Abstract
Introduction:
Pulmonary arterial hypertension is a progressive haemodynamic disease with high morbidity and mortality. Of the different treatments available, the prostacyclin analogues are the drugs of choice for high-risk patients, with treprostinil being the most commonly used drug in Argentina.
Methodology:
The objective of this study is to perform a retrospective evaluation of the efficacy and safety of subcutaneous treprostinil in regular clinical practice in Argentina in 51 patients with pulmonary arterial hypertension after 12 months of follow-up.
Results:
The results showed that treatment with subcutaneous treprostinil is associated with a significant improvement in different clinical efficacy parameters: 65% reduction in advanced functional class (p < 0.0001), 130-m increase in the 6-min walk test (p < 0.0001), 65% reduction in the pro B-type natriuretic peptide value (–531 pg/dL; p < 0.0001), significant reduction of 15.7% in pulmonary vascular resistance [–1.3 wood units (WU); p < 0.0001], improved cardiac index with an increase of 16.7% (+0.4 L/min/m2; p = 0.002), as well as a high survival rate (92%) and a 44% incidence of combined events (mortality, heart failure, syncope and/or lung transplantation), without a significant increase in previously reported adverse events. The risk stratification evaluation according to ESC/ERS guidelines showed a significant decrease in the proportion of patients at high risk after the treatment period (p = 0.004).
Conclusions:
These real-world results corroborate the efficacy and safety of subcutaneous treprostinil, even at high doses, and open up the possibility of improving its current use in clinical practice as a first-line therapy, especially in high-risk patient profiles.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a multifactorial and clinically progressive disease with a low incidence and prevalence.
PAH is characterised by presenting a mean pulmonary arterial pressure (mPAP) value of > 20 mmHg, pulmonary vascular resistances (PVR) ⩾ 2 Wood units (WU) and pulmonary capillary wedge pressure (PCWP) ⩽ 15 mmHg, and with pulmonary vascular hypertension. 1
PAH has high morbidity and mortality rates, in which right ventricular dysfunction is a relevant prognostic indicator. 2 The survival of this population has improved significantly in recent decades as a consequence of greater biological knowledge and the introduction of new therapeutic strategies, among other factors. Among the different treatments available, the parenteral prostanoids are the pharmacological strategy of choice in the high-risk population (class I indication), since scientific evidence and clinical experience have demonstrated that prostanoids have a positive impact on event-free survival rate and on quality of life.3,4 Despite these positive results, prostanoids present a high rate of adverse events (AEs), 3 whereby the risk–benefit ratio of this treatment must be properly balanced on a patient-specific basis.
Treprostinil is the most commonly used prostacyclin analogue in Argentina, with more than 15 years of experience in clinical practice. However, few articles reporting results of treprostinil use in this country have been published hitherto. 5
In this scenario, this study focuses on the effects of subcutaneous treprostinil (scTRE), a route of administration that is possible due to a greater stability and half-life of this prostacyclin analogue, especially when compared with the synthetic prostacyclin epoprostenol. 6 The objective was to describe scTRE use in regular clinical practice conditions in Argentina by means of the analysis of data from patients with a confirmed diagnosis of PAH, at 12 months of follow-up of treatment with treprostinil.
Methodology
Study population
The data of the patients analysed in this study were taken from the scTRE in pulmonary hypertension (TRES-HP) national registry. The information was obtained between January 2018 and August 2019 through seven different health centres in the Argentinian Republic (Buenos Aires and Santa Fe). The data were collected using a closed questionnaire. All patients provided their consent for the inclusion of their data in the registry knowing that it could be used for investigational purposes. The study was supervised and approved by the ethics committees of all the sites involved and it was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice Guidelines recommendations.
The study included consecutive patients from urban environment with a confirmed diagnosis of PAH for more than 6 months and right cardiac catheterisation with a mPAP ⩾ 25 mmHg, PVR ⩾ 3 WU and PCWP ⩽ 15 mmHg (according to 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension) 4 who were on stable treatment with scTRE for a period equal to or greater than 12 months or who required the discontinuation of treatment due to a cardiovascular event or AE. The baseline variables were recorded before the beginning of the infusion of treprostinil by the investigators, and the kinetics of administration during the study were quantified.
Study variables
Demographic (gender and age) and clinical [aetiology, syncope and heart failure (HF)] variables were recorded. HF was defined, accordingly to guidelines, 7 as an elevated intracardiac pressure and/or inadequate cardiac output at rest and/or during exercise due to a structural and/or functional abnormality of the heart that produces characteristic symptoms like breathlessness, ankle swelling or fatigue that may be accompanied by signs such as elevated jugular venous pressure, pulmonary crackles and peripheral oedema.
Several functional data items such as functional class (FC), the 6-min walk test (6MWT) and biochemical variables (red blood cell and platelet count and serum biomarkers such as the pro B-type natriuretic peptide (pro-BNP) were collected. The pro-BNP values were defined as described in the European Society of Cardiology (ESC)/European Respiratory Society (ERS) guidelines.
Echocardiographic variables were also recorded according to the recommendations of the international guidelines, including right ventricular systolic function (RVSF) as defined by the investigator, and tricuspid annular plane systolic excursion (TAPSE). The haemodynamic variables included cardiac index (CI), right atrial pressure (RAP) and pulmonary vascular resistance (PVR). The data were recorded by the investigators according to regular clinical practice in baseline conditions and at 3, 6, 9 and 12 months after the beginning of the infusion of treprostinil.
Of the aforementioned variables, the following were defined as risk variables: FC III/IV (advanced FC), deterioration in RVSF, reduced TAPSE (⩽ 16 mm), CI ⩽ 2 L/min/m2 and RAP ⩾ 14 mmHg. The treatment efficacy primary outcome was defined as the combined cardiovascular events occurring in the course of the 12-month post-infusion period (overall mortality, hospitalisation due to HF, syncope and/or lung transplant (with or without heart transplant depending on the patient’s condition and the cause of the PAH).
With regard to safety, the AE associated with the administration of the drug (pain at the injection site, phlogosis, local infection, thrombocytopenia and systemic effects) were evaluated and described.
Statistical analysis
The normality of the variables was determined taking into account different criteria such as symmetry of the histogram curve, the differences between mean and median and the kurtosis value. The Shapiro–Wilk test was also used to ascertain the normality of the dataset analysed.
The qualitative variables were described as frequencies and percentages. The quantitative variables were expressed as mean and standard deviation (SD) or mean and interquartile range (IQR) depending on whether distribution was normal or not. A bivariate analysis was conducted using the Student’s t-test, Chi-square test, Wilcoxon test, Mann–Whitney U test or McNemar test depending on the distribution and the characteristics of the variables. The odds ratio (OR) and the 95% confidence interval (95% CI) were estimated. Complementary to this, the effect of treprostinil in the distribution and classification of the patients according to their FC and the ESC/ERS guidelines was analysed throughout the treatment period. Moreover, the deterioration in RVSF and TAPSE depending on FC was analysed by means of a Chi-square test.
In addition, a descriptive survival analysis was performed using Kaplan–Meier (K-M) curves for the overall mortality (survival) and mortality variables depending on whether the patients were given monotherapy or had combined treatment from the beginning. K-M analysis was also performed to assess the event-free rate for the combined variable of mortality, hospitalisation for HF, syncope and/or lung transplant (with or without heart transplant depending on the patient’s condition and the cause of the PAH).
A comparative analysis was performed using the log-rank test for the combined variable (death, HF, syncope and/or lung transplantation) depending on the treatment received by the patient (combined or not). These analyses were also performed for clinical variables such as FC or the pro-BNP value, as well as according to the dose of treprostinil taken by the patient.
Finally, Cox proportional hazards regression models were used to explore the association between the occurrence of the combined event (death, HF, syncope, and/or lung transplantation) and treatment with treprostinil. The same analysis was also used to explore the association between the occurrence of the combined event and demographic variables (gender, age) or clinical variables such as advanced FC, deterioration in the RVSF, reduced TAPSE, low CI, elevated RAP, the pro-BNP value and the result of the 6MWT.
Statistical significance was established for all analyses with a value of p < 0.05, and the STATA 14 programme (StataCorp LLC, Texas, USA) was used for calculations.
Results
Efficacy results
A total 64 consecutive patients from an urban environment were included in the TRES-HP registry (Figure 1) and a total of 51 patients diagnosed with PAH were included in the analysis, with a mean age of 51 years (±16), a predominance of women (79.5%) and different aetiologies (Table 1). Among the baseline clinical characteristics, the presence of heart failure was observed in 43 (84%) of the patients, and syncope in 23 (45%).

Consort diagram.
Baseline characteristics and aetiology of the study patients.
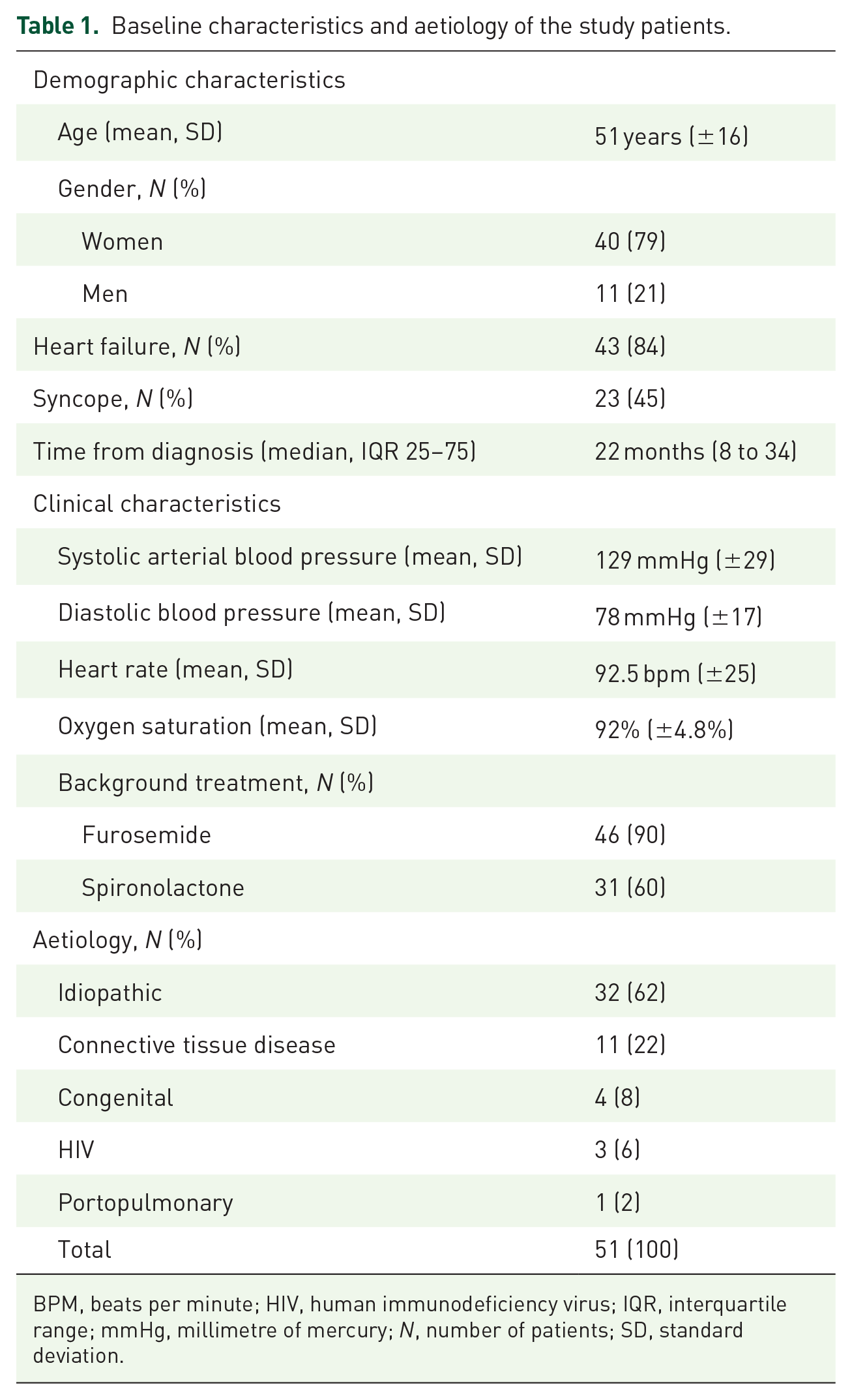
BPM, beats per minute; HIV, human immunodeficiency virus; IQR, interquartile range; mmHg, millimetre of mercury; N, number of patients; SD, standard deviation.
Baseline treatment was combined in 32 (62.7%) of the patients included in the study. The combined treatments that were given together with scTRE include endothelin receptor antagonists (ERA) such as ambrisentran, bosentan and macitentan and phosphodiesterase 5 inhibitors (PDE5) such as tadalafil and sildenafil, as well other inhaled prostanoids. The remaining 19 (37.3%) study patients came from baseline treatment with monotherapy (sildenafil, bosentan or macitentan), which was maintained throughout the study together with the scTRE treatment. A complete description of the distribution of the patients depending on the type of baseline treatment at the beginning of the study is provided in Table 2. All the patients also began treatment with scTRE at baseline and maintained it for a period of equal to or more than 12 months, barring the cases in which the treatment was discontinued due to AEs. Treatment with inhaled prostanoids was discontinued according to routine clinical practice after starting the scTRE administration.
Distribution of the patients according to the baseline therapy at the beginning of the study.

One patient was treated with sildenafil and ambrisentan for 11 months, while the other was treated with tadalafil and macitentan along with the inhaled prostanoid for 14 months.
Baseline characteristics for the functional, biochemical and haemodynamic variables, together with the echocardiographic parameters, are provided in Table 3, which also describes the relationship between the variables at baseline and after the 12 months of treatment.
Clinical and functional parameters at baseline and after the 12-month treatment period with scTRE.

6MWT, 6-min walk test; 95% CI, 95% confidence interval; CI, cardiac index; ERS, European Respiratory Society; ESC, European Society of Cardiology; FC, functional class; IQR, interquartile range; LEI, left ventricle eccentricity index; mPAP, mean pulmonary arterial pressure; Pro-BNP, pro B-type natriuretic peptide; PVR, pulmonary vascular resistance; RAP, right atrial pressure; RVSF, right ventricular systolic function; SD, standard deviation; TAPSE, tricuspid annular plane systolic excursion; WU, wood units.
p < 0.05. **Mean mPAP values was calculated based on information from 44 patients. There were no data at 12 months for the remaining patients.
With regard to the echocardiographic parameters, attention should be drawn to a deteriorated RVSF at baseline in 82% of the patients (mild 12%, moderate 47% and severe 23%) and an average TAPSE of 16.1 (SD: ±0.6), with 58.7% of the patients presenting a diminished value below 16 mm. The baseline haemodynamic parameters presented a mean RAP of 11.9 mmHg (SD: ±5), with RAP > 14 mmHg in 30.9% of the patients. CI was 2.4 L/m/m2 (IQR 25–75: 2–2.8), with CI ⩽ 2 L/min/m2 in 35.9% of the patients, whereas the median PVR was 8.3 WU (IQR 25–75: 6–15).
After 12 months of treatment, statistically significant differences were found for advanced FC, the 6MWT, pro-BNP, mPAP, TAPSE, RVSF, PVR and CI (p < 0.05 for all cases) with regard to the baseline values. Of these results, it should be underlined that the mean TAPSE value improved significantly when compared with the baseline level (19.5 mm; SD: ±0.6; p = 0.001), with 22.5% of patients with reduced TAPSE (<16 mm) [Figure 2(a)]. Another relevant finding was the significant quarterly increase in the distance travelled in the 6MWT with a final increment of 130 m (95% CI: 84 to 143; p < 0.0001) after 12 months of treatment versus the baseline value [Figure 2(b)]. The pro-BNP value also fell significantly after the period of treatment with treprostinil (−531 pg/dL; 95% CI: −409 to −829; p < 0.0001) [Figure 2(c)].

Effect of the treprostinil treatment on different variables at baseline and during the follow-up period (after 3, 6, 9 and 12 months of treatment). (a) Effect of the treatment with treprostinil on the 6-min walk test (6MWT). (b) Effect of the treatment with treprostinil on pro B-type natriuretic peptide (pro-BNP). (c) Effect of the treatment with treprostinil on tricuspid annular plane systolic excursion (TAPSE) value.
Patient distribution by FC throughout the treatment period is displayed in Figure 3. At the baseline, the distribution of FC was as follows: FC I 0%, FC II 10.2%, FC III 65.5% and FC IV 24.3%. By contrast, the following distribution was observed after 12 months of treatment: FC I 13.4%, FC II 53.3%, FC III 28.9% and FC IV 4.5%. These results showed that the percentage of patients with advanced FC diminishes significantly throughout the treatment period, with a total of 17 patients 33 with advanced FC after 12 months of treatment versus the 46 patients (90%) with advanced FC at baseline level (p < 0.0001).

Patient distribution by FC throughout the 12-month treatment period with treprostinil.
The analysis of the deterioration in RVSF according to FC showed that more than 90% of the patients with FC III or FC IV presented a deterioration in RVSF compared with 20% of the patients with FC II (Chi-square = 19.2; p < 0.0001). The analysis of the change between baseline and final TAPSE presented no statistical significance.
The evaluation of the risk stratification at baseline according to the ESC/ERS score showed that 46 (90%) patients had a high risk, while 5 (10%) patients had an intermediate risk. After 12 months of treatment with treprostinil, a high risk was observed in 20 (39.2%) patients, an intermediate risk in 17 (33.3%) patients and a low risk in 14 (27.5%) patients. These results show that the percentage of high-risk patients according to ESC/ERS classification was reduced by more than half after 12 months of treatment with treprostinil, with the change between baseline and the final high-risk percentage of patients being statistically significant (p = 0.004) as shown in Table 4. Overall, the ESC/ERS risk was reduced by 0.78 (95% CI: 0.55 to 1.02; p < 0.001), as shown in Table 3.
Risk stratification at baseline and after the 12-month follow-up period according to ESC/ERS guidelines.

ERS, European Respiratory Society; ESC, European Society of Cardiology; N, number of patients.
p < 0.05. **It cannot be calculated since baseline is equal to 0.
The descriptive survival analysis based on K-M curves for the overall mortality showed a survival rate of 92% after 12 months [Figure 4(a)], whereas the K-M curve for the combined variable of overall mortality, hospitalisation for HF, syncope and/or lung transplantation presented an incidence density of 43.1%, meaning that 22 patients had at least one of the events included in the combined variable [Figure 4(b)]. The survival analysis according to whether the patients received monotherapy or combined treatment from the beginning did not show significant differences between groups (baseline characteristics of both groups were comparable, see Supplementary Table 1). This result was maintained irrespective of whether the survival analysis was performed taking only overall mortality into account or the combined event variable. However, it is important to emphasise that the survival analysis for the combined variable according to treatment (monotherapy versus combined therapy) approached statistical significance (Chi-square = 3.25; p = 0.07), indicating that combined baseline therapy might hypothesise better long-term outcomes.
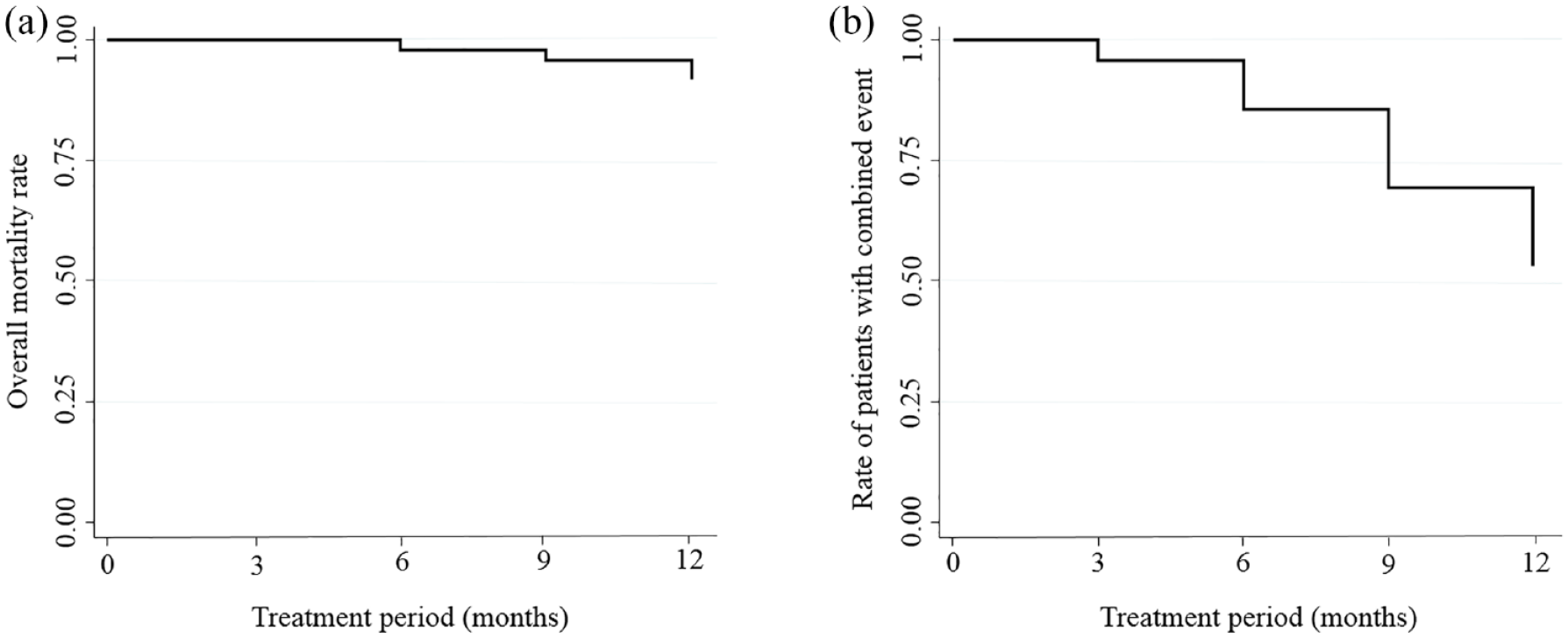
Survival analysis by Kaplan–Meier curves. (a) Overall mortality (survival) during the treatment period. A survival rate of 92% after 12 months is observed with four deaths. The deaths occurred: one after 6 months of treatment, another one after 9 months and the remaining two after 12 months. The causes were: two cases of sudden death, one case of heart failure and another from infection. Three patients were at high risk and one at intermediate risk according to ESC/ERS guidelines. (b) Survival for the combined variable of mortality, hospitalisation for heart failure, syncope and/or lung transplantation. An overall incidence of 43.1% is observed; this means that 22 patients presented at least one of the events included in the combined variable during the treatment period (17 patients suffered from heart failure, 4 patient deaths and 1 patient presented recurrent syncope).
On the contrary, the results also showed that patients with high pro-BNP values (Chi-square = 10.50; p = 0.001) presented a significantly greater incidence of combined events (mortality, HF, syncope and/or transplantation), with 21 (53%) patients affected, compared with the patients with low pro-BNP levels, in whom the incidence was zero [Figure 5(a)]. In the case of FC, patients with advanced FC during follow-up also displayed a greater incidence of combined events (Chi-square = 4.12; p = 0.04), with 12 (80%) patients affected versus the 10 (30%) cases for the milder functional classes [Figure 5(b)]. The Cox proportional hazard regression model corroborated these results since, among all the variables analysed, a statistically significant association was only found between the occurrence of the combined event and these two variables: high pro-BNP values [hazard ratio (HR) = 1.0003; 95% CI: 1.000011 to 1.000772; p = 0.04] and advanced FC (HR = 3.3; 95% CI: 1.4 to 7.7; p = 0.005). Therefore, high pro-BNP values and advanced FC could be associated with a greater risk of mortality, HF, syncope and/or lung transplantation.

Survival analysis by Kaplan–Meier curves for the combined variable of mortality, hospitalisation for heart failure, syncope and/or lung transplantation during the treatment period. (a) Analysis according to pro- BNP values. There is a significantly greater incidence (21, 53%) of combined events in the patients with high levels of pro-BNP after the 12 months of treatment. (b) Analysis according to functional class (FC) levels. Patients with advanced FC present a greater incidence of combined events (20; 43.4%) versus the group without advanced FC (2; 40%) after 12 months of treatment.
Table 5 presents the quarterly increase in the dose of treprostinil, reaching an average value of 33.8 ng/kg/min after 12 months (SD: ±11.5). The sample was divided in two groups, lower and higher doses, according to the median dose of scTRE (30 ng/kg/min). The incidence of mortality was 17% in the group with lower doses of treprostinil (mean dose: 27.7 ng/kg/min) compared with the group with higher doses (mean dose: 35.5 ng/kg/min), in which no event was reported. These data suggest that high doses of scTRE would be associated with a greater reduction in cardiovascular events. Despite this, the results of the comparative analysis showed that there is no statistically significant association between the dose of treprostinil and the occurrence of the combined event, although the log-rank test result (Chi squares = 3.31; p = 0.07) approached statistical significance.
Evolution of the mean dose of scTRE throughout the 12 months of treatment.
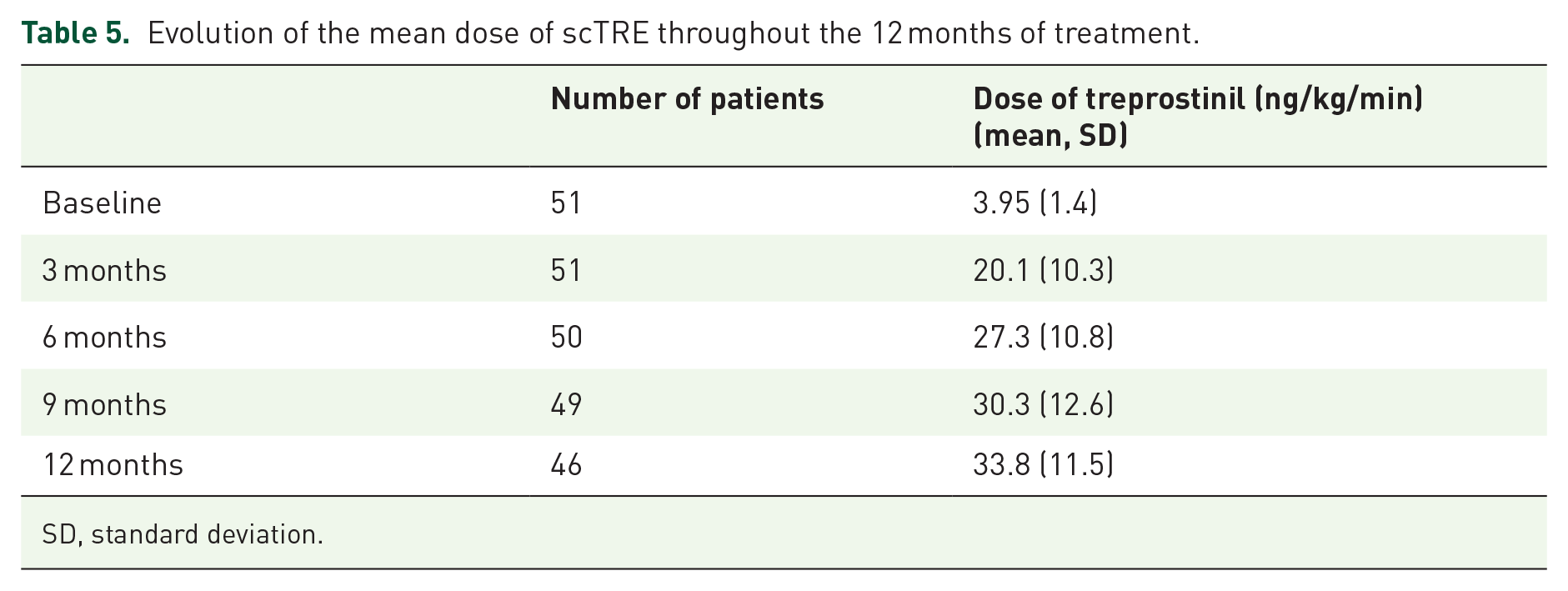
SD, standard deviation.
Safety results
The study’s mortality rate was 8%, with two cases of sudden death, one case of heart failure and another for an infectious condition. The overall discontinuation rate was 2% (one) and it was due to the presence of treatment-derived AEs. The most frequent AEs were local pain (42; 81.6%), phlogosis (37; 73.4%), systemic effects (8; 16%) local infection (5; 10%), bleeding (2 cases, one gastrointestinal bleeding and the other haematuria; 4%) and thrombocytopenia (2; 4%). The reported systemic effects were rush, flashing, headache, mandibular pain, lower limb pain and pruritus (one patient per symptom).
Discussion
Prostanoids are used extensively in the treatment of PAH, and abundant clinical experience with them has been accrued over time (>20 years) in different pathological scenarios [idiopathic, hereditary, toxic PAH, collagen diseases, human immune deficiency virus (HIV) and congenital heart disease] and functional classes. 8 Several meta-analyses have been published about specific PAH treatment, 9 with the greatest body of evidence being based on trials with parenteral prostanoids. The outcomes showed that the treatment of PAH is related to a 43% reduction in the relative risk of combined events and mortality [control group = 3.8% versus treatment group = 1.5%; risk ratio (RR) = 0.57; 95% CI: 0.35 to 0.92; p = 0.023] after 14 weeks of follow-up. 10
Despite the extensive and proven benefits of prostanoids, there is a great deal of information that suggests that these drugs are under-used and relegated mainly to very advanced disease stages in which they are used basically as rescue strategies. This under-use of the parenteral prostanoids is rendered apparent by the REVEAL registry,11,12 which showed that only 43% of the patients who died from PAH had received treatment with them, a value which reaches 60% of the population if patients that progress to FC IV are included.
There are few worldwide reports on real clinical evidence with treprostinil, particularly when it is used at high doses. This lack of published data is particularly accentuated in Argentina, where there are hardly any reports on the use of and experience with prostacyclin analogues. For these reasons, this study with 12 months of follow-up heralds the first reference in the world and is particularly relevant because scTRE is the prostanoid with which there is greatest experience in clinical practice in the region, particularly in the high-risk PAH patients, probably because it presents higher thermal stability and adequate half-life, therefore permitting sustained effects over time and consequently fewer complications when it is abruptly discontinued than intravenous epoprostenol. 6 Moreover, the inclusion and analysis of the first 51 patients with PAH from different centres allows us to obtain data about real clinical practice with treprostinil in Argentina. In addition, the most represented subgroups in the registry are the idiopathic (62%) of the patients included in the study) and the connective tissue disease (22%) population, in line with other existing PAH registries in Latin America.13,14
Our study results are comparable to the findings of a randomised clinical trial conducted by Simonneau et al., 15 which included 470 patients with PAH (58% idiopathic, 19% collagen diseases and 23% congenital heart diseases) and 89% of them in advanced FC (III–IV) with baseline characteristics similar to those of our own data. In the 12-week follow-up period in the aforementioned clinical trial, significant benefits were observed in the distance travelled in the 6MWT (increment of 16 m, p = 0.003), in the combined clinical events rate for the haemodynamic parameters (MPP, PVR and CI) and also in quality of life–related aspects.
Our study suggests that pharmacological treatment with scTRE is associated with an improvement in several of the parameters analysed in real clinical practice. With regard to the clinical and functional parameters (FC and 6MWT, respectively), it should be emphasised that the magnitude of the benefits for these variables is greater than that which was found in the aforementioned study. 15 In the case of the 6MWT, a 130-m increment was observed in the distance travelled (p < 0.0001) versus the 16 m reported in the clinical trial, whereas the reduction in the proportion of patients with advanced FC was 65% (p < 0.0001) in our study compared with the 22% reduction found in the aforementioned trial. These differences could be due to the exposure time to treprostinil and the infusion dose, as well as the fact that 62% of the patients in our study were on a combined treatment regimen from the beginning.
Information about the evaluation of echocardiographic parameters in clinical studies is scant. These studies also underline the high prevalence of RV systolic dysfunction (82.9%) in baseline conditions. In our study, a non-significant improvement was observed in RVSF (82.9 versus 73.6%; p = 0.09) after the 12 months of follow-up, although a significant increase of 21% (+3.4 mm; p = 0.001) for the TAPSE value after 12 months was observed. Another one of the relevant findings is the significant 65% reduction in the pro-BNP value (−531 pg/dL; p < 0.0001), which is a marker of parietal stress and congestion of the cardiac chambers, thus highlighting the impact on subclinical heart failure. Moreover, our registry points to a significant improvement in the haemodynamic variables, such as the improvement in the CI, with an increase of 16.7% (+0.4 L/min/m2; p = 0.002) and a significant fall of 15.7% in PVR (−1.3 WU; p < 0.0001). A reduction in RAP was also observed, although it did not reach significance (−1.1 mmHg; p = 0.08). Despite this, the level of improvement obtained in our study is greater when compared with the results from other previous works with treprostinil, in which CI increased by 0.12 L/min/m2. However, it is comparable to the results obtained in other prostanoids meta-analysis (+0.32 L/min/m2).9,16
In addition, the aforementioned clinical trial 15 demonstrated that the clinical effects are concordant with the dose of treprostinil given, with the infusion of doses above 13.8 ng/kg/min showing greater benefits, although the average dose in the trial was 9.3 ng/kg/min. Although the maximum optimal dose varies depending on each patient, in most cases it ranges from 20 to 40 ng/kg/min, though there are reports of doses of up to 80 ng/kg/min. Our work showed a baseline dose of 3.95 ng/kg/min that reached, on average, 20 ng/kg/min after 12 weeks of follow-up and a value of 33 ng/kg/min after 12 months. This high dose could account for the positive impact and magnitude of the different risk variables analysed.
Previous to the development of specific PAH therapies, patients had a considerable low life expectancy as reported in the National Institute of Health (NIH) registry with a median survival of 2.8 years and survival rates of 68%, 48% and 34% at 1, 3 and 5 years, respectively. 2 After the development of specific PAH treatments, most contemporary registers reported 3-year survival rates of between 58.2% and 84% worldwide.13,17–23 In addition, long-term treatment survival has been reported in the REVEAL registry with survival rates of 85%, 68% and 49% at years 3, 5 and 7, respectively. 24 Similarly, the recently published Argentine RECOPILAR registry reported an 82.8% 3-year survival rate. 25
Long-term outcome of 860 PAH patients treated with scTRE was validated in an observational clinical trial, 26 followed by the addition of other PAH therapies, if needed, in which a sustained clinical improvement and a survival of 87% and 64% after 1 and 4 years, respectively, were observed. In this trial, 85% of patients were in FC III or IV at baseline, and 17% of the patients required active treatment with bosentan or sildenafil, underlining the need for combined therapy in a high percentage of patients. 26 Similarly, 111 patients with severe PAH in treatment with scTRE experienced overall survival rates of 84%, 53% and 33% at 1, 5 and 9 years, respectively. The latter increased up to 57% in patients who were able to tolerate treatment greater than 6 months, and bosentan or sildenafil were added only in five patients. 27
Our study showed a 1-year survival rate of 92% in a similar advanced PAH population, with a combined events rate of 43.1% after 12 months of follow-up, a higher survival rate compared with the previously mentioned studies. These differences in the clinical outcomes may be due to the predominance of the combined treatment received by 62% of the population (tadalafil and ambrisentan with elective regimen) in baseline conditions and even to the fact that one subgroup of patients entered with three base treatment drugs. It could also be due to differences in exposure time and the high dose of treprostinil during follow-up.
These efficacy data for treprostinil to improve survival in patients with PAH when combined with other treatments highlight the current preference among clinicians for adding prostanoids to the therapeutic strategy as an additive molecule in patients who do not fulfil the objectives during follow-up, with a greater casuistry in the group with a previous dual pharmacological regimen.
Importantly, in our study, the percentage of high-risk patients according to the ESC/ERS classification was reduced by more than half after 12 months of treatment with treprostinil, while 27.5% of patients achieved a low-risk status. This is in line with other previously reported data with combination therapy including parenteral prostanoids in advanced treatment naïve PAH patients,28–30 although the upfront strategy led to a more pronounced improvement of risk status and contrasts with those achieved with dual oral combination therapy in high-risk patients. 31 These data highlight the importance of initial combination therapy including parenteral prostanoids for high-risk patients, consistent with the treatment recommendations of current guidelines.4,32
The most salient frequent AEs associated with treprostinil reported in other studies include the presence of pain and/or local phlogosis at the infusion site, which affects 85% of patients and required discontinuation in 8% of the cases. 15 These findings are similar to those found in our study, with a rate of pain and/or phlogosis approaching 80%, indicating that the adverse effects are maintained irrespective of the dose of treprostinil used.
Despite the favourable results obtained; the study presents certain limitations that must be taken into account. These mainly include the fact that it is a retrospective study without a control group and in which the evaluation of the variables in the evolution may also be influenced by the specific concomitant treatments as well as the small sample size. Therefore, it would be recommendable to emphasise the need for large prospective studies to validate the data already obtained.
However, it is important to stress that this is one of the first studies to retrospectively analyse data from a national registry in Argentina regarding the use of scTRE and its long-term efficacy effects. Moreover, our study constitutes one of the few real-world international reports on real clinical evidence with treprostinil, particularly with its use at high doses, demonstrating its effectiveness in improving cardiac functional parameters and patient survival rates without causing secondary effects other than the ones already known.
Conclusion
The results of the data analysis of the TRES-HP registry demonstrate that the combination of scTRE with specific background therapy in PAH is associated with a significant improvement in different efficacy parameters after 12 months (FC, 6MWT, pro-BNP, PVR and CI values), a significant improvement in risk status according to ESC/ERS guidelines and a high survival rate (92%), with AEs similar to those reported in trials already published, albeit with an average dose of 33.8 ng/kg/min, higher than the dose studied to date.
These results open up the possibility to improve the current use of scTRE in clinical practice by using it as first-line therapy in combination with other drugs instead of as rescue treatment, especially in high-risk patient profiles, improving their survival rate and quality of life.
Supplemental Material
sj-doc-1-tar-10.1177_17534666221132735 – Supplemental material for Real-world evidence of subcutaneous treprostinil use in pulmonary arterial hypertension in Argentina
Supplemental material, sj-doc-1-tar-10.1177_17534666221132735 for Real-world evidence of subcutaneous treprostinil use in pulmonary arterial hypertension in Argentina by Adrián Lescano, Guillermo Giacommi, Cristian E. Botta, Julieta Soricetti, Manuel Rodriguez, Paul Vargas Mielles and Fabián Diez in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We would like to thank Ianina Fernandez and Federico Stroschein for their constant collaboration and general support during the study. We would like to thank Nerea Toro, Maite Artés (Adelphi Targis S.L.) and Gabriela Bacchini (Ferrer Internacional) for their support in the medical writing process. We also wish to thank the Ferrer Internacional for its scientific advice and general support in the course of the project.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
