Abstract
Eosinophilic granulomatosis with polyangiitis (EGPA), as a heterogeneous component of antineutrophil cytoplasmic antibody-associated vasculitis, may be induced by a series of environmental and genetic factors, involved with a variety of immune cells and immune components, and presented with various clinical manifestations, with multiple organs and systems (respiratory, skin, heart, kidney, nerve, etc.) involved. The choice of glucocorticoid (GC) dosage and immunosuppressant in traditional treatment strategies varies greatly from individual to individual and is not universally applicable in all the EGPA phenotype spectrum, especially in relapsing or refractory diseases. With the understanding of the heterogeneity of EGPA, a variety of therapeutic approaches are emerging and improving the traditional treatment model. In this review, we summarized the heterogeneity of EGPA etiology and pathogenesis. Clinical and pathological manifestations of the same organ involved also show significant differences and there are even gender differences. Biological treatments that mainly target type 2 inflammatory pathways are widely used in clinical practice for remission induction and maintenance of EGPA. Targeted biological therapy has shown excellent performance in reducing GC dosage and controlling symptoms and recurrence. However, a large number of high-quality randomized controlled studies are still under research for relapsing or refractory EGPA with special organ involvement. We believe that EGPA has a highly heterogeneous phenotype spectrum, and the treatment patterns targeting key molecules in the pathogenesis are of great value for individual treatment of EGPA.
Plain language summary
1. Eosinophilic granulomatosis with polyangiitis is a rare disease characterized by inflammation of small and medium blood vessels.
2. Manifestations of the disease are diverse, and multiple organs may be involved. A variety of immune cells (cells that are part of the immune system and help fight infections and diseases) and mediators (molecules) secreted by these cells are involved in the development of diseases.
3. Traditional treatments use powerful medicines such as glucocorticoids (a class of steroid hormone) and immunosuppressants (drugs that inhibit your immune system from attacking healthy cells) to control the symptoms. It has certain curative effect but with side effects.
4. There is a more favorable treatment model emerging. The drugs targeting specific immune cells or mediators are applied and help reduce the dosage of glucocorticoids to achieve better disease control. This model allows doctors to choose a tailored treatment according to the characteristics of the patient’s disease to achieve individual treatment.
Keywords
Introduction
Eosinophilic granulomatosis with polyangiitis (EGPA), previously known as Churg-Strauss syndrome, 1 is a rare type of antineutrophil cytoplasmic antibody (ANCA) associated vasculitis (AAV) and characterized by asthma, eosinophilia and necrotizing vasculitis of medium-to-small arteries. 2 The general population prevalence of EGPA is 10–14 cases per million,3,4 and the incidence is 0.5–6.8 cases per million people per year, varying with geographical location and classification criteria.5 –8 The cause of EGPA is currently unclear, and a combination of environmental and hereditary factors may play a role. With genetic and environmental triggers, various immune cells, as well as some cytokines and chemokines participate in the complex pathophysiological processes of EGPA.9,10 The heterogeneity of pathogenesis contributes to distinct clinical manifestations. Three disease phases are described previously: The prodromic phase, which mainly exhibits allergic disease such as asthma and chronic rhinosinusitis and may last for several years; the eosinophilic phase with eosinophilia and end-organ involvement; and the vascular phase with prominent small-vessel vasculitis lesions in the organ system, such as mononeuritis multiplex and glomerulonephritis. 11 The three distinct but sometimes overlapping phases, and the fact that some patients do not even experience one of them, add to the heterogeneity of EGPA clinical presentations. The prognosis for EGPA is generally favorable with the use of corticosteroids and immunosuppressants. 12 However, some patients suffer from recurrent disease, and the prognosis varies depending on the extent and degree of organ damage. 13 The heterogeneous etiology, pathogenesis, and clinical manifestations urgently require individual treatments for EGPA. In the present review, we summarized the potential etiology of EGPA, including drug-related etiology, heterogeneous manifestations in different organs and genders, and complex pathogenesis. Then we thoroughly reviewed the potential molecular targets of EGPA and the emerging targeted therapies. By integrating the latest clinical drug practice and randomized controlled studies, we revealed the application and efficacy of different targeted biological drugs in different therapeutic stages of EGPA and clinical phenotypes. The therapeutic differences between the targeted biologics as well as traditional immunosuppressants were also included. Our study provides a comprehensive frame for understanding the complexity of EGPA and offers valuable references for the individualized treatment of EGPA.
Heterogeneity
Etiology
Environmental factors
The study of AAV has shown that various environmental factors contribute to the predisposition of AAV, 14 but the effects are neither always consistent nor apply to all AAV phenotypes. As for the etiology of EGPA, a study found that increased latitude and reduced ambient ultraviolet radiation are associated with susceptibility to EGPA. 15 Occupational exposures such as silica, farming, or organic solvents were found related to an increased tendency to EGPA, whereas smoking is related to a lower risk. 16 In addition, multiple exposures show a multiplier effect on the incidence of EGPA. Various exposures exert distinct susceptibility to EGPA subtypes. A high risk of ANCA-positive EGPA was found by exposure to silica and farming, whereas a higher risk of ANCA-negative EGPA by exposure to organic solvents. Smoking showed a stronger protective effect against ANCA-negative EGPA. 16 A suppressive effect on mucosal immunity and modification of local microecology by smoking may explain the protective effect.17 –19 In another study of primary systemic vasculitis with 16 patients with EGPA included, farming exposure was not associated with the risk of EGPA. 20 The difference in EGPA sample size and profile of matched controls may account for the discrepancy.
Medication factors
The agents used for asthma treatment were found associated with the development of EGPA. The leukotriene receptor antagonists (LTRAs) were highly controversial. According to data from the United States Food and Drug Administration pharmacovigilance database, LTRAs (montelukast or zafilukast) accounted for 90% of suspected drug-associated EGPA cases. 21 This phenomenon may be explained by corticosteroid reduction and exposure to potential EGPA during the medication. Also, the timing of LTRA administration beyond the mentioned situations was found associated with EGPA onset, indicating a complicated pathogenesis of EGPA. 21 A case-series study that enrolled patients who took montelukast before progressing to EGPA but had no history of oral corticosteroids found that these patients experienced no EGPA relapse after discontinuing montelukast, increasing the potential association between LTRAs and EGPA pathogenesis. 22 In another study, montelukast, as well as other asthma control medications showed an increased risk of developing EGPA within 3 months, indicating the upgrade asthma therapy before EGPA onset as a possible confounding factor. 23 The long-term medication of montelukast should be of concern. Cases of severe asthma patients developing EGPA during anti-type 2 (T2) immune monoclonal antibodies treatment were also published. A multinational cohort reported that 5.7% of EGPA patients developed the disease during anti-T2 therapy for severe asthma, including omalizumab, benralizumab, mepolizumab, dupilumab, and reslizumab. 24 More rigorous study design, rather than a case series, may be an important way to reveal the association between drug medication and EGPA.
Microbial factors
Microbial infection also plays a role in the pathogenesis of EGPA. Aspergillus infections, including Candida and Fusarium, may be triggers for EGPA susceptibility, as they have been found in patients with both allergic bronchopulmonary aspergillosis (ABPA) and EGPA. 25 A small number of cases reported that ABPA occurred before or simultaneously with EGPA, suggesting a potential susceptibility of EGPA caused by fungi. 25 EGPA patients with positive hepatitis B core antigen faced a significantly higher risk of relapse. 26
Genetic factors
Genetic variation plays a vital role in EGPA pathogenesis and predisposes individual genetic susceptibility. Variants in the human leukocyte antigen (HLA)-DR gene, which gets involved in CD4+ T-cell lymphocyte responses are closely related to EGPA. Specifically, HLA-DRB4 and HLA-DRB1*07 may be involved in the onset of EGPA, while HLA-DRB3 and HLA-DRB1*13 act as protective factors. 27 Interleukin (IL)-10 polymorphisms also show an association with the pathogenesis of EGPA, in which the IL-10.2 haplotype correlates with an elevated expression of IL-10 and ANCA-negative EGPA. 28 In addition, several genes may explain differences in disease characteristics associated with ANCA status. The genome-wide association study reported the association of HLA-DQ gene with ANCA-positive EGPA, indicating an eosinophilic autoimmune disease with vasculitis, while thymic stromal lymphopoietin (TSLP), glycoprotein (GP)A33, and interferon regulatory factor (IRF)1/IL-5 genes, which are involved in mucosal responses seemed to be associated with ANCA-negative EGPA. 29
Pathogenesis
T lymphocytes
In the pathophysiology of EGPA, T2 response is a key contributor to eosinophilia and symptoms. EGPA patients showed a higher level of activated CD4+ T cells than the healthy controls. 30 Secretion of IL-5, IL-4, and IL-13 by T-helper (Th)2 cells increased in both peripheral blood and bronchoalveolar lavage fluid of EGPA and was associated with EGPA disease activity.31 –33 IL-5 has strong activity in prompt eosinophil generation, maturation, tissue migration, and activation by binding to IL-5 receptors on the surface of eosinophils. 34 The IL-4/IL-13 pathway also exerts the effect of eosinophil migration and accumulation in the blood. The pathogenesis of EGPA is also associated with Th1 and Th17 responses. A decreased number of Treg cells and increased Th17 cells and Th17/Treg ratio were found in EGPA patients. 35 Both Th1 and Th17 cells secrete a large amount of IL-5 in the periods of active EGPA. 35 Th1 and Th17 cell-mediated pathways are also responsible for granulomatosis, vasculitis, and neutrophilic inflammation. Th17 lymphocytes promote neutrophil recruitment and activation. In turn, neutrophils activated by myeloperoxidase (MPO)-ANCA showed elevated expression of IL-6, IL-17, and IL-23, which favor T-cell differentiation toward a Th17 phenotype36,37 (Figure 1).
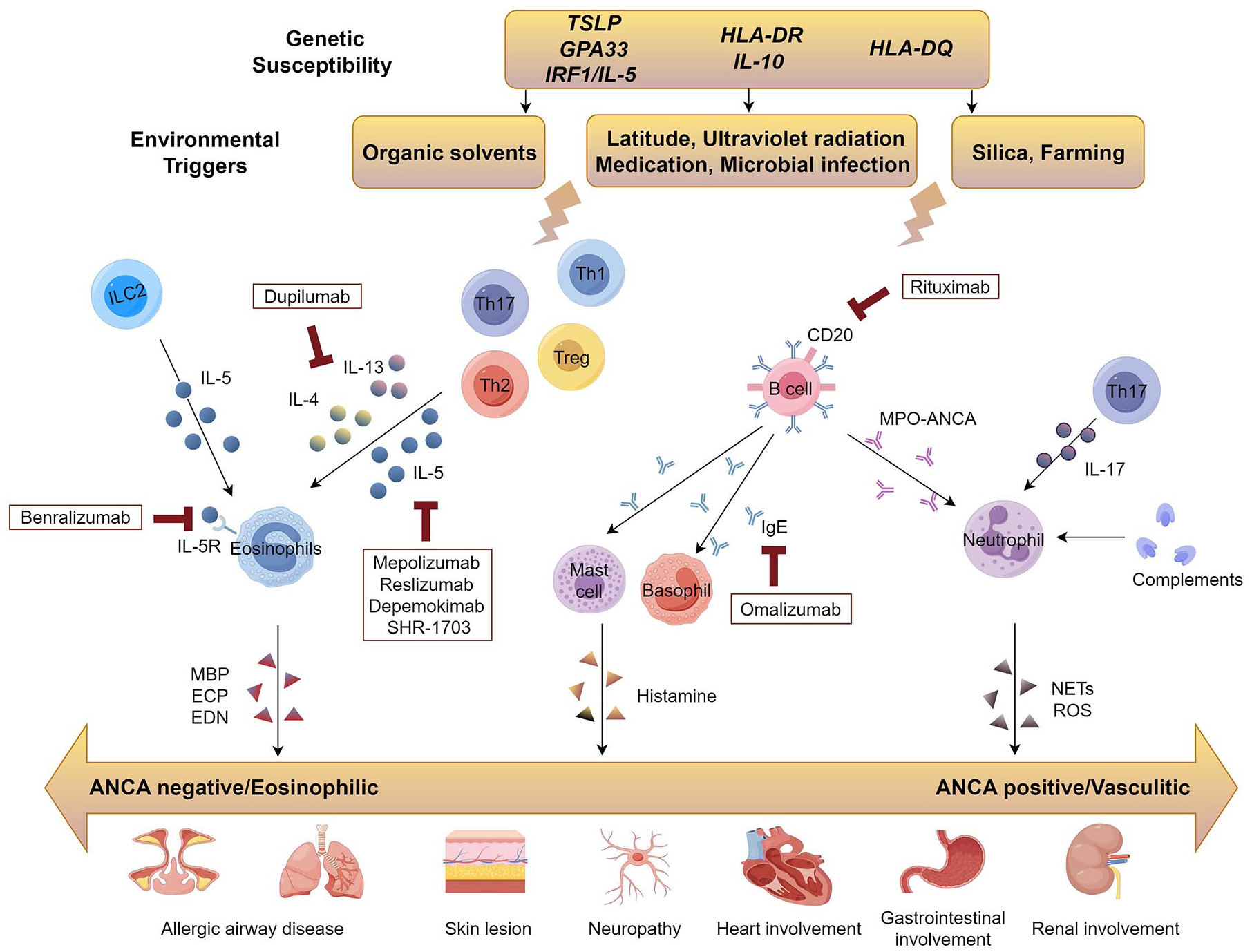
Immunopathogenesis and potential targets of EGPA for biological therapy. Various genes influence the susceptibility to EGPA and have distinct effects on EGPA subsets. Triggered by environmental factors, both innate immune response and adaptive immune response are activated and participate in the activation of eosinophils and neutrophils, mediating the pathogenesis of both ANCA-negative and ANCA-positive EGPA. In addition to conventional GC therapy, several biologics may be able to play a therapeutic role by targeting specific molecules and blocking their signaling pathways.
B lymphocytes
There is evidence to support the contribution of B cells and the humoral immune response in the EGPA. An increased percentage of B cells presenting a positive cluster of differentiation (CD) 80, CD27, or CD95 and fewer CD19-positive B cells were found in frequently relapsing EGPA, which is associated with a lower serum IgG concentration compared to those with less frequent relapses. 38 B cells promote EGPA autoimmunity by producing a series of antibodies, including ANCA (especially MPO-ANCA in EGPA) stimulated by T cells. Elevated serum total immunoglobulin (Ig) E concentrations and the presence of an Ig E-containing immune complex are often observed in EGPA patients. 39 Elevated serum IgG4 levels in EGPA cases are found associated with an increased number of organ involvement and active EGPA.30,40 In addition, numerous Treg cells, IgG4-riched plasma cells, and immune complexes were also exhibited in EGPA lung biopsies.30,41
Type 2 innate lymphoid cells
Type 2 innate lymphoid cells (ILC2s), mediating innate immune response, are also increased in peripheral blood of active EGPA and the level of ILC2s was found to correlate with eosinophil count and disease activity. 42 ILC2s are also an important source of IL-5 and IL-13, which as mentioned above, and involved in the eosinophilia of EGPA. On the other hand, ILC2s secrete IL-33, which involves active vasculitis in EGPA. 42 As for tissue pathology, ILC2s may play a role in regulating the crosstalk between epithelial cells and the immune system. 43
Eosinophils
Eosinophil is an important effector cell in EGPA pathogenesis with adaptive and innate immune response described above as a prelude. Functional changes were observed in eosinophils. Increased expression of CD69 and CD25 on the cell surface suggested cell activation. 44 Evidence of apoptotic pathway dysfunction was also found, manifested by decreased expression of several pro-apoptotic genes and increased levels of circulating CD95, which acts as an inhibitor of Fas-mediated apoptosis.45,46 Eosinophil activation and degranulation mediate cytotoxicity, tissue and vascular injury via various granule proteins such as major basic protein (MBP), eosinophil-cationic protein (ECP), and eosinophil-derived neurotoxin (EDN).47,48 In addition, eosinophilic activation is accompanied by increased IL-25 expression, which mediates a vicious cycle by promoting their proliferation and T2 response. 49
Endothelial cells
Eotaxin-3 produced by endothelial cells is a highly eosinophil-specific chemoattractant, which may be involved in EGPA and act as the marker of disease activity. 50 In addition, the well-known epithelial-derived alarmins (TSLP, IL-25, IL-33, and soluble ST2) were found elevated in the serum of EGPA.42,51 These cytokines may lead to changes in vascular permeability and the subsequent eosinophilic infiltration and vasculitic injury by mediating the activation of the innate immune response. 51
Neutrophils
In the pathogenesis of AAVs, ANCA induces neutrophil degranulation and production of oxygen-reactive species, causes increased adhesion of neutrophils to glomeruli and endothelial cells, and aggravates glomerulonephritis and vasculitis through immune complex deposition. 9 Complement activation also plays an important role in vasculitis. Complement 5a (C5a) as a potent neutrophil chemotactic agent, is involved in neutrophil activation by binding to the C5a receptor on neutrophils. 52 Recent studies suggest that neutrophil extracellular traps generated by neutrophil activation may be involved in the pathogenesis of AAV by providing ANCA.9,53 Given the potential heterogeneous pathogenesis of EGPA compared to other AAV types, further research is urgent to explore the role of neutrophils in EGPA pathogenesis.
Clinical manifestations
Respiratory manifestation
The respiratory system is commonly affected in EGPA and pulmonary involvement is predominantly seen, which was reported as 34%–76% in EGPA patients. 54 Chronic recurrent sinusitis and nasal polyposis are common manifestations of the upper respiratory tract. Asthma is a prominent manifestation of EGPA, with a prevalence of 95%–100%. Asthma may present several years earlier than other systemic symptoms. 55 Computed tomography (CT) findings of lung involvement have been reported in many AAV cohorts with only a few EGPA patients included in the cohorts, thus the findings do not reflect the true characteristics of EGPA.54,56,57 A recent EGPA cohort, involving 46 patients with EGPA, reported 82.6% abnormal parenchymal findings on high-resolution CT. 58 Bronchial wall thickening (69.6%) and mosaic perfusion (63.0%) were the prominent manifestations, followed by gross glassy turbidity, bronchodilatation, mucus plugging, and solid changes. 58 Thickening of the bronchial wall is a common feature of eosinophilic asthma, so the mark may be attributed to the eosinophilic mechanism in EGPA and act as a marker for initiating eosinophilic-targeted therapy.
Skin manifestation
In cohorts of AAV, 40%–70% of EGPA exhibited skin involvement, which was at least two times higher compared to the frequency in granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA).59,60 Common cutaneous manifestations included palpable purpura, urticarial rash, papules, nodules, cutaneous ulcerations, and other nonspecific rash.59 –61 The distribution range of the lesions also varied among patients, with upper and lower extremities, trunk as well as generalized distribution being found. Skin lesions in EGPA tend to be more common in the lower extremities rather than in the head and neck. 62 Purpura and petechiae were usually seen on the lower extremities, while nodules and papules on extensor extremities such as the elbows. 60 Leukocytoclastic vasculitis, extravascular necrotizing granuloma, and perivascular eosinophils infiltrate were the most frequent cutaneous histopathological features.59,60,63 Extravascular necrotizing granuloma was often found in the lesion of erythematous nodules and papules, while leukocytoclastic vasculitis was more frequently identified in the lesion of purpuric papules with obvious eosinophils infiltrate. 63 It is important to note that skin lesions and pathological manifestations in EGPA patients are not entirely specific and may be partly due to overlap with other diseases. 62 In addition, these clinical and pathological manifestations of skin do not exist in isolation but may appear simultaneously in the same patient or change throughout the disease.60,64 EGPA patients with cutaneous manifestations are more likely to present with systemic involvement and symptoms, but patient outcomes are not affected. 62
Heart and vascular alterations
Cardiovascular involvement is one of the indicators of poor prognosis of EGPA, indicating frequent disease relapse, propensity to intensive care admission, and reduced survival rate.65 –67 The involvement form mainly includes arterial occlusion disease, venous thrombosis, and cardiac involvement, with great variation in the prevalence of each part. 68 Common manifestations of cardiac involvement include pericardial effusion, myocardial involvement, and heart failure. 69 According to the heterogeneity of pathophysiological characteristics, eosinophilic myocarditis, and chronic inflammatory cardiomyopathy are considered two distinct subtypes of cardiac involvement, and the prognosis of the former is poorer. 70 Most patients with cardiac involvement present with ANCA negative and eosinophilia,70 –72 suggesting the dominant mechanism of eosinophilic inflammation. In addition to macrovascular involvement, microvascular alterations are also found in the nail bed and retina of AAV patients.73,74 The most frequent findings of the nail bed were neoangiogenesis, capillary loss, microhemorrhages, and bushy and enlarged capillaries. The changes of nailfold capillary were mostly nonspecific, while higher rates of these abnormalities seemed to be associated with active AAV. 73 Decreased retinal vessel density was also found to be related to a higher vasculitis damage index. 74 Microvascular damage should not be ignored as part of assessment of systemic vasculitis activity. EGPA, as a heterogeneous and rare type of AAV, also requires more exclusive research.
Gastrointestinal involvement
Gastrointestinal involvement occurs in approximately 20%–50% of EGPA patients, with the most common gastrointestinal symptoms being abdominal pain, diarrhea, and ulcers.75,76 Perforation and stenosis are relatively rare. These gastrointestinal lesions are thought to be the result of ischemia caused by vasculitis. However, histology of biopsy specimens of the gastrointestinal tract usually shows only eosinophilic infiltration, and vasculitis is uncommon. 75 Gastrointestinal involvement was also included in the assessment of poor prognosis and may develop life-threatening complications. 55
Nervous involvement
More than 50% of patients have peripheral nervous system involvement in the early vasculitis stage, characterized by paresthesia and muscle weakness and eventually developing into mononeuritis multiplex.77,78 ANCA positive, arthritis or joint pain, and higher Birmingham Vasculitis Activity Score (BVAS) were found to be independently associated factors for peripheral neuropathy in EGPA. 79 Sural nerve biopsies in patients with EGPA-associated neuropathy suggested that peripheral neuropathy was associated with ischemia and inflammation caused by vasculitis in MPO-ANCA–positive patients, whereas eosinophilic vascular occlusion and ischemia and eosinophil-mediated tissue injury dominated in MPO-ANCA–negative patients. 80
Renal involvement
EGPA renal lesions are less common than GPA and MPA. The study reported that 84% of patients with renal involvement were ANCA positive, and the most common presentation was acute renal failure. 81 The most common renal pathological manifestation is necrotizing oligo-immune glomerulonephritis, which is associated with ANCA positive, whereas renal pathological changes are less common in ANCA-negative patients. 81 Although rare, renal involvement is an important adverse prognostic factor in EGPA patients and poses a significant burden on the treatment and life of EGPA patients, so larger studies are needed to better characterize kidney involvement.
Ophthalmic manifestations
Of the ocular manifestations found in AAV patients, 10.8% contributed to EGPA, which was significantly lower than GPA (86.2%) but higher than MPA (3.0%). 82 Conjunctivitis and retinal vasculitis were the most common features of EGPA.82,83 Scleritis, uveitis, dacryocystitis, and/or lacrimal duct obstruction were uncommon in EGPA. Ocular damage and complications that were either irreversible or not associated with active disease were cataracts and visual impairment, which were thought to be the cause of drug toxicity rather than the disease itself for long-term use of high doses of glucocorticoids (GCs). 82
Gender difference
A study enrolled 170 EGPA patients and reported gender differences in clinical presentation and prognosis of EGPA. 84 Renal and severe gastrointestinal involvement were more frequent in the male, while more allergic manifestations including allergic rhinitis and asthma were found in female patients. 84 These differences were more pronounced before the age of 55. In addition, male patients showed more obvious active disease evaluated by BVAS and a poor prognosis assessed by Five Factor Score compared with females. 84 Similar findings were found in our previous study, in which a cluster of EGPA patients characterized by male and high serological immune abnormalities exhibited a lower incidence of allergic rhinitis and asthma, but more active and severe disease compared with the other clusters dominated by females or less immune abnormalities. 85 This finding further suggests sex differences in EGPA and the important role of immune disorders in EGPA pathogenesis.
Individualized treatments
Glucocorticoids
GCs are usually used throughout remission induction and maintenance (Table 1). GC monotherapy could reach a remission rate of over 90% in remission induction of new-onset or relapsing EGPA patients without organ-threatening or life-threatening manifestations.12,86 An initial dose of 1 mg/kg/day prednisone or equivalent (max 60–80 mg/day) is prescribed to patients, followed by a quick dose halving after 2–4 weeks, a slightly slow reduction during 3–6 months, and a minimum maintenance dose (7.5–10 mg/day or less) or discontinuance. 87 There is no universally accepted tapering strategy for GCs, nor is the duration of remission maintenance, which commonly lasts no less than 18 months. 87 In the 2021 guideline of the American College of Rheumatology/Vasculitis Foundation, the patient’s clinical condition, preferences, and values are advised as the factors to determine the duration of GC therapy. 88 It is worth noting that EGPA, as a rare heterogeneous group in AAV, is often accompanied by organ involvement that has frequently aggravated manifestations (e.g., asthma and ear, nose, and throat (ENT) symptoms) and requires GCs control. Thus, GC reduction in EGPA patients tends to be slower in the real world. To minimize GC toxicity, the lowest effective dose is preferred. This increases the need for individualized treatment and multidisciplinary management of EGPA.
Characteristics of treatment options for EGPA.

AZA, azathioprine; CD20, cluster of differentiation 20; CTX, cyclophosphamide; EGPA, eosinophilic granulomatosis with polyangiitis; ENT, ear, nose, and throat; IFN-α, interferon-α; Ig, immunoglobulin; IL, interleukin; IVIG, intravenous immunoglobulin; MMF, mycophenolate mofetil; MTX, methotrexate; TSLP, thymic stromal lymphopoietin.
Immunosuppressive therapies
Conventional immunosuppressive agents
Conventional synthetic disease-modifying anti-rheumatic drugs (DMARDs) are commonly used in the treatment of EGPA, exerting the effect of immunosuppression based on evidence available from patients with GPA and MPA (Table 1). For EGPA patients without organ-threatening or life-threatening manifestations, immunosuppressants in combination with high-dose GCs showed no significant efficacy difference from GC monotherapy in this population.86,89 Several observational studies found the efficacy and safety of methotrexate (MTX) and mycophenolate mofetil (MMF) in remission induction treatments of non-severe EGPA. However, the generality of these results requires further verification, as only a small number of EGPA patients were enrolled.90,91 As for remission induction of EGPA patients with life-threatening or organ-threatening manifestations, Cyclophosphamide (CTX) in combination with high-dose GCs is recommended. 92 Six pulsed doses of CTX to reach remission are considered acceptable for no further benefit was found by increasing the doses. 93 In the retrospective study, azathioprine (AZA) or MTX allowed for more than half of remission rates in both remission induction of non-severe EGPA and remission maintenance of severe EGPA. 94 MTX had a significantly lower cumulative GC dose during maintenance of remission compared with AZA, however, MTX was associated with more asthma and ENT relapses. 94 MTX also showed twice the risk of adverse reactions compared to AZA, mainly including gastrointestinal disturbances and high transaminase levels. 94 High-quality evidence for the use of conventional synthetic DMARDs in EGPA is scarce and well-designed randomized controlled trials (RCTs) are urgently needed given the well-tolerated and lower-cost nature of the agents.
Rituximab
B-cell clearance therapy by monoclonal anti-CD20 antibody rituximab is an effective treatment option in GPA and MPA; however, it was poorly studied in EGPA. In a systematic literature review with 171 patients who received rituximab as induction therapy, 36%–100% of the patients reached remission in 6–36 months of observation duration after rituximab initiation, and also GCs dose was effectively reduced. 95 It seems that ANCA-positive patients respond better to rituximab than ANCA-negative patients. For maintenance therapy, regular rituximab administration showed significantly reduced recurrence rates compared with those administrated on demand. 95 However, findings from the high-quality RCT of rituximab for remission induction suggested that rituximab was not superior to conventional treatment strategies in inducing remission of EGPA (REOVAS ClinicalTrials.gov number, NCT02807103). 96 Currently, another RCT is being actively carried out to compare rituximab with AZA in remission maintenance of EGPA (MAINRITSEG ClinicalTrials.gov number, NCT03164473), hopeful to provide more credible evidence for immunotherapy of EGPA (Table 2).
Clinical trials of targeted biological therapies for EGPA.

AZA, azathioprine; CD20, cluster of differentiation 20; CTX, cyclophosphamide; EGPA, eosinophilic granulomatosis with polyangiitis; ENT, ear, nose, and throat; IL, interleukin; JAK1, Janus kinase 1; TSLP, thymic stromal lymphopoietin.
Targeted biological therapies
Mepolizumab
Mepolizumab, a monoclonal antibody against IL-5, inhibits IL-5 signaling in eosinophils and the subsequent pathophysiological changes (Table 2).
The efficacy of mepolizumab in relapsing or refractory EGPA patients was produced in a multicenter, double-blind, parallel-group, phase III trial (MIRRA ClinicalTrials.gov number, NCT02020889), in which a significantly higher proportion of patients treated with mepolizumab (300 mg/4 weeks, for 52 weeks) reached disease remission (defined as a BVAS of 0 and prednisone dose ⩽4 mg), along with a longer cumulative remission period, lower annualized relapse rates and reduced GCs use compared with the placebo group. 97 Subsequently, mepolizumab was recommended for relapsing or refractory EGPA in the latest EULAR recommendations for the management of AAV. 92 Mepolizumab is primarily used in combination with GCs in remission induction of non-severe EGPA or as an additional treatment to maintain remission, with prominent efficacy in GCs reduction and respiratory symptom improvement.98,99 A phase III RCT comparing mepolizumab-based regimen to conventional therapeutic strategy (CTX or AZA) for remission induction of EGPA is recruiting (E-merge ClinicalTrials.gov number, NCT05030155). The regimen of sequential rituximab and mepolizumab was used to induce and maintain remission of EGPA patients, and the results showed that patients were well tolerated and both the systemic and respiratory symptoms were effectively improved. 100 There is still a large knowledge gap in the indication of mepolizumab for EGPA treatment. Practical evidence is still lacking in several settings, including the remission induction and remission maintenance of the severe active EGPA, efficacy in eosinophilic manifestations except respiratory system or refractory vasculitic manifestations, and reduction of immunosuppressive therapy while maintaining remission. 101 Fortunately, the observation studies are under recruiting to assess the treatment effect of mepolizumab on nasal microbial disturbance, local and systemic immune response, as well as the acute phase of EGPA neuropathy (ClinicalTrials.gov number, NCT06298448, JPRN-jRCT1061230097). These studies are expected to unlock more indications for mepolizumab treatment for EGPA. As for the dosage of mepolizumab for EGPA, retrospective observational studies have shown that both 100 mg/4 weeks and 300 mg/4 weeks had reached comparable response rates, with similar significant reductions in BVAS score, prednisone dose, and eosinophil counts. 98 Indeed, in the real world, more patients opt for doses of 100 mg/4 weeks.100,102 This may suggest that flexible dose adjustment is feasible in EGPA therapy, but a large number of high-quality clinical trials are still needed to explore the indications for dose adjustment to achieve individualized precision therapy.
Reslizumab
Reslizumab, another monoclonal antibody against IL-5, was previously approved for the treatment of severe eosinophilic asthma. 103 Different from mepolizumab, reslizumab is administered intravenously with the dose adjusted by weight, which is more friendly to dosage adjustment in obese patients compared with a fixed dosage of mepolizumab. In the open-label, pilot study, reslizumab (3 mg/kg) was used in 10 EGPA patients under stable dose of prednisone/immunosuppressant therapies except CTX (RITE ClinicalTrials.gov number, NCT02947945). The results showed that reslizumab was well tolerated by the patient and contributed to a significant reduction in daily oral GCs and improvement of BVAS (Table 2). 104 It is worth pointing out that asthma symptoms remained the most common cause of exacerbations. This result is in contrast to a previous prospective cohort where reslizumab was administered to nine refractory, oral GCs-dependent EGPA patients with severe eosinophilic asthma, which reached significant GCs-sparing effects and improvements in asthma symptom control and quality of life scores. However, there was no improvement in BVAS after reslizumab. 105 Both of the studies have a bias for the absence of a control group and a small number of subjects, thus large RCTs are needed to determine the efficacy of reslizumab for EGPA.
Depemokimab and SHR-1703
Both depemokimab and SHR-1703, are emerging long-acting anti-IL-5 monoclonal antibodies with higher affinity for IL-5 and an extended half-life,106,107 and are considered for the treatment of EGPA. The phase III trial assessing depemokimab and mepolizumab in EGPA (OCEAN ClinicalTrials.gov number, NCT05263934) and the phase II/phase III trial assessing SHR-1703 and placebo are in the recruitment phase (ClinicalTrials.gov number, NCT05979051) (Table 2).
Benralizumab
Different from the targeted monoclonal antibodies mentioned above, benralizumab works by blocking the IL-5 receptor and activating NK cell-mediated eosinophilic killing effect, which enables the depletion of eosinophils both in circulation and tissues. 101 In the phase II trials of benralizumab, good tolerance and efficacy in reducing acute exacerbations and GCs doses of the therapy in EGPA were demonstrated (BITE ClinicalTrials.gov number, NCT03010436). 108 In the phase III trials enrolling patients with relapsing or refractory EGPA, benralizumab was noninferior to mepolizumab for the remission induction of EGPA (MANDARA ClinicalTrials.gov number, NCT04157348) (Table 2). Simultaneously, data from the multicenter retrospective cohort showed that benralizumab treatment was effective in improving respiratory and systemic symptoms in real-world clinical practice.109,110
Dupilumab
Dupilumab has been approved for the treatment of moderate to severe eosinophilic asthma, chronic rhinosinusitis without nasal polyps, and atopic dermatitis by blocking IL-4 and IL-13 signaling pathways of type 2 immune response.111,112 In relapsing or refractory EGPA, dupilumab was administrated to deal with ENT or asthma manifestations with a complete response rate of 41%, however, 31% of the patients experienced an EGPA relapse, due to generally elevated blood eosinophils in patients who received dupilumab. 113 The elevation of blood eosinophil levels may contribute to either an intrinsic effect of dupilumab or a simultaneous reduction of glucocorticoid due to improved symptoms. 114 Thus, the application of dupilumab in EGPA should be cautious about eosinophilic elevation and the risk of relapse.
Omalizumab
Omalizumab, a human monoclonal antibody targeting Fc fragments of Ig E in circulation and preventing their interaction with basophils and mast cells, has prominent effects on allergic diseases.
Evidence on the use of omalizumab for the treatment of EGPA is limited. Several case series reported the efficacy of omalizumab in EGPA with severe asthmatic manifestations. 115 In the retrospective cohort study with the longest follow-up, up to 6 years, 18 EGPA cases treated with omalizumab benefited from the reduction of GCs doses, and the frequency of asthma exacerbations decreased while lung function improved. 116 On the other hand, omalizumab was reported as a risk factor for EGPA, which may be related to GC reduction, the potential direct effect of omalizumab could neither be ignored. 24 In a retrospective European collaborative study, omalizumab showed poor performance in GC-sparing effect and overall remission than mepolizumab. 99 Overall, the value of omalizumab in EGPA may be limited, given the excellent performance of various anti-IL-5 agents.
Tezepelumab
Tezepelumab, a human monoclonal antibody that targets upstream signaling of the T2 inflammatory pathway by inhibiting TSLP, has demonstrated efficacy in the treatment of asthma. A recent study reported the off-label tezepelumab treatment in two patients with severe refractory EPGA-related asthma and the result showed a reduced BVAS and increased Asthma Control Test scores, indicating the potential therapeutic effect of tezepelumab in EGPA. Also, a phase II RCT is under recruiting to explore the efficacy and mechanism of tezepelumab in EGPA (RACEMATE ClinicalTrials.gov number, NCT06230354) (Table 2).
Other potential therapies
Interferon-α (IFN-α), a known immunomodulator, was reported in small case series and showed a certain efficacy as well as transient adverse events of EGPA. 117 However, the application of IFN-α should be cautious until high-quality evidence is available. The efficacy of high-dose intravenous immunoglobulin has been demonstrated in a variety of autoimmune and systemic inflammatory diseases. Thus, the human IgG (drug code GB-0998) is under an ongoing phase III trial to explore the efficacy and safety in residual neurological neuropathy of EGPA after corticosteroid therapy (JPRN-jRCT2041220136). The small-molecule inhibitor, NS-229, targeting tyrosine-protein kinase Janus kinase 1 (JAK1), is also being validated in phase II trials (ClinicalTrials.gov number, NCT06046222) (Table 2).
Conclusion
Given the high clinical heterogeneity of EGPA, the selection of targeted therapies that target specific molecules based on their pathophysiological and immune characteristics is critical for the improvement and prognosis of EGPA. The understanding of etiology and pathogenesis will help to discover biomarkers related to EGPA phenotype and disease activity, and further facilitate the selection of individualized treatment regimens. The emerging biologic agents and the indications and efficacy of EGPA therapy summarized in the review may provide a reference for individualized drug selection and management decisions for EGPA. Further studies are needed to elucidate the long-term safety and efficacy of novel EGPA treatments and to identify indications for personalized treatments based on the individual circumstances of EGPA patients.
