Abstract
On 30 January 2020, the World Health Organization (WHO) declared the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) epidemic a public health emergency of international concern. The viral outbreak led in turn to an exponential growth of coronavirus disease 2019 (COVID-19) cases, that is, a multiorgan disease that has led to more than 6.3 million deaths worldwide, as of June 2022. There are currently few effective drugs approved for treatment of SARS-CoV-2/COVID-19 patients. Many of the compounds tested so far have been selected through a drug repurposing approach, that is, by identifying novel indications for drugs already approved for other conditions. We here present an up-to-date review of the main Food and Drug Administration (FDA)–approved drugs repurposed against SARS-CoV-2 infection, discussing their mechanism of action and their most important preclinical and clinical results. Reviewed compounds were chosen to privilege those that have been approved for use in SARS-CoV-2 patients or that have completed phase III clinical trials. Moreover, we also summarize the evidence on some novel and promising repurposed drugs in the pipeline. Finally, we discuss the current stage and possible steps toward the development of broadly effective drug combinations to suppress the onset or progression of COVID-19.
Keywords
Key Messages
Several FDA-approved drugs have been repurposed to help treating COVID-19. Some of these, such as molnupiravir, remdesivir, dexamethasone, tocilizumab, and baricitinib have shown promising clinical results, displaying some level of efficacy in randomized controlled studies. New repurposing approaches are being tested and have displayed preclinical efficacy in animal models. Efforts are ongoing to define the best drug combinations to treat SARS-CoV-2–infected patients, both in the early stages and during severe COVID-19.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic is the most significant global health crisis as the 1918 influenza outbreak, and it has caused enormous health and economic devastation around the world. 1 The causative agent of COVID-19, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is a single-stranded 5’-capped positive-sense RNA virus2,3 that was identified as a novel strain of group 2b of beta coronaviruses. 4 The viral genome ranges in size from 26 to 32 kb and contains 14 open reading frames (ORFs)2,3 that encode for both structural and nonstructural proteins, as well as for several accessory proteins (Figure 1). The structural proteins include a helical nucleocapsid surrounding the viral genome, the spike (S) glycoprotein, which is essential for viral attachment and entry, and the membrane (M) and the envelope (E) proteins that are mainly required for viral assembly and viral envelope formation.5,6 The nonstructural proteins include key enzymes necessary for viral replication, such as the viral proteases and the RNA-dependent RNA polymerase (RdRp). Owing to their particular importance as druggable targets, these enzymes are described in further detail in the main body of the review. Another potential pharmacologic target is the S-glycoprotein, which has also been heavily investigated as the main target of antibodies, elicited by both natural infection and by vaccination. This is the case for all the main vaccine platforms that have proven most successful so far, such as vaccines based on mRNA (BNT162b2 and Spikevax) or adenoviral vector vaccines (ChAdOx1-S and Ad26.COV2.S). 7 Therefore, the evolution of the S-glycoprotein remains closely monitored to track the development of mutations that may favor immune evasion (Figure 1). In this regard, some of the so-called variants of concern (VOCs), that is, B.1.1.7 (alpha), B.1351 (beta), P.1 (gamma), B.1.617.2 (delta), and B1.1.529 (omicron) 8 are already characterized by increased transmissibility and severity, or as compared with the original strain.9,10 This, in turn, can partially explain the variable re-infection, hospitalization, and death rates observed during the multiple waves of infection. 11

Structural features of SARS-CoV-2 and its main VOC. Key mutations in the S-glycoprotein are displayed for each VOC.
While the symptoms of SARS-CoV-2 infection, if present, vary greatly between individuals, the development of severe COVID-19 is usually accompanied by three stages: (a) early infection, which includes the various steps of viral replication (Figure 2) and is associated with mild symptoms; (b) pulmonary involvement, in which there is adaptive immunity stimulation and the predominance of respiratory symptoms; and (c) hyper inflammation, which is associated with immune dysregulation and often includes the Acute Respiratory Distress Syndrome (ARDS). In addition, COVID-19 patients have been reported to display a circadian clinical course, with deterioration after initial improvement, which is consistent with a delayed and exaggerated immune activation.12,13 This hyperactivation can lead to a ‘cytokine storm’, possibly accompanied by dysregulation of pulmonary macrophages, dendritic cells, and leading to ARDS in these patients. 14 In line with this, multiple inflammatory cytokines and chemokines [interleukin (IL)-1, IL-6, IL-7, IL-8, IL-9, IL-10, granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon (IFN), interferon-gamma-induced protein (IP-10), monocyte chemoattractant protein-1 (MCP-1), and macrophage inflammatory protein-1 (MIP-1)] are elevated in the blood of COVID-19 patients, especially those requiring admission to an intensive care unit (ICU).15,16 Consequently, the hyperinflammatory response to COVID-19 shares biochemical similarities with the macrophage activation syndrome, implying that targeting the innate immune system could be a viable technique to reduce disease severity. 17

Schematic depiction of the SARS-CoV-2 life cycle. (A) SARS-CoV-2 binds to the surface receptor, angiotensin-converting enzyme 2 (ACE-2), on the host cell and releases its RNA genome in the cytoplasm
Although the morbidity and mortality of the infection have been significantly reduced by the implementation of broad vaccination campaigns, 18 SARS-CoV-2 transmission is still ongoing leading to further hospitalizations and deaths. It is therefore generally accepted that effective therapies will be an indispensable complement to vaccination strategies. At present, only few drugs are approved at least in some countries for use against SARS-CoV-2 infection. These include antiviral formulations (molnupiravir, nirmatrelvir/ritonavir, remdesivir, several monoclonal antibodies) or medicaments to decrease immune hyperactivation (dexamethasone, tocilizumab, baricitinib) [sources: WHO https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-therapeutics-2022.2; United States Food and Drug Administration (FDA): https://www.fda.gov/drugs/emergency-preparedness-drugs/coronavirus-covid-19-drugs].
Notably, with the important exception of nirmatrelvir, the approved drugs in the list, as well as several drug candidates, were devised following a drug repurposing approach. In comparison with the
A plethora of repurposed drugs have been tested as the beginning of the SARS-CoV-2 outbreak, several of which had been already proposed during the earlier, and more contained, epidemics of SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV). 21 Although the disappointing clinical results of some repurposed compounds have dampened the early hope that an effective therapy could be immediately devised, some drugs – such as molnupiravir, remdesivir, baricitinib, dexamethasone, and tocilizumab – have shown benefit in some late stage randomized clinical trials. These drugs have so far been mostly tested in monotherapy, although previous experience with RNA viruses, or retroviruses, such as hepatitis C virus (HCV) 22 or human immunodeficiency virus (HIV), 23 respectively, suggests that a potent combination therapy might be required to reproducibly decrease morbidity and mortality over time.
The current review provides an up-to-date outline of the most relevant repurposing attempts against SARS-CoV-2/COVID-19, including information on their mechanisms of action, pharmacokinetics, safety as well as their most relevant preclinical and clinical results. Moreover, the potential future developments in drug repurposing against SARS-CoV-2 are discussed, with particular focus on the possibility of combining effective compounds in a potent cocktail of antiviral and immune-modulating compounds.
Antivirals
Drugs that inhibit key steps of the viral life cycle are generally regarded as the most straightforward and least toxic option to counteract acute viral infections. Their use is often more effective during the early stages of the infection, but they can be considered for later stages as well, if residual viral replication is suspected to fuel persistent inflammation. Some of the antiviral drugs repurposed for SARS-CoV-2 treatment act through direct inhibition of the activity of key viral enzymes, including two of the most successful repurposing attempts so far (molnupiravir and remdesivir) (Figure 3), while others modulate some cellular factors necessary for viral replication. The sections below describe in detail the rationale and results of the most relevant antivirals tested so far against SARS-CoV-2.

Summary and mechanism of action of the RdRp inhibitors described in the review.
Molnupiravir
Molnupiravir (also known as EIDD-2801 and MK-4482, commercial name: Lagevrio) is an orally available prodrug that inhibits RNA viral replication (Figure 3) by inducing catastrophic mutations during transcription by the viral RdRp.
24
The drug was initially developed by Emory University’s Drug Innovation Ventures for treatment of influenza.
25
while Merck and Ridgeback Biotherapeutics spearheaded the attempts to repurpose it for the prevention and treatment of COVID-19.
Remdesivir
Remdesivir (GS-5734, commercialized as Veklury) is a 1’-cyano-substituted adenosine nucleotide analogue prodrug, and broad-spectrum antiviral with potential activity against a variety of RNA viruses. 30 Remdesivir acts by inhibiting the viral RdRp (Figure 3) and had been initially tested against Ebola Virus infection, without clinical success. 31 Remdesivir is in turn a derivative of the GS-441524 nucleoside analogue, which is used for treating feline infectious peritonitis caused by infection with feline coronavirus. 32 The intracellular delivery of GS-441524 and remdesivir is enhanced by the Pro-Tide technology. 33 Moreover, the conversion of remdesivir to an active metabolite, as with molnupiravir and other nucleoside analogues requires intracellular phosphorylation. In line with this, the addition of a monophosphate prodrug to the nucleoside dramatically increased its intracellular transport and activation 34 and was one of the significant achievements that allowed the development of this drug.
In terms of efficacy, it has been demonstrated that remdesivir potently reduces SARS-CoV-2 replication
The clinical results of remdesivir have been mixed. On one hand, remdesivir was the first FDA-approved medicine (https://www.fda.gov/media/137564/download.) for treating hospitalized COVID-19 patients. This was based on a preliminary report from a randomized, double-blind, placebo-controlled study enrolling 1062 hospitalized patients with COVID-19. The study showed that time to recovery and respiratory tract infections were significantly lower in treated patients. 36 On the other hand, subsequent reports failed to highlight a significant effect of remdesivir for moderate or severe COVID-19,37,38 including the interim analysis of the WHO Solidarity trial, which included 2750 patients. 39 More recently, however, an early three-day course of remdesivir in patients with mild/moderate COVID-19 was shown to decrease the likelihood of hospitalization and death. 40
Despite these promising results, the widespread early adoption of remdesivir might be impaired by its administration route (parenteral), which limits its use outside hospital settings, and by its suboptimal
Favipiravir
Favipiravir (T-705, commercialized as FabiFlu) is a purine nucleic acid analogue which can inhibit the RdRp (Figure 3) of several RNA viruses, including norovirus, Ebola virus, and influenza virus. 44 Favipiravir was approved for treatment of influenza virus in Japan in 2014.
Inside cells, favipiravir is converted to an active phosphoribosylated form [favipiravir-ribofuranosyl-5’ triphosphate (RTP)], which is recognized as a substrate by the RdRp, inhibiting its activity, including that of SARS-CoV-2 (Figure 3). 45 The recommended therapeutic dose of favipiravir, according to the Japanese Association for Infectious Diseases, is 1800 mg twice a day on day 1, then 800 mg twice a day for 7 days, with the possibility of continuing for up to 14 days if needed. 46 Multiple clinical trials testing favipiravir in patients with COVID-19 have been conducted so far, with some, including an open-label phase III study, suggesting that favipiravir might accelerate recovery time, 47 disappearance of symptoms, 48 and viral clearance. 49 Mixed effects, however, were obtained when analyzing time to reverse transcription polymerase chain reaction (RT-PCR) negativization, and other trials did not observe any statistical benefit,50,51 or only moderate benefit on symptoms such as pyrexia and cough. 52
The overall quality of evidence for favipiravir is still low, with few randomized placebo-controlled trials published so far. Recent meta-analyses have attempted to quantify a potential benefit of favipiravir based on the clinical evidence accumulated so far. These analyses could not highlight an improvement in mortality rate or mechanical ventilation,53,54 but left open the possibility that favipiravir might facilitate viral clearance and prevent hospitalization in patients with mild or moderate COVID-19. 54
Ribavirin
Ribavirin (RBV) (l-β-
Lopinavir/ritonavir
Lopinavir was suggested, along with other HIV protease inhibitors, such as darunavir, as a candidate drug to target the main protease (MPro) of SARS-CoV-2. In particular, lopinavir was tested in its common co-formulation (marketed as Kaletra) with the cytochrome P450 3A (CYP3A) inhibitor ritonavir. Ritonavir is known to increase the activity of several protease inhibitors
61
by decreasing liver first-pass metabolism.
61
Interestingly, during the SARS-CoV epidemic in 2003, lopinavir had been shown to inhibit the main protease of the virus
Chloroquine and hydroxychloroquine
Apart from malaria, chloroquine (CQ) and hydroxychloroquine (HCQ) are approved by the FDA for treatment of autoimmune diseases such as systemic lupus erythematosus and rheumatoid arthritis. 66 The repurposing of CQ and HCQ as antivirals has been proposed mainly based on their ability to impair endosomal acidification, which is required for the activation of endosomal proteases responsible for the initiation of virus/endosome fusion. 66
Although during the early stages of the pandemic, CQ/HCQ were shown to impair SARS-CoV-2 replication
Amodiaquine
Amodiaquine (AQ), is like, CQ/HCQ, another antimalarial derivative of the quinoline class of chemicals. AQ was proposed as a compound with a similarly pleiotropic, but more potent, antiviral activity as compared with CQ/HCQ.
75
Ivermectin
Ivermectin is a widely used antiparasitic medication that is approved by the FDA for both veterinary and human medicine.
79
The repurposing of ivermectin for SARS-CoV-2 use was proposed due to the well-established broad antiviral activity of this drug
Immune modulators
The severity of COVID-19 is determined largely by a dysregulated immune response, potentially leading to a cytokine storm and damage to multiple organs. 91 It is not surprising, therefore, that many of the drug repurposing approaches have focused on compounds able to decrease immune hyperactivation (Figure 4). Although some broad-spectrum weak antivirals, such as CQ/HCQ and ivermectin, can also play a role in immune function, the following sections will detail only the most popular approaches specifically focused on drugs whose primary or only role is immunomodulatory.

Inhibition of SARS-CoV-2-mediated cytokine storm by immune-modulating drugs. A hyperinflammatory response or ‘cytokine storm’ has been proposed as a significant pathogenetic contributor and as a cause of ARDS in COVID-19 patients. Toll-like receptors (TLRs) in macrophages recognize ssRNA from viruses such as SARS-CoV-2 and activate nuclear factor B (NF-κB) signaling,
Dexamethasone
Dexamethasone is a synthetic corticosteroid immunosuppressant that is widely used to reduce inflammation and immune responses in a plethora of conditions, particularly autoimmune, respiratory, and severe allergic syndromes. 92 Owing to its low cost and high potency, dexamethasone was quickly proposed to treat COVID-19 patients and was the first drug to show a significant benefit in the large RECOVERY trial. 93 Specifically, this trial demonstrated that a moderate dose of dexamethasone (6 mg daily for 10 days intravenous administration) can decrease mortality in hospitalized patients with COVID-19 and respiratory failure who required supplemental oxygen or mechanical ventilation. 94
A subsequent trial further demonstrated that intravenous dexamethasone plus standard of care, compared with standard of care alone, resulted in a statistically significant increase in the number of ventilator-free days (days alive and free of mechanical ventilation) over 28 days in patients with moderate COVID-19 or ARDS. 95 Moreover, also a short course of dexamethasone was demonstrated to decrease inflammation levels (as shown by lower C-reactive protein levels) and accelerate recovery time. 96 Based on this evidence, dexamethasone (and corticosteroids in general) were the first drugs recommended by the WHO, in November 2020, for the treatment of severe or critical COVID-19 (COVID-19: living guideline WHO). Dexamethasone is now routinely used to manage the cytokine storm and hyperinflammation in COVID-19 patients, and it is also recommended as a treatment for immune thrombocytopenic purpura caused by COVID-19. 97
Tocilizumab
Tocilizumab (TCZ, commercialized as Actemra) is a humanized monoclonal antibody (mAb) that belongs to the immunoglobulin G1K subclass and binds to the soluble and membrane-bound interleukin-6 receptor (IL-6R) 98 (Figure 4). The repurposing of TCZ was driven by its well-known clinical efficacy, pharmacokinetics and favorable safety profile for treatment of rheumatoid arthritis. 99 Apart from rheumatoid arthritis, TCZ is also used to treat cytokine release syndrome/systemic inflammatory response syndrome that can occur as a side effect of cancer immunotherapy 100 or chimeric antigen receptor (CAR)-T therapy. 101 Importantly, IL-6 levels have been linked to ARDS severity and outcome in COVID-19 patients. 102 In line with this, TCZ has been recommended as an effective treatment for hospitalized COVID-19 patients, especially for those who display high levels of circulating IL-6. 103
In a study conducted in China during the early stages of the pandemic, TCZ treatment improved oxygen intake, lung lesion opacity and lymphocyte counts, and decreased the inflammatory marker C-reactive protein. 104 All patients were discharged on average 15.1 days after receiving TCZ. 104 Moreover, another open-label, noncontrolled trial conducted in China showed that a single intravenous dose of 400 mg TCZ can be effective in COVID-19 patients with mild-to-moderate ARDS. 105 On the contrary, placebo-controlled phase III studies showed mixed results of TCZ in hospitalized patients with severe COVID-19 pneumonia.106,107 Heterogeneous results were also observed in other randomized trials.108–114 Despite these mixed results, recent meta-analyses suggest that TCZ use might be associated with lower mortality, 115 and the WHO has recommended its use in COVID-19 patients (COVID-19: living guideline WHO).
Baricitinib
Baricitinib (commercial name: Olumiant) is an anti-inflammatory drug that is approved for treatment of rheumatoid arthritis. 116 At the molecular level, baricitinib acts as a selective inhibitor of the Janus kinase 1 and 2 (JAK 1/2) (Figure 4) at nanomolar concentrations. 117 Through its inhibitions of JAKs, baricitinib impairs the downstream activation of signal transducers and activators of transcription (STATs), thus impairing their nuclear translocation and the response of immune cells to pro-inflammatory cytokine secretion. 118 Owing to its anti-inflammatory effect, favorable route of administration (oral), and safety, 119 baricitinib was proposed as a therapeutic candidate for COVID-19. After proving effective in resolving lung inflammation in SARS-CoV-2–infected rhesus macaques, 120 baricitinib, plus standard of care, proved able to reduce mortality of hospitalized COVID-19 patients in a randomized placebo-controlled phase III clinical trial. 121 Recently, in January 2022, the WHO has included baricitinib as one of the recommended therapeutic options for treatment of severe COVID-19, a list previously including corticosteroids and TCZ (COVID-19: living guideline WHO).
Acalabrutinib
Acalabrutinib (also known as ACP-196, marketed as Calquence) is an irreversible covalent inhibitor of Bruton’s tyrosine kinase (BTK) (Figure 4). Three BTK inhibitors – ibrutinib, zanubrutinib, and acalabrutinib – are approved by the FDA to treat B-cell malignancies such as chronic lymphocytic leukemia and mantle B-cell lymphoma. 122 Acalabrutinib is a second generation BTK inhibitor and is characterized by better oral absorption, higher selectivity, and lower adverse effects, as compared with ibrutinib.122,123 Interestingly, before the outbreak of SARS-CoV-2, BTK inhibitors have been shown to protect against severe lung injury in preclinical research.124,125
At the molecular level, BTK inhibition might mitigate COVID-19 by decreasing macrophage activation. Macrophages contribute to the COVID-19 inflammatory response through phagocytic absorption of viral particles or cellular debris harboring viral ssRNA. Using double-stranded areas in its secondary structure, the viral ssRNA can bind to TLR7 and TLR8, as well as TLR3, thus recruiting and activating BTK. 126 BTK in turn can activate NF-κB signaling, leading to the generation of pro-inflammatory cytokines and chemokines and potentially to a cytokine storm by boosting monocyte/macrophage and neutrophil recruitment during the late stages of severe COVID-19 infection 127 (Figure 4).
Early data on the off-label clinical use of acalabrutinib showed improved oxygenation, normalization of IL-6 levels, and reduction of lymphopenia in patients with severe COVID-19. 128 In this small study, 8/11 treated patients in the supplementary oxygen cohort could be discharged on room air and 4/8 patients in the mechanical ventilation cohort could successfully undergo extubation. 128 Unfortunately, in subsequent phase II randomized trials (CALAVI program, NCT identifiers: NCT04380688 and NCT04346199), acalabrutinib did not decrease mortality and respiratory failure (https://www.astrazeneca.com/media-centre/press-releases/2020/update-on-calavi-phase-ii-trials-for-calquence-in-patients-hospitalised-with-respiratory-symptoms-of-covid-19.html). Therefore, the use of acalabrutinib, as well as that of other BTK inhibitors, is currently not recommended for treatment of COVID-19.
Fostamatinib
Severe COVID-19 is often associated with thrombocytopenia.
129
Fostamatinib (marketed as Tavalisse or Tavlesse) is an FDA-approved inhibitor of the spleen tyrosine kinase (SYK) and is typically used to prevent chronic immune-mediated destruction of platelets.
130
In several immune cells, such as, neutrophils, platelets, monocytes, and macrophages, SYK signaling occurs
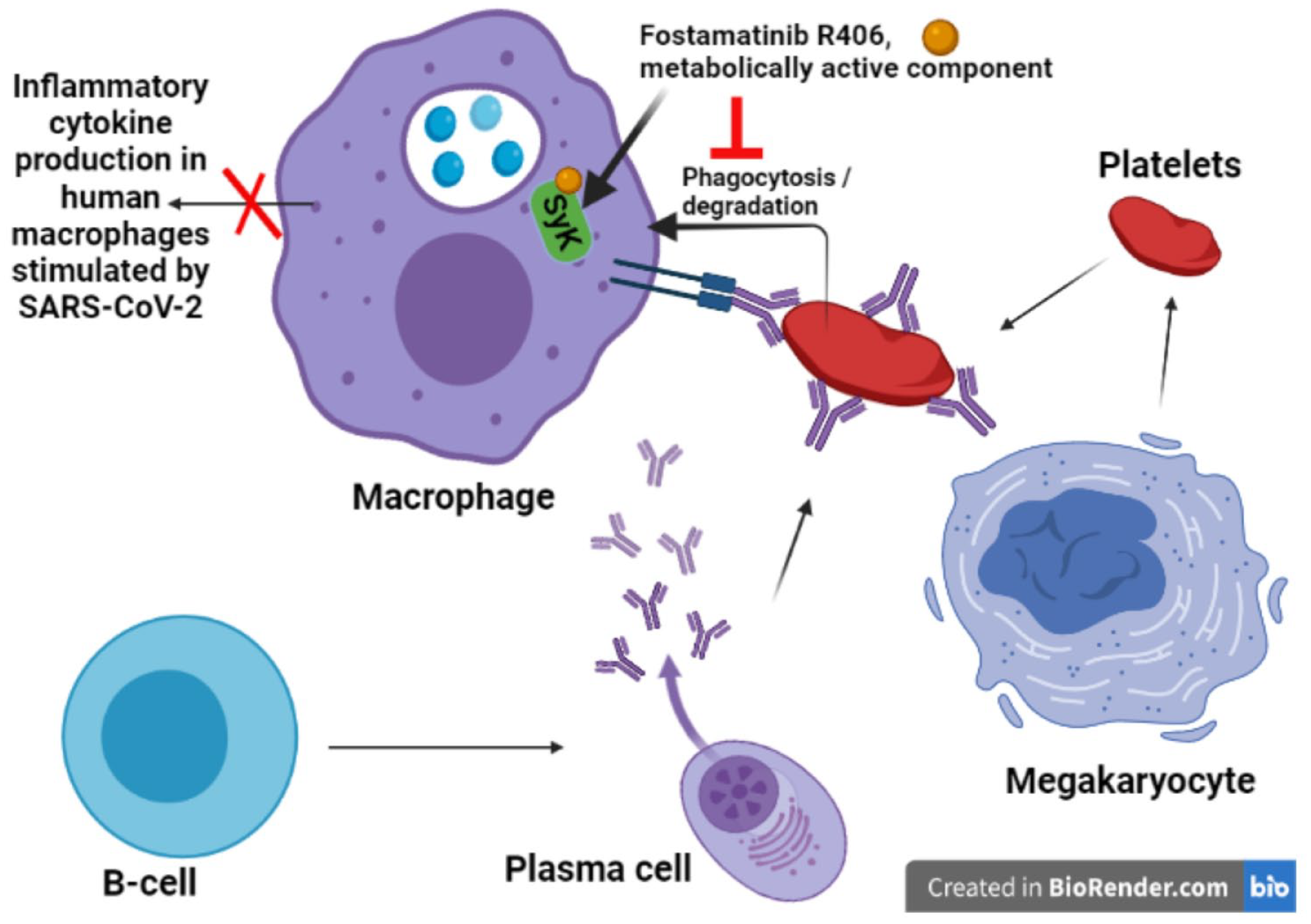
Activity of fostamatinib in decreasing thrombocytopenia. The metabolically active component of fostamatinib, R406, inhibits the enzyme spleen tyrosine kinase (SYK). This prevents the destruction of platelets induced by the SYK-dependent cytoskeletal reorganization during phagocytosis.
In line with this, a randomized placebo-controlled trial of fostamatinib, plus standard of care, was associated with lower mortality and faster recovery in hospitalized COVID-19 patients. 136 Despite these promising results, the number of enrolled patients was small, and definitive evidence of the efficacy of fostamatinib is still lacking. An ongoing phase III clinical trial is expected to provide additional data on the potential of fostamatinib in treating COVID-19. 137
Drug combinations
Combining effective, or partially effective compounds might provide several advantages over monotherapies. Combinations of antivirals could increase the barrier to resistance mutations and extend the time window in which treatment is effective, potentially leading to full suppression of viral replication. Previous experience with other RNA viruses 22 or retroviruses 138 suggests that a combination of two/three antivirals might be required to achieve reproducible results in wide patient cohorts and across different viral subtypes/variants. On the contrary, only sporadic cases of drug resistance mutations have been observed so far in SARS-CoV-2, 139 possibly due to the short period of administration or of evolutionary constraints of the viral enzymes targeted. Moreover, data on drug–drug interaction and efficacy of antiviral combinations in SARS-CoV-2–infected patients are still scarce. 140 Initial guidelines recommended avoiding the combination of remdesivir and CQ/HCQ, due to potential toxicity. In addition, combining favipiravir and HCQ did not provide any clinical benefit in hospitalized patients with moderate-to-severe COVID-19. 141 Similarly disappointing results were obtained by combining HCQ and the antibiotic azithromycin. 142 On the contrary, a combination of lopinavir/ritonavir and IFN-β1b showed more effective than single treatments in an open-label, randomized, phase II trial enrolling hospitalized adults with mild or moderate COVID-19. 143
Encouraging results were also obtained by associating lopinavir/ritonavir and IFN-α with RBV. 30 In another trial, however, the combination of RBV and IFN did not appear to improve clinical results in COVID-19 patients. 144 Indeed, current guidelines indicate that the combination of RBV and IFN should be avoided in patients with COVID-19 (National Institute of Health, 2020; WHO, 2021b). More generally, the evidence of the benefit of combination treatments including IFN for SARS-CoV-2 patients is limited, and the use of IFN can be associated with significant toxicities. 145 Also attempts to further increase the efficacy of the combination by adding HCQ and umifenovir (i.e. another repurposed drug used to inhibit fusion of RNA viruses) 146 were not successful. 147 Similarly unsuccessful was the attempt to potentiate the effect of RBV by repurposing the direct acting antivirals sofosbuvir and daclatasvir, which are typically used to HCV infection. 148 Despite this, other trials suggest that combining sofosbuvir and daclatasvir with a standard of care including other repurposed compounds – such as HCQ, remdesivir, ivermectin, or lopinavir/ritonavir – might have a marginal benefit.149,150 The evidence is, however, still insufficient to support the addition of sofosbuvir and daclatasvir to antiviral combinations for treating COVID-19 patients.
Overall, clinical data on purely antiviral combinations have been underwhelming so far. Some recent lines of preclinical evidence suggest that other repurposed compounds in the pipeline might help to fill this gap. In this regard, a recent study showed that the use of pyrimidine biosynthesis inhibitors, such as brequinar, can significantly potentiate the effect of the RdRp inhibitors remdesivir and molnupiravir in a mouse model of SARS-CoV-2 infection. 151 Although brequinar is not approved by the FDA and can be associated with high toxicity, 152 the dose used in the study was well tolerated and effective.
In another study, conducted by some of us, the CYP3A inhibitors cobicistat was shown to decrease viral replication and disease progression in a Syrian hamster model of SARS-CoV-2 infection, as well as to increase the potency of remdesivir.
153
The doses of cobicistat required to achieve an antiviral effect were higher than those employed to boost the effect of HIV protease inhibitors in antiretroviral therapy, thus potentially explaining lack of effect of the lower doses that had been observed in the early phases of the pandemic, when SARS-CoV-2 patients received cobicistat with darunavir in the standard formulation.
154
Of note, although this
Other approaches at drug combinations, typically used in more advanced stages of COVID-19, include associating antivirals and immune-modulating drugs or a cocktail of immune-modulating drugs. Most trials of this type have been conducted by associating remdesivir with immune modulators such as the TCZ or other monoclonal antibodies targeting pro-inflammatory pathways. None of these combinations, however, proved a significant clinical benefit in hospitalized patients with COVID-19 156 (https://www.roche.com/media/releases/med-cor-2021-03-11). On the contrary, the combination of remdesivir and baricitinib accelerated clinical improvement in hospitalized COVID-19 patients as compared with remdesivir only. 157 These promising results led to the approval of Emergency Use Authorization of this drug combination by the FDA. Similarly, positive results were obtained in an observational clinical trial by testing the combination of baricitinib with corticosteroids, which led to improved pulmonary function. 158
Conclusion
The Herculean efforts in drug repurposing have led to the identification of several antiviral and immune-modulating compounds to treat SARS-CoV-2/COVID-19. Although many of the proposed compounds could not pass rigorous clinical scrutiny, a subset of drugs proved able to decrease the mortality and morbidity of the infection, thus confirming the potential of the drug repositioning approach as a fast-response framework to confront sudden viral outbreaks or novel epidemics. Despite their promise, drug combinations have so far yielded only sporadic or early-stage results (Figure 6). Research is ongoing on the optimal compounds/formulations that could allow to improve current treatments at both the early and the advanced infection stages.

Mechanism of action, effectiveness, and stage of development of the drug combinations described in the review. Color codes and bar lengths indicate, respectively, the available results/approval status and stage of development of each drug combination.
