Abstract
Background:
Perioperative heparin-free anticoagulation extracorporeal membrane oxygenation (ECMO) for lung transplantation is rarely reported.
Objective:
To evaluate the impact of a heparin-free strategy on bleeding and thrombotic events, blood transfusion, and coagulation function during the early perioperative period and on prognosis, and to observe its effect on different ECMO types.
Design:
A retrospective cohort study.
Methods:
Data were collected from 324 lung transplantation patients undergoing early perioperative heparin-free ECMO between August 2017 and July 2022. Clinical data including perioperative bleeding and thrombotic events, blood product transfusion, coagulation indicators and 1-year survival were analysed.
Results:
Patients were divided in venovenous (VV; n = 251), venoarterial (VA; n = 40) and venovenous-arterial (VV-A; n = 33) groups. The VV group had the lowest intraoperative bleeding and thoracic drainage within 24 h postoperatively. Vein thrombosis occurred in 30.2% of patients within 10 days postoperatively or 1 week after ECMO withdrawal, and no significant difference was found among the three groups. Double lung transplantation, increased intraoperative bleeding, and increased postoperative drainage were associated with vein thrombosis. Except for acute myocardial infarction in one patient, no other serious thrombotic events occurred. The VV-ECMO group had the lowest demand for blood transfusion. The highest prothrombin time and the lowest fibrinogen levels were observed in the VA group during ECMO run, while the highest platelet counts were found in the VV group. Both intraoperative bleeding and thoracic drainage within 24 h postoperatively were independent predictors for 1-year survival, and no thrombosis-related deaths occurred.
Conclusion:
Short-term heparin-free anticoagulation, particularly VV-ECMO, did not result in serious thrombotic events or thrombosis-related deaths, indicating that it is a safe and feasible strategy for perioperative ECMO in lung transplantation.
Keywords
Introduction
Lung transplantation is the mainstay for managing end-stage lung disease. 1 Extracorporeal membrane oxygenation (ECMO) has been increasingly used in lung transplantation to provide intraoperative extracorporeal respiratory and/or circulatory support and shows significant beneficial effects, including superior survival, decreased incidence of primary graft dysfunction (PGD), and acceptable complications.2–5 However, intraoperative bleeding remains a major clinical concern, and severe postoperative bleeding is associated with decreased 60-day survival after lung transplantation.6,7 Additionally, large-volume blood product transfusion and a high frozen plasma to red blood cell ratio are associated with worsened 72-h PGD scores and mortality.8,9
The interface between blood and non-biological ECMO circuit elements activates coagulation pathways, resulting in the consumption of both prothrombotic and anti-thrombotic factors and increasing the rate of thrombosis and bleeding. 10 Therapeutic anticoagulation is recommended by the Extracorporeal Life Support Organization when possible. 11 During lung transplantation, intraoperative ECMO usually requires pre-emptive intravenous administration of unfractionated heparin to achieve systemic heparinization.2–5 However, the optimal heparin dose and application mode of intraoperative ECMO anticoagulation are controversial. 3 Anticoagulation increases risks of intraoperative and postoperative bleeding and the requirement for blood product transfusions. Coatings with good biocompatibility on ECMO circuit surfaces were designed to decrease the risks of bleeding and thrombosis. Heparin-free anticoagulation ECMO is increasingly used in critically ill patients with a high bleeding risk.12–15 Low-intensity anticoagulation ECMO with low molecular weight heparin reduces thrombotic events during lung transplantation. 16 Heparin-free venoarterial (VA) ECMO strategy does not increase the incidence of perioperative thrombotic events or bleeding requiring reoperation. 17 In addition, a case series reported the perioperative use of heparin-free anticoagulation ECMO in lung transplantation with a high risk of haemorrhage. 18 Lung transplant recipients in China are typically older patients with poor coagulation and larger surgical trauma; therefore, routine use of heparin inevitably increases bleeding risk. In August 2017, our centre initiated a heparin-free anticoagulation strategy for ECMO during the intraoperative and early postoperative period. No significant adverse clinical events were recorded in the initial patients. Subsequently, this strategy became routine protocol in our centre.
In this study, we analysed the clinical data of lung transplantation patients supported with perioperative heparin-free anticoagulation ECMO over a 5-year period. We aimed to determine the impact of heparin-free ECMO on bleeding and thrombotic events, blood transfusion and coagulation function during the early perioperative period and on prognosis. We also assessed the effect of heparin-free anticoagulation on different ECMO types.
Methods
Patient selection
This study was registered in the Chinese Clinical Trial Registry (No. ChiCTR2300076634). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19 Data on lung transplantation recipients admitted to the China-Japan Friendship Hospital, from August 2017 to July 2022, were retrospectively collected. All donor lungs were derived from voluntary donors assigned by the China Organ Transplant Response System. The inclusion criteria were as follows: (1) age > 18 years and (2) intraoperative ECMO support with a heparin-free strategy. The following exclusion criteria applied: (1) preoperative ECMO support >24 h, (2) death within 24 h after surgery. Figure 1 shows the study flow chart. No anticoagulant or antiplatelet drugs were administered 24 h before surgery in all patients. Sodium citrate anticoagulation was administered to all patients requiring renal replacement therapy during ECMO.

Flow chart of the Cohort.
ECMO implantation, management and withdrawal
The application of preoperative bridging ECMO was determined by the intensive care unit (ICU) physicians. The usage and types of intraoperative ECMO were based on discussions between surgeons, anaesthesiologists and ICU physicians, and the application and withdrawal indications for ECMO were shown in the Supplemental Material. ECMO was established percutaneously under ultrasound guidance with the Seldinger technique using puncture catheters (Maquet, Germany). For venovenous (VV) ECMO, the femoral vein on one side served as the drainage end, and the right internal jugular vein was the reflux end. For VA ECMO, the femoral vein on one side was the drainage end, and the femoral artery on the other side was the reflux end. The membrane oxygenator kit was supplied by Sorin Company (Italy) or Maquet. The heparin-free anticoagulation was administered. After surgery, patients were transferred to the ICU and ECMO withdrawal was evaluated every morning. Coagulation indicators, including platelet counts activated clotting time, prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen and D-dimer, were routinely evaluated. The femoral artery required surgical repair for VA and VV-A ECMO patients. Within 2 days of ECMO withdrawal, thromboprophylaxis was administered after assessing the risk of bleeding from the chest and other sites. Thromboprophylaxis was started with a fixed dose of nadroparin calcium 4100 IU qd or enoxaparin 2000 IU qd, and adjusted by clinical evaluation.
The heparin-free anticoagulation strategy was defined as no heparin or heparin derivatives for ECMO anticoagulation during surgery, in the first 3 days after surgery (if ECMO run > 72 h) or during the postoperative ECMO run period (if ECMO run ⩽ 72 h). In addition, for ECMO established for bridge to lung transplantation within preoperative 24 h, no heparin was used before surgery.
Data collection
We collected information including demographics, medical history, lung transplantation data, ECMO duration, PGD grade 3 at 72 h, and 1-year survival. Use of ECMO with bilateral pulmonary oedema on chest X-ray image was graded as grade 3 according to the current PGD scoring system. 20 Moreover, coagulation indicators, bleeding and thrombotic events, and blood product transfusions were recorded. The coagulation indicators were monitored from pre-surgery to 2 days after ECMO withdrawal. Day 1 was defined as the time of routine blood collection on the morning after ECMO withdrawal. Bleeding and thrombotic events were recorded from ECMO initiation to 7 days post-ECMO withdrawal (if ECMO run ⩽3 days) or within the first 10 days after ECMO run (if ECMO run >3 days). Bleeding events included the amount of intraoperative bleeding, the amount of thoracic drainage within 24 h postoperatively, intracranial haemorrhage (ICH), significantly visible gastrointestinal bleeding, and bleeding at puncture sites. Thrombotic events included ultrasound-determined thrombosis in the internal jugular vein and extremity vessels, and severe thrombotic events such as limb necrosis, acute myocardial infarction (AMI), acute cerebral infarction, pulmonary embolism and mesenteric arteriovenous thrombosis. Data on blood product transfusion during surgery and within 24 h postoperatively were collected.
Statistical analysis
All data were analysed using IBM SPSS Version 27.0 software (SPSS, Chicago, IL, USA). Data with a normal distribution were presented as the mean ± standard deviation, and student’s t-test was used for between-group comparisons. For skewed-distribution data, variables were expressed as the median (25–75th percentiles), and the Kruskal–Wallis and Mann–Whitney U tests were used for between-group comparisons. The paired sample t-test or Wilcoxon signed-rank test was used for self-control data. Qualitative variables were compared using a continuity table followed by the chi-square test. Generalized estimated equation was used for dynamic analysis. The optimal thresholds were determined by the receiver operating characteristic (ROC) curves according to the Youden index. Using optimal thresholds, Kaplan–Meier survival curves were plotted, and the log-rank tests were applied for comparisons of survival distribution. Binary logistic regression was used to identify the variables associated with vein thrombosis and 1-year survival. All statistical tests were two-tailed, and p-values <0.05 were considered statistically significant.
Results
Clinical characteristics
This study included 324 lung transplantation recipients with intraoperative ECMO support. Based on ECMO types, patients were divided into VV, VA and VV-A groups, and VV-ECMO was the main support mode (Table 1). Overall, interstitial lung disease was the primary cause of lung transplantation, although the causes varied among the three groups. The VV group had the lowest proportion of cases of double lung transplantation and the shortest ECMO duration, ventilator time and ICU stay. In addition, the lowest pulmonary arterial pressure was observed in the VV group (Supplemental Table 1).
Patient demographic data and clinical characteristics.
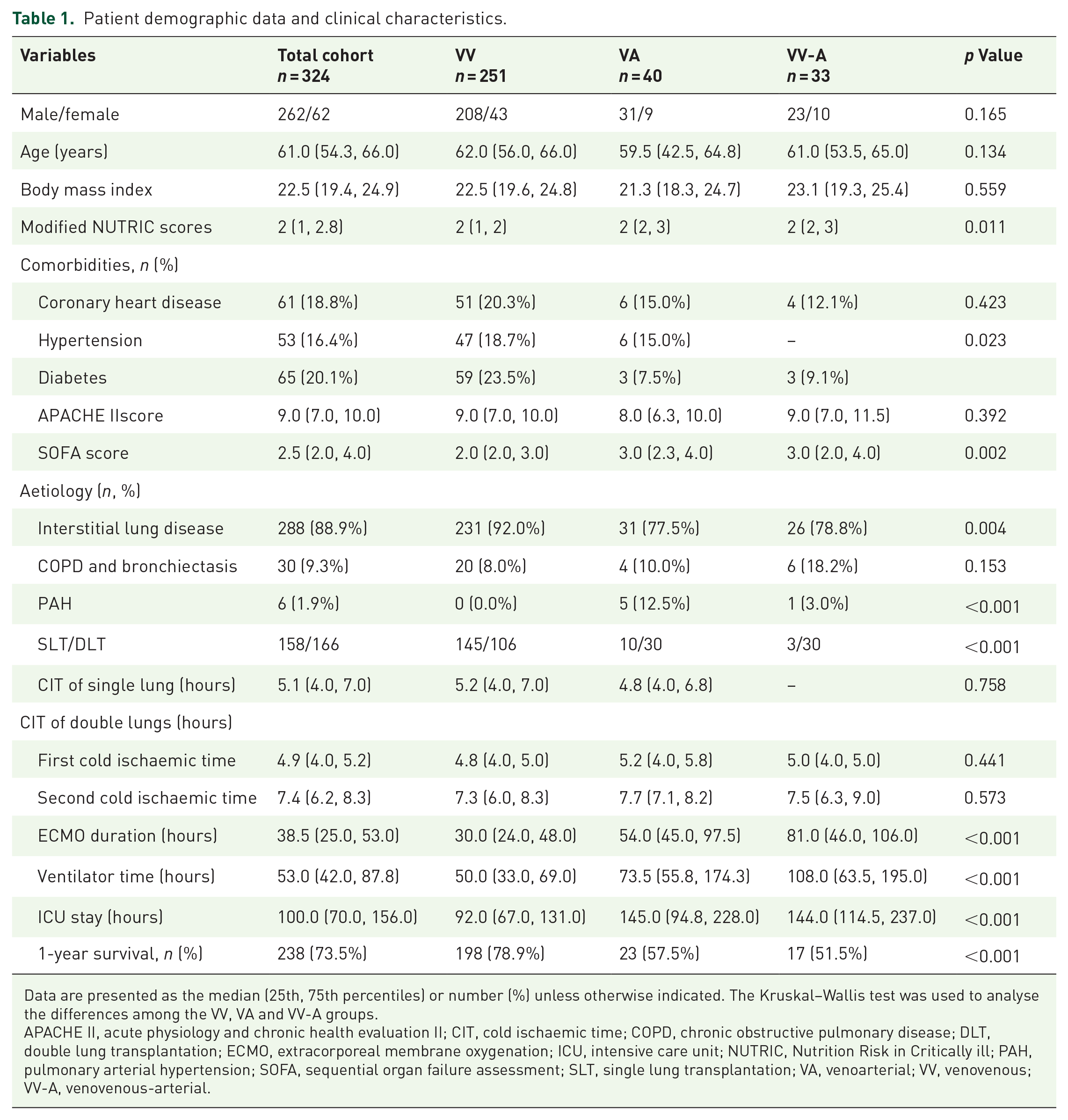
Data are presented as the median (25th, 75th percentiles) or number (%) unless otherwise indicated. The Kruskal–Wallis test was used to analyse the differences among the VV, VA and VV-A groups.
APACHE II, acute physiology and chronic health evaluation II; CIT, cold ischaemic time; COPD, chronic obstructive pulmonary disease; DLT, double lung transplantation; ECMO, extracorporeal membrane oxygenation; ICU, intensive care unit; NUTRIC, Nutrition Risk in Critically ill; PAH, pulmonary arterial hypertension; SOFA, sequential organ failure assessment; SLT, single lung transplantation; VA, venoarterial; VV, venovenous; VV-A, venovenous-arterial.
Bleeding and thrombotic events and the risk factors of vein thrombosis
Compared with VA and VV-A groups, the VV group had the least intraoperative bleeding and thoracic drainage of blood within 24 h postoperatively (Table 2). In addition, seven patients including four in the VV group and three in the VV-A group, underwent emergency thoracotomy for haemostasis due to massive postoperative thoracic bleeding. Except for one ICH in the VA and VV-A groups respectively, there were no other severe extra thoracic haemorrhage events.
Bleeding and thrombotic events.

VA, venoarterial; VV, venovenous; VV-A, venovenous-arterial.
Vein thrombosis occurred in 30.2% of patients within 7 days after ECMO withdrawal (if ECMO run ⩽ 3 days) or within the first 10 days after ECMO run (if ECMO run > 3 days), and no significant differences were found in different sites among the three groups. The rate of femoral artery thrombosis was similar between the VA and VV-A groups (15.0% vs 15.2%). Except for AMI in one patient, no other severe thrombosis events occurred.
Risk factors for vein thrombosis were evaluated using logistic regression, including variables shown in Table 1, blood product transfusion, intraoperative bleeding and postoperative bleeding. ROC analyses showed that thoracic drainage within postoperative 24 h and intraoperative bleeding had a good discriminatory ability for predicting vein thrombosis (Supplemental Figure 1A). After adjustment for age and sex, double lung transplantation, intraoperative bleeding (cut-off value ⩾ 375 ml), and thoracic drainage within 24 h postoperatively (cut-off value ⩾ 555 ml) were identified as risk factors for vein thrombosis (Table 3).
Risk factors for vein thrombosis.

CI, Confidence Internal; DLT, double lung transplantation; SLT, single lung transplantation; OR, odds ratio.
Transfusion of blood products
Overall, 67.3% of patients had intraoperative blood transfusions (Table 4). However, only 42.9% of patients required allogeneic transfusion. Compared with the VA and VV-A groups, the VV group had the least transfusion of both packed red blood cells (PRBCs) and frozen plasma. Within 24 h after surgery, 62.3% of patients required allogeneic transfusion. The VV group had the lowest demand for PRBCs, Fresh frozen plasma, and platelets both in terms of transfusion rates and transfusion volumes.
The transfusion of blood products.

FFP, fresh frozen plasma; PRBCs, packed red blood cells; VA, venoarterial; VV, venovenous; VV-A, venovenous-arterial.
Dynamic changes in coagulation indicators
To explore the effects of short-term heparin-free ECMO on the coagulation system, we tried to exclude potential factors affecting coagulation (Supplemental Figure 2) and observe the changes in coagulation indicators from pre-surgery to day 2 after ECMO withdrawal (Figure 2). APTT, PT and D-Dimer levels increased and then decreased, although levels did not return to preoperative levels on day 2 after withdrawal. Fibrinogen levels first decreased and then returned to preoperative levels on day 1 after ECMO withdrawal. Platelet count decreased after ECMO run and did not return to preoperative levels 2 days after withdrawal. Overall, coagulation indicator and haemoglobin level trends were consistent among the three groups. Except for higher PT and lower fibrinogen levels during ECMO run in the VA group compared with the VV group, no significant difference was found among the three groups at every time point. Furthermore, the highest platelet counts after ECMO run were observed in the VV group.
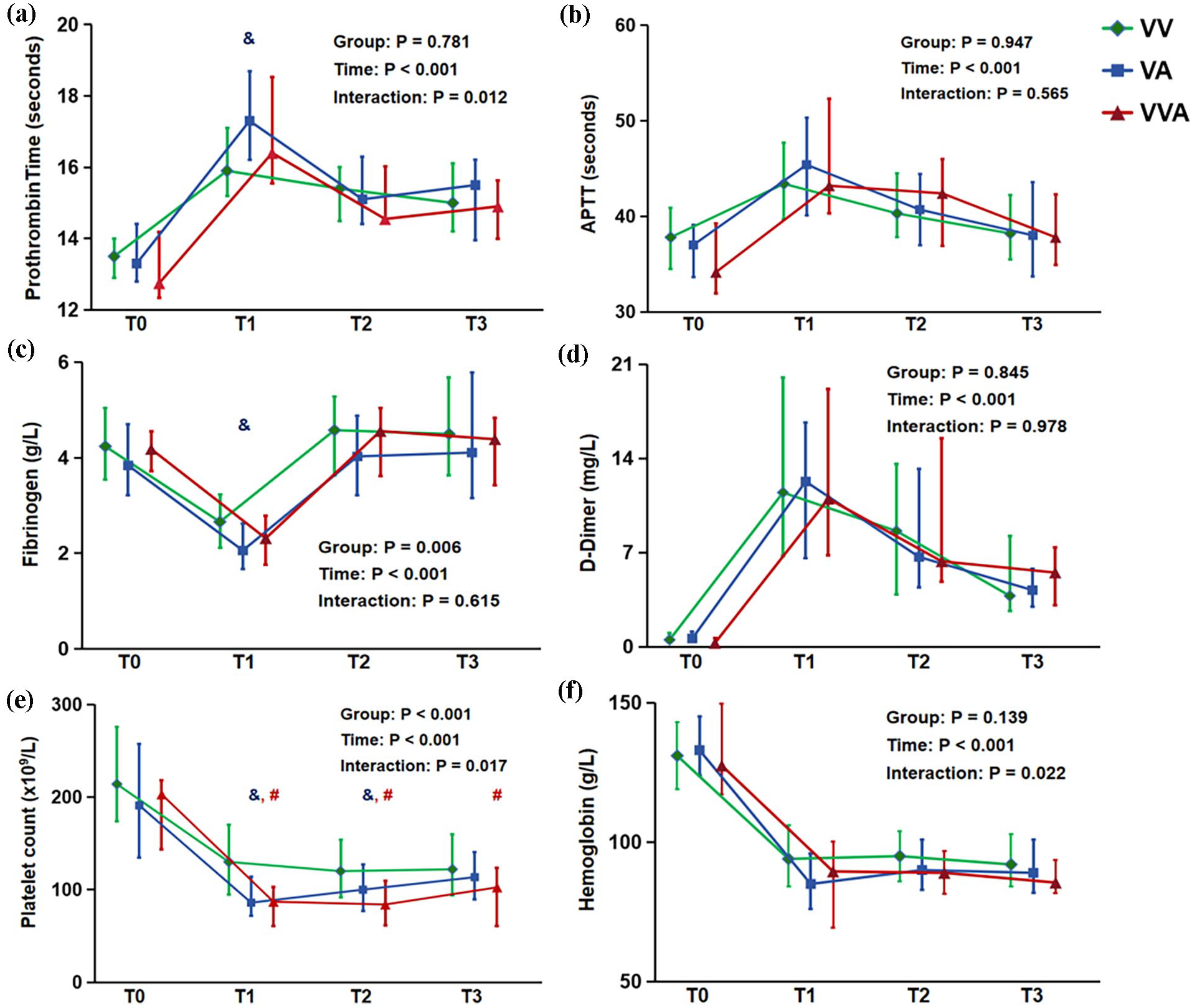
The impact of heparin-free anticoagulation strategy on the coagulation system. (a)–(e): The dynamic changes of prothrombin time, APTT, fibrinogen, D-Dimer and platelet counts from ECMO run to day 2 after ECMO withdrawal. (f) The dynamic changes in haemoglobin levels.
Death causes and prognostic factors of 1-year survival
Supplemental Table 2 lists the specific causes of death. Overall, severe infection was the primary cause of death. Two patients died of ICH on postoperative days 2 and 10, and 4 patients died of severe thoracic haemorrhage. However, no thrombosis-related death occurred.
Prognostic factors of 1-year survival were evaluated, including variables shown in Table 1, blood product transfusion, and intraoperative and postoperative bleeding. ROC analyses showed that thoracic drainage within 24 h postoperatively and intraoperative bleeding had discriminatory ability for predicting 1-year survival (Supplemental Figure 2(B)). In univariable and multivariable models, thoracic drainage within 24 h postoperatively (cut-off value ⩾ 685 ml) showed prognostic ability (Table 5). Comparatively, intraoperative bleeding (cut-off value ⩾ 475 ml) showed the prognostic ability in univariable, not multivariable models. Using the designated cut-off value, Kaplan–Meier curves were plotted. Lung transplantation patients with intraoperative bleeding and thoracic drainage within 24 h postoperatively that was higher than the above optimal thresholds had a lower probability of 1-year survival (Figure 3(a) and (b)). Furthermore, ECMO type and platelet transfusion also affected 1-year survival (Figure 2(c) and (d)).
Predictive factors for 1-year survival.

CI, Confidence Internal; OR, odds ratio; VA, venoarterial; VV, venovenous; VV-A, venovenous-arterial.

Kaplan–Meier curves for 1-year survival after lung transplantation based on different prognostic factors. (a) and (b) According to optimal thresholds of intraoperative bleeding and thoracic drainage within postoperative 24 h determined by receiver operating characteristic curves based on 1-year survival, Kaplan–Meier survival curves showed statistically different outcomes. (c) ECMO types; (d) Platelet transfusion.
Correlation between bleeding and clinical outcomes
Both intraoperative bleeding and thoracic drainage within postoperative 24 h showed a low positive correlation with ECMO duration, ventilator time and ICU stay (Supplemental Figure 3).
The PGD grade 3 at 72 h and perioperative bleeding
At postoperative 72 h, 49 (15.1%) patients including 18 (7.2%) in the VV group, 12 (30.0%) in the VA group and 19 (57.6%) in the VV-A group, had PGD grade 3. PGD grade 3 rates were compared based on the above thresholds of intraoperative bleeding (475 ml) and thoracic drainage (685 ml). PGD grade 3 rates were lower in patients with less intraoperative bleeding (8.4% vs 22.2%, p = 0.001) or less postoperative thoracic blood drainage (7.8% vs 27.5%, p < 0.001). Furthermore, patients with PGD grade 3 had increased intraoperative bleeding (400 (200, 800) ml vs 600 (400, 1000) ml, p = 0.001) and postoperative thoracic haemorrhage (530 (380, 790) ml vs 900 (590, 1660) ml, p < 0.001).
Discussion
To the best of our knowledge, the impact of perioperative heparin-free anticoagulation ECMO on coagulation indicators, bleeding and thrombotic events, and prognosis in lung transplantation have not been previously examined. The main findings of this study were as follows. (1) Compared with VA and VV-A groups, the VV group had the least intraoperative bleeding and thoracic drainage within 24 h postoperatively. (2) Venous thrombosis occurred in nearly one-third of lung transplantation patients, and severe extra thoracic bleeding events were rare. (3) The VV group had the lowest demand for blood product transfusion. (4) Coagulation indicators exhibited similar trends among the three groups, while the VV group had the highest platelet counts during ECMO run, and the VA group had the highest PT and the lowest fibrinogen levels. (5) Few patients died of severe bleeding, and no thrombosis-related death developed. (6) Intraoperative bleeding and thoracic drainage within 24 h postoperatively were independent predictors for 1-year survival. (7) Patients with less intraoperative bleeding and thoracic drainage had a lower incidence of PGD grade 3.
In the absence of pre-existing coagulopathy, haemostatic dysfunction during ECMO occurs due to exposure of blood to non-biologic surfaces of the ECMO circuit and shear stress.10,21 Mechanical forces provoke activation of platelets and coagulation factors, fibrinogen deposition, adherence to device surfaces, and thrombin generation. Thus, the coagulation system in patients undergoing ECMO is initially tilted toward a procoagulatory state. For intraoperative ECMO, most centres administer intravenous unfractionated heparin to achieve heparinization. The dose differs from 40 to 75 IU/kg or 3000–5000 IU.2,5,22,23 However, lung transplantation patients in China have different degrees of bleeding risk, which manifest as extensive pleural adhesions and extensive trauma during intraoperative lung separation, leading to significant bleeding.4,24–26 Thus, lower heparin doses tend to be administered in China. 4
In this cohort using a heparin-free strategy over a 5-year period, intraoperative bleeding appeared to be lower than that previously reported in China and other countries.4,24–28 Different ECMO types have different impact on bleeding and thrombotic events,29–31 and different transfusion demands. 31 We found that the VV group showed the least intraoperative bleeding and thoracic drainage. Only seven patients experienced emergency thoracotomy for haemostasis due to postoperative severe thoracic bleeding, and this ratio was significantly lower than that reported in other studies.4,32 Furthermore, except for two patients with ICH, there no severe extra thoracic haemorrhage events were recorded. Thus, the heparin-free strategy appears to contribute to reducing perioperative bleeding.
Vein thrombus occurred in nearly one-third of the patients, and no significant difference was found based on ECMO type or involved vein. The reported incidence of early vein thrombosis varies from 17.3% to 29.9% among the different centres.4,33–35 Previous studies included patients without ECMO support which was an independent risk factor of vein thrombosis. In the current study, except for AMI in one patient, no severe thrombosis event such as pulmonary embolism or thrombosis-related deaths occurred. Thus, the short-term heparin-free strategy was not associated with a significant increase in the incidence of vein thrombosis and thrombosis-related death risk. Arterial thrombosis occurred only in the punctured femoral artery, and no cases of limb necrosis occurred.
Consistent with the relatively few perioperative bleeding, intraoperative and postoperative early allogeneic transfusion demand also appeared to be lower than in previous studies.4,9,25,28 Massive intraoperative transfusion and inappropriate blood component transfusion ratio have been previously associated with adverse clinical outcomes.8,9 Therefore, it is important to further investigate whether a heparin-free strategy to reduce blood product demand reduces the incidence of adverse clinical outcomes.
We explored the effects of heparin-free ECMO on the coagulation system. Some potential factors affecting the coagulation system such as renal replacement and rethoracotomy were excluded. Overall, changes in the coagulation indicators were comparable among the three groups. Notably, higher PT and lower fibrinogen levels were observed during ECMO run in the VA group, and platelet counts were persistently higher in the VV group, while there were no significant differences in haemoglobin levels in the three groups. This is consistent with the finding of less bleeding and lower blood transfusion demand in the VV group.
In this study, severe infection was the leading cause of death in lung transplantation patients undergoing perioperative heparin-free anticoagulation ECMO, which is consistent with data from the China Lung Transplantation Registry. 36 Bleeding was not the primary direct cause of death. However, pathophysiologic changes and treatment in response to bleeding may potentially affect clinical outcomes. Both intraoperative bleeding and thoracic drainage within 24 h postoperatively had a positive correlation with ECMO duration, ventilator time and ICU stay, and were independent predictors of 1-year survival. Patients with less intraoperative bleeding and reduced thoracic drainage had a lower incidence of PGD grade 3. Therefore, minimizing perioperative bleeding and serious bleeding events is of great significance to improve clinical outcomes in lung transplantation patients. In addition, patients in the VV group had the shortest ECMO duration, ventilator time, and duration of ICU stay. In this study, single lung transplantations were performed on a case-by-case basis, taking into account factors such as age, frailty, overall disease severity and availability and suitability of donor lungs. The results may partly be attributed to more single lung transplantations in the VV group, which aligns with previous studies.37,38
Recent years have seen numerous advances in circuit technology, including surface coatings, reduction in surface area and priming volume, and pump technology, all of which optimize clinical application and reduce thrombus formation.39–41 In this study, the Sorin system was coated with phosphorylcholine, and the Maquet system was coated with heparin. We consider that the optimized materials and biological coatings helped slow down or reduce thrombosis, supporting the use of a short-term heparin-free strategy. We did not acquire accurate data on the thrombi of the ECMO systems. Data from some patients revealed ECMO system thrombi in nearly 60% of patients. Most visible thrombi were <5 mm in diameter, some thrombi had the appearance of a thin strip, and very few had the appearance of large flakes. Post-membrane blood gas analysis showed that the presence of thrombi did not affect oxygenator function and oxygenator replacement was not required. Notably, despite a high incidence of thrombi, no significant pulmonary embolism events or thrombosis-related deaths were observed in the first year after surgery.
In patients with high bleeding risk, ECMO has been safely performed without continuous heparin infusion for ⩾3 days or with delayed heparin anticoagulation (>5 days), and mid-to-long-term ECMO support was possible without an increased risk of thrombotic complications or oxygenator failure.12,14,15 Some centres have attempted to use heparin-free anticoagulation ECMO during lung transplantation in selected cases.2,18 Our results on heparin-free anticoagulation ECMO support during the perioperative period of lung transplantation support the safety and feasibility of a short-term heparin-free strategy.
This study had some limitations. First, this was a retrospective observational cohort study. Sample size calculation was not performed for this retrospective study and the limited number of samples may affect the statistical significance of the results. Second, some coagulation monitoring indicators and techniques, including thromboelastography, anti-Xa and antithrombin III, which could have helped better analyse coagulation function, were not routinely performed. Third, owing to the postoperative prolongation of ECMO, detailed PGD data were not analysed. Fourth, we were unable to obtain sufficient baseline data on the circulatory variables, aside from pulmonary arterial pressure. This limitation prevented a comprehensive assessment of patients’ circulatory status which may have affected bleeding and thrombotic outcomes. However, interstitial lung disease was the primary indication for lung transplantation; 97.8% of patients did not require preoperative ECMO support, and selective operation was performed after preoperative evaluation. Therefore, the preoperative circulatory status was generally stable. Fifth, the huge difference in the number of patients included in the study groups may create issues in interpreting the results.
Conclusion
The heparin-free anticoagulation ECMO contributes to reducing early perioperative bleeding and blood product transfusion requirements and does not increase thrombosis-related deaths. Intraoperative bleeding and thoracic drainage within 24 h postoperatively were independent predictors of 1-year survival. This study supports the safety and feasibility of the short-term heparin-free anticoagulation strategy for perioperative ECMO in lung transplantation, particularly for VV-ECMO.
Supplemental Material
sj-doc-1-tar-10.1177_17534666241273012 – Supplemental material for The impact of early perioperative heparin-free anticoagulation for extracorporeal membrane oxygenation on bleeding and thrombotic events in lung transplantation: a retrospective cohort study
Supplemental material, sj-doc-1-tar-10.1177_17534666241273012 for The impact of early perioperative heparin-free anticoagulation for extracorporeal membrane oxygenation on bleeding and thrombotic events in lung transplantation: a retrospective cohort study by Zhijiang Qi, Sichao Gu, Xin Yu, Zeyu Zhang, Xiaoyang Cui, Changlong Li, Min Li and Qingyuan Zhan in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241273012 – Supplemental material for The impact of early perioperative heparin-free anticoagulation for extracorporeal membrane oxygenation on bleeding and thrombotic events in lung transplantation: a retrospective cohort study
Supplemental material, sj-docx-2-tar-10.1177_17534666241273012 for The impact of early perioperative heparin-free anticoagulation for extracorporeal membrane oxygenation on bleeding and thrombotic events in lung transplantation: a retrospective cohort study by Zhijiang Qi, Sichao Gu, Xin Yu, Zeyu Zhang, Xiaoyang Cui, Changlong Li, Min Li and Qingyuan Zhan in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
