Abstract
Lung and hematopoietic stem cell transplantation are therapeutic modalities in chronic pulmonary and hematological diseases, respectively. One of the complications in these patients is the development of bronchiolitis obliterans syndrome (BOS). The efficacy and safety of available treatment strategies in BOS remain a challenge. A few mechanisms have been recognized for BOS in lung transplant and graft-versus-host disease (GVHD) patients involving the TH-1 and TH-2 cells, NF-kappa B, TGF-b, several cytokines and chemokines, and cysteinyl leukotrienes (CysLT). Montelukast is a highly selective CysLT receptor antagonist that has been demonstrated to exert anti-inflammatory and anti-fibrotic effects in abundant experiments. One area of interest for the use of montelukast is lung transplants or GVHD-associated BOS. Herein, we briefly review data regarding the mechanisms involved in BOS development and montelukast administration as a treatment modality for BOS, and finally, the possible relationship between CysLTs antagonism and BOS improvement will be discussed.
Plain language summary
Lung and bone marrow transplantation are therapeutic modalities in chronic diseases of the lungs and the blood, respectively. One of the complications in these patients is the development of Bronchiolitis obliterans syndrome (BOS). The efficacy and safety of available treatment strategies in BOS remain a challenge. A few mechanisms for BOS in lung transplant and graft-versus-host disease (GVHD) patients involving many immune components have been recognized. Cysteinyl leukotrienes are products of plasma membrane phospholipids that increase smooth muscle contraction, microvascular permeability, and airway mucus secretion. Montelukast is a highly selective cysteinyl leukotriene receptor blocker demonstrated to exert anti-inflammatory and anti-fibrotic effects. One area of interest for the use of montelukast is in lung transplant- or GVHD-associated BOS. In this article, we briefly review data regarding the mechanisms involved in BOS development and montelukast administration as a treatment modality for BOS. Finally, the possible relationship between cysteinyl leukotriene inhibition and BOS improvement will be discussed.
Introduction
Lung transplantation and hematopoietic stem cell transplantation (HSCT) are therapeutic modalities in chronic pulmonary and hematological diseases, respectively, with a growing trend globally.1,2 One of the complications in these patients is the development of bronchiolitis obliterans syndrome (BOS). Being a category of chronic lung allograft dysfunction (CLAD), BOS is considered an important cause of morbidity and mortality, decreased quality of life, and use of healthcare resources in the lung transplant population, and is known to develop in up to 50% of lung transplant patients by the fifth post-transplant year. 3 On the other hand, BOS in HSCT patients is particularly recognized in the context of chronic graft-versus-host disease (GVHD), affecting almost 10% of this population. GVHD-associated BOS is a rare but important complication usually occurring between 100 days and 2 years after HSCT, 4 mainly the allogenic type.
Diagnosis of CLAD-BOS after lung transplantation is based on the detection of a persistent at least 20% decrease in forced expiratory volume in 1 s (FEV1) from post-transplant measurements for more than 3 weeks, and obstruction found in spirometry (FEV1/forced vital capacity (FVC) ratio < 70%), with no evidence of restriction.5,6 It is important to exclude other possible causes. GVHD-BOS is defined as FEV1<75% predicted with a 2-year decline of at least 10% and FEV1/FVC ratio < 70% or the fifth percentile predicted. Investigations and patients’ clinical features should not show evidence of respiratory infections.4,7,8 The clinical presentation of BOS resembles obstructive lung disease detected as a decline in and progressive airflow obstruction.6,9
One of the challenges regarding BOS is the efficacy and safety of available treatment strategies. In lung transplant patients, several non-pharmacologic (such as lymphoid irradiation and extra-corporeal photopheresis) and pharmacologic interventions such as immunosuppressive modification, azithromycin, and montelukast have been suggested and used for so long. Other treatment options including alemtuzumab, Janus Kinase (JAK) inhibitors, and thymoglobulin have also been introduced.4,5,10 The last treatment that might be considered is re-transplantation. Also, a few other agents are widely under investigation such as antifibrotics and inhaled liposomal cyclosporine.11,12 In GVHD patients, management approaches consist of systemic corticosteroid use, fluticasone–azithromycin–montelukast combination therapy, inhaled long-acting bronchodilators, extra-corporeal photopheresis, rituximab, and finally lung transplantation.1,4,13
Montelukast has been demonstrated to exert anti-inflammatory and anti-fibrotic effects. 14 One area of interest for the use of montelukast is in lung transplant or GVHD-associated BOS. 15 However, no review of studies stating the therapeutic effect of montelukast in lung transplant and HSCT patients exists; nor any data focusing on the rationale for its use in BOS and possible mechanisms leading to its potential effects are available.
In this review, we briefly highlight the mechanisms involved in BOS development; then we will focus on reports of montelukast administration as a treatment modality for BOS both in lung transplant and HSCT patients, and finally discuss the possible relationship between cysteinyl leukotrienes (CysLTs) antagonism and BOS improvement.
Method
To collect data regarding the administration of montelukast in post-lung transplant and post-GVHD BOS, first, we chose the keywords and designed the search strategy for our review as presented below: [montelukast OR Singulair OR leukotriene antagonist OR leukotriene receptor antagonist OR cysteinyl leukotriene receptor antagonist OR CysLT receptor antagonist] AND [bronchiolitis obliterans OR BO OR bronchiolitis obliterans syndrome OR BOS OR chronic lung allograft dysfunction OR CLAD OR CLAD-BOS OR lung transplant OR lung transplantation OR graft-versus-host disease OR GVHD OR hematopoietic stem cell transplantation OR HSCT]
The search was carried out using PubMed, Google Scholar, and Scopus in January 2023 for studies in the English language.
Duplicate or irrelevant articles, editorials, commentaries, and review articles were excluded from the search result, and among the references selected based on their relevance, finally 10 were used for the review (Figure 1). In cases where a review article was found, the cited references were also reviewed and utilized in our study.
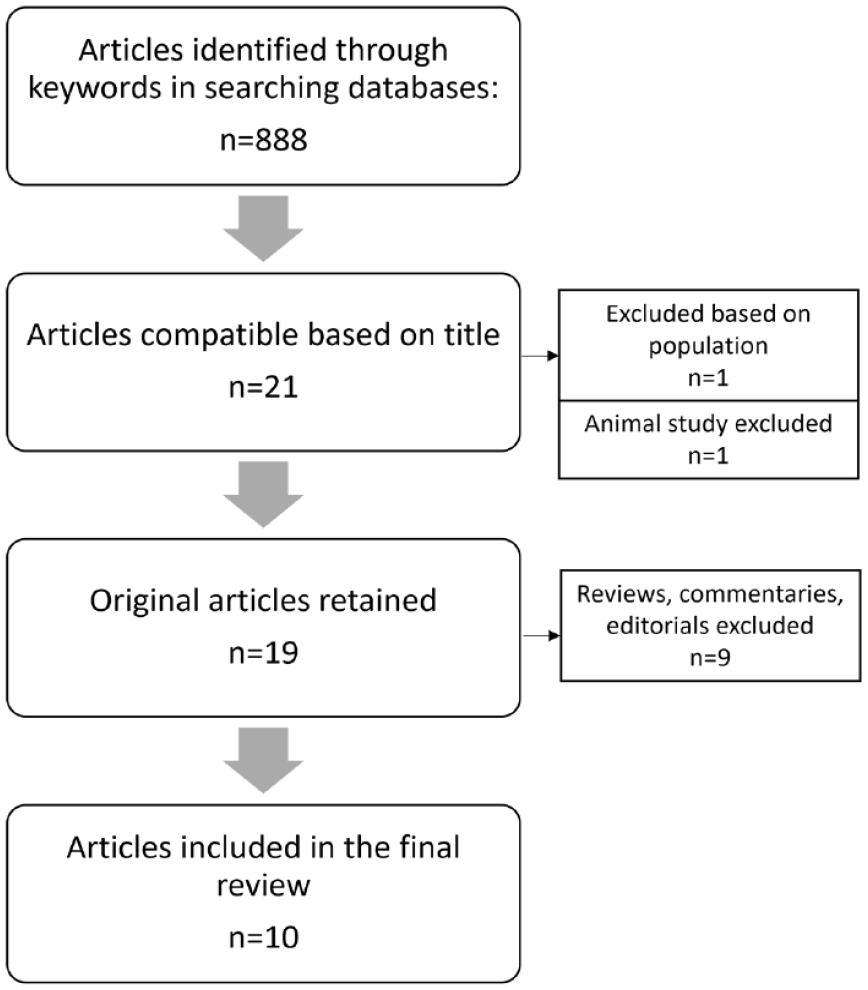
Flowchart of included studies in the review of the effect of montelukast in bronchiolitis obliterans syndrome.
Pathophysiology and etiology of BOS
BOS is the major phenotype of chronic lung allograft dysfunction in lung transplant patients and a pulmonary manifestation of GVHD after HSCT. Besides, bronchiolitis obliterans has previously been detected in some inflammatory conditions and auto-immune disorders such as rheumatoid arthritis or post-infection in children.16,17 BOS is the inflammation of epithelial cells and subepithelial structures of the small airways, leading to fibroproliferation. Histologically, it is defined as patchy submucosal fibrosis involving the respiratory bronchioles leading to their occlusion.
Pathobiology and immunology of CLAD-BOS
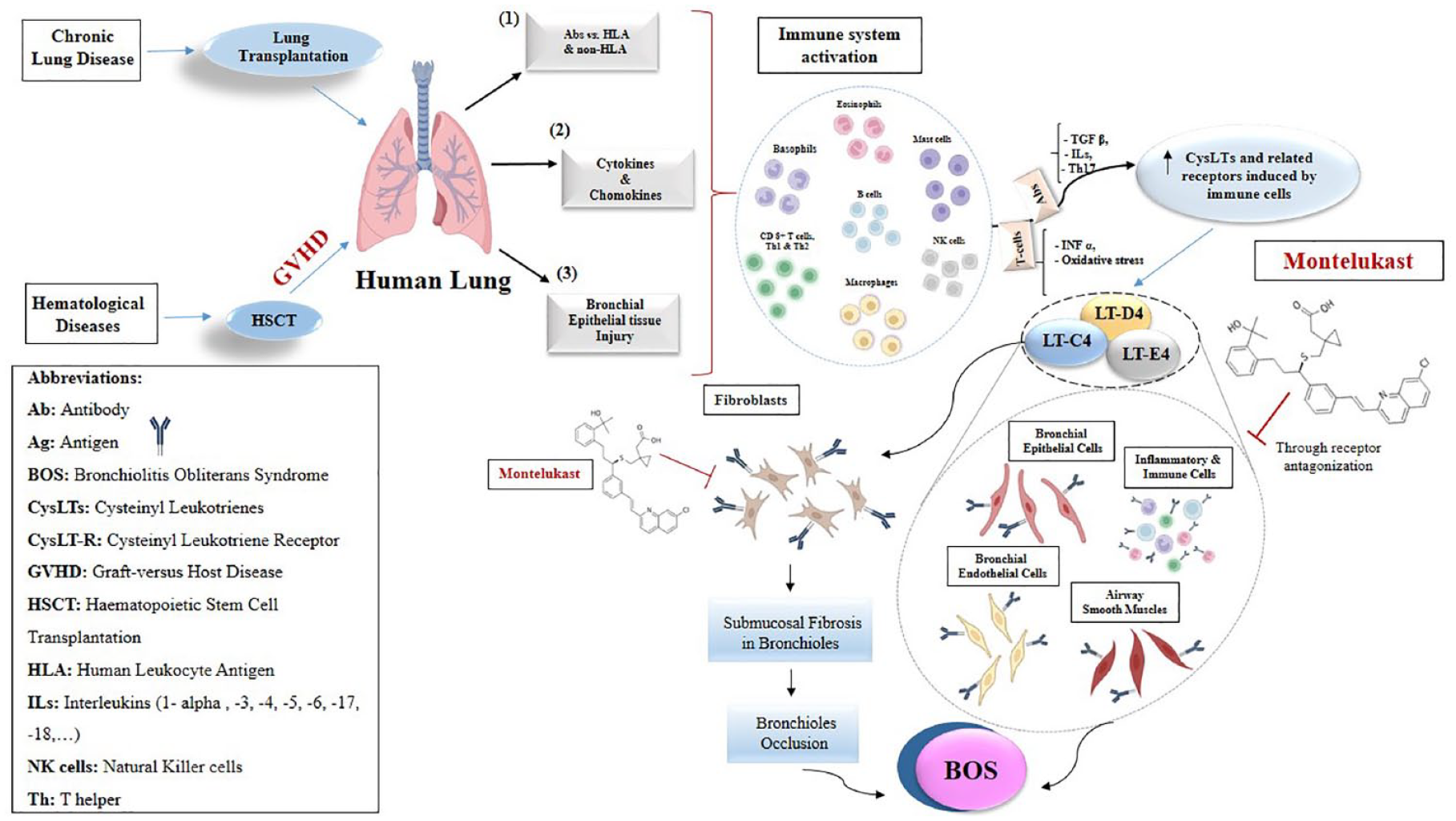
A few mechanisms have been recognized for BOS development in lung transplant patients. Alloimmunity is one of the important pathways related to T-cell activation and T Helper-1 cells. Humoral immunity is also known to be involved in BOS where the development of human leukocyte antigen (HLA) and non-HLA antibody development is correlated with an increased risk of BOS. One concept is that autoimmunity involving autoreactive T-cell proliferation and auto-antibodies production after the first phase of tissue injury mediates fibroproliferative events. This also involves auto-immune induction with collagen type (V) reactivity as an antigen and TH-17 pathway. Besides, lipopolysaccharide and toll-like receptors, especially TLR4 and CD14, are part of innate immunity contributing to the pathobiology of BOS in lung transplant patients. 9
Several mediators such as matrix metalloproteinase (MMP), interleukin (IL)-8, IL-17, neutrophils, Hyaluronan, Alpha-defensins and transforming growth factor (TGF)-beta, IL-1-alpha, and oxidative stress pathway are important in the pathogenesis of BOS. Microbes and viruses have also been demonstrated to play a role in BOS.6,9 One study showed that inhibition of nuclear factor (NF)-kB activation in a mouse model attenuated lung injury in the allograft rejection process. Also, it has been speculated that high mobility group box protein 1 (HMBG1) can exacerbate fibrosis leading to BOS via NF-kB signaling in a mouse model of tracheal transplantation.
In addition, CysLTs have been reported to exist in high levels in broncho-alveolar fluid and blood of transplanted animals. Also, Tu et al. suggested that LT B4 might play a role in BOS development following animal models of lung grafting in rats. 18 Exposing the human bronchial epithelial cell line to LT C4 resulted in a significantly increased expression of collagen I and smooth muscle actin and decreased cytokeratin, contributing to epithelial to mesenchymal transition in the process of BOS. 19 Mechanisms involved in the process of BOS development are briefly depicted in Figure 2.

Mechanisms involved in the process of bronchiolitis obliterans development. Several mediators have been recognized to be involved in BOS in lung transplant and graft-versus-host disease including the TH-1 and TH-2 cells, CD8+ T cells, NF-kappa B, TGF-b, various immune cells (such as neutrophils and monocytes), TNF-alpha, several cytokines and chemokines, and cysteinyl leukotrienes (CysLT).
Pathobiology and immunology of GVHD-BOS
No exact mechanism has been introduced for BOS development in GVHD following HSCT. We know that chronic GVHD displays autoimmune and fibrotic features, while the underlying mechanism for acute GVHD involves mostly inflammatory components. The immune pathophysiology related to chronic GVHD is complex, with the interplay of donor T-cells, B cells and auto-antibody production, and other donor cells, pre-fibrotic and fibrotic lesions. High levels of B-cell activating factor in plasma of GVHD patients have been recognized. Besides, the development of fibrosis in various parts of the body is the hallmark of GVHD with TGF-beta, tumor necrosis factor (TNF)-alpha, and platelet-derived growth factor as major mediators in this process.20,21
Increased levels of CD4+ and CD8+ T cells are hypothesized to be involved in GVHD-associated BOS. TH1 cells, when activated, release interferon-gamma, and TH2 cells secrete IL-4, IL-5, and IL-13. Other interleukins such as IL-13 and IL-17 are also suggested to have a role in this pathway. Furthermore, several cytokines and chemokines seem to be involved in the development of BOS based on studies.
It has been speculated that CysLTs are involved in the process of GVHD-associated BOS. Also, macrophages, and to a lesser extent CD4+ T cells and neutrophils are found in the bronchoalveolar lavage (BAL) of these patients. Leukotriene receptors’ expression on macrophages/monocytes, eosinophils, granulocytes, and T cells was shown to be elevated. Also, in moderate to severe BOS, CD8+ T cells were predominant. 22
Cysteinyl leukotrienes
Cysteinyl leukotrienes refers to LT C4, D4, and E4, being mainly produced by cells of innate immunity system, such as basophils, eosinophils, mast cells, monocytes/macrophages through stimulation caused by pro-inflammatory cytokines or various other receptors, and are structurally different from LTs A4 and B4. 23 CysLTs also interact with G-protein-coupled receptors on lymphocytes and to a lesser extent, neutrophils. CysLT1 receptor is widely expressed on different immune and inflammatory cells in addition to some structural cells such as airway smooth muscles, epithelial and endothelial cells, and fibroblasts. However, the distribution of the CysLT2 receptor is limited compared to the former receptor. 24 There are also data presenting the role of CysLTs in regulating vascular leakage and edema along with cytokine signaling. Having been known to hyperstimulate many acute and chronic inflammatory conditions, CysLTs introduced a target for immunopathogenesis and treatment of many inflammatory disorders, especially asthma. Up to now, three antagonists of CysLTs have been introduced including Pranlukast, Zafirlukast, and Montelukast. This review will focus on the current and potential application of montelukast in BOS.
Montelukast
Montelukast (C35H35CINNaO3S), a highly selective CysLT receptor antagonist, is an orally dosed drug. It is mainly a LTD4 antagonist.25,26 Montelukast binds with high affinity to the CysLT receptors that are found in macrophages and smooth muscle cells of the airways, blocking the leukotrienes’ physiologic effects. 27 This drug has been used in chronic asthma, allergic rhinitis, and exercise-induced bronchoconstriction, and recently in neurodegenerative diseases. 28 Montelukast is regarded as generally safe, although neuropsychiatric effects have been reported with the use of this drug, including depression, anxiety, sleep problems, agitation, and paresthesia; therefore, it holds a US boxed warning for these serious adverse effects and patients should be regularly monitored for mood and behavior changes. 29 It is suggested that neuropsychiatric side effects of montelukast are more commonly observed in children and adults with a previous history of psychiatric disorders. However, these symptoms usually disappear following drug discontinuation. 28 Other potential adverse events are headache, fever, upper respiratory tract or gastrointestinal symptoms, dermatologic manifestations, arthralgia and myalgia, hepatobiliary signs, and infections. 30
CysLTs antagonism exerts anti-inflammatory effects through two mechanisms. First, by interacting with innate immune cells such as macrophages/monocytes, mast cells/basophils, eosinophils, and to a lesser extent with neutrophils, through CysLT receptor type 1. Second, the anti-inflammatory properties of CysLT antagonists are also mediated through a non-CysLTR1-dependent effect on immune cells. These non-CysLT-dependent pathways include but are not limited to the inhibition of 5-lipoxygenase, prostaglandin E synthase, NF-kB, purinergic P2Y receptors, and cyclic nucleotide phosphodiesterase. 24 It is previously described that NF-kB signaling inhibition results in diminished release of several pro-inflammatory cytokines and mediators such as IL-6, IL-8, IL-10, TNF-alpha, and Monocyte chemoattractant protein (MCP)-1. It also increased glutathione and superoxide dismutase, the antioxidants that were known to reduce mortality in animal models of lung inflammation.23,31
Anti-inflammatory and anti-fibrosis effects of montelukast
Montelukast has been demonstrated to exert anti-inflammatory and anti-fibrotic effects in animal studies. In an experiment using bleomycin-induced inflammatory and oxidative stress lung injury in rats, montelukast treatment reversed the increased levels of TNF-alpha and TGF-beta along with the fibrosis and collagen deposition in lungs which had been caused by bleomycin. 32 Plus, Henderson et al. stimulated inflammation and fibrosis with widespread collagen deposition under airway epithelial cells and lung interstitium. In this study, montelukast lessened fibrosis, interleukin expression, and chronic inflammatory and allergic events. 14 Montelukast also decreased oxidative stress and pro-inflammatory mediators in mice and led to diminished cytokine concentrations in the lung. 33 On the other hand, montelukast treatment in an animal model of lung transplant led to decreased LT B4 receptor concentration, causing a preventive effect on post-lung transplant fibrosis. 18
Montelukast use in BOS
History and use of montelukast in CLAD-BOS
There are a few studies evaluating the effect of montelukast on BOS in post-lung transplant patients. Verleden et al. conducted a pilot study with 11 patients receiving montelukast and 11 controls. Montelukast was started 3 weeks following BOS diagnosis, and all patients used azithromycin either before or simultaneously with montelukast. All patients developed BOS stage < 3 and BAL neutrophilia < 15%. Results showed that the FEV1 decline rate decreased in the montelukast-treated group but not in controls (p < 0.001). No difference was obvious between slow and fast decliners. 34
In a conference report of a retrospective study by Obi et al. (1996–2009) comparing FEV1 before and after treatment with montelukast, 63 patients developed BOS and 28 of them received montelukast. FEV1 change between these groups was not significant but survival was better in the montelukast group (53 versus 23 months, p < 0.001). 35
A small randomized clinical trial comparing montelukast with placebo in addition to azithromycin administration revealed high graft loss in patients. Montelukast was not shown to affect lung function significantly, but in BOS stage 1 patients, the FEV1 decline rate was decreased. 36
Another single-center retrospective study by Gottlieb et al. was performed, in which 38 patients, mostly received double lung transplants, took montelukast 10 mg at least for 3 weeks as a rescue therapy for progressive BOS. All patients had received azithromycin in advance. Follow-up continued for 74 days. The response was defined as FEV1>110% baseline and no response as FEV1<90% baseline. Only four of the patients were responsive, and others either progressed or died. 37
A retrospective study was performed including 153 patients who had received azithromycin for at least 3 months, and now started montelukast for 3 months and more. FEV1 decline rate lessened significantly after 3 and 6 months; interestingly, an improvement in survival was also observed. In this study, rapid decliners, restrictive allograft syndrome (RAS) phenotype, and patients with high neutrophils in BAL did not respond to montelukast treatment. 38
History and use of montelukast in GVHD-BOS
Reuven et al. conducted a pilot study enrolling 19 patients with chronic GVHD that was refractory, starting montelukast 10 mg daily for a median of 12 months along with their standard immunosuppression regimen. It was shown that in three of five patients with lung involvement, improvement of lung function occurred. However, generally, 79% of patients showed improvements in their GVHD manifestations. The effect was seen in milder stages. Also, no side effects were reported relating to montelukast. 39
A prospective phase II trial involving 25 patients with post-allogenic-HSCT chronic GVHD and a history of 3 months with moderate to severe BOS investigated the effect of montelukast 10 mg treatment after 6 months. Montelukast appeared to be well-tolerated and halted the need for immunosuppression or BOS therapy escalation. The results also revealed treatment success in all patients, regarded as an improved or stable FEV1, or no more than15% decline in FEV1 predicted. Besides, scores related to mood, energy, and breathing, and also 6-minute walking test distance were boosted. The authors also reported improvement or stability of other manifestations of GVHD. The overall survival was 87% after 2 years. Furthermore, insomnia was the only adverse event reported by the patients during the study period. 22
A combination regimen consisting of budesonide/formoterol, montelukast, and N-acetylcysteine was investigated in BOS after HSCT which led to increased FEV1 and FVC and decreased residual volume significantly following 3 months of therapy. 40 Another regimen consisting of fluticasone, azithromycin, and montelukast was evaluated in a clinical trial study, which was demonstrated to be well-tolerated and efficient in terms of improved quality of life and avoiding pulmonary function decline. The same regimen had been reported in a case series compared to corticosteroid treatment, showing positive but not significant effects on FEV1, but fewer side effects in contrast to the control group, and led to less prednisone exposure. 41 These regimens did not cause serious side effects. 40 Interestingly, Williams et al. reported that although CysLT levels could not be measured in BAL fluid possibly owing to a low half-life in the lung, LT E4 (final CysLT) in 24-h urine samples of nearly half of patients was elevated, and interestingly, lower FEV1 and more severe BOS was associated with higher levels of LT E4. 42
Discussion
An important but insidious cause of increased morbidity and mortality in lung transplant and HSCT patients is BOS. Therefore, efforts have been made by researchers to investigate new treatments to improve outcomes in this population and to overcome challenges regarding the safety and efficacy of currently available therapies. Owing to diverse mechanisms and mediators involved in BOS development following lung and hematopoietic stem cell transplants, numerous agents each of which affect a part of the pathways leading to BOS, have been introduced and implemented in the therapeutic approach, while many others targeting specifically the immune pathways such as inhibitors of B-cell or Janus kinase are experimental.43–45 Montelukast has been suggested as a therapeutic option in patients with different stages of BOS, mostly refractory to first-line therapies post-lung transplant and GVHD. 4 Table 1 shows a summary of the studies including experiments on the effect of montelukast treatment in BOS. Montelukast mostly interferes with the final steps in immune pathways which cause the inflammation and fibrosis of BOS, for instance, by antagonizing the CysLT receptors on immune cells and smooth muscle cells of the airways or inhibiting LTC4 production by cells of the immune system which finally leads to a diminished cytokeratin and collagen level. Besides, montelukast tends to affect CD 8+ T cells and also hampers inflammation and fibrosis through NF-kB, TNF-alpha, IL-6, MMP, and oxidative stress suppression, minimizing the function of interleukins and chemokines.33,46–50 All these are part of the immune response activation and disruption of regulatory mechanisms and activation of leukocytes and T helper cells, which is assumed to be similar between CLAD-BOS and GVHD-BOS. We generally presume that the trigger for CLAD-BOS could range from tissue injuries from acute cellular rejection or a response to bacterial and viral infections, for example, Pseudomonas and community-acquired respiratory viruses. On the other hand, the early process that contributes to GVHD-BOS mainly contains central tolerance failure and B-cell activation progressing to autoantibodies production. 4 Cumulatively, although the early phase of immune response initiation in BOS in lung transplant and HSCT patients is not identical, the final processes share similar characteristics. This hypothesis might be the rationale for a relatively identical clinical and experimental approach to BOS management following lung transplant and GVHD.
Table of studies on montelukast use in BOS following lung transplant and GVHD.

6MWT, 6-minute walking test; BOS, bronchiolitis obliterans syndrome; CysLTs, cysteinyl leukotrienes; FEV1, forced expiratory volume in 1; GVHD, graft-versus-host disease.
The primary limitation in our review was the low quality and quantity of the available studies on this topic, thus, larger basic studies and clinical trials with more strength are warranted to evaluate the effectiveness of montelukast in BOS and discover the involved mechanisms in this indication of the drug. Despite this potential limitation, we have carefully included all the relevant in vitro and in vivo studies in our review.
Conclusion
In conclusion, considering the recognized role of CysLTs in BOS pathobiology, along with other mediators and cytokines that are known to be inhibited by montelukast, this drug has been proposed as a therapeutic option to stabilize or improve lung function. Montelukast is a safe treatment, although some drug interactions may exist. Adverse effects of concern reported following montelukast use for different indications are neuropsychiatric events which should be considered and discussed carefully before drug prescription.
Finally, besides the discovery of novel therapies that can prove advantageous, repurposing drugs for other indications even in a combination regimen with other treatments, following basic and clinical experiments, paves the way for identifying safe and effective treatments for this serious complication. The value of these experiments is especially emphasized due to the known mechanisms, usually lower prices, and better availability.
Footnotes
Acknowledgements
None.
