromised hosts and is linked to higher mortality compared to usual CAP.
Introduction
Community-acquired pneumonia (CAP) represents a significant global health concern, contributing to substantial morbidity and mortality, particularly among high-risk demographics such as the elderly, young children, and immunocompromised individuals. 1 Glucocorticoids are frequently prescribed to mitigate inflammation and alleviate symptoms in severe CAP cases.2 –4 However, the impact of cumulative glucocorticoid dose (CGD) on mortality risk in immunocompromised patients who develop CAP during glucocorticoid use remains insufficiently explored. While certain studies have suggested potential beneficial effects of glucocorticoid therapy in specific patient cohorts,2,3 others have highlighted apprehensions regarding possible adverse outcomes, including immunosuppression and heightened susceptibility to secondary infections.5,6
In patients with chronic obstructive pulmonary disease (COPD), previous studies have shown a dose-dependent relationship between oral glucocorticoid dosage and mortality. Specifically, a dose of 10 mg/day of prednisone equivalent was associated with an increased risk, while a dose of 15 mg/day further elevated the relative risk. 7 In patients with cardiogenic shock, short-term systemic glucocorticoid use was significantly associated with 90-day all-cause mortality. Although this association varied across different subgroups, overall, the use of glucocorticoids was linked to higher mortality rates. 8 These findings imply a potential dose-dependent correlation between glucocorticoid utilization and unfavorable outcomes. Furthermore, prolonged glucocorticoid use may lead to enduring adverse effects that significantly impact a patient’s quality of life, persisting even after treatment cessation. 9 Given the complex and incompletely understood relationship between these factors, our study sought to explore the intricate association between CGD and mortality risk in immunocompromised patients who develop CAP while undergoing glucocorticoid use. By doing so, we aim to provide novel insights into the multifaceted interplay between glucocorticoid therapy and mortality risk, with implications for the formulation of evidence-based treatment strategies within this vulnerable patient population.
Methods
Study design and subjects
This study leveraged data from the publicly accessible database, www.Datadryad.org, which allows unrestricted use for research and educational purposes. 10 The cohort comprised 716 patients who had received either oral or intravenous glucocorticoid therapy. These patients were recruited from six secondary and tertiary academic hospitals in China over a 5-year period from January 2013 to December 2017. 11 Diagnosis of CAP was made based on the criteria set forth by the American Thoracic Society and the Infectious Disease Society of America.12,13 Inclusion criteria for the study required patients to have been administered oral or intravenous glucocorticoids prior to hospital admission, to have a confirmed diagnosis of CAP at admission or during hospitalization, and to be at least 16 years of age. Exclusion criteria encompassed patients with non-infectious pulmonary conditions such as lung cancer, interstitial lung diseases unrelated to infection, pulmonary embolism, or heart failure, as well as those unable to provide consent. 11 The study flow chart is shown in Figure 1. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
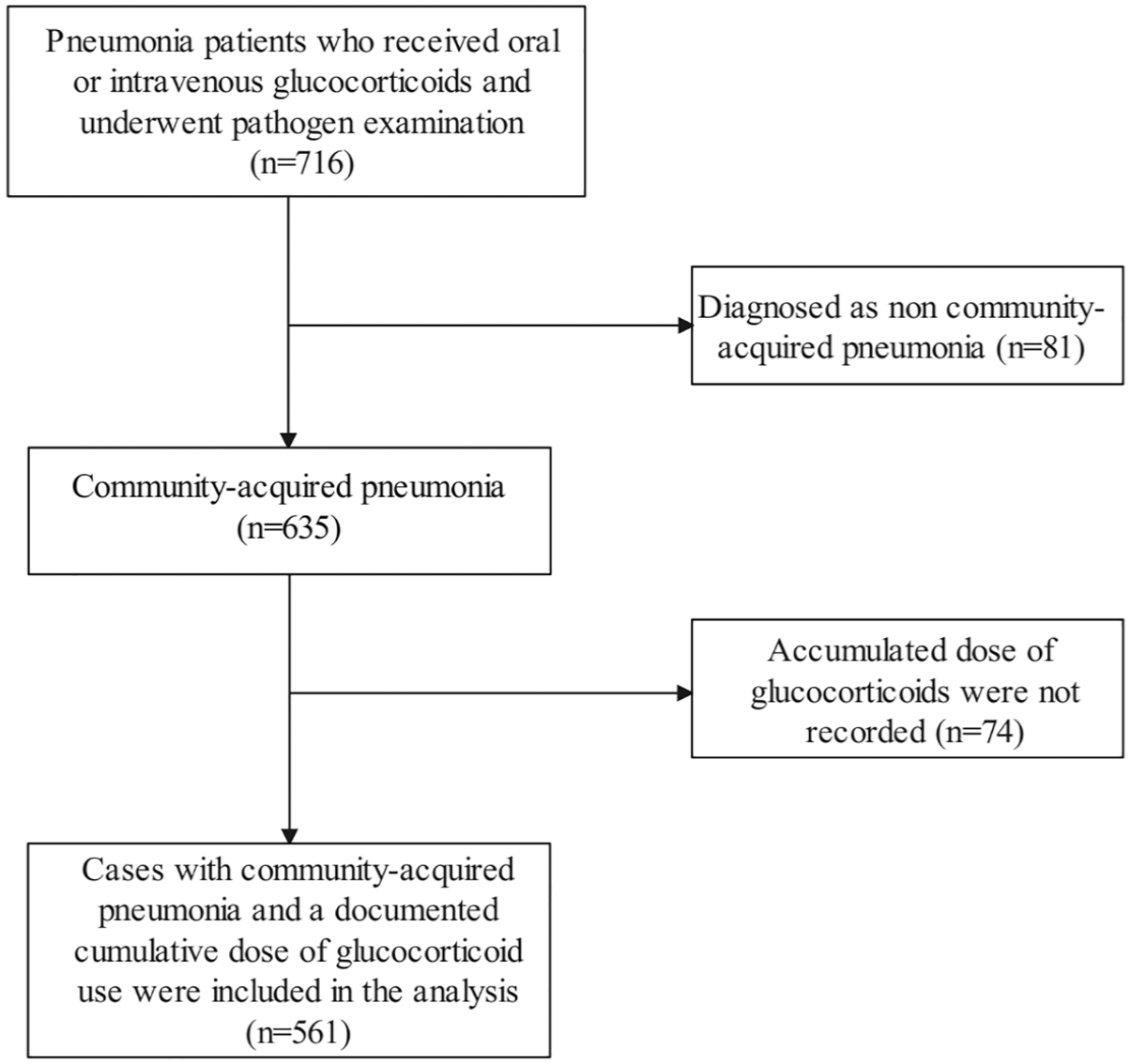
Study flow chart.
Data collection
Comprehensive demographic data were gathered, including information on gender, age, smoking status, and history of alcohol use. The presence of co-morbid conditions was also recorded, encompassing COPD, asthma, idiopathic interstitial pneumonia, interstitial lung disease, hypertension, coronary heart disease, diabetes mellitus, nephrotic syndrome, chronic renal failure, connective tissue disease, cerebrovascular disease, and malignancies. Pneumonia-specific characteristics and treatment details were meticulously documented. This included etiological data derived from sputum and/or bronchoalveolar lavage samples, CURB-65 scores, and therapeutic interventions such as antibiotics, antiviral agents, anti-Aspergillus treatments, extracorporeal membrane oxygenation (ECMO), continuous veno-venous hemofiltration (CVVH), mechanical ventilation, vasoactive drugs, and other immunosuppressants (methotrexate, cyclosporine, cyclophosphamide, tacrolimus, sirolimus, and azathioprine).
Microbiological samples, including blood, sputum, and bronchoalveolar lavage (BAL), were collected for the identification of bacterial, fungal, and viral pathogens. BAL specimens were immediately aliquoted and transported to the laboratory for processing. Bacterial cultures were incubated at 35°C in a 5%–10% carbon dioxide atmosphere for 48 h, with extended incubation for suspected Nocardia infections. Fungal cultures were incubated at 27°C for 5 days. Pathogen identification was performed using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry or the BACTEC 9102 system. Respiratory viral and atypical pathogens were detected by PCR, and galactomannan was assessed using the Platelia Aspergillus assay. 11
In addition, a range of hematological and biochemical parameters were collected. These included white blood cell count, neutrophil count, lymphocyte count, hemoglobin levels, platelet counts, and levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), and serum creatinine. High-dose glucocorticoid usage was defined as the administration of ⩾30 mg/day of prednisolone or an equivalent glucocorticoid within the 30 days preceding hospital admission. 11
Statistical analysis
The baseline characteristics of the study participants were summarized using descriptive statistics. Categorical variables were presented as frequencies (percentages), while continuous variables were expressed as medians (interquartile ranges, IQRs). To compare differences between groups, appropriate statistical tests were employed. Specifically, unpaired t-tests or Wilcoxon rank-sum tests were used for continuous variables, and Pearson’s chi-squared tests or Fisher’s exact tests were used for categorical variables, as deemed appropriate. Multiple imputation was utilized to address missing values. To investigate the association between CGD and the risk of mortality, with adjustment for potential confounders, a generalized additive model with smoothing curve fitting was employed. Furthermore, an adjusted two-piecewise linear regression analysis, coupled with the log-likelihood ratio test, was utilized to identify any threshold effect of CGD on the mortality risk. Survival analysis, including Kaplan–Meier analysis and the log-rank test, was performed to compare the differences in survival rates between high and low glucocorticoid dose groups. To assess the independent relationship between CGD and the risk of pneumonia mortality, adjusting for potential confounders, Cox regression analysis was conducted. Two distinct criteria were employed to select the covariates for adjustment: Criteria I: A covariate-discrimination algorithm was utilized, whereby variables were included if their introduction into the basic model or removal from the complete model resulted in a ⩾10% change in the regression coefficient. The covariates meeting this criterion were etiology, neutrophils, CURB-65 score, and persistent lymphocytopenia. Criteria II: Clinically relevant variables, such as age, gender, and the use of other immunosuppressants, were adjusted based on clinical considerations. All statistical analyses were performed using R software (version 3.5.1, the R Foundation for Statistical Computing, Vienna, Austria), and a p-value < 0.05 was considered statistically significant.
Results
Among the 561 CAP patients, 296 (52.8%) were male, and 280 (49.9%) were aged over 60 years. Pathogen examination confirmed the presence of pathogens in 410 out of 561 cases, with Cytomegalovirus (CMV) identified as the most prevalent single pathogen (114/410), followed by Pneumocystis jirovecii pneumonia (PCP) (58/410). In addition, 63 cases were found to have co-infection with both CMV and PCP. In our study, co-infection was observed in 58.3% of the cases. We found that both the 30-day mortality rate (27.5% vs 17.1%) and the 90-day mortality rate (31.9% vs 19.9%) were significantly higher in patients with co-infection compared to those with single-pathogen infections (p < 0.05). The median CGD administered was 4 g (interquartile range (IQR) 2.16–8.80 g). The admission rate to the intensive care unit was 43.3% (243/561). Among patients who died within 90 days, 51.05% were high-dose glucocorticoid users, compared to 33.73% among survivors. The mortality rates at 30 days, 60 days, and 90 days were 22.28%, 25.13%, and 25.49%, respectively. Detailed demographic and clinical characteristics of the patients and their pneumonia presentation are shown in Table 1. The characteristics of patients stratified by high and low doses of glucocorticoids are presented in Supplemental Table 1.
Clinical characteristics of patients with community-acquired pneumonia receiving oral or intravenous glucocorticoids.
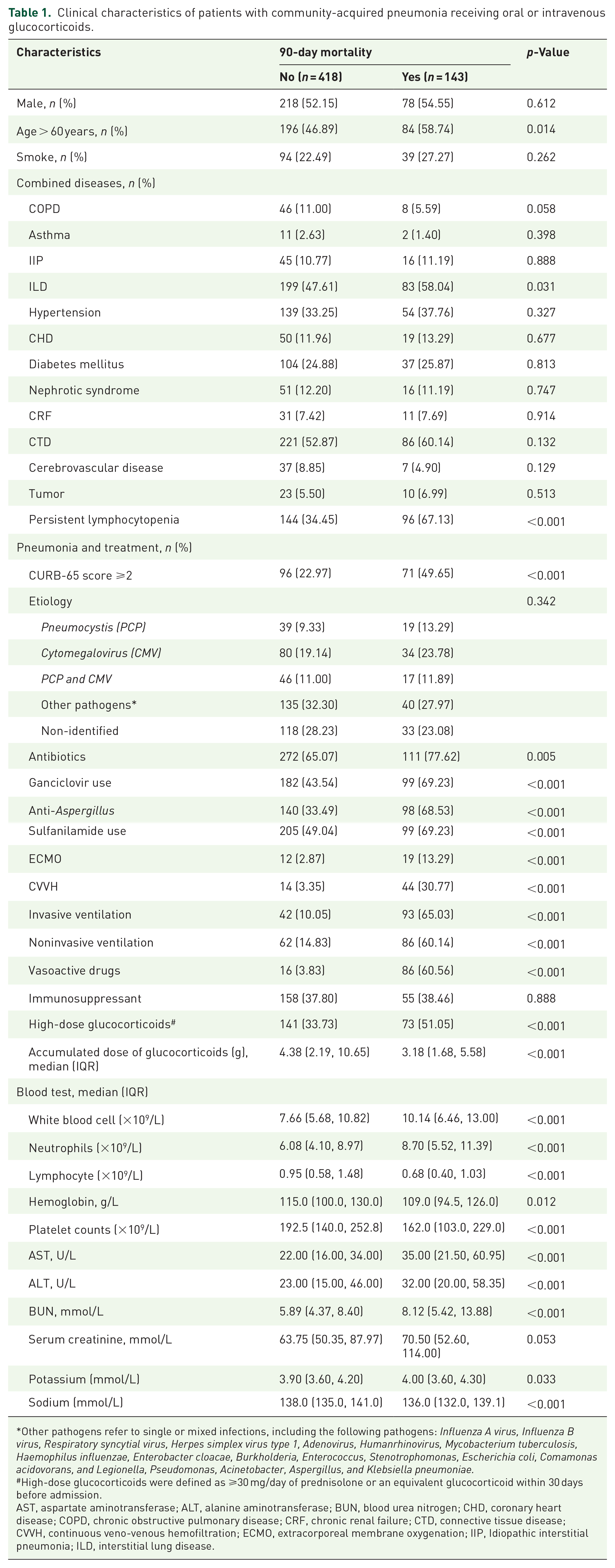
Other pathogens refer to single or mixed infections, including the following pathogens: Influenza A virus, Influenza B virus, Respiratory syncytial virus, Herpes simplex virus type 1, Adenovirus, Humanrhinovirus, Mycobacterium tuberculosis, Haemophilus influenzae, Enterobacter cloacae, Burkholderia, Enterococcus, Stenotrophomonas, Escherichia coli, Comamonas acidovorans, and Legionella, Pseudomonas, Acinetobacter, Aspergillus, and Klebsiella pneumoniae.
High-dose glucocorticoids were defined as ⩾30 mg/day of prednisolone or an equivalent glucocorticoid within 30 days before admission.
AST, aspartate aminotransferase; ALT, alanine aminotransferase; BUN, blood urea nitrogen; CHD, coronary heart disease; COPD, chronic obstructive pulmonary disease; CRF, chronic renal failure; CTD, connective tissue disease; CVVH, continuous veno-venous hemofiltration; ECMO, extracorporeal membrane oxygenation; IIP, Idiopathic interstitial pneumonia; ILD, interstitial lung disease.
After adjusting for potential confounders such as age, gender, other immunosuppressants, pneumonia etiology, neutrophil count, CURB-65 score, and persistent lymphocytopenia, a non-linear relationship between CGD and 90-day mortality was observed (Figure 2). This relationship was also evident at 30-day and 60-day mortality (Supplemental Figure 1). Further hierarchical analysis indicated no significant association between CGD and 90-day mortality across subgroups defined by age (>60 years), gender, presence of comorbidities (such as COPD, asthma, interstitial lung disease, coronary heart disease, hypertension, diabetes, nephrotic syndrome, cerebrovascular disease, connective tissue disease, tumors), and CAP severity (Figure 3).
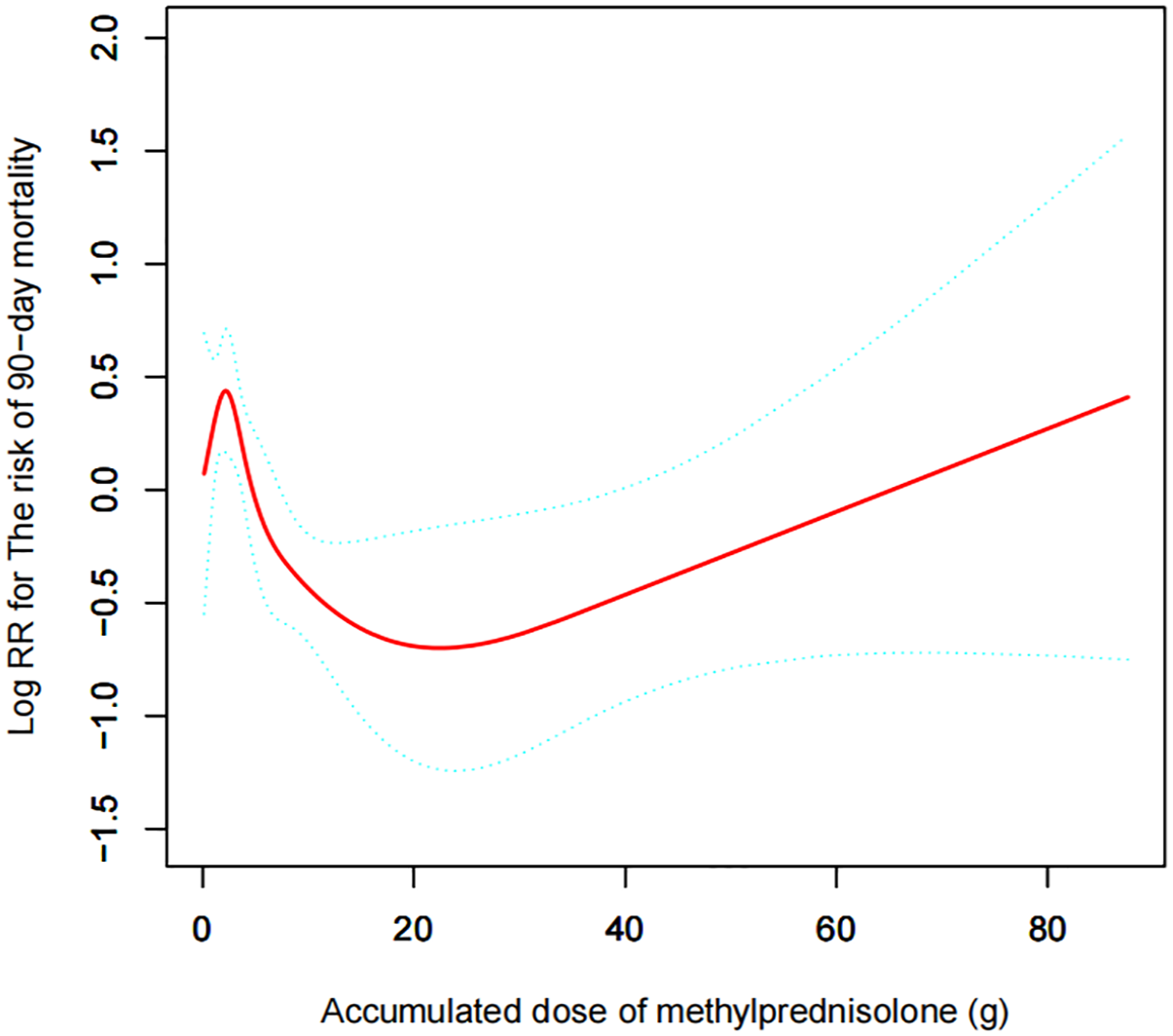
A smooth fitting curve illustrating the association between cumulative glucocorticoid dosage and the 90-day mortality risk in patients with community-acquired pneumonia. Adjustments were made for age, gender, etiology, other immunosuppressants (methotrexate, cyclosporine, cyclophosphamide, tacrolimus, sirolimus, and azathioprine), neutrophils, CURB-65 score, and persistent lymphocytopenia.
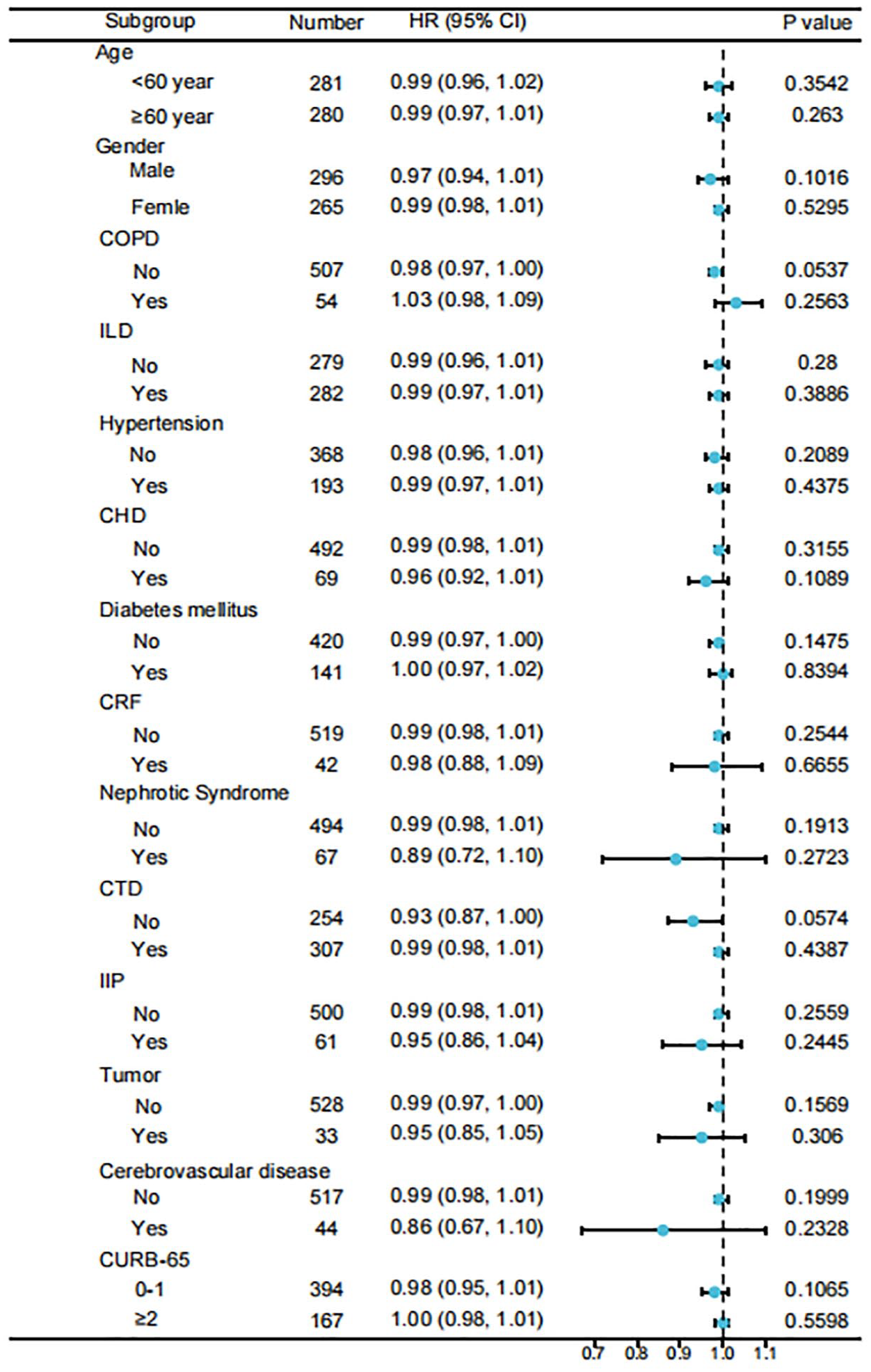
Stratification analysis of cumulative glucocorticoid dosage with the mortality risk in immunocompromised patients with community-acquired pneumonia.
Exploring a potential threshold effect, our analysis revealed an inflection point at 20 g of methylprednisolone associated with increased 90-day mortality risk, with similar findings at 30- and 60-day mortality risk (Table 2). Stratifying methylprednisolone into two subgroups (20–40 g and >40 g), Cox regression analysis demonstrated an adjusted hazard ratio for mortality of 5.164 (95% CI: 1.16–22.99) for patients with methylprednisolone exceeding 40 g compared to those with CGD between 20 and 40 g (Table 3).
Threshold effect of accumulated dose of methylprednisolone (g) on the risk of mortality in immunocompromised patients with community-acquired pneumonia.

Adjust for: age, gender, etiology, other immunosuppressants (methotrexate, cyclosporine, cyclophosphamide, tacrolimus, sirolimus, and azathioprine), neutrophils, CURB-65 score, and persistent lymphocytopenia.
Cox regression analysis of high cumulative methylprednisolone dosage on the risk of mortality in immunocompromised patients with community-acquired pneumonia.

Adjust for: age, gender, etiology, other immunosuppressants (methotrexate, cyclosporine, cyclophosphamide, tacrolimus, sirolimus, and azathioprine), neutrophils, CURB-65 score, and persistent lymphocytopenia.
Furthermore, high-dose glucocorticoid users exhibited significantly lower 30-day, 60-day, and 90-day survival rates compared to those receiving low-dose glucocorticoid (Figure 4, p < 0.001).

Kaplan–Meier analysis and comparison of mortality between high- and low-dose glucocorticoid groups.
Discussion
The present study identifies a complex, nonlinear relationship between CGD and mortality risk across 30-, 60-, and 90-day intervals. Notably, initial increments in CGD (<20 g of methylprednisolone) were associated with a reduced risk of mortality. However, beyond a critical threshold (20 g of methylprednisolone), there was a pronounced escalation in mortality risk.
Long-term corticosteroid use leads to immune suppression, thereby increasing the risk of secondary infections, including CAP. 15 Studies have shown that the incidence of CAP is significantly higher in immunosuppressed individuals.15,16 A study found that the prevalence of CAP in immunosuppressed patients was 6.6%, compared to just 1.7% in non-immunosuppressed patients. Moreover, immunosuppressed individuals exhibit a notably higher 30-day all-cause mortality rate. 15 During hospitalization, they are also more susceptible to developing sepsis and other complications. 15 Furthermore, prolonged corticosteroid therapy may alter the pathogen profile, with certain pathogens, such as fungi and mycobacteria, becoming predominant causes of infection. 15 These findings further emphasize that long-term corticosteroid use increases the risk of secondary infections through its immune-suppressive effects. In the context of long-term corticosteroid use, an important question arises: is the CGD associated with an increased risk of mortality in patients? A study focusing on patients with rheumatoid arthritis demonstrated a dose–response relationship between CGD and both all-cause mortality and cause-specific mortality. As the cumulative dose increased, the risk of death rose significantly. 17 However, this relationship exhibited a dose-saturation effect, where the risk of death no longer increased significantly once a certain cumulative dose threshold was reached. 17 Similarly, another study indicated that long-term use of high-dose corticosteroids was significantly associated with an increased risk of mortality in asthma patients, with the risk also escalating in proportion to the dose. 18 Patients with a history of prolonged glucocorticoid use often present with increased disease severity and higher mortality rates upon developing CAP compared to non-immunosuppressed individuals. 19 This highlights the heightened vulnerability of patients with compromised baseline health, necessitating tailored management approaches. Furthermore, the variability in treatment response and potential adverse effects associated with glucocorticoid therapy underscores the importance of careful patient selection to optimize therapeutic outcomes. 19 In our study, we focused on patients with a history of prolonged glucocorticoid use, a cohort predisposed to CAP due to their immunosuppressed status. Exploring the relationship between CGD and mortality within this population carries significant clinical implications. Our findings indicate that the 30- to 90-day mortality rates among these patients range from 22% to 25%, consistent with previously reported data. Previous studies have noted an overall mortality increase of 20% in severe CAP patients, with higher rates observed in cases involving shock (22%), invasive mechanical ventilation (25%), or both (30%). 20
There is a subtle effect of corticosteroid use on the body’s immune status. The use of corticosteroids at certain doses can enhance the activity of CD4 T cells, B lymphocytes, and NK cells in postmenopausal women, thereby contributing to the restoration of normal immune function. 21 However, the use of high-dose corticosteroids also carries the risk of impairing immune function. 22 We identified a nonlinear relationship where CGD exceeding 20 g significantly increased mortality risk among long-term users of oral or intravenous glucocorticoids. This observation may help explain the varying conclusions of different studies regarding the impact of glucocorticoid therapy on mortality risk—either attenuating or exacerbating it. Importantly, this threshold effect persisted across diverse patient profiles, irrespective of underlying conditions or pneumonia severity. Notably, mortality risk can increase up to fivefold when CGD exceeds 40 g compared to doses below 20 g. These findings underscore the critical importance of judicious dosing strategies in CAP management, particularly for patients requiring prolonged glucocorticoid therapy.
Limitations
Nevertheless, our study has several limitations that require careful consideration. First, its retrospective observational design introduced potential bias by indication, where the decision to administer glucocorticoids may be influenced by underlying patient factors that also impacted mortality risk. Second, delays in pathogen identification post-admission could potentially increase the risk of nosocomial infections, thereby affecting overall mortality outcomes. Third, our assessment of CGD did not account for inhaled glucocorticoids, commonly used in conditions such as asthma and COPD. This omission may underestimate the true mortality risk in these patient populations. The absence of immune function markers, such as CD4 and other immunoglobulins, is also a limitation of this study. In addition, the lack of relevant data on steroid use in severe pneumonia limited our ability to investigate its potential impact. Furthermore, our study is the absence of data regarding the proximity of the most recent steroid dose to the onset of pneumonia, as well as whether steroids were continued during the course of the CAP episode. Moreover, detailed information and categorization of specific immunosuppressive drugs, such as rituximab, cyclophosphamide, plaquenil, or low-dose methotrexate, were not available, nor were detailed data on conditions like severe asthma, nephrotic syndrome, or autoimmune diseases, which limited our ability to analyze the impact of CGD on adverse outcomes in these patient groups. As a result, we are unable to perform sensitivity analysis on this aspect. The findings are therefore limited to interpreting the association between cumulative glucocorticoid use and mortality risk without differentiating between specific types of immunosuppressive drugs. These factors may have influenced the outcomes, and future research should address these aspects to provide a more comprehensive understanding.
Conclusion
In conclusion, our findings underscore the critical role of CGD in influencing mortality risk among patients receiving oral or intravenous glucocorticoids and subsequently developing CAP. Specifically, CAP developing shortly after the administration of high-dose steroids is associated with pathogens commonly found in immunocompromised individuals and is associated with increased mortality compared to usual CAP. Clinicians should consider the cumulative effects of glucocorticoids on patient outcomes when managing the immunocompromised population. Future research should aim to further elucidate these complex relationships to optimize the use of glucocorticoids in the management of CAP.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251332085 – Supplemental material for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia
Supplemental material, sj-docx-1-tar-10.1177_17534666251332085 for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia by Saibin Wang, Qian Ye and Yijun Sheng in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251332085 – Supplemental material for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia
Supplemental material, sj-docx-2-tar-10.1177_17534666251332085 for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia by Saibin Wang, Qian Ye and Yijun Sheng in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-tif-3-tar-10.1177_17534666251332085 – Supplemental material for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia
Supplemental material, sj-tif-3-tar-10.1177_17534666251332085 for From treatment to threat: the fatal impact of cumulative glucocorticoid dosage on outcomes in immunocompromised patients with community-acquired pneumonia by Saibin Wang, Qian Ye and Yijun Sheng in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
