Abstract
Background:
Guidelines advocate pulmonary function testing (PFT) in preoperative evaluation before lung resection. Although forced expiratory volume in 1 s (FEV1) and diffusing capacity of the lungs for carbon monoxide (DLCO) are recommended, they are often poor predictors of complications.
Objectives:
Determine if PFT testing results other than FEV1 and DLCO are associated with post-operative complications. We hypothesized that other PFT test results may improve the prediction of post-operative complications.
Design:
Retrospective cohort study of a single institution.
Methods:
We analyzed patients who underwent anatomic lung resections from 1/2007 to 1/2017. Percent predicted post-operative (ppo) PFT values were calculated for each test result. Outcome of interest was any post-operative complication. Wilcoxon rank-sum and multivariable regression were used to determine the relationship of PFT results to post-operative complications.
Results:
We analyzed 922 patients who underwent anatomic lung resections. Complications occurred in 240 (26.0%) patients, and mortality occurred in 12 (1.3%) patients. In univariate analysis, predicted and percent predicted post-operative (ppo) forced vital capacity (FVC), FEV1, FEF2575, DLCO, DLCO/VA, and VC values were predictors of post-operative complications. Multivariable logistic regression found no independent relationship of test results to post-operative complications, likely reflecting the collinearity of PFT results.
Conclusion:
Our findings suggest that non-traditional PFTs, such as FVC, may enhance risk stratification for post-operative complications following anatomic lung resection. Notably, traditional parameters like FEV1 and DLCO were not independently predictive, highlighting the need to reconsider their role in isolation. These findings highlight the need to reconsider how PFT are used in surgical risk stratification given high levels of collinearity.
Plain language summary
When doctors plan lung surgery, they use tests to check how well the lungs work. Two common tests are FEV1, which measures how much air you can blow out in one second, and DLCO, which checks how well your lungs move oxygen into your blood. But sometimes these tests don’t do a good job of predicting if a patient will have problems after surgery. In our study, we looked at other lung tests to see if they could help doctors better predict who might have trouble after surgery. We studied 922 patients who had lung surgery at our hospital over 10 years. We used the usual tests (FEV1 and DLCO), and we also looked at other tests like forced vital capacity (FVC), which measures how much air a person can blow out after taking a deep breath. We found that these extra tests can help give a better picture of a patient’s lung health and risk of complications. For example, FVC was helpful in predicting problems after surgery. But when we looked at all the test results together, their usefulness decreased because some of the tests measure similar things. Our study shows that while the usual lung tests are helpful, adding other lung tests could make it easier to predict problems before surgery. This might help doctors decide which patients are good candidates for surgery and which patients need different types of care.
Background
Surgical resection is currently the gold standard for the treatment of early-stage lung cancer. Surgery alone can be curative and has advantages over stereotactic radiation in that there is a lymphadenectomy performed at the time of resection, tissue for molecular analysis, and minimal residual radiographic defect remaining post-resection. However, not all patients are suitable surgical candidates due to comorbidities and chronic lung disease. Chronic lung disease is present in over 50% of newly diagnosed lung cancer patients. 1
Given the high prevalence of chronic lung disease in this population, pulmonary function testing (PFT) plays a critical role in perioperative risk assessment. Both the American College of Chest Physicians and the European Respiratory Society recommend risk stratification algorithms that rely on parameters such as forced expiratory volume in 1 s (FEV1), diffusion capacity of carbon monoxide (DLCO), and cardiopulmonary exercise testing.2,3 However, despite the widespread use of these guidelines, there is growing evidence that traditional PFT parameters, such as FEV1 and DLCO, may not reliably predict post-operative pulmonary complications in certain patient subgroups.4,5 Previous studies have indicated that some patients with seemingly poor pulmonary function can still tolerate surgery without significant complications, while others with better PFT scores may experience adverse outcomes. This suggests that FEV1 and DLCO alone may not fully capture the complexity of pulmonary risk in this heterogeneous population.6,7
To address this gap, our study aims to explore whether additional, non-traditional PFT metrics—such as forced vital capacity (FVC) and mid-expiratory flow rates—can improve perioperative risk stratification. By expanding the set of pulmonary function parameters under consideration, we seek to identify whether these additional measures provide more nuanced insights into a patient’s ability to tolerate lung resection, potentially leading to more individualized and accurate risk assessments. We hypothesize that incorporating these non-traditional PFTs could enhance our understanding of perioperative risk, helping to ensure that patients who may benefit from surgery are accurately identified, while those at higher risk of complications are appropriately managed.
Methods
We retrospectively analyzed a prospectively maintained database of patients who underwent anatomic lung resections from January 2007 to January 2017 at a tertiary-care academic medical center. The database included preoperative comorbidities, operative details, and post-operative complications. These data were supplemented with PFT results. Non-traditional PFTs were defined as any results other than FEV1 or DLCO, and included absolute values and “percent predicted” values (which are adjusted for patient characteristics according to standard nomograms) for: FVC, FEV1/FVC ratio, forced expiratory flow at 25%–75% (FEF2575), total lung capacity (TLC), residual volume (RV), and vital capacity (VC). Percent predicted post-operative (ppo) values for PFT values were calculated based on the amount of lung that would be remaining following anatomic surgical resection, assuming that there were 19 functional segments of the lung preoperatively. 3
All patients undergoing anatomic lung resection were included in the analysis. Anatomic lung resection was defined as segmental resection, lobectomy, bilobectomy, or pneumonectomy. Procedure type was defined as minimally invasive if the entire procedure was done using minimally invasive techniques (thoracoscopic or robotic), and conversions to open were defined as thoracotomy. Patients were excluded from the analysis if PFT results were not available. In addition to studying the entire cohort of patients and evaluating the PFT results individually, we also performed a subgroup “sensitivity” analysis of patients with “complete” PFT results available, defined as having results for FVC, FEV1, FEF25-75, DLCO, TLC, and VC.
The primary outcome of interest was post-operative complications. Complications were defined as adverse events which occurred within 30 days of the index surgical procedure or during the index hospitalization and included all post-operative deaths. A post-operative complication was recorded based on widely used definitions from the Society for thoracic surgeons and was not graded by severity. 8 Major morbidity was defined as pneumonia, acute respiratory distress syndrome, proctoscopy pleural fistula, pulmonary embolus, initial vent support greater than 48 h, reintubation, respiratory failure, tracheostomy, myocardial infarction, or unexpected return to the operating room, based on previous studies. 8
Statistical analysis
Descriptive statistics were performed to determine if patients receiving non-traditional PFTs were different from those receiving only FEV1 or DLCO recorded. Due to the non-normality of the data, PFT test results were compared using the Wilcoxon rank-sum test to determine if PFT results were associated with the occurrence of complications. To reduce possible bias, multivariable logistic regression was performed based on the reverse selection of these t-test results, with an alpha ⩽0.05 considered significant. Collinearity was assessed using variable influence factor (VIF) calculation. All data were analyzed using STATA/IC, Version 16 (StataCorp, College Station, TX, USA) and R (R Core Team, Vienna, Austria). 9 The study was approved by the hospital’s Institutional Review Board (CC439: CHRV0053). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 10 The STROBE checklist is submitted as a Supplemental File.
Results
There were 922 patients who underwent anatomic lung resection during the study period, of which 872 (94.6%) had available PFT results. Of the 872 patients, 393 (45.1%) were men and 479 (54.9%) were women with a median age of 67.6 years. There were 63 (7.2%) segmental resections, 701 (80.4%) lobectomies, and 34 (3.9%) bilobectomies, and 74 (8.5%) pneumonectomies. Of these anatomic resections, 626 (71.8%) were performed with a minimally invasive technique.
Post-operative complications occurred in 230 patients (26.4%), major morbidity occurred in 54 (6.2%), and mortality occurred in 11 (1.3%). “Non-traditional” PFTs results in availability varied, with 698 patients (80.1%) having at least one non-traditional result reported. The variability of non-traditional PFT results is shown in Table 1. The patient characteristics and clinical outcomes for the 698 patients with at least one non-traditional result compared to the remaining cohort of 174 patients are shown in Table 2. The two groups were similar in regards to age, sex, comorbidities, or clinical outcome.
Availability of each PFT test result among 872 patients with at least one PFT result.

FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; PFT, pulmonary function testing; RV, residual volume; TLC, total lung capacity.
Description of cohort relative to availability of non-traditional PFT results. Data presented as n (%) or mean (S.E.). n = 872 unless noted. Comparison by chi square or t-test.

ASA, American Society of Anesthesiologists; PFT, pulmonary function testing.
Among patients with PFTs available, we analyzed which PFT tests were associated with post-operative complications. The association of PFT results and post-operative complications is shown in Table 3. Predicted and percent predicted post-operative (ppo) FVC, FEV1, FEF2575, DLCO, DLCO/VA, and VC values were significant predictors of post-operative complications. The logistic regression relationship of each ppoPFT result is shown in Table 4. Notably, the percent predicted post-operative FVC showed a similar discriminatory ability (c-statistic 0.604) relative to “traditional” PFT values of ppoFEV1 (c-statistic 0.626) and ppoDLCO (c-statistic 0.619) (Figure 1). However, multivariable logistic regression found no independent relationship of test results to post-operative complications in a multivariable regression of all factors, likely because of collinearity of many test results (Table 5) A receiver operating characteristic curve analysis of the multivariable logistic regression shows a c-statistic of 0.664. To reduce the risk of possible selection bias we also performed a subgroup analysis of 442 patients with complete PFTs available. The description of that cohort is described in Supplemental Table 1 and is grossly similar to the larger cohort of patients. An analysis of PFT results and their association with post-operative complications is shown in Supplemental Tables 2 and 3 and again shows that in univariate logistic regression analysis, percent predicted postop values of FVC, FEV1, FEV1/FVC, FEF 2575, DLCO and TLC were associated with postop complications, whereas vital capacity was not.
Association of PFT results and post-operative complications. Data shown as median (IQR), and comparison performed with Wilcoxon rank-sum test. “Unadjusted” refers to test result and “percent predicted post-operative” value is adjusted based on the number of segments removed during the procedure.
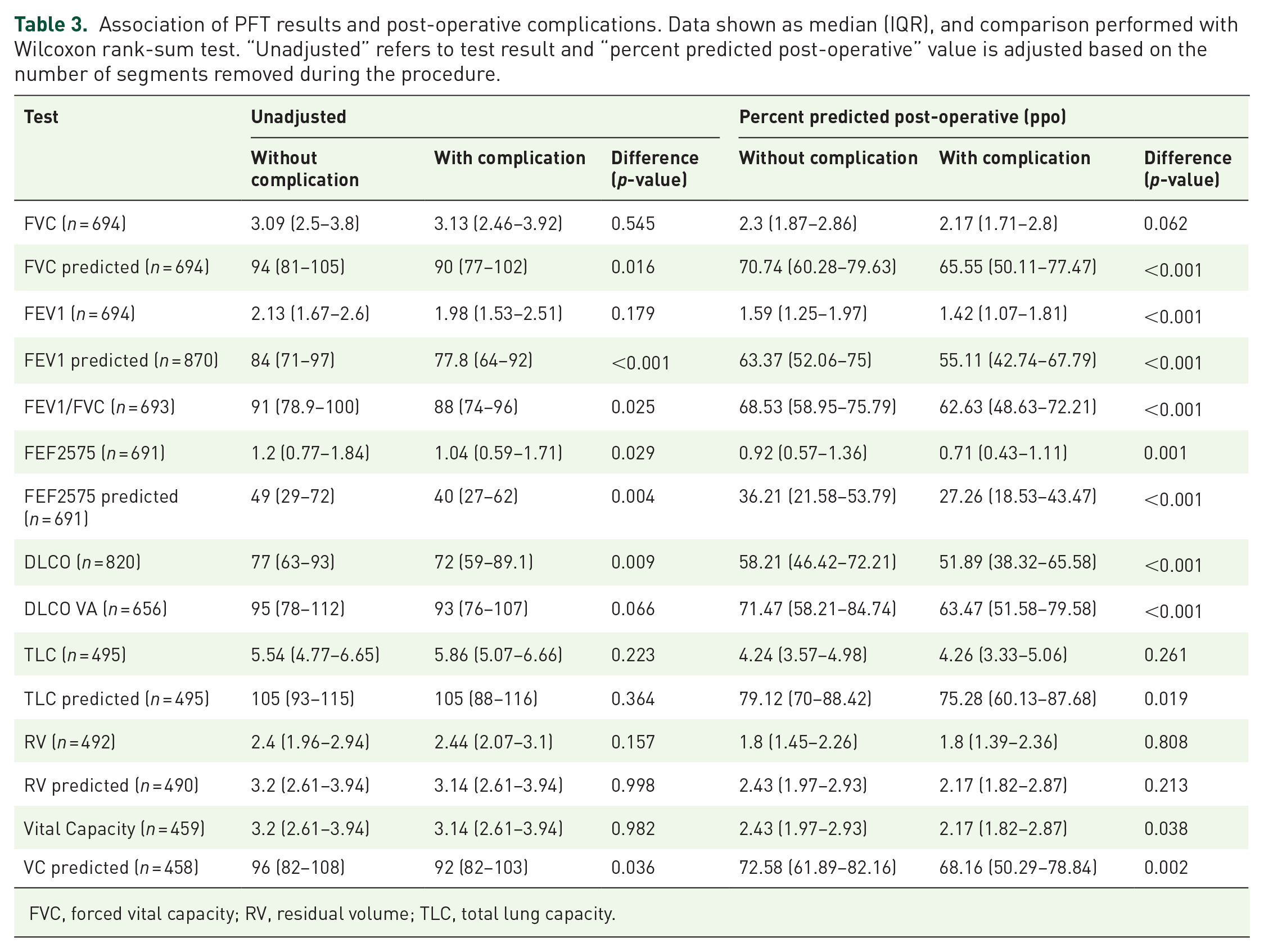
FVC, forced vital capacity; RV, residual volume; TLC, total lung capacity.
Individual logistic regression of the relationship of percent predicted post-operative (ppo) pulmonary function test result to post-operative complications. Also shown are number of patients in the analysis (n) and the c-statistic for the association.


Receiver operating characteristic curves for individual percent ppo pulmonary function test result to post-operative complications.
Multivariable logistic regression of the relationship of percent predicted post-operative (ppo) pulmonary function test result to post-operative complications. N = 442 patients with all listed test results available.

FVC, forced vital capacity; TLC, total lung capacity; VIF, variable influence factor.
Discussion
PFT is recommended for perioperative risk stratification in all patients considered for anatomic surgical resection.2,3 Current guidelines may be overly stringent in defining “high risk” as some studies have suggested that pulmonary resection can be safely performed in these patients.4,6,7,11 This means that some patients who would tolerate an anatomic resection may not be offered surgery and could potentially receive suboptimal care. Furthermore, some patients may be at higher risk than is suggested by limiting risk stratification to only FEV1 and DLCO. We hypothesized that evaluating non-traditional PFT results could offer improved risk stratification for patients undergoing anatomic lung resection as most values obtained during PFT testing are not considered in risk stratification models. Our results show that other PFT results can be used for risk stratification.
Pulmonary function and post-operative outcomes were first correlated in the 1950s. Since that time, FEV1 has been repeatedly found to be an independent predictor of post-operative morbidity and mortality.12–14 We then later learned that DLCO was a predictor of post-operative outcomes independent of FEV1. 14 Studies have suggested that looking at both factors is crucial, in that 40% of patients with a FEV1>80% will have a DLCO < 80% and 7% of those will have a DLCO < 40%, suggesting that these patients would likely be high risk for an operation. 14 Our analysis, however, suggests a more complex association between lung function and post-operative complications. By limiting risk assessment to FEV1 and DLCO alone, we may overlook the full scope of a patient’s capacity to tolerate surgery. This is supported by the improved c-statistic observed when incorporating additional PFT parameters in the multivariable analysis. While FEV1 and DLCO remain critical components of risk assessment, our findings highlight the need for a more comprehensive evaluation that considers the broader array of pulmonary function parameters. Furthermore, risk stratification should not rely solely on PFTs but also incorporate other factors, such as frailty, comorbidities, and functional status, to provide a more accurate and holistic estimation of perioperative risk.
As a “non-traditional” PFT result, FVC has been shown to predict poor outcomes in other settings, including idiopathic pulmonary fibrosis and amyotrophic lateral sclerosis (ALS).15–19 In ALS, FVC is an indicator of survival and disease progression. 15 FVC is also part of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) definition of chronic obstructive lung disease, which considers a post-bronchodilator FEV1/FVC ratio of < 0.70 as diagnostic of Chronic Obstructive Pulmonary Disease (COPD). 18 We believe this study is the first to demonstrate a relationship between FVC and perioperative outcomes in patients undergoing anatomic lung resection. Physiologically, this finding is reasonable because FVC provides a broad measure of lung volume and respiratory mechanics. Unlike FEV1, which primarily reflects airway obstruction, FVC reflects the total volume of air that can be exhaled after maximal inhalation. This means that FVC abnormalities can indicate both obstructive and restrictive lung diseases, offering a more comprehensive view of the patient’s overall respiratory function. In the context of lung resection, where both lung volume and pulmonary mechanics are critical to recovery, FVC may provide important insights that go beyond what is captured by FEV1 and DLCO alone. Patients with restrictive lung disease, in particular, may have a preserved FEV1 but a reduced FVC, signaling reduced lung compliance or increased stiffness. This could put them at higher risk for post-operative complications, as their lungs may not tolerate the reduction in functional lung volume after surgery. Conversely, patients with a high FVC may have better lung reserve and could be more resilient during the perioperative period. Thus, FVC adds nuance to risk stratification by capturing a broader spectrum of pulmonary abnormalities, which may explain its potential predictive value in this context. Nevertheless, further validation is needed to confirm these findings and to determine whether FVC should be routinely integrated into preoperative risk assessments for lung resection.
The Society of Thoracic Surgeons (STS) has developed a risk model that looks at comorbidities and other preoperative factors that may aid in predicting post-operative outcomes.8,20 While these measures are essential, we believe that additional insight can be gained from other PFT parameters, such as FVC, which may help to improve the accuracy of risk stratification. Our findings suggest that non-traditional PFT results, like FVC, could be used to refine patient selection and better predict post-operative outcomes. However, we do not advocate using FVC as a definitive criterion for decision-making in clinical practice at this time. Instead, FVC could be considered as part of a holistic evaluation that includes multiple clinical parameters. For instance, a low FVC in a patient already considered at high risk might provide additional support for caution, while a strong FVC in a borderline-risk patient could offer reassurance. Further studies are necessary to validate the role of FVC and other non-traditional PFTs in risk stratification before they can be routinely integrated into clinical practice.
Limitations
This study has several weaknesses. As a retrospective study, we cannot account for patients with abnormal lung function who were not offered surgery, potentially introducing selection bias. It is possible that some patients with poor PFT results, such as severely reduced FEV1 or DLCO, were excluded from surgery preoperatively based on clinical judgment. Consequently, the study population may represent a selection of patients deemed “fit” for surgery, thus skewing the post-operative outcomes toward those with more favorable PFT results. This could limit the generalizability of our findings, particularly for higher-risk patients who were never offered surgery. Furthermore, because our study focuses only on patients who underwent anatomic lung resection, it is possible that those with poor pulmonary function who might have been considered too high-risk for surgery are underrepresented in our analysis. This inherent selection bias may reduce the ability to fully explore the predictive value of PFTs across the entire spectrum of lung function. To better understand the applicability of non-traditional PFTs like FVC, future prospective studies should include all patients being evaluated for lung resection, regardless of whether surgery is ultimately offered, to capture a broader risk profile. Furthermore, not every patient had FVC results available, which may offer additional bias. We believe that this effect is likely minimal as there was no demographic or outcome difference between those patents with and without “non-traditional” PFT results. Surgical procedures were predominately thoracoscopic, which may have affected the relationship of PFT results and complications. As such, these results may not be generalizable to a broader population of patients, or at least where thoracotomy was performed with higher frequency. Furthermore, other risk factors for surgery were not included in this analysis, which may bias the results. For example, age and other comorbidities are not included and are likely unmeasured confounders in the outcome of interest. The purpose of this study was not to create a “perfect” risk model for surgical complications but rather to evaluate how PFT results should be considered in risk stratification, and which additional variables may be of interest. Additionally, while our analysis adjusted for the extent of resection using percent predicted post-operative (ppo) values, it is possible that certain procedure-related risks, such as those associated with technical aspects of the surgery, may not be fully captured by the ppo calculation. These additional factors may contribute to the overall risk of complications but were not explicitly included in the model.
A strength of the study is that the majority of the literature on outcomes and PFTs was published in the era of thoracotomy and this analysis includes a relatively large sample size of patients undergoing thoracoscopic procedures.2,13,14 Minimally invasive surgery decreases morbidity and mortality, and is performed with increasing frequency in the United States. 21 Therefore, these results may more accurately predict the risk of complications in patients undergoing minimally invasive anatomic lung resection relative to findings from the “landmark” studies prior to widespread use of thoracoscopic techniques.22 –24
Conclusion
Although PFT is performed frequently, current risk stratification models do not utilize all available data obtained from the test. Furthermore, FVC is incorporated into the Gold criteria for COPD and is associated with poor outcomes in other disease processes, but is not commonly included in surgical risk stratification. Herein, we show that abnormal FVC is correlated with adverse events. We recommend further study of FVC in surgical patients and advocate for including FVC in the future studies of perioperative risk.
Supplemental Material
sj-jpg-1-tar-10.1177_17534666241305954 – Supplemental material for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review
Supplemental material, sj-jpg-1-tar-10.1177_17534666241305954 for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review by Christopher W. Towe, Avanti Badrinathan, Alina Khil, Christine E. Alvarado, Vanessa P. Ho, Aria Bassiri and Philip A. Linden in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpg-2-tar-10.1177_17534666241305954 – Supplemental material for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review
Supplemental material, sj-jpg-2-tar-10.1177_17534666241305954 for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review by Christopher W. Towe, Avanti Badrinathan, Alina Khil, Christine E. Alvarado, Vanessa P. Ho, Aria Bassiri and Philip A. Linden in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpg-3-tar-10.1177_17534666241305954 – Supplemental material for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review
Supplemental material, sj-jpg-3-tar-10.1177_17534666241305954 for Non-traditional pulmonary function tests in risk stratification of anatomic lung resection: a retrospective review by Christopher W. Towe, Avanti Badrinathan, Alina Khil, Christine E. Alvarado, Vanessa P. Ho, Aria Bassiri and Philip A. Linden in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
