Abstract
Background:
REMIT is the first real-world study of mepolizumab effectiveness in patients with severe asthma (SA) in Taiwan.
Objectives:
The primary objective evaluated changes in clinically significant exacerbations (CSEs; defined as use of oral corticosteroids (OCS) or emergency department (ED) visits and/or hospitalizations) in the 12 months pre- and post-mepolizumab treatment. Secondary objectives assessed changes in the number of CSEs requiring ED visits/hospitalizations and daily maintenance OCS (mOCS) dosage 12 months pre- and post-mepolizumab treatment. Three- and four-component clinical remissions were analyzed based on OCS-free, exacerbation-free, and asthma control (± stability in lung function).
Design:
REMIT was a retrospective, observational, self-controlled study analyzing patients in Taiwan with SA who were newly prescribed subcutaneous mepolizumab 100 mg Q4W.
Methods:
Data were extracted from records of 15 medical centers in Taiwan for patients indexed between November 1, 2018 and October 31, 2020.
Results:
A total of 170 patients were included: mean age at index date, 58.7 years; 53.5% female; 100% Chinese; 7.1% with chronic rhinosinusitis with nasal polyps, 1.8% with eosinophilic granulomatosis with polyangiitis, 1.2% with hypereosinophilic syndrome; and 55.7% with blood eosinophil count >300/µL. Pre-treatment, 71.2% had ⩾2 exacerbations, and 28.7% were on mOCS; 75.3% had no prior biologic treatment, and 24.7% had switched from other biologics. Most patients (80.0%) completed ⩾10 mepolizumab doses. Following the first mepolizumab administration (index date), CSEs reduced by 46.0% (rate ratio (RR): 0.545, 95% confidence interval (CI): 0.418–0.710; p < 0.0001) in the 12 months post-index. Exacerbations requiring ED visits/hospitalization reduced by 46.9% (RR: 0.531, 95% CI: 0.349–0.808; p = 0.0031). Median mOCS dose reduced by 100% by end of study and 81.8% of patients discontinued mOCS post-treatment. After 1 year of mepolizumab treatment, 28% and 23% patients achieved three- and four-component clinical remission, respectively.
Conclusion:
Mepolizumab use in a patient population in Taiwan with SA significantly reduced CSEs and mOCS use in routine clinical practice.
Plain language summary
• Patients with severe asthma can experience flare-ups, when symptoms become worse than usual and may require extra treatment
• Severe asthma is often treated with corticosteroids, which are oral medications (not inhalers) that can reduce asthma flare-ups, but with side effects that can impact patients’ quality of life
• Mepolizumab is a promising treatment for severe asthma. Clinical trials have shown that mepolizumab reduces flare-ups and corticosteroid use in patients with severe asthma, reducing corticosteroid side effects
• We wanted to know how well mepolizumab controls severe asthma in Chinese patients in clinical practice
• We looked at data from 170 patients with severe asthma in Taiwan who were given mepolizumab for the first time, with injections once every 4 weeks
• We compared the number of flare-ups 1 year before treatment with mepolizumab to the number of flare-ups after 1 year of mepolizumab
• After 1 year of mepolizumab, we looked at:
○ Changes in flare-ups by measuring corticosteroid use and number of emergency department visits/hospitalizations
○ Whether patients reached high level disease control (known as remission), which is when a patient does not need corticosteroids, does not experience flare-ups, has no severe asthma symptoms, and/or has no changes in lung function
• We found that, after taking mepolizumab:
○ The average number of flare-ups in patients was almost halved
○ The number of flare-ups needing emergency department visits/hospitalizations was reduced by nearly half
○ All patients stopped taking their regular daily corticosteroids. More than three quarters of patients stopped using any corticosteroids
○ 28% of patients met 3 of the remission criteria, 23% met all 4 criteria
• In Chinese patients with severe asthma, mepolizumab is effective at reducing flare-ups and corticosteroid use, helping patients achieve remission

Introduction
Severe asthma (SA), a heterogeneous condition that involves eosinophil and basophil recruitment and activation, occurs in 3%–10% of patients with asthma.1,2 The 2022 Global Initiative for Asthma defines patients with SA as those who have uncontrolled asthma despite adherence to high-dose inhaled corticosteroids (ICS)/long-acting beta-2 agonists (LABA) therapy and treatment of other contributory factors. 1 Conventional therapies, including ICS-LABA and oral corticosteroids (OCS), are effective at controlling SA in most patients; however, the detrimental side effects of such treatments may leave patients with increased hospitalizations and overall poorer quality of life compared with patients with well-controlled asthma.3 –5
Mepolizumab is a first-in-class humanized monoclonal antibody that binds with high affinity to interleukin (IL)-5 and blocks its binding to and activation of the IL-5 receptor. 6 IL-5 is a core cytokine in type 2 inflammation that is central to the development, maturation, and activation of eosinophils, a type of white blood cell implicated in the pathogenesis of SA with an eosinophilic phenotype.7,8 Evolving evidence suggests IL-5 has an impact on other cell types beyond eosinophils, leading to epithelial barrier dysfunction, airways remodeling, and disease progression in patients with asthma.9 –11
The clinical efficacy of IL-5 inhibition through treatment with mepolizumab has been demonstrated in multiple randomized controlled trials when added to standard-of-care (high-dose ICS and an additional controller). Such randomized controlled trials include the phase III MENSA and MUSCA trials, which found a decrease in annual clinical significant exacerbation (CSE) rates by 53% and 58%, respectively,12,13 as well as the SIRIUS trial, where patients on maintenance oral corticosteroids (mOCS) experienced a 32% reduction in CSEs and improvements in asthma control when treated with mepolizumab compared to placebo. 14
The effectiveness of mepolizumab has also been demonstrated in several real-world studies.15,16 In the REALITI-A study, results from the interim analysis showed that 1 year of treatment with mepolizumab was associated with a 69% reduction in the annual rate of CSEs (risk ratio: 0.31, 95% confidence interval (CI): 0.27–0.35) and a 77% reduction in the annual rate of CSEs requiring hospitalization/emergency department (ED) visits (risk ratio: 0.23, 95% CI: 0.18–0.30). In addition, the findings showed that 34% of mepolizumab-treated patients were able to discontinue mOCS use. 16 In the 2-year analysis, reduction in the annual rate of CSEs (74%) and CSEs requiring hospitalization/emergency room visits (79%) was sustained. 17 Most importantly, all patients were able to reduce their daily median mOCS dose by 100%. 17
From these studies, mepolizumab has demonstrated efficacy in patients with SA; however, real-world studies in patients with an Asian background are lacking, including those with a Chinese ethnicity. No consensus has been established on the assessments and criteria required to determine clinical remission in Asian patients with SA.
In 2016, mepolizumab was approved for treating SA in Taiwan and has been reimbursed by Taiwan National Health Insurance since November 2018 (Table S1). Although the criteria were updated in 2020, patients seeking reimbursement still require pre-approval in order to be eligible and an evaluation of treatment response is conducted at week 32 of treatment. 18
The REMIT study will help healthcare professionals to better understand patient profiles in Taiwan where mepolizumab is approved and also to provide insight on the use of the drug in Chinese populations.
Objectives
The primary objective of this study was to evaluate changes in the annualized rate of CSEs 12 months before and after treatment with mepolizumab. CSEs were defined as exacerbations requiring the use of OCS (including intravenous or intramuscular corticosteroids) and/or ED visits and/or hospitalizations.
Secondary objectives of this study were to describe changes in the annualized rate of CSEs requiring ED visits/hospitalization after treatment with mepolizumab, compared to the 12-month period prior to treatment with mepolizumab, and to describe changes in daily mOCS dosage after treatment with mepolizumab, compared to mOCS daily dose prior to mepolizumab initiation.
As exploratory objectives, we described changes in blood eosinophil count, forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC), and asthma control test (ACT) score before and after treatment with mepolizumab initiation. In addition, clinical remission after mepolizumab treatment was explored as a post hoc analysis and was defined as three- and four-component clinical remission based on OCS-free (including mOCS-free, where mOCS was defined as the use of OCS for ⩾28 days), exacerbation-free, and asthma control (± stability in lung function) during the 12-month post-index follow-up period. 19 Criteria for clinical remission endpoints are defined in detail in Table S2.
Methods
Study design
REMIT was a retrospective, self-controlled cohort study that analyzed observational data from patients with SA 12 months before and after being prescribed mepolizumab 100 mg subcutaneously every 4 weeks for the first time, as chosen by their physician. A patient’s index date was defined as the date of first mepolizumab administration; patients with an index date between November 1, 2018 and October 31, 2020 were analyzed in this study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 20
Data source and collection
Data were collected retrospectively from patient medical records beginning from the reimbursement approval date (November 1, 2018), including the 12 months before mepolizumab initiation (inclusive of the index date) and 12 months after the index date through to lost to follow-up, death, discontinuation (defined as a gap of >90 days between injections), start of another biologic, or end date of 12-month follow-up. Medical records included inpatient, outpatient, prescription, laboratory, radiology, and administration records. Electronic case report forms were used to collect data from medical records and reviewed by site nurses/investigators. Diagnoses were coded using International Classification of Diseases (ICD)-9/10 revisions.
Study sites and population
Fifteen sites met the eligibility criteria for study inclusion of having at least 10 eligible patients (Table S3). Patients who met all of the following criteria were included in the study: treatment initiation with mepolizumab between November 1, 2018 and October 31, 2020, treatment initiation with mepolizumab at least 12 months prior to the data extraction date, clinical diagnosis of asthma, age ⩾18 years at index date, and at least 12 months of medical records available prior to initiation of mepolizumab. Patients were excluded from the study population if they met any of the following criteria: mepolizumab injection during the 12 months prior to the index date, participation in an interventional clinical trial in which the treatment regimen and/or monitoring was dictated by a protocol during the previous 12 months, and missing data on date of first mepolizumab treatment (e.g., if treatment was initiated in another clinic).
Variables
Mepolizumab persistence definition
Mepolizumab persistence was defined as the duration from first mepolizumab administration to 28 days following last administration without a 90-day gap between two administrations until the end of follow-up.
Outcome definitions
Full definitions for the outcomes of interest are provided in Table 1. In brief, the primary outcome of interest was CSE, defined as patients with SA requiring either the use of systemic corticosteroids or an ED visit and/or hospital admission related to asthma exacerbation. For patients on mOCS therapy (defined based on Taiwan reimbursement criteria 18 ), a new prescription for OCS for at least double the current maintenance dose and days supply of ⩾3 days was considered a CSE. Any CSEs separated by less than 7 days were treated as a continuation of the same exacerbation.
Summary of outcome definitions.
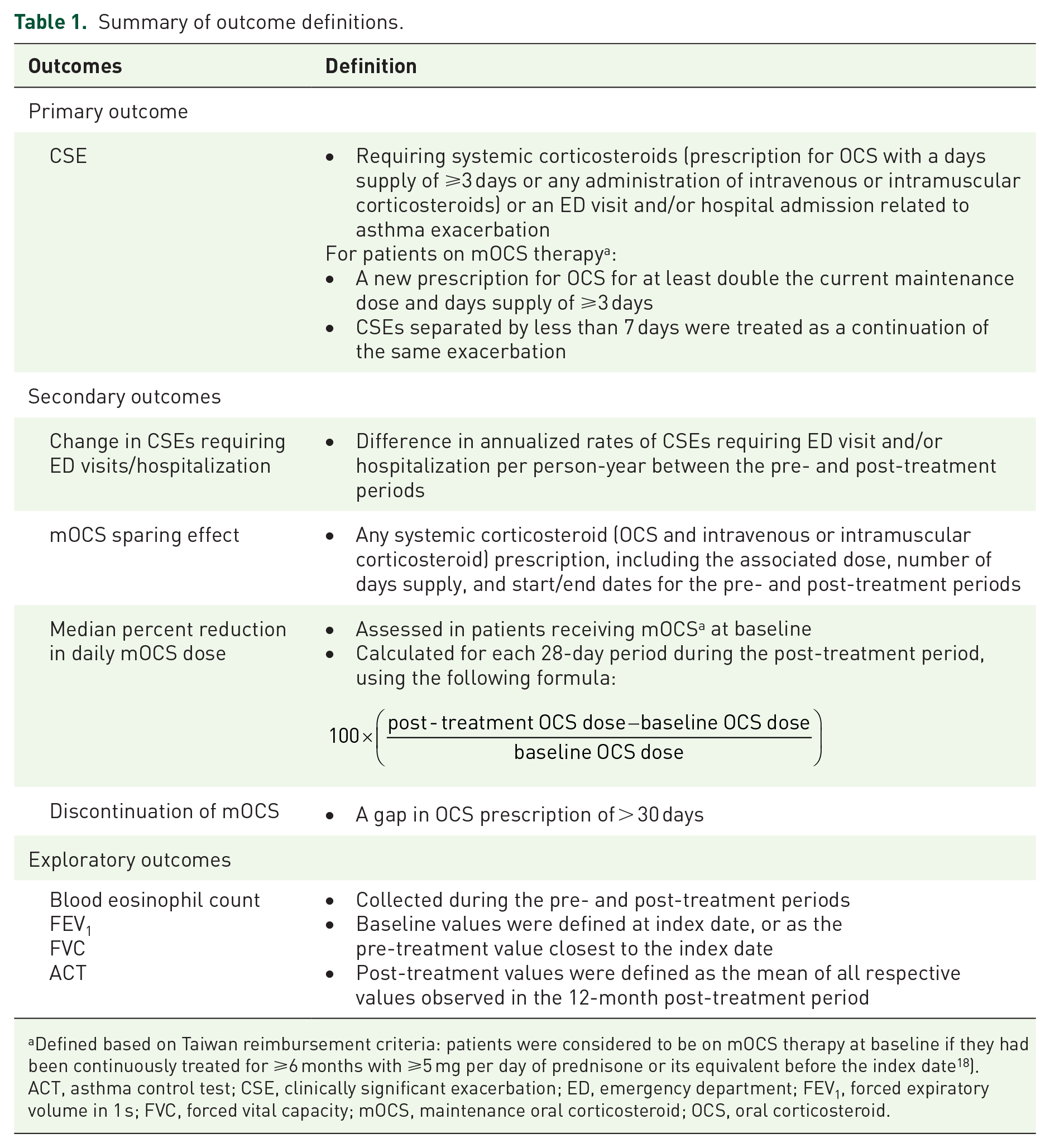
Defined based on Taiwan reimbursement criteria: patients were considered to be on mOCS therapy at baseline if they had been continuously treated for ⩾6 months with ⩾5 mg per day of prednisone or its equivalent before the index date 18 ).
ACT, asthma control test; CSE, clinically significant exacerbation; ED, emergency department; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; mOCS, maintenance oral corticosteroid; OCS, oral corticosteroid.
The secondary outcomes were changes in annualized rates of CSEs requiring ED visits and/or hospitalization and mOCS sparing effect between the pre- and post-treatment periods. For exploratory outcomes, all available blood eosinophil counts, FEV1 (L) and FVC (L) values, as well as ACT scores were collected during the pre- and post-treatment periods.
Other variable definitions include patient, clinical, and treatment characteristics, which were described using the value recorded at index date or the pre-treatment value closest to the index date. Treatment characteristics collected include concomitant use of asthma medications (assessed during the index date and the 12-month periods before and after to determine the proportion of patients who used any/each treatment and the number of administrations) and included drug name, start/end dates, dose/frequency, and administration route, and were categorized by drug treatment class; categories were not mutually exclusive and reflected all medications taken by a patient during the study period (pre-treatment, index, and post-treatment), regardless of whether such medications were received alone or combined. Treatment characteristics also included the evolution of doses of ICS-containing treatment (average doses of ICS in all ICS-containing treatments were analyzed at 3-month intervals during the 12-month periods before and after mepolizumab initiation); ICS doses for ICS/LABA and ICS/LABA/long-acting muscarinic antagonist (LAMA) were analyzed separately. Finally, the distribution of mepolizumab administrations was also collected, which was defined as the distribution of patients based on the number of mepolizumab administrations in the 12-month period after treatment initiation.
Sample size
Sample size was calculated using the most conservative estimate for reductions in exacerbations based on results from prior publications, such as the MENSA, MUSCA, and REALITI-A trials (42%–69% reduction),12,13,16,21 and with the assumption that the number of exacerbations per year follows a negative binomial distribution with a dispersion parameter κ = 0.8. 22 A study with 12 months of mepolizumab treatment designed to detect a 42% decrease in exacerbation rate with 90% power at the two-sided 5% level would require at least 73 patients.
Statistical analyses
The primary and secondary outcomes involving CSE rates were analyzed using a negative binomial regression via generalized estimating equations (GEEs) with covariates of time period (pre- and post-treatment). The logarithm of observation time in each period was used as an offset variable. The variance of the rate ratio (RR) estimate was corrected for within-subject correlation. Descriptive statistics and paired t tests or Wilcoxon signed-rank tests were used to describe other secondary outcomes and all exploratory outcomes. The annualized rate of CSEs was calculated as the number of exacerbations observed over the pre- and post-treatment periods per person-year of observation time.
Pre-treatment mOCS dose was calculated for each patient as the mean daily mOCS dose (expressed as prednisone equivalent dose, mg/day) in the 27 days before and inclusive of the index date; OCS dose was assumed to be 0 mg/day if no OCS medications were present on any given day.
Post-treatment mOCS dose was calculated as the mean daily OCS doses for each patient for every 28-day period in the post-treatment period. In patients classified as receiving mOCS at baseline, the median percent reduction in daily dose of mOCS in the post-treatment period was calculated for each 28-day period using the following formula: (100 × (post-treatment OCS dose − baseline OCS dose)/baseline OCS dose).
Post hoc analyses were conducted to assess the proportion of patients who achieved clinical remission based on OCS-free (including mOCS-free defined as the use of OCS for ⩾28 days), exacerbation-free, and asthma control (± stability in lung function) during the 12-month follow-up period after mepolizumab treatment (Table S2). Patients who died during the study period were removed from the clinical remission analysis.
Validity and sensitivity analyses
When a patient discontinued mepolizumab, their CSEs were censored and the rate was annualized in the analyses of the total population. Given that censoring of CSE data after mepolizumab discontinuation could potentially bias the study to change its conclusion, validity analyses were performed to examine whether data censoring on the annualized CSE rate would produce different statistical results that were significant enough to alter the study conclusion.
Sensitivity analyses were also performed to examine the robustness of our real-world results for the following circumstances: (1) patients who did not have post-treatment data collected between May and August 2021 (to eliminate the potential impact of COVID-19 (e.g., longer interval between administrations or reporting of exacerbations)); (2) patients who received exactly eight administrations of mepolizumab; and (3) patients for whom follow-up data on mepolizumab were not available for the full 12 months after first mepolizumab administration (“Rationale for sensitivity analyses” section in Supplemental Material).
For all three circumstances, sensitivity results were compared with the primary analysis results to determine whether they were significant enough to alter the study conclusion by using a Negative Binomial Regression via GEEs and Z test.
Results
Patient disposition
Across 15 centers, 175 patients with SA were identified by their first mepolizumab administration between November 16, 2018 and October 30, 2020 (Figure 1); 5 patients were excluded from the study as their asthma diagnosis occurred less than 6 months prior to the index date with undocumented or unverifiable exacerbation records.

Patient disposition.
In total, 170 patients were included in the analyses from the northern, central, and southern regions of Taiwan. The patients were divided into two subgroups based on predefined criteria: biologic-naïve (n = 128 (75.3%)) and biologic-switch (n = 42 (24.7%)) patients. The biologic-naïve subgroup comprised all patients in the total population who had not received biologics in the 12-month pre-treatment period; the biologic-switch subgroup comprised all patients in the total population who had switched from other biologics within the 12-month pre-treatment period.
Patient demographics and clinical characteristics
In the total population (Table 2), the mean (standard deviation (SD)) age of the patients was 58.7 (14.8) years, with 63.5% (n = 108) aged ⩾55 years; 53.5% (n = 91) of patients were female. Mean (SD) age at asthma diagnosis was 52.4 (14.4) years, with 7.1% (n = 12) and 30.0% (n = 51) as current and past smokers, respectively. In the pre-treatment period (i.e., 12 months prior to mepolizumab administration), 71.2% (n = 121) of patients experienced ⩾2 exacerbations in the past 12 months. During the baseline period, 72.9% (n = 124) patients had a prescription for any OCS use, while 28.7% (n = 48) were on mOCS. Baseline median blood eosinophil level was 336.0 cells/μL, with 55.7% (n = 93) of patients having >300 cells/μL. The most common comorbidities were chronic obstructive pulmonary disease (COPD; 42.4%), which may be due to the percentage of current and past smokers in this study (7.1% and 30.0%, respectively), and gastroesophageal reflux disease (GERD; 29.4%).
Baseline characteristics at index date for total, biologic-naïve, and biologic-switch populations.
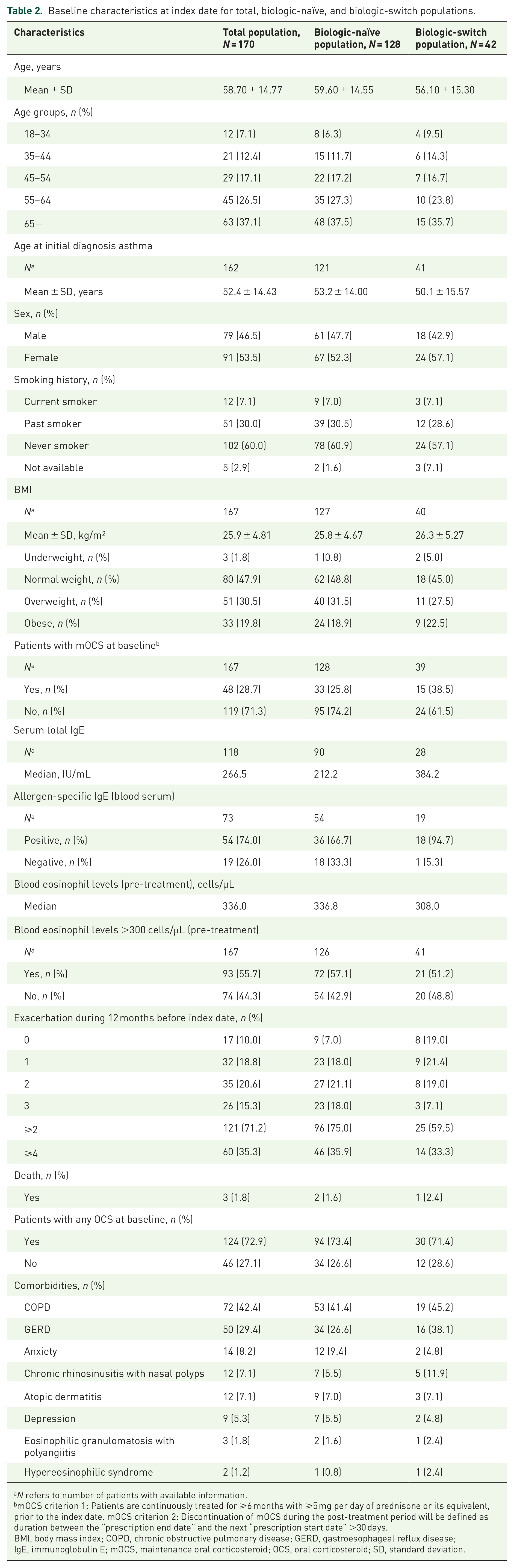
N refers to number of patients with available information.
mOCS criterion 1: Patients are continuously treated for ⩾6 months with ⩾5 mg per day of prednisone or its equivalent, prior to the index date. mOCS criterion 2: Discontinuation of mOCS during the post-treatment period will be defined as duration between the “prescription end date” and the next “prescription start date” >30 days.
BMI, body mass index; COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; IgE, immunoglobulin E; mOCS, maintenance oral corticosteroid; OCS, oral corticosteroid; SD, standard deviation.
In the subgroup populations of biologic-naïve and biologic-switch patients, there were 52.3% and 57.1% female patients, respectively. Prior to mepolizumab administration, 75.0% and 59.4% biologic-naïve and biologic-switch patients experienced ⩾2 exacerbations in the past 12 months. The proportion of biologic-naïve and biologic-switch patients on mOCS was 25.8% and 38.5%, respectively. Median eosinophil levels in the biologic-naïve and -switch patients were 336.8 and 308.0 cells/μL, respectively, with 57.1% and 51.2% of patients in each group having >300 cells/μL. Similar to the total population, the most common comorbidities were COPD (41.4% and 45.2%) and GERD (26.6% and 38.1%) for biologic-naïve and biologic-switch patients. All other characteristics in the biologic-naïve and biologic-switch patients were similar, such as age, age at diagnosis of asthma, body mass index, smoking status, and ethnicity.
Treatment regimens at index date
In the total population, 76.5% (n = 130) of patients received ICS-containing treatments alone or in combination with other asthma medications before mepolizumab administration (Figure S1). The most common treatments were ICS/LABA/LAMA; 52.4% (n = 89)), ICS/LABA (24.1% (n = 41)), leukotriene receptor antagonist (30.6% (n = 52)), and prednisone (29.4% (n = 50)).
In the biologic-naïve and biologic-switch populations, approximately three-quarters of patients in both subgroups (77.3% and 73.8%, respectively) received at least one ICS-containing medication either alone or in combination with other asthma medications. The median ICS dose, which was calculated as fluticasone propionate–equivalent doses, was 400 μg/day for both subgroups. The proportion of patients receiving each asthma medication at the index date showed small differences between biologic-naïve and biologic-switched patients; however, more biologic-switch patients (40.5%) received a leukotriene receptor antagonist at the index date than biologic-naïve patients (27.3%).
Dose evolution of ICS-containing medications (expressed as fluticasone propionate–equivalent doses)
Between the 12-month pre-index period and baseline, there was a modest increase in the median ICS dose of any ICS-containing medications for the total population (156 μg/day vs 400 μg/day) and biologic-naïve subgroup (140 μg/day vs 400 μg/day); however, there was no change in the biologic-switch group (400 μg/day vs 400 μg/day). Between baseline and the 12-month post-index period, a gradual decline was observed in the total population (400 μg/day vs 241 μg/day), biologic-naïve subgroup (400 μg/day vs 241 μg/day), and biologic-switch subgroup (400 μg/day vs 178 μg/day). A decrease in ICS dose was observed in the total population and biologic-naïve groups at 3 months post-index; however, a decrease was not observed in the biologic-switch group until 9 months post-index and the decrease continued through to the 12-month post-index period (Figure S1).
Primary outcome
Annualized CSE rates
All patients were included in the analyses except for one whose data could not be annualized due to missing records in 12-month post-index follow-up period. As a result, only 169 patients were included in the total population. Among the 169 patients, the annualized CSE rate was reduced by 46.0% from 3.02 per person-year pre-treatment to 1.65 per person-year post-treatment (RR (95% CI): 0.55 (0.45–0.71); p < 0.0001). This difference decreased to 47.6% from 3.19 per person-year pre-treatment to 1.67 per person-year post-treatment (RR (95% CI): 0.52 (0.41–0.68); p < 0.0001) in the biologic-naïve group (n = 128), and a 26.1% reduction in annualized CSE rate was seen in the biologic-switch group (n = 41) from 2.51 per person-year pre-treatment to 1.86 per person-year post-treatment (RR (95% CI): 0.74 (0.38–1.44); p = 0.3749) (Figure 2).

Annualized CSE rate 12-month before and after the index date and annualized rate of exacerbations related to ED visit/hospitalization.
Secondary outcomes
Annualized rate of CSEs requiring ED visits/hospitalization
In the total population (n = 169), the annualized rate of CSE requiring ED visits/hospitalization was reduced by 46.9% from 1.28 per person-year pre-treatment to 0.68 per person-year post-treatment (RR (95% CI): 0.53 (0.35–0.81); p = 0.0031). In the biologic-naïve subgroup (n = 128), the annualized rate of CSEs requiring ED visits/hospitalization was reduced by 40.6% from 1.41 per person-year pre-treatment to 0.84 per person-year post-treatment (RR (95% CI): 0.59 (0.38–0.93); p = 0.0214). In the biologic-switch subgroup, the annualized CSE rate requiring ED visits/hospitalization were reduced by 63.9% from 0.88 per person-year pre-treatment to 0.32 per person-year post-treatment (RR (95% CI): 0.36 (0.07–1.72); p = 0.2014) (Figure 2).
Mepolizumab persistence
In the 12-month post-index period, 80% of patients (n = 136) received ⩾10 doses of mepolizumab (Table S4 and Figure S2). At day 364, 81.8% of patients were still receiving mepolizumab without discontinuation (Table S4 and Figure S2).
Exploratory outcomes
mOCS sparing effect
At index date, 48 patients (28.7%) were on mOCS before mepolizumab administration; all 48 patients (100%) discontinued mOCS by the end of the 12-month post-index period (Figure 3). The daily median mOCS dose was reduced by 100% from a median of 5 mg/day (interquartile range (IQR): 5.0–10.0) from pre-treatment period to 0 mg/day (IQR: 0.0–5.0) at the ninth 28-day period until end of study during the post-treatment period. The decline was evident by the third 28-day period (i.e., first quarter) post-index date and continued through the 12-month post-index period.

Sparing of mOCS during 12-month post-index period for (a) total, (b) biologic-naïve, and (c) biologic-switch populations.
At the index date, more biologic-switch patients were on mOCS than the naïve patients (38.5% (n = 15) vs 25.8% (n = 33), respectively). Of those who received mOCS, the median dose was higher in the biologic-switch than that in the naïve patients (10 mg vs 5 mg, respectively). By the end of the 12-month post-index period, 100% of patients no longer required mOCS, regardless of whether the patient was biologic-naïve or biologic-switch.
Blood eosinophil levels
After mepolizumab administration, median blood eosinophil levels reduced by 89% from 336 to 36 cells/μL, which was observed by the first quarter after the index date and maintained through the 12-month post-index period (Figure 4).

Exploratory outcomes: median eosinophil counts at quarterly intervals during the 12-month post-index period for the (a) total, (b) NP, and (c) SP populations; (d) mean ACT score; and (e) mean lung function at index date and end of 12-month post-index period.
Asthma control and lung function
After mepolizumab administration, the mean ACT score of the total population improved by 3.1 points from 17.2 ± 3.89 points to 20.2 ± 2.92 points, which was both clinically and statistically significant (p < 0.0001). FEV1 (mean ± SD) at the end of the 12-month post-index period increased significantly by 0.1 L from 1.6 ± 0.7 L to 1.7 ± 0.7 L (p = 0.0011) and FVC (mean ± SD) improved significantly by 0.1 L from 2.4 ± 0.84 to 2.5 ± 0.81 L (p = 0.0301); however, these results were not clinically meaningful (Figure 4).
Clinical remission
A total of 167 patients were included in the clinical remission analyses after excluding 3 patients who died during the study period. Patients achieving each criterion for clinical remission are summarized in Table 3. The three-component clinical remission rate ranged from 23.0% to 28.0%, with the highest rate reflected in criterion 1 (no CSEs during the 12-month follow-up, no OCS over the last 28 days of the 12-month follow-up, and an ACT score ⩾20 at the last observed data of the 4th quarter of the 12-month follow-up period) (Figure 5). The four-component clinical remission rate ranged from 14.8% to 23.4%, with the highest rate reflected in criterion 6 (no CSEs during the 12-month follow-up, no OCS over the last 28 days of the 12-month follow-up, an ACT score ⩾20 at the last observed data of the 4th quarter of 12-month follow-up, and stabilized lung function (⩽100 mL decrease from baseline in FEV1) during the 12-month follow-up period) (Figure 5).
Patients achieving clinical remission endpoints in the total, biologic-naïve, and biologic-switch populations.

Definition of mOCS is an OCS course of ⩾28 days.
ACT, asthma control test; CSE, clinically significant exacerbation; ED, emergency department; FEV1, forced expiratory volume in 1 s; mOCS, maintenance oral corticosteroids; OCS, oral corticosteroid.

Clinical remission component criteria for the total population.
Validity and sensitivity analyses
Our results showed that the annualized CSE rate conclusion was not influenced by censored data in the total population. Similarly, the annualized CSE rate conclusion remained unchanged in sensitivity tests that excluded patients who did not have post-index data collected between May–August 2021, those who received exactly 8 doses of mepolizumab (i.e., did not continue mepolizumab beyond 32 weeks), or those with an annualized CSE rate in the total population (Figure S3).
The exclusion of patients who did not have post-index data collected between May and August 2021 resulted in declines of 52%, 59%, and 23% in CSEs from pre- to post-treatment in the total, biologic-naïve, and biologic-switch groups, respectively.
Declines of 48%, 51%, and 33% were observed in CSEs from pre- to post-treatment in the total, biologic-naïve, and biologic-switch groups, respectively, when patients who did not continue mepolizumab beyond 32 weeks were excluded.
When patients with an annualized CSE rate were excluded, the decline in CSEs from pre- to post-treatment were 57%, 55%, and 71% in the total, biologic-naïve, and biologic-switch groups, respectively, with the biologic-switch group result being statistically significant compared with the primary analysis (26% vs 71%, p = 0.0002).
Discussion
The REMIT study was a retrospective, self-controlled cohort study conducted to assess the real-world effectiveness of mepolizumab in a patient population with SA in Taiwan. This study demonstrated that the effectiveness of mepolizumab translates to a real-world patient population in Taiwan, with a decrease of 46.0%–71.2% in the annualized CSE rate. The findings of this study are likely to translate to patients with SA in Taiwan, as patients were recruited from across the northern, central, and southern regions of Taiwan.
Our real-world findings largely aligned with those found in global clinical trials, such as the MENSA, 12 SIRIUS, 14 and MUSCA 13 trials, where the rate of CSEs were reduced by 47%–53% in MENSA, 32% in SIRIUS, and 58% in MUSCA compared with placebo in patients with SA. Real-world studies have also been conducted in Spain (REDES), 23 United States,24,25 Japan, 26 Australia, 27 and across multiple countries (REALITI-A).16,28 Similar to the clinical trials, these studies have that found mepolizumab decreases the rate of CSEs in patients with SA. The highest reduction was observed in the REDES study where there was a reduction in CSEs up to 77.5% in the 12 months after mepolizumab initiation. 23
In this study, there was a modest increase in the median ICS dose of any ICS-containing medications during the 12-month pre-index period in the total population and biologic-naïve subgroup, which peaked at the index date, followed by a steady decrease during the 12-month post-index follow-up period. In the biologic-switch subgroup, the median ICS dose was maintained between the 12-month pre-index period and baseline; however, the median ICS dose decreased during the 12-month post-index follow-up period. These observations suggest that patients in this study may have had uncontrolled asthma before the index period; however, their need for ICS-containing medication was reduced in the 12 months after initiating mepolizumab.
According to the validity and sensitivity analyses, our primary outcome was not influenced by any of the following three circumstances identified: (1) patients who did not have post-treatment data collected in May to August 2021; (2) patients who received exactly eight administrations of mepolizumab; and (3) patients for whom follow-up data on mepolizumab were not available for the full 12 months after first mepolizumab administration. Our analyses confirmed that none of the circumstances were statistically significant to alter the conclusion of annualized CSE rate (Figure S3).
Exacerbations requiring ED visits/hospitalization were reduced by 40.6%–63.9% after mepolizumab initiation. Our findings are similar to that reported in the MENSA trial, where the annual rate of CSEs requiring ED visits/hospitalization reduced by 32%–61% in patients treated with mepolizumab compared with placebo. 12 In addition, findings from real-world studies, such as REALITI-A, found a 76%–77% reduction in CSEs requiring hospitalization,16,28 while the REDES study observed a decrease of 79.4%. 23 Multiple real-world studies have observed similar results with annual CSE rates ranging from 38.0%–77.5% to 55.8%–83.0% for CSE requiring hospitalization.16,23,24,26,28,29
By the end of this study, 81.8% of patients continued mepolizumab administration without discontinuation. The high degree of treatment compliance may due to the enforced requirement of mepolizumab administration at hospitals and the prerequisite condition for continual treatments in order to satisfy Taiwanese reimbursement criteria.
All patients with mOCS at baseline (n = 48) discontinued by end of the study. In the SIRIUS trial, the median mOCS dose was reduced by 50% in patients treated with mepolizumab compared with placebo. 14 In the real-world REDES study, 47.8% of patients were able to discontinue mOCS after 12 months with the mean daily mOCS dose reducing by 59.9% at 12 months after baseline. 23 In the REALITI-A study, the median mOCS dose decreased by 52%–75% compared to baseline, with 34%–43% of patients discontinuing mOCS at weeks 53–56.16,28
Blood eosinophil changes were similar across the populations, indicating that prior biologic use did not impact the effectiveness of mepolizumab in reducing eosinophil counts. In addition, blood eosinophil count decreased significantly from as early as the first quarter after the index date and continued through the 12-month post-index period. This observation is likely due to mepolizumab’s mechanism of action, which directly targets the IL-5 pathway to reduce eosinophil levels in both blood and airways. 6 Although no correlation tests were performed in the REMIT study between blood eosinophil levels and the rate of CSE, the correlation between blood eosinophil levels and CSE improvement has been demonstrated previously in clinical trials and post hoc analyses.12,30 This suggests that the decrease in the annualized CSE rate may be associated with the decreased eosinophil count in this study.
Interestingly, 55.7% of patients in this study had >300 cells/μL at baseline despite having received ⩾6 months of OCS treatment prior to administration of mepolizumab. This has also been observed in other studies, particularly in patients with late-onset asthma (after ⩾40 years of age), frequent exacerbations, and non-atopic asthma. 31
In our study, the FEV1 and FVC values were stable by the fourth quarter of the post-treatment period, with a mean statistically significant improvement of 100 mL. Clinical trials have found that mepolizumab either did not significantly improve FEV113,32 or only marginally improved FEV1 after treatment (<200 mL) compared with placebo. 12 However, a recent meta-analysis involving 13 real-world observational studies found slight improvements in FEV1. 33 Our results are consistent with published reports, which add to the current body of evidence indicating that eosinophilic airway inflammation correlates with CSEs and OCS responsiveness more than variable airflow limitations. 34 However, it is important to recognize that delays in treatment and airway remodeling can influence lung function results in patients with SA; thus, early treatment is essential.
In the total population, ACT improved by an average of 3 points, which was clinically and statistically significant in our study. This is supported by the meta-analysis of 13 real-world studies, which studied the effectiveness of mepolizumab in severe eosinophilic asthma, and observed an improvement in ACT score of 6.52 after 6–12 months. 33
In this study, rate annualization of the 12-month post-index period was performed in patients with missing data. Biologic-switch patients were nearly three times more likely than biologic-naïve patients to have missing data (41.5% vs 14.8%). Results from the sensitivity analyses showed that patients with annualized CSE rates confounded the CSE rate of the biologic-switch group, with a significantly higher decrease in CSE rate in the subgroup excluding patients with annualized CSE rates compared with the subgroup including patients with annualized CSE rates. This indicates that patients who discontinued mepolizumab are unlikely to benefit further from mepolizumab.
After 1 year of mepolizumab treatment, our study showed that up to 28% and 23% of patients achieved three- and four-component clinical remission, respectively. Similarly, a post hoc analysis of three- (N = 260) and four-component (N = 144) clinical remission in the REDES study showed that after 1 year of mepolizumab treatment, 38% of patients achieved three-component clinical remission (OCS-free, exacerbation-free, and ACT score ⩾20 points) and 30% of patients achieved four-component clinical remission (OCS-free, exacerbation-free, ACT score ⩾20 points, and predicted post-bronchodilator FEV1 ⩾80%). 35 Although the remission criteria differ slightly, both real-world post hoc analyses showed that at least one-quarter and one-fifth of patients achieved three- and four-component clinical remission, respectively, after 1 year of mepolizumab treatment.
In other studies where lung function was a component of clinical remission assessment, lung function was considered stabilized when FEV1% predicted post-bronchodilator was ⩾80% or <10% decline from baseline. 36 Similarly, the present study considered a ⩽100 mL decrease from baseline in FEV1 as lung function stabilization because a numerical reduction in FEV1 does not necessarily translate to actual deterioration of lung function.
Limitations
Several limitations should be acknowledged in this study. For instance, missing data from incomplete medical records or incorrect coding assignments may have affected the accuracy of comorbidity diagnoses, such as COPD, which may have been overestimated if patients presented with any obstructive airway condition that was considered to be COPD. Other comorbidities, such as nasal polyps, may also not be accurate as they were determined mainly by respiratory physicians rather than specialists. In addition, the prescription data may not have reflected actual medication use and did not provide information on medication adherence. Furthermore, subgroup sample sizes were not adequate for CSE rate analyses, and forced expiratory flow between 25% and 75% data, which is considered a more sensitive measure of lung dysfunction, is unavailable in this study.
Conclusion
The retrospective, self-controlled REMIT study demonstrated a significant reduction in annualized CSEs by mepolizumab in a real-world patient population with SA in Taiwan. Mepolizumab also decreased the annualized rate of CSEs requiring ED visits/hospitalization, reduced daily median mOCS dose, and showed high persistence in all populations. These findings align with those of previous international clinical trials and real-world studies. Our post hoc analysis provides the first real-world evidence that a subset of patients in Taiwan with SA were able to achieve the three-component clinical remission after 1 year of mepolizumab treatment. Overall, our real-world study supports the clinical effectiveness of mepolizumab in routine clinical practice in Taiwan for patients with SA and suggests that this may also apply to other regions where the majority of the population is of Chinese ethnicity.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241308406 – Supplemental material for A real-world study to evaluate effectiveness of mepolizumab in treating severe asthma in Taiwan (REMIT)
Supplemental material, sj-docx-1-tar-10.1177_17534666241308406 for A real-world study to evaluate effectiveness of mepolizumab in treating severe asthma in Taiwan (REMIT) by Shih-Lung Cheng, Shu-Min Lin, Chung-Kan Peng, Ming-Cheng Chan, Sheng-Yeh Shen, Ping-Hung Kuo, Chien-Hao Lai, Chou-Chin Lan, Chung-Yu Chen, Ching-Hsiung Lin, Kuang-Ming Liao, Po-Hao Feng, Jiin-Torng Wu, Yu-Feng Wei, Xiaomeng Xu, Rafael Alfonso-Cristancho, Tina Lai, Aldo Navarro, Dominique Milea and Diahn-Warng Perng in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank Joe Tsai and Hui Chi Liu from Formosa Biomedical Technology for the development of the clinical study report. Medical writing and editorial support for this manuscript were provided by Rebecca Yao, PhD, and Joyce Lee, PhD, CMPP™ from Nucleus Global, an Inizio company. The authors would also like to thank William Zhang for his contributions to this manuscipt.
Correction (February 2025):
The name of one of the co-authors has been corrected in the article. For more details, please see the correction notice (10.1177/17534666251322341).
Author’s note
An abstract for this study was presented at the American Thoracic Society Meeting, Washington, DC, USA (May 19–24, 2023).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
