Abstract
Background:
During the novel coronavirus disease 2019 (COVID-19) pandemic raging around the world, the effectiveness of respiratory support treatment has dominated people’s field of vision. This study aimed to compare the effectiveness and value of high-flow nasal cannula (HFNC) with noninvasive ventilation (NIV) for COVID-19 patients.
Methods:
A comprehensive systematic review
Results:
In total, nine studies [one randomized controlled trial (RCT), seven retrospective studies, and one prospective study] totaling 1582 patients were enrolled in the meta-analysis. The results showed that the incidence of IMV, number of deaths (no time-limited), and length of ICU stay were not statistically significant in the HFNC group compared with the NIV group (
Conclusion:
For COVID-19 patients, the use of HFNC therapy is associated with the reduction of the number of deaths at day 28 and length of hospital stay, and can significantly improve oxygenation index (PaO2/FiO2) at 24 h. However, there was no favorable between the HFNC and NIV groups in the occurrence of IMV. NIV group was superior only in terms of ventilator-free days.
Introduction
The outbreak of novel coronavirus disease 2019 (COVID-19) has caused untold harm and challenges to people in more than 200 countries and territories around the world. To this day, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) continues to ravage the planet, with more than 270 million people infected with COVID-19 worldwide. 1 Pneumonia caused by SARS-CoV-2 differs from community-acquired pneumonia (CAP) and hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia in that it is highly transmissible from person to person and can even infect people asymptomatically.2,3 Patients with severe COVID-19 can trigger acute respiratory distress syndrome (ARDS),4,5 which can progress to acute respiratory failure (ARF), manifested by severe hypoxemia and dyspnea. What’s more, hypoxemia is associated with more rapid disease progression and higher mortality. Currently, the choice of ventilation support treatment for patients with COVID-19 is particularly important.
Traditionally, when encountering severe respiratory diseases, medical staff would first think of invasive mechanical ventilation (IMV), because years of experience have shown that it is a potential intervention to save the lives of acute patients. However, years of clinical experience have taught us that IMV is a risk factor for the development of ventilator-associated pneumonia (VAP).
6
Long-term IMV is a major risk factor for patients with hospital-acquired infections, which may worsen the patient’s condition and is associated with a high mortality rate in hospital.
7
Noninvasive ventilation (NIV) can reduce intubation rate in COVID-19 patients with ARF, shorten length of stay, and reduce in-hospital mortality. NIV is the delivery of a specific flow of fresh gas to the lungs without an invasive endotracheal airway. It can both give a certain flow of oxygen alone
The fact that COVID-19 will continue to exist and expand its impact has been recognized by experts around the world. According to our data search, the amount of meta-analysis that compares the ventilation strategies of COVID-19 patients is very limited. Therefore, it is essential to compare the various ventilation treatment strategies suitable for patients with COVID-19. On this premise, we conducted this systematic review and meta-analysis, with the purpose of comparing HFNC with NIV and exploring which one can better reduce the occurrence of IMV and the death at 28 days of COVID-19 patients.
Methods
Search strategy
This systematic review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We followed the PRISMA checklist to complete the current meta-analysis. To identify studies comparing the efficacy of HFNC with conventional oxygen therapy in COVID-19 patients, two investigators (Y.W.H. and X.W.) systematically searched PubMed, Web of Science, Cochrane, Scopus, WHO database, China Biology Medicine Disc (SINOMED), and China National Knowledge Infrastructure (CNKI) databases for relevant articles published before 20 October 2021. The whole retrieval process is shown in Figure 1. The search terms are as follows: ((High-flow Nasal Cannula OR HFNC OR High flow nasal cannula therapy OR nasal high flow OR high flow nasal therapy OR high flow oxygen therapy OR high flow therapy OR HFNO OR high flow nasal oxygen [Title/Abstract])) AND (2019-nCoV OR nCoV-2019 OR novel Coronavirus 2019 OR SARS-CoV-2 OR COVID-19 OR coronavirus OR coronavirus covid-19 OR nCoV OR corona virus [Title/Abstract]). For different databases, we will use different search formulas to avoid omissions.

PRISMA flow diagram of search strategy and included studies.
Study selection
The retrieved literature was managed using EndNote X9 (Thomson Reuters, NY, USA). After excluding duplicates and nonclinical studies, we (Y.W.H., N.L., and X.H.Z.) screened the literature for titles, abstracts, and keywords, respectively. In this process, we rated the studies by using the starring feature of EndNote X9. Two investigators marked the studies with low relevance as ‘one star’, the controversial research that required two investigators to re-screen them marked as ‘two or three stars’, and the undoubted study is ‘four or five stars’. The studies with one star can be excluded at once by a single investigator, two or three stars’ studies need extra evaluation by all the investigators, and the research with four or five stars can be included in this meta-analysis. We used the modified methodological index for nonrandomized studies (MINORS) score12,13 to evaluate the quality of nonrandomized controlled trials (NRCTs) and excluded studies with a total score of ⩽12. Finally, two researchers identified the included literature based on the full text. When the results of the two researchers diverged, the opinion of a researcher (W.H.M.) was used to reach a consensus. Figure 1 includes a screening process to illustrate the number of studies that we exclude at each stage.
Eligibility criteria
For the inclusion of this systematic review and meta-analysis, studies had to meet the following criteria: randomized controlled trials (RCTs), observational cohort studies, or retrospective studies that compare the efficacy of HFNC with conventional oxygenation therapy in the patients with COVID-19. There was no restriction in terms of the type of conventional oxygenation therapy. Excluded studies had the following characteristics: case reports or case series, guidelines, expert consensus, animal experiments, protocol, reviews, meta-analysis, conference abstract, letters, comments, experiences, survey, and clinical trials with different observational indicators.
Risk of bias assessment
Three investigators (Y.W.H., N.L., and X.H.Z.) used the Cochrane collaboration tool to assess the risk of bias of studies. This information was recorded and evaluated in RevMan 5.4 (Review Manager, Versio 5.4, The Cochrane Collaboration, 2014). The risk of bias summary is shown in Figure 2. When researchers disagree on the biased analysis of the same study, another researcher (W.H.M.) will make the decision.

Risk of bias graph: review authors’ judgments about each risk of bias item presented as percentages across all included studies.
Data extraction
Three investigators independently extracted the relevant information and data. All differences are double-checked, and W.H.M. was responsible for handling different points of view. According to the modified MINORS score, we will analyze the data included in the NRCT and complete the quality assessment (Table 1). We extracted the following data based on the characteristics of the included studies – groups, nationals, type of hospital, ages, gender, body mass index (BMI), and comorbidities – and summarized in Table 2. And also, we extracted the intervention characteristics of studies in Table 3.
Modified MINORS score of all eligible NRCT.

MINORS, methodological index for nonrandomized studies; NRCT, nonrandomized controlled trial.
Only studies with scores ⩾12 can be included in the meta-analysis.
Evaluated patients’ characteristics (
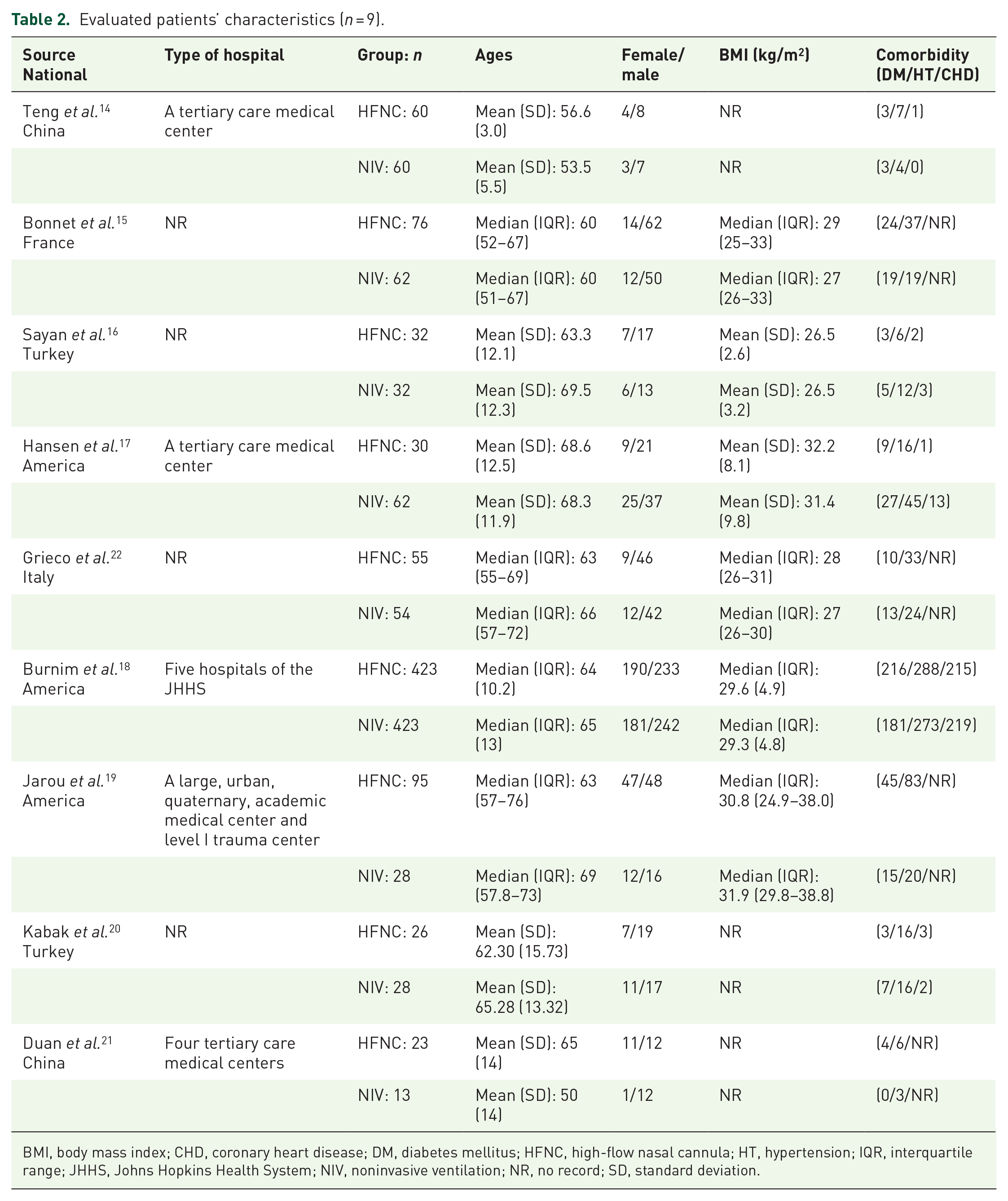
BMI, body mass index; CHD, coronary heart disease; DM, diabetes mellitus; HFNC, high-flow nasal cannula; HT, hypertension; IQR, interquartile range; JHHS, Johns Hopkins Health System; NIV, noninvasive ventilation; NR, no record; SD, standard deviation.
Evaluated intervention characteristics (

CPAP, continuous positive airway pressure; FiO2, fraction of inhaled oxygen; HFNC, high-flow nasal cannula; NIV, noninvasive ventilation; NR, no record; PEEP, positive end-expiratory pressure; P/F, partial oxygen pressure/fraction of oxygen saturation; RR, respiratory rate; SpO2, peripheral arterial oxygen saturation.
Outcomes
The primary outcome is the number of deaths at day 28. The secondary outcomes are the occurrence of IMV, the number of deaths (no time-limited), length of ICU and hospital stay, ventilator-free days, and oxygenation index [partial pressure of arterial oxygen (PaO2)/fraction of inhaled oxygen (FiO2)] at 24 h.
Statistical analysis
RevMan 5.4 computer software was used for all data analysis in this study. For all the dichotomous data that need to be analyzed, we chose to report odds ratio (OR). And for continuous variables, the mean and standard deviation (SD) should be reported. If the median and interquartile range (IQR) are reported in the study, it can be converted into the mean and SD through formulas.
RevMan 5.4 also reported the heterogeneity of the data while producing the forest plot. For heterogeneity test
Results
The protocol for this review has been published in Prospero, and the registration number is CRD42021289413.
Literature search findings
We searched six databases with a total of 2778 studies (PubMed: 772, Embase: 693, Web of Science: 898, Scopus: 155, Cochrane: 238, WHO: 22). Two researchers used EndNote X9 to remove 499 duplicate studies and 5 editorial chapters. Since COVID-19 cases have been gradually reported from December 2019, our search time is defined as December 2019 until now, and 4 studies beyond the limitation have been removed. Three researchers (Y.W.H., N.L., and X.H.Z.) reviewed the titles and abstracts, and checked the full text of 241 studies. Finally, nine studies were included in the systematic review and meta-analysis. The search and screening process is shown in Figure 1.
Risk of bias assessment and study quality
Since only one RCT can be retrieved, the inclusion criteria of this study are not limited to prospective studies. We use the MINORS to evaluate the quality of NRCT based on the researches of Vinuela
Study and patient characteristics
In nine studies, 1582 patients were included in the meta-analysis. The investigators extracted the characteristics of the patients in the included studies, containing groups, nationals, type of hospital, ages, gender, BMI, and comorbidity (diabetes mellitus, hypertension, and coronary heart disease), in Table 2. In the study of Jarou
Intervention characteristics
The researchers extracted intervention characteristics of included studies; the main content are the oxygenation strategy and concomitant medications of the NIV and HFNC groups (Table 3). Only the study of Burnim
Meta-analysis and synthesis
Number of deaths at day 28
Five studies with 1227 patients (NIV group: 619; HFNC group: 608) reported the number of deaths at day 28. The pooled mean difference (MD) (95% confidence interval, CI) was 1.62 (1.24, 2.14) units,

(a) The number of deaths at day 28 and (b) the number of deaths (no time-limited).
Number of deaths (no time-limited)
Three studies with 213 patients (NIV group: 69; HFNC group: 144) reported the number of deaths. The pooled MD (95% CI) was 1.41 (0.72, 2.74) units,
Occurrence of IMV
Five studies with 380 patients (NIV group: 176; HFNC group: 204) reported the occurrence of IMV. The pooled MD (95% CI) was 1.21 (0.45, 3.29) units,

The occurrence of IMV.
Length of ICU stay
Seven studies with 580 patients (NIV group: 262; HFNC group: 318) reported the length of ICU stay. The pooled MD (95% CI) was –1.06 (–3.19, 1.07) units,

(a) The length of ICU stay and (b) the length of hospital stay.
Length of hospital stay
Four studies with 308 patients (NIV group: 120; HFNC group: 188) reported the length of hospital stay. The pooled MD (95% CI) was 1.62 (0.05, 3.19) units,
Ventilator-free days
Four studies with 381 patients (NIV group: 196; HFNC group: 185) reported the ventilator-free period (days). The pooled MD (95% CI) was –2.26 (–3.18, –1.35) units,

The ventilator-free days.
Oxygenation index (PaO2/FiO2) at 24 h
Three studies with 101 patients (NIV group: 42; HFNC group: 59) reported the oxygenation index (PaO2/FiO2) at 24 h. The pooled MD (95% CI) was –34.76 (–41.00, –28.51) units,

The oxygenation index (PaO2/FiO2) at 24 h.
Discussion
Three years have passed since the outbreak of COVID-19, which has shown the world how frightening and aggressive it can be. Early epidemiological studies5,25,26 stated that 8.2% of cases suffer from rapid and progressive respiratory failure, similar to ARDS. In most severe cases, patients with COVID-19 require high probability of needing IMV, which is associated with high mortality. 4 Therefore, patients often need different degrees of noninvasive respiratory support treatment to maintain the arterial partial pressure of oxygen at a normal level. Our systematic review and meta-analysis yielded one RCT and eight high-quality NRCTs to evaluate the efficacy of different NIV strategies in the patients with COVID-19. By comparing the HFNC group with the NIV group, we found that the HFNC group was significantly better than NIV concerning the number of deaths at day 28, length of hospital stay, and oxygenation index at 24 h. The comparison between the HFNC and NIV groups was not statistically significant in terms of the incidence of IMV, number of deaths (no time-limited), and length of ICU stay. NIV group was superior only for the outcome of ventilator-free days.
How to improve patients’ survival rate, reduce the occurrence of IMV, and improve the prognosis of patients are the questions that always have been discussed since the outbreak of the COVID-19. Severe COVID-19 patients usually undergo ARF. Therefore, the use of various NIMV methods to maintain the patient’s FiO2 at a high level has become the global consensus.27,28 NIV is an oxygen therapy modality that emerged in the 1990s of the last world.
29
Esteban
What bothers us most is the high mortality of COVID-19 patients. Both the NIV and HFNC have been demonstrated to significantly reduce mortality and enhance the prognosis of patients. Thus, we used the number of deaths at day 28 as the primary outcome of our meta-analysis. According to our analysis of five included studies, we can easily find a significant reduction in the number of deaths at day 28 in COVID-19 patients using HFNC as ventilation treatment [OR = 1.62, 95% CI = (1.24, 2.14),
Investigating the effect of different NIV treatments on reducing the occurrence of IMV is also a necessary outcome of importance for patients with COVID-19. We included five studies to analyze which of HFNC and NIV was more meaningful to reduce the occurrence of IMV. Unfortunately, the results we made comparing HFNC with NIV were not statistically significant [OR = 1.21, 95% CI = (0.45, 3.29),
Our study found that HFNC also has obvious advantages over NIV in terms of reducing the length of hospital stay [mean difference = 1.62, 95% CI = (0.05, 3.19),
Interestingly, the NIV group has a significant advantage over the HFNC only in the aspect of ventilator-free days [mean difference = –2.26, 95% CI = (–3.18, –1.35),
To our knowledge, this is the first systematic review and meta-analysis to explore efficacy and value of these two types of oxygen strategies for patients with COVID-19. This study was created through an extensive search, iterative screening, and data extraction. We provide a comprehensive overview of the comparison between HFNC and NIV, with the aim of evaluating its true efficacy and benefit to patients by including thorough various assessment criteria. Meanwhile, we have taken into account the potential limitations of this meta-analysis. First, despite an extensive literature search, the limited number of relevant RCTs led to the inclusion of only one RCT and eight high-quality NRCTs. Although they passed the quality assessment, this may affect the accuracy of the results. Furthermore, COVID-19 patients in the eight included studies had different reasons for receiving ventilation therapy. Some of the studies included COVID-19 patients with ARF, and some have ARDS. Patients with different medical backgrounds receiving the same ventilation treatment may cause bias in the study outcomes. Finally, the timing of treatment as well as the pattern of treatment also vary between HFNC and NIV, which further increases heterogeneity.
Conclusion
For COVID-19 patients, the use of HFNC therapy is associated with the reduction of the number of deaths at day 28 and length of hospital stay, and can significantly improve oxygenation index (PaO2/FiO2) at 24 h. However, there was no favorable option in the comparison between the HFNC and NIV groups in the occurrence of IMV. NIV group was superior only for the outcome of ventilator-free days. Large samples and high-quality clinical studies are still needed to evaluate different ventilation strategies for patients with COVID-19.
Supplemental Material
sj-docx-1-tar-10.1177_17534666221087847 – Supplemental material for High-flow nasal cannula versus noninvasive ventilation in patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tar-10.1177_17534666221087847 for High-flow nasal cannula versus noninvasive ventilation in patients with COVID-19: a systematic review and meta-analysis by Yuewen He, Na Liu, Xuhui Zhuang, Xia Wang and Wuhua Ma in Therapeutic Advances in Respiratory Disease
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
