Abstract
Background:
The anti-Mycobacterium avium complex (MAC) antibody test measures levels of IgA antibody against the glycopeptidolipid (GPL) core in the bacterial cell walls and is a useful clinical indicator of nontuberculous mycobacterium pulmonary disease (NTM-PD). However, it is not currently possible to diagnose the disease using anti-MAC antibodies alone.
Objectives:
The study aim was to assess the efficacy of the combination of anti-MAC antibodies and clinical findings for diagnosing potential NTM-PD.
Methods:
This cross-sectional study included 938 patients tested using the anti-MAC antibody. NTM-PD was diagnosed by multiple positive cultures of the same species in sputum samples. Multivariate logistic regression models were used to identify the clinical factors related to NTM-PD.
Results:
Overall, 19.6% (184/938) of participants were diagnosed with NTM-PD. In multivariate analysis, positive anti-MAC antibodies, low body mass index, absence of malignancy, and cavity-forming lung lesions were significantly associated with NTM-PD at diagnosis. The positive rates of the anti-MAC antibody test were 79.4% (135/170) for MAC and 55.6% (5/9) for Mycobacterium abscessus complex, respectively.
Conclusions:
Bronchoscopic examinations should be performed especially in certain types of individuals from whom sputum samples cannot be obtained. Anti-MAC antibodies are also positive in patients other than those harboring MAC, but the rate may be low because of the different components in GPLs.
Introduction
Tuberculosis is an interhuman-transmission disease caused by the pathogenic bacteria Mycobacterium tuberculosis, whereas nontuberculous mycobacterial (NTM) infection is considered to be nontransmissible from human to human. Nontuberculous mycobacterium pulmonary disease (NTM-PD) used to be rarer than pulmonary tuberculosis, but the number of patients diagnosed with NTM-PD has been increasing over the last three decades. 1 The reasons for this increase are unclear; however, improvement of culture technology, awareness of the disease, and changes in lifestyle may be contributing to the widespread infection. Recent studies have shown that immunological or genetic defects contribute to susceptivity and prognosis in NTM-PD. 2 The progression of NTM-PD depends on individuals. Some patients have a long-term stable course of infection, whereas others progress to respiratory failure. However, it remains difficult to predict disease progression in routine practice, and early diagnosis and long-term follow-up are needed.
There are >200 species of NTMs, among which approximately 20 are known to cause infections in humans. 3 Especially in Japan, Mycobacterium avium complex (MAC) – including Mycobacterium avium and Mycobacterium intracellulare – are the most common bacterial species accounting for approximately 90% of cases, followed by Mycobacterium kansasii and Mycobacterium abscessus complex. 4 Unlike M. tuberculosis, NTMs can be detected in environmental soil, dust, and water. Thus, the diagnosis of NTM-PD is not as simple as that of pulmonary tuberculosis, and diagnostic criteria have been established. 5 The anti-MAC antibody has been available since 2011 as an aid in diagnosis and is a serum test that measures levels of immunoglobulin A (IgA) antibody against the glycopeptidolipid (GPL) core in the bacterial cell walls. Rapidly growing mycobacteria (RGM), including M. abscessus complex, also contain GPLs. Contrarily, M. tuberculosis and M. kansasii, which are the major causative agents of lung infection, do not carry GPLs. It has been reported that although the sensitivity of the anti-MAC antibody test in patients with NTM-PD was approximately 70%, it was inadequate. 6 In this study, we investigated the efficacy of combining anti-MAC antibodies with clinical factors to estimate NTM-PD.
Materials and methods
Population and data collection
This cross-sectional study was conducted from November 2017 to March 2021 to investigate the effectiveness of anti-MAC antibodies combined with clinical features in routine practice for diagnosing NTM-PD at Showa University Fujigaoka Hospital. Anti-MAC antibody tests were performed in patients with NTM-PD suspected on the basis of the clinical course and imaging studies. All clinical data were collected from the patients’ medical records. The estimated glomerular filtration rate (eGFR) of each patient was calculated.7,8 Chronic kidney disease (CKD) was defined as an eGFR <60 ml/min/1.73 m2. A cavity-forming lung lesion was defined as a gas-filled space within consolidation, a mass lesion, or a nodule on computed tomography imaging. Human immunodeficiency virus (HIV) antibody tests were not performed because of the low HIV infection rate in Japan. 8 The study protocol was approved by the Institutional Ethics Committee of Showa University (approval no. F2021C025). The requirement to obtain informed consent from the patients was waived because of the retrospective nature of this study.
Anti-GPL-core IgA antibody test
The anti-MAC antibody test used a serum IgA antibody against the GPL core and an enzyme-linked immunosorbent assay kit (Capilia MAC Ab ELISA; Tauns, Shizuoka, Japan) according to the manufacturer’s guidelines. The cutoff value was 0.7 U/ml.
Isolation of mycobacterium and diagnosis of NTM-PD
Sputum samples were collected from each patient at least twice, whenever possible. Even in cases where sputum samples were difficult to obtain, inducing sputum extraction, such as via a nebulizer, was attempted. The Ziehl–Neelsen staining or fluorescence staining was used to perform the smear. Clinical isolates were cultured in mycobacterial growth indicator tubes and in 2% Ogawa solid medium. To distinguish NTM strains, DNA–DNA hybridization (DDH Mycobacteria; Kyokuto Pharmaceutical, Tokyo, Japan) was used until March 2019, and matrix-assisted laser desorption ionization time of flight mass spectrometry was used after April 2019, as reported previously. 9 Polymerase chain reaction tests (Cobas TaqMan MAI; Roche Diagnostics, Tokyo, Japan) were also performed to identify MAC and M. tuberculosis. According to the latest criteria of the American Thoracic Society (ATS) and the Infectious Disease Society of America (IDSA), 5 diagnosis requires at least two positive sputum cultures of the same species or a single positive culture from a specimen obtained by bronchoscopy. In this study, the diagnosis of NTM-PD was limited to multiple positive sputum cultures of the same species combined with clinical symptoms and imaging study findings to ensure constant diagnostic accuracy.
Statistical analysis
All data are expressed as the mean value ± standard deviation for continuous variables or as percentages for categorical variables. The Mann–Whitney rank-sum test was used to compare group mean values. Pearson’s Chi-square test or Fisher’s exact test was used for the univariate analysis of the association between two categorical variables. A logistic regression model was used to evaluate the adjusted effects of multiple variables, and the findings were presented as odds ratios (ORs) with 95% confidence intervals (CIs). Statistical significance was set at p < 0.05. A receiver-operator characteristic (ROC) analysis was conducted to determine a cutoff value using the Youden Index. All statistical analyses were performed in JMP software version 16.0 (SAS Institute, Cary, NC, USA).
Results
Patient characteristics
First, all cases in whom anti-MAC antibodies were measured based on suspicion of NTM-PD during the period were extracted. As shown in Figure 1, 1572 patients were enrolled in this study; of these, 618 who were not tested using the anti-MAC antibody within 1 month of the first visit were excluded for the purpose of evaluating the usefulness of measuring anti-MAC antibodies at the first visit. As mentioned above, 16 patients diagnosed by bronchoscopy were excluded to unify diagnostic criteria of NTM-PD (Supplemental Table 1). Finally, 938 patients were included in this study. The patient characteristics are shown in Table 1. The 596 females and 342 males ranged in age from 15 to 95 years. The body mass index (BMI) ranged between 10.9 and 31.8 kg/m2. There were 266 smokers and 672 never smokers, and 30.5% (286/938) were positive for anti-MAC antibodies. The mean C-reactive protein level was 1.5 mg/dl. One hundred and six patients were taking inhaled corticosteroids for asthma and chronic obstructive pulmonary disease, and 21 were taking oral corticosteroids (⩾10 mg/day of prednisolone) for various indications. Ninety-seven patients had diabetes mellitus, 196 had various malignancies (Supplemental Table 2), and 216 had CKD. Eighty-three patients had an autoimmune disease (Supplemental Table 3), and nine received ⩾10 mg prednisolone daily. Cavity lesions were detected in 61 patients.
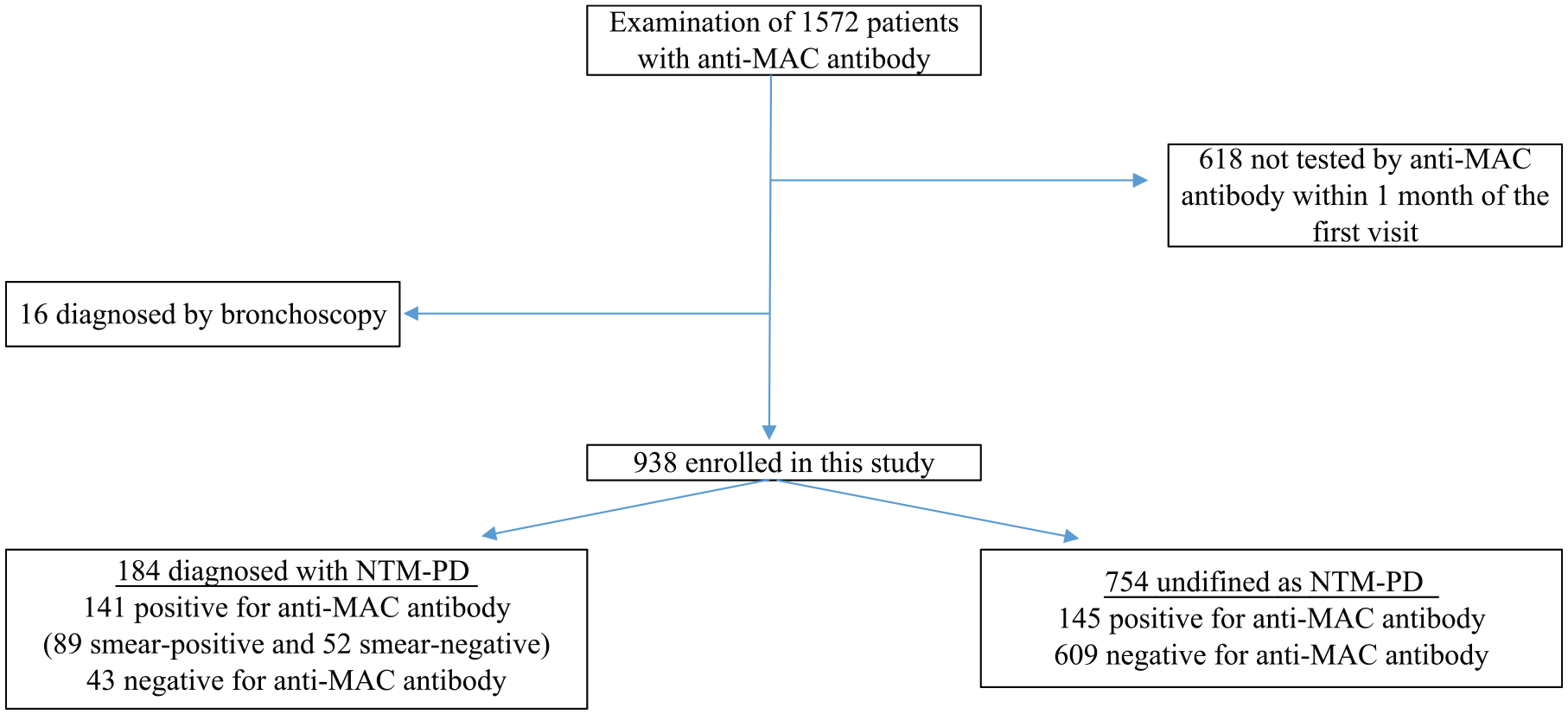
Study cohort recruitment flowchart.
Association of each variable with NTM pulmonary disease.

BMI, body mass index; CKD, chronic kidney disease; MAC, Mycobacterium avium complex; NTM, nontuberculous mycobacterial; SD, standard deviation.
More than 10 mg of prednisolone per day.
p < 0.05 was considered statistically significant.
As shown in Figure 1, NTM-PD was identified in 19.6% (184/938) patients. Of these, 76.6% (141/184) were positive for anti-MAC antibodies. Conversely, 19.2% (145/754) had positive anti-MAC antibodies in the cohort that was not definitively diagnosed. Figure 2 shows the values of anti-MAC antibodies in a positive case of the test. The values of anti-MAC antibodies in patients with NTM-PD were independently higher than those in the undefined diagnosis group, which comprised all the patients who did not reach a definitive diagnosis of NTM-PD because of a lack of sputum production or low levels of bacterial content in the sample (p < 0.0001), whereas the ranges were overlapping. Of the 141 well-defined patients confirmed with positive anti-MAC antibodies, 63.1% (89/141) were smear-positive and 36.9% (52/141) were negative. As shown in Figure 3, there was a significant difference in the values of anti-MAC antibodies between the two groups (p < 0.0318). An ROC analysis was performed for the anti-MAC antibodies to distinguish patients with NTM-PD from undefined individuals. Figure 4 showed a cutoff of 0.31 U/ml identified NTM-PD with 85.3% sensitivity, 76.5% specificity, and 0.844 of area under the curve.

Anti-MAC antibody levels in positive cases of the test. The Mann–Whitney rank-sum test showed significant differences in anti-MAC antibody levels between the definitive diagnosis group (N = 141) and the undefined diagnosis group (N = 145). Values are presented as the mean value (standard deviation).

Anti-MAC antibody levels in patients with NTM-PD confirmed with a positive test. The Mann–Whitney rank-sum test showed significant differences in the levels of anti-MAC antibody between the smear-positive group (N = 89) and smear-negative group (N = 52). Values are presented as the mean value (standard deviation).

Receiver-operating characteristic curves of anti-MAC antibodies for NTM-PD diagnosis. Area under the curve result for anti-MAC antibodies was 0.844.
Statistical analysis of NTM-PD and clinical features
In univariate analyses, NTM-PD was significantly associated with sex, BMI, smoking status, anti-MAC antibody, C-reactive protein, malignancy, autoimmune disease, and cavity lesions (Table 1). However, age, corticosteroid usage, diabetes mellitus, and CKD were not significantly associated with NTM-PD.
Multivariate analyses were used to control for potential confounding effects of these variables. Table 2 presents the logistic regression models of factors related to NTM-PD. The frequency of positive diagnosis increased significantly with low BMI (OR = 0.9096; 95% CI = 0.8502–0.9711). Anti-MAC antibodies were frequently associated with a positive diagnosis (OR = 12.4183; 95% CI = 8.1102–19.0148). The absence of malignancy (OR = 0.5562; 95% CI = 0.3189–0.9699) and presence of cavity lesion (OR = 2.9537; 95% CI = 1.4881–5.8625) were associated with a positive diagnosis.
Logistic regression models of NTM pulmonary disease.

BMI, body mass index; CI, confidence interval; CKD, chronic kidney disease; MAC, Mycobacterium avium complex; NTM, nontuberculous mycobacteria; OR, odds ratio.
More than 10 mg of prednisolone per day.
p < 0.05 was considered statistically significant.
Culture isolates of mycobacterium
Table 3 presents the mycobacterial isolations in patients with NTM-PD. Of these patients, 88% (162/184) were found with single-species isolation of MAC, including 140 strains of M. avium and 22 of M. intracellulare. Single-species isolation of M. abscessus complex and M. kansasii were identified in nine and two patients, respectively. A total of 4.3% (8/184) duplicate species were isolated, all of which contained MAC. One case each of Mycobacterium chelonae, Mycobacterium fortuitum, and Mycobacterium gordonae were detected. Moreover, M. tuberculosis was identified in 13 patients, all of whom had no superinfection with NTMs (Supplemental Table 4).
Culture isolates.

Correlation between mycobacterial isolation and anti-MAC antibody
Table 4 presents the anti-MAC antibody status in the definitive diagnosis cohort. The anti-MAC antibody test was positive for MAC, M. abscessus complex and M. gordonae and negative for M. tuberculosis, M. kansasii, M. chelonae, and M. fortuitum. The positive rates were 79.4% (135/170) in MAC and 55.6% (5/9) in M. abscessus complex. Table 5 presents the patients with NTM-PD except for MAC isolation. Six patients, consisting of five with M. abscessus complex (antibody range = 0.93–12.67 U/ml) and one with M. gordonae (No. 151, antibody level = 0.75 U/ml) tested positive for anti-MAC antibodies. Eight patients tested negative for anti-MAC antibodies; of these eight patients, M. abscessus complex was detected in four, M. kansasii in two, M. chelonae in one, and M. fortuitum in one.
Correlation between culture isolates and anti-MAC antibody.
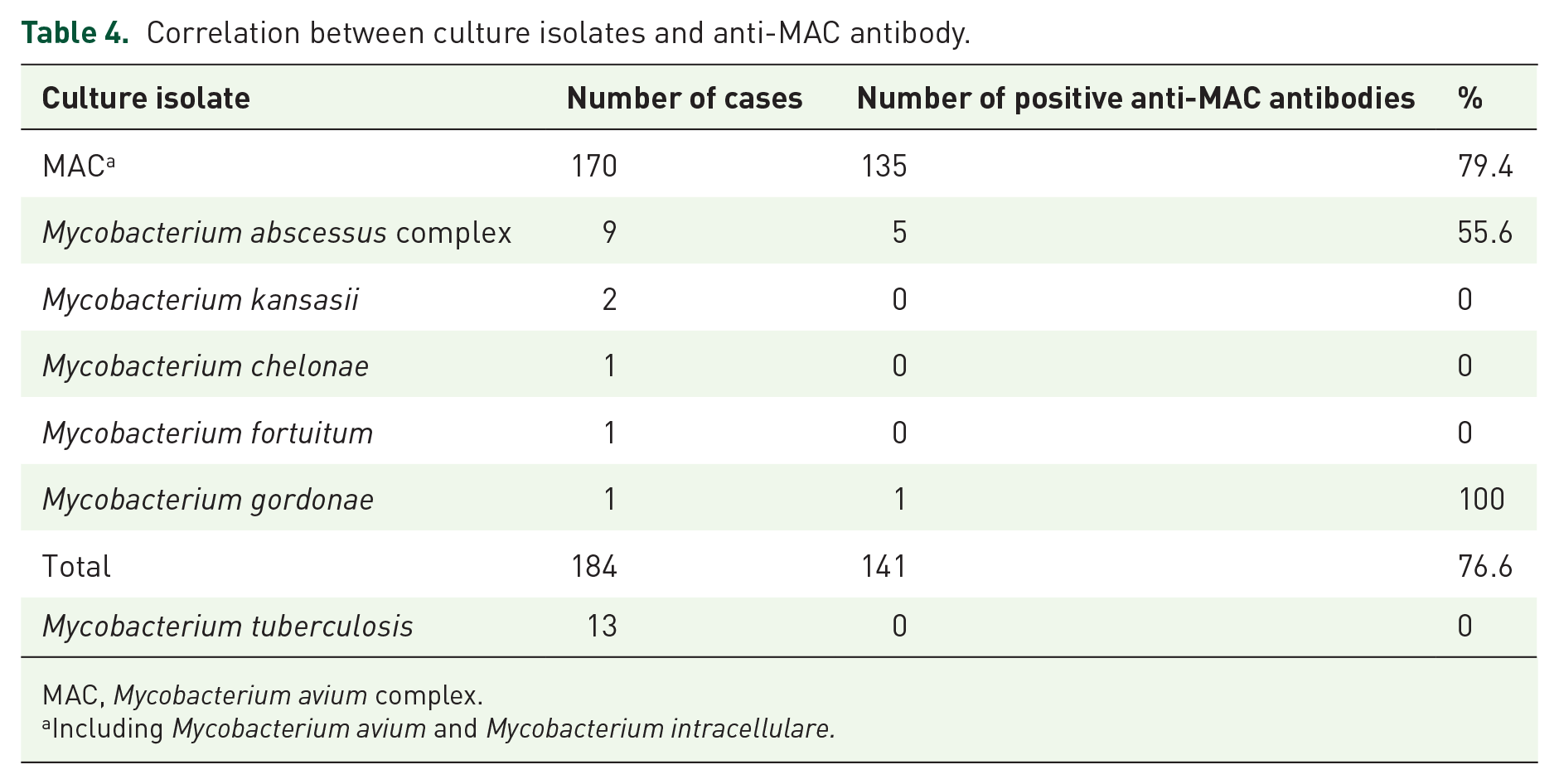
MAC, Mycobacterium avium complex.
Including Mycobacterium avium and Mycobacterium intracellulare.
Patients with NTM pulmonary disease except for MAC isolation.

BMI, body mass index; CKD, chronic kidney disease; MAC, Mycobacterium avium complex; NTM, nontuberculous mycobacteria.
More than 10 mg of prednisolone per day.
Discussion
In this study, the efficacy of combining anti-MAC antibody test results with clinical findings in predicting the presence of NTM-PD was analyzed. Overall, 19.6% of the study population was diagnosed with NTM-PD according to the latest ATS/IDSA guideline. 5 The value of anti-MAC antibodies was significantly higher in the patients with NTM-PD than in the patients for whom a definitive diagnosis could not be reached, which included those who could not be diagnosed because of a lack of sputum production or low levels of bacterial content in the sample. Likewise, the value of anti-MAC antibodies has been reported to be associated with disease activity, treatment outcomes, and disease progression.10,11 Sputum smear-positive results have been reported to be significantly associated with antibody positivity, 12 consistent with the results of this study. These data suggest that the antibody-based results correlated with the bacterial count. In this study, the ROC analysis indicated the cutoff of anti-MAC antibody was 0.31 U/ml, consistent with findings reported in the literature.13,14 In routine practice, however, 0.7 U/ml is recommended as the cutoff with a high regard for specificity.
Our multivariate analysis showed that NTM-PD was significantly associated with positive anti-MAC antibodies, low BMI, absence of malignancy, and cavity lesions at the time of diagnosis. Although immunological status is affected by both obesity and low BMI, a clear relationship between obesity and NTM infection has not been reported. Several studies have reported that low BMI was closely related to NTM-PD.15–19 It remains unknown if low BMI leads to mycobacterial infection or if weight loss is caused by the inflammation; however, both are possible. This study’s data suggest that careful attention at the initial visit should be paid to patients with low BMI suspected to have NTM-PD. Similarly, whether or not malignancy or NTM infection occurs first remains controversial, despite several investigations of this association.10,20–24 In general, comorbidities, including HIV infection, 25 smoking, 26 diabetes mellitus, 27 CKD, 28 autoimmune diseases, 29 and malignancy,10,20–24 may lead to an NTM infection–related immunodeficiency. Conversely, antineoplastic effects induced by mycobacterial infection have also been noted. 30 Cavity-forming lung lesions are caused by various disorders, including autoimmune disease, malignancy, and infectious disease. 31 There are no specific imaging findings for NTM-PD, but the presence of cavity lesions is associated with worse prognosis,32,33 as in the case of tuberculosis. Although the progression of NTM-PD depends on individuals, treatment initiation should be decided when cavity lesions are observed. This study indicated that NTM-PD was not significantly associated with the presence of autoimmune disease or malignancy when combined with an anti-MAC antibody test. In addition, the anti-MAC antibody test results were negative for all tuberculous cases. Hence, a combination of cavity lesions and a positive anti-MAC antibody test result can be useful for diagnosing potential NTM-PD. Bronchoscopic examinations should be performed, especially in patients with positive anti-MAC antibodies, low BMI, absence of malignancy, and presence of cavity-forming lung lesions when sputum samples cannot be obtained as the ATS/IDSA guideline states that a single positive culture from a specimen obtained by bronchoscopy can be used as an alternative diagnostic method. 5 Progression has been reported in NTM-PD cases diagnosed by bronchoscopic methods and in those diagnosed by sputum samples. 34
The positive rate of anti-MAC antibodies was 79.4% (135 of 170) in MAC, which was consistent with the finding in a previous study. 6 Positive conversion has also been reported in cases of RGM infection.6,35 In this study, positive conversions were detected in a patient with M. abscessus complex and M. gordonae infections, but not in M. chelonae and M. fortuitum infections. M. abscessus complex is a major pathogen of RGM in East Asia, including Japan, 4 whereas M. gordonae is not classified as an RGM. GPLs are components of the bacterial cell wall in RGM, but their composition differs from that of MAC. 36 Therefore, the positive rates for anti-MAC antibodies can be lower in RGM than in MAC. Similarly, positive conversions may be observed in cases of M. chelonae and M. fortuitum infections because of the RGM classification. Although M. gordonae is a slow-growing mycobacteria in common with MAC, it has no GPLs in the cell walls. 37 In case No. 151, sputum samples were cultured repeatedly; however, only M. gordonae, but not MAC, was isolated. Moreover, the value of anti-MAC antibody was close to the cutoff value. These data suggest a false-positive antibody test result or a superinfection harboring minimal MAC counts in this case. Recently, it has been reported that a single positive sputum culture test and a positive anti-MAC antibody test can be used to definitively diagnose NTM-PD. 35 However, the probability of duplication of other mycobacterial infections, in addition to MAC infection, should not be ignored because multiple infections were observed in 4.3% (8/184) of the definitive cases in this study. In summary, a positive anti-MAC antibody test is not necessarily indicative of MAC infections and may indicate infectious disorders caused by other NTMs in some cases.
This study had some limitations that should be considered when interpreting the results. First, the study was conducted at a single facility, which might have reduced the generalizability of the findings. Second, the immunological and genetic statuses were not analyzed because of the retrospective nature of this study.
Conclusion
This study showed that anti-MAC antibody testing and clinical factors obtained in routine practice could be used to estimate NTM-PD. The findings suggest performing bronchoscopic examinations in patients with anti-MAC antibody positivity, low BMI, absence of malignancy, and presence of a cavity-forming lung lesion. Large clinical studies should be designed to confirm antineoplastic effects of NTM-PD. Anti-MAC antibodies can be positive in patients not harboring MAC and in patients with multiple mycobacterial infections; thus, extra attention is warranted when diagnosing pulmonary disease caused by MAC in patients with a positive antibody test result.
Supplemental Material
sj-xlsx-1-tar-10.1177_17534666221138002 – Supplemental material for Combination of anti-glycopeptidolipid-core IgA antibody and clinical features for diagnosing potential nontuberculous mycobacterium pulmonary disease in routine practice
Supplemental material, sj-xlsx-1-tar-10.1177_17534666221138002 for Combination of anti-glycopeptidolipid-core IgA antibody and clinical features for diagnosing potential nontuberculous mycobacterium pulmonary disease in routine practice by Takuya Iwasaki, Fumihiro Yamaguchi, Makoto Hayashi, Hitoshi Kobayashi, Kento Hirata, Kenta Miyo, Chika Kondo, Mamiko Kanzaki, Kazusawa Tei, Takashi Abe, Shunsuke Sakakura, Daisuke Inoue, Yohei Yamazaki, Hidetsugu Tateno, Takuya Yokoe and Yusuke Shikama in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors are grateful to Haruka Kitano, Mina Hiraiwa, Yo Shiratori, Shota Onozaki, Mari Nakamoto, Saori Kawamura, Miku Kosuge, Kenji Atarashi, Hidekazu Cho, Shohei Shimizu, Akira Fujishima, and Yuki Osakabe for their assistance in the interpretation of the results and critical review of the manuscript. The authors thank Maruzen (![]() ) for English language editing.
) for English language editing.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
