Abstract
Approximately 1–2 per 1000 pregnancies are complicated by venous thromboembolism (VTE). VTE includes deep vein thrombosis (DVT) and pulmonary embolism (PE) and the diagnostic management of pregnancy-related VTE is challenging. Current guidelines vary greatly in their approach to diagnosing PE in pregnancy as they base their recommendations on scarce and weak evidence. The pregnancy-adapted YEARS diagnostic algorithm is well tolerated and is the most efficient diagnostic algorithm for pregnant women with suspected PE, with 39% of women not requiring computed tomographic pulmonary angiography. Low-molecular-weight heparin is the first-choice anticoagulant treatment in pregnancy and should be continued until 6 weeks postpartum and for a minimum of 3 months. Direct oral anticoagulants should be avoided in women who want to breastfeed. Management of delivery needs a multidisciplinary approach in order to decide on an optimal delivery plan. Neuraxial analgesia can be given in most patients, provided time windows since last low-molecular-weight heparin dose are respected. Women with a history of VTE are at risk of recurrence during pregnancy and in the postpartum period. Therefore, in most women with a history of VTE, thromboprophylaxis in subsequent pregnancies is indicated.
The reviews of this paper are available via the supplemental material section.
Keywords
Introduction
Venous thromboembolism (VTE) includes deep vein thrombosis (DVT) and pulmonary embolism (PE). Pregnancy-related VTE remains one of the main causes of maternal death during pregnancy in developed countries in spite of the use of thromboprophylaxis in women at increased risk. 1 Pregnant women have a 4–5-times higher risk of developing VTE compared with non-pregnant women of the same age2,3 with an estimated incidence of VTE of approximately 1–2 per 1000 pregnancies.1,2,4 A hypercoagulable state, vascular damage and venous stasis all contribute to the increased risk of VTE.5,6 Roughly half of pregnancy-related VTE episodes occur during pregnancy, with a more or less similar distribution among the trimesters, and the other half during the 6-week postpartum period. As a result, the daily absolute risk is highest during the postpartum period, as the antepartum period lasts much longer than the postpartum period.7,8
The diagnostic management of DVT and PE in pregnancy is challenging due to the fact that pregnant women often have clinical symptoms of the legs or symptoms that could point to PE, such as shortness of breath or tachycardia, which may well be consequences of physiological changes occurring during normal pregnancy. 9 Because missing a diagnosis of DVT or PE may have severe or even fatal consequences, in clinical practice the index of suspicion for VTE is low. This is illustrated by the low VTE prevalence of approximately 5% in most studies focusing on pregnant patients with suspected PE, compared with 15–25% in the non-pregnant population.10,11 Imaging tests to confirm PE diagnosis are associated with radiation and may be harmful to both mother and fetus, whereas clinical decision rules and D-dimer testing had, until recently, not been proven useful in the diagnosis of suspected PE in pregnancy. Two contemporary studies have investigated a diagnostic algorithm in women with suspected PE during pregnancy.12,13 Overall, current guidelines vary greatly in their approach to diagnosing pulmonary embolism in pregnancy as they base their recommendations on scarce and weak evidence, with the most recent European Society of Cardiology (ESC) guideline based on the two aforementioned recent studies.7,14–16
In this review we provide an overview of guidelines and contemporary best practices of management of PE in pregnancy.
Case
A 35-year-old woman presented at the emergency department with mild dyspnea and thoracic pain on the left side that worsened with deep inhalation for 1 day, without cough, hemoptysis, or leg symptoms. She was 22 weeks pregnant, and had recently returned from holiday after an 11-h flight. She had a positive family history for VTE; her mother experienced a DVT in the postpartum period.
On physical examination she appeared short of breath and in pain, with a respiratory rate of 20 excursions per minute, blood pressure of 115/70 mmHg, regular pulse rate of 95 beats per minute, temperature of 38.1° C, and transcutaneous oxygen saturation of 95%. She had a normal body mass index of 23.9 with a weight of 80 kg and height of 183 cm. There were no abnormal findings at auscultation of her lungs and heart. Her legs showed no swelling, redness or edema. Laboratory examination revealed a C-reactive protein level of 50 mg/l, normal hemoglobin level of 12.89 g/dl, elevated D-dimer of 2.1 µg/ml, and mild leukocytosis of 12.0 × 109 mg/l. Compression ultrasonography of both legs including visualization of the iliac veins showed no abnormalities. Because of suspicion of PE, a computed tomography pulmonary angiography (CTPA) scan was performed and showed multiple bilateral pulmonary embolism with a normal right ventricular/left ventricular ratio.
Current guidelines for management of acute VTE in pregnancy
Guidelines on VTE are widely divergent and differences in recommendations are listed in Table 1. Until recently, there was no consensus about the role of D-dimer and clinical decision rules in diagnostic management or PE in pregnancy because of the lack of strong evidence for validated diagnostic algorithms. Thus evidence is of low grade quality and recommendations are weak. The American Society of Hematology (ASH) 2018 VTE in pregnancy states that the role of D-dimer testing and clinical prediction rules is limiting and the need for radiologic test in pregnant women with suspected PE and suspected DVT need to be investigated in well-designed management studies. 7 The guidelines of the American Thoracic Society/Society of Thoracic Radiology (ATS/STR) 2011 and Royal College of Obstetricians and Gynaecologists (RCOG) 2015 suggest not to use D-dimer to exclude PE in pregnancy,15,17,18 whereas the ESC 2014 guideline states that D-dimer measurement may be used to rule out PE in pregnancy. 19 Only in the ESC 2019 update have the two prospective management studies been taken into consideration. 16 This guideline states that D-dimer measurement and clinical prediction rules should be considered to diagnose or rule out PE during pregnancy or the postpartum period. 16
Recommendations guidelines.

ASH, American Society of Hematology; CT, computed tomography; CTPA, computed tomography pulmonary angiography; CXR, chest X-ray; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; INR, international normalized ratio; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; UFH, unfractionated heparin; VKA, vitamin K antagonist; V/Q, ventilation-perfusion; VTE, venous thromboembolism.
Diagnostic management of VTE in pregnant patients
Pregnant women often have non-specific symptoms, such as shortness of breath or tachycardia at exertion. Given the fact that VTE is a major cause of maternal morbidity and mortality and the consequences of missing the diagnosis may be devastating, the clinical threshold to test pregnant women with the suspicion of acute PE should be low. Clinical decision rules to determine pretest probability of suspected PE in non-pregnant women, such as the Wells score and revised Geneva scores are widely used clinical tools that have been extensively validated in the non-pregnant population.20,21 A low or unlikely pretest probability combined with a low D-dimer level can safely exclude the diagnosis of PE without imaging in the non-pregnant population, and adjustments of D-dimer cut-off to increasing age (age-adjust) or with varying D-dimer cut-offs depending on presence of certain clinical criteria (YEARS) have been made to make this strategy very efficient.11,22–24
Until recently, imaging was the cornerstone for diagnosis of PE in pregnant women. 25 The most widely used imaging techniques to rule out or diagnose PE in the non-pregnant population is CTPA. However, whether CTPA should be the first choice in pregnant women is controversial and debated, and guidelines vary in their recommendations (Table 1). Although CTPA results in the lowest radiation exposure to the fetus, for the pregnant woman it may result in higher radiation exposure to proliferating breast tissue than ventilation-perfusion (V/Q) scanning. Interestingly, a retrospective cohort study evaluated the risk of early-onset breast cancer after CTPA or V/Q scanning in pregnancy and in the postpartum period compared with non-exposed controls. 26 The primary outcome of the study was a first diagnosis of breast cancer more than 366 days after the delivery date. The median duration of follow up was 5.9 for CTPA, 7.3 for V/Q scanning and 11.1 years in the unexposed control group. The risk of breast cancer was 7.0–7.1 per 10,000 women and did not differ between those exposed to CTPA, V/Q scanning and unexposed women. However, the duration of follow up was too short to determine potential risk increases on the long-term. 26 A recent systemic review found both CTPA and V/Q scanning to be appropriate for ruling out PE during pregnancy. 27 Both scanning techniques were below the safety threshold of 100 mGy, associated with fetal radiation complications. The decision whether to use CTPA or V/Q can hence be based on user convenience and local availability, as acute V/Q scanning is not widely available. 27
The prevalence of inconclusive test results for both imaging techniques is relatively high due to the physiological changes during pregnancy and therefore specifically designed protocols for pregnant women are required. In the aforementioned systematic review 30 studies were included, in which 2535 pregnant women underwent V/Q scanning and 1774 patients were investigated with CTPA. The pooled rate of an inconclusive result with V/Q scanning was 14% [95% confidence interval (CI): 10–18%, I 2 90.30%] and 12% with CTPA (95% CI: 6–17%, I 2 93.86%). 27
Some guidelines suggest performing CTPA over V/Q scanning in case there is an abnormal chest X-ray (CXR).15,16 In a post hoc analysis of the ARTEMIS study the diagnostic value of CXR for the diagnosis of PE in pregnant women was investigated. 28 An abnormal CXR was found more frequently in patients with confirmed PE than in patients in whom PE was ruled out. However, performing of routine use of CXR in all patients with suspected PE prior to CTPA is not recommended. 28
Until recently, studies that investigated the potential of clinical decision rules or D-dimer testing to exclude PE in the pregnant population were being retrospective in nature and of limited quality. 29 D-dimer levels increase over the course of pregnancy and are often above the widely used D-dimer threshold of 0.5 µg/ml. 10 A recent multicenter cohort study for in pregnant women with suspected PE, augmented with additional PE cases (DiPEP) found little diagnostic value of clinical decision rules and D-dimer and the authors concluded that these should not be used in pregnancy and the postpartum period. 30
However, in the past year two large, revolutionary prospective multicenter management studies on optimizing the diagnostic management of pregnant women who are suspected of PE were published.12,13 The first was a large multicenter management outcome study and included 395 women from 11 centers in France and Switzerland. PE was considered ruled out in women with a low or intermediate pretest probability based on the revised Geneva score and D-dimer <0.5 µg/ml. All women who had a higher pretest probability or D-dimer of 0.5 µg/ml or higher underwent bilateral compression ultrasonography (CUS), including visualization of the iliac veins. When proximal DVT was diagnosed, there was no indication for further pulmonary examination. When CUS was negative, women were referred for a CTPA or V/Q scan if the CTPA was non-conclusive. PE was confirmed in 28 (7.1%) of 395 women; 7 women had a positive CUS and in 21 women, PE was diagnosed by CTPA or V/Q scan. The primary endpoint was symptomatic VTE after 3 months of follow up in patients in whom the diagnosis was ruled out at presentation and who were left untreated. There were no events and the rate of symptomatic VTE was 0.0% (95% CI, 0–1.0%) after 3 months. In 46 (11.6%) of 395 women who had a low or intermediate clinical probability and a D-dimer <0.5 µg/ml, PE could be ruled out and CTPA could be avoided.
The second study (ARTEMIS) was based on the YEARS algorithm for non-pregnant women 23 and assessed the YEARS algorithm in pregnant women with a suspicion of PE. 13 This prospective management study included 498 pregnant women with a suspicion of PE in 18 participating hospitals in The Netherlands, France and Ireland. In all women the YEARS criteria (clinical sign of DVT, hemoptysis, and PE as the most likely diagnosis) were assessed and D-dimer testing was performed. PE could be ruled out without imaging if none of the three YEARS criteria was present and the D-dimer was <1.0 µg/ml, or if one or more YEARS criteria were present and D-dimer was less <0.5 µg/ml. The full algorithm is displayed in Figure 1. In patients in whom one of the YEARS criteria was ‘signs of DVT’, CUS, including visualization of the iliac vein, was performed and if a DVT was diagnosed, no CTPA was performed as the diagnosis of VTE was already confirmed. In all other cases CTPA was performed. At time of presentation, PE was diagnosed in 20 patients (4.0%). The primary endpoint was the cumulative incidence of confirmed symptomatic VTE during a 3-month follow-up period in patients in whom PE was ruled out and anticoagulant treatment was withheld based on the pregnancy-adapted YEARS algorithm. During follow up, one patient was diagnosed with a proximal DVT 12 weeks after the initial presentation. Hence, the failure rate was 0.21% (95% CI 0.04–1.2). The efficiency of the strategy was high, as CTPA was avoided in 195 (39%) patients. The efficiency varied per trimester: CTPA could be avoided in 65% of women who were in the first trimester of pregnancy, in 46% in the second trimester and in 32% in the third trimester.
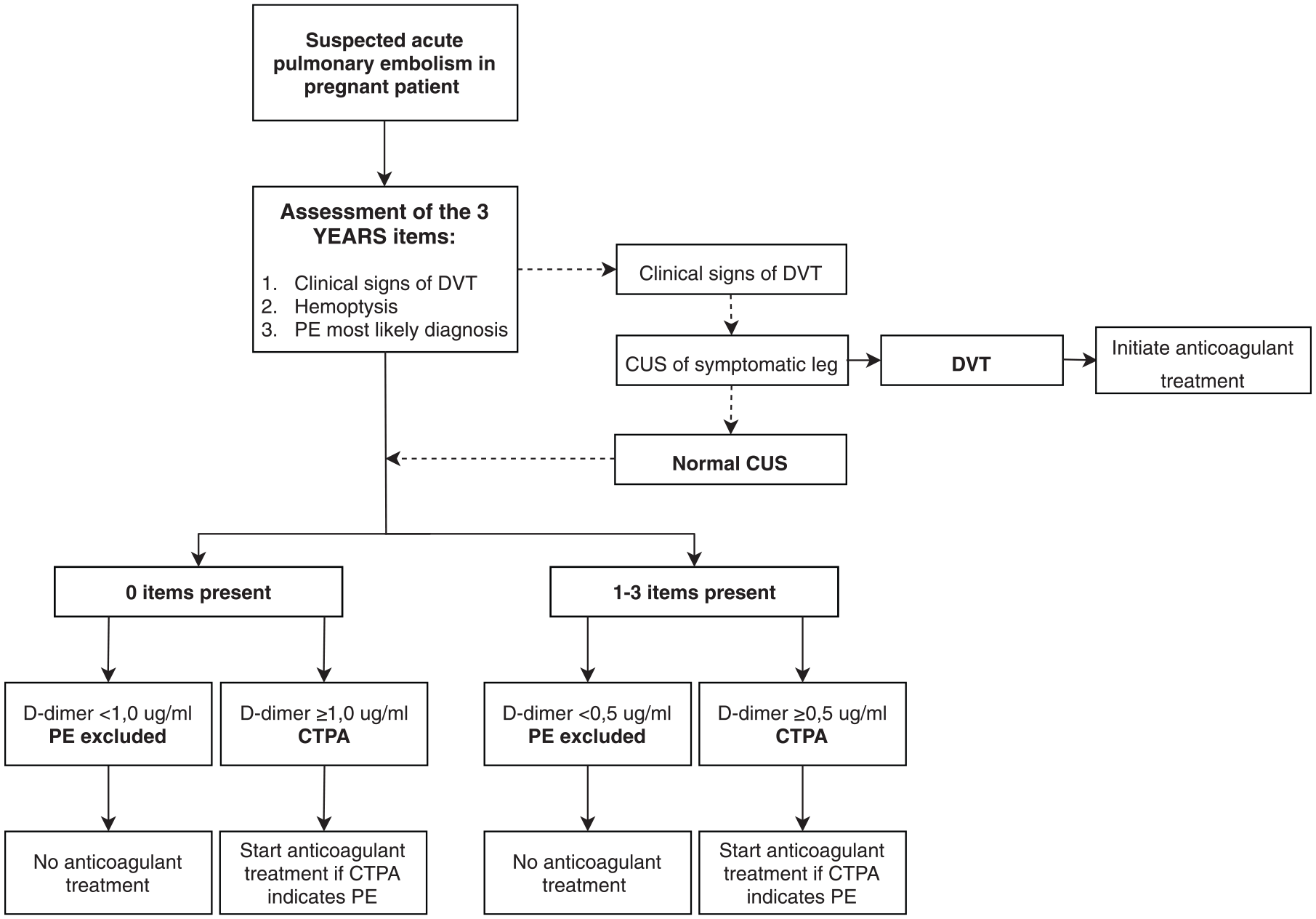
Pregnancy-adapted YEARS algorithm for diagnosis of PE in pregnant women
A post hoc analysis of the previously mentioned Swiss–French prospective management study 31 assessed the accuracy and safety of the pregnancy-adapted YEARS algorithm in women with suspected PE. 12 Also in this analysis, the algorithm proved to be well tolerated with no VTE occurring during follow up (0%, 95% CI 0–3.9). CTPA would have been avoided in 77 of 371 (21%) of women, which is lower than the 39% in the original study but still substantial.
The observed failure rates of these two large prospective management studies are in line with the proposed criteria for confirming the safety of PE diagnostic management studies by the International Society on Thrombosis and Haemostasis, 32 in which the recommended safety threshold varies depending on PE prevalence. Assuming a prevalence of 5%, the proposed failure rates should not exceed 0.70 with an upper limit of the 95% CI of 1.85.
We conclude that the pregnancy-adapted YEARS diagnostic algorithm is well tolerated and the most efficient diagnostic algorithm for pregnancy women with suspected PE.
Case continued
Our patient was treated with a therapeutic dose of dalteparin once daily based on body weight at the time of diagnosis. At 38 + 3 weeks of gestational age she delivered a healthy son 25 h after the last injection of low-molecular-weight heparin (LMWH). The estimated amount of blood loss was 300 ml. LMWH at full dose was resumed 12 h after delivery after assessment of normal vaginal blood loss.
Treatment of acute pulmonary embolism in pregnancy
Heparins, including LMWH and unfractionated heparin (UFH), can be safely used in pregnant women (Table 2). 7 Heparins do not pass the placenta, nor are they associated with teratogen effects on the fetus. LMWH is the first-choice anticoagulant treatment in pregnancy and is preferred over UFH due to its superior tolerability and convenient profile since frequent monitoring of activated partial thromboplastin time (aPTT) is not required and the risk of heparin-induced thrombocytopenia (HIT) is lower.7,33,34
Choice of anticoagulants during pregnancy and breastfeeding.

Outside the scope of this review, please see Scheres et al. 35
DOAC, direct oral anticoagulant; LMWH, low-molecular-weight heparin; UFH, unfractionated heparin; VKA, vitamin K antagonist.
Treatment regimens of LMWH for pregnant women are based on the available evidence in the non-pregnant population. Hence, both a once daily dose regimen and a twice daily regimen of therapeutic-dose LMWH is suggested. 7 We generally choose a once daily regimen as this requires fewer subcutaneous injections. It is unknown whether the pre-pregnancy weight should be used or the actual body weight to establish the appropriate dose of LMWH; in our institution we use actual body weight. Guidelines suggest not to monitor anti-factor Xa levels to guide dosing of LMWH as there is no evidence that anti-factor Xa level monitoring and subsequent dose adjustments leads to improved patient outcomes. 7 Although we agree that there is no evidence, for treatment of acute VTE we do monitor anti-factor Xa levels 4 h after injection with target levels of anti-factor Xa between 0.8 and 1.6 with a once daily regimen at about bimonthly intervals, and we measure platelets alongside. We instruct women to inject in the morning so that they reach a 4-h peak level of anti-factor Xa for blood sampling during office hours. Monitoring of anti-factor Xa levels in obese patients or with renal dysfunction in the pregnant population were not discussed in the guidelines. 7 We cap the dose to the maximum allowed dose per day.
LMWH leads to side effects such as bruising, local skin reactions and, as with any anticoagulant, increases the risk of bleeding. Skin reactions and bruising are common side effects of LMWH and are frequently observed in pregnant patients. However, they seem to be underreported in studies, with rates varying widely between studies from 1.8% to 42%. 35 Heparin-induced skin lesions are the consequences of delayed type IV hypersensitive reactions and a switch to another type of LMWH should be considered if skin reactions are experienced as burdensome.36,37 In the majority of women, skin reactions will disappear after a switch to another type of LMWH. A switch to a non-heparin anticoagulant such as danaparoid sodium or Fondaparinux can be considered if all types of LMWH have not led to the desired effect. Danaparoid does not cross the placenta and is the first choice in pregnant women in whom HIT occurs. 38 HIT is a very rare complication and the incidence is very low in pregnant patients who use LMWH (<0.1%). 39 Only a few cases of HIT have been described.40,41 Heparin-induced osteoporosis has been thought to have a risk as high as 2% with long-term use of UFH, 42 but in a systematic review of 2777 pregnant women, only 1 case (0.04%) of an osteoporotic fracture was identified 39 and in a case series only 9 cases of heparin-induced osteoporosis were reported. 43 Data about the use of Fondaparinux in pregnancy are limited but it has been observed that Fondaparinux crosses the placenta in small amounts. 44 Therefore the use of Fondaparinux during the first trimester is not recommended.44,45 The risk of bleeding antepartum is low but will increase at the time of delivery and with obstetrical procedures. 35
Other types of anticoagulants are contraindicated in pregnancy. Vitamin K antagonists (VKAs; warfarin, acenocoumarol, phenprocoumon) cross the placenta and use during pregnancy is associated with congenital abnormalities. In particular during the 6th to 12th weeks of pregnancy there is an increased risk of abnormalities because the fetus is vulnerable to vitamin K deficiency. 46 VKAs inhibit the formation of vitamin K-dependent proteins needed in cartilage and bone formation. This leads to warfarin embryopathy, which has been reported in 5–6 % in case series of pregnant women using VKAs for the indication of mechanical heart valves. 47 The use of VKAs also increases the risk of fetal loss in the first trimester. During delivery there is an increased risk of fetal cerebral hemorrhage. 48
In recent years, direct oral anticoagulants (DOACs) have replaced VKAs as first choice for the treatment of VTE. 7 There is currently little evidence about the safety of DOACs during pregnancy.49,50 Animal models have shown that DOACs pass the placenta and were also found in breastmilk.51,52 Several small-scale studies have looked into the effect of DOACs in pregnancy in women wanting to become pregnant. In a review of 223 women who were exposed to DOACs (apixaban, dabigatran, edoxaban and rivaroxaban) in pregnancy, congenital abnormalities were observed in 7 of the 137 live-born children, with 3 considered as embryopathy. 53 The International Society on Thrombosis and Hemostasis (ISTH) recommends against the use of DOACs during pregnancy and also advises on the use of anticoagulants other than DOACs in women trying to become pregnant. 54
Systemic thrombolysis must be considered in pregnant patients with life-threatening hemodynamic instability due to massive PE. A recent literature review summarized 23 case reports on the use of systematic thrombolysis as treatment for massive PE in pregnant patients. 55 No maternal deaths occurred, and fetal deaths were reported in 2 of 23 (9%) cases. Bleeding complications were reported in 9 of 23 (39%) cases of which 5 of 23 (22%) were major bleeding. The authors concluded that thrombolysis could possibly be beneficial with regard to pregnancy outcomes with a relatively low risk of complications. Obviously, publication bias must be taken into account. The ASH guideline 2018 suggests administering systemic thrombolytic therapy in addition to anticoagulant therapy in pregnant women with acute PE and life-threatening hemodynamic instability. 7
It is uncertain whether the bleeding risk associated with catheter-directed thrombolysis is lower than systemic thrombolysis, even in the non-pregnant population. 16 Therefore, we do not use this option routinely. Experience with inferior vena cava (IVC) filters in pregnant women is limited. A systematic review of published cases suggests that IVC filters seem to be effective in pregnancy and that the complication rates in pregnant women are similar to non-pregnant women, but clearly, publication bias must be considered. 56 We limit the use of IVC filters in pregnancy to rare cases where anticoagulation is absolutely contraindicated for a prolonged period of time (not just for delivery).
Management of delivery
Options for delivery include unplanned spontaneous delivery, planned delivery and elective cesarean section in women using anticoagulants, and the choice depends strongly on local preferences and experiences. 57 The ASH 2018 guidelines suggest scheduled delivery with prior discontinuation of anticoagulant therapy for pregnant women using therapeutic doses of LMWH, and against scheduled delivery with discontinuation of anticoagulation for women who are using prophylactic doses of LMWH. 7 These recommendations are based on very low certainty in evidence about effects and there was disagreement within the panel.
Potential consequences of an unplanned spontaneous delivery are the increased risk of bleeding and limiting availability to neuraxial analgesia and anesthesia. Studies vary with regard to the observed bleeding rate and are of low quality. 35 In a systemic review, 2777 pregnant patients who used prophylactic and therapeutic dose of LMWH were included. 39 Maternal bleeding (>500 ml) occurred in 52 (2.0%) pregnancies. Of these 52 bleeding events, 24 occurred during the postpartum period. Another systematic review could retrieve rates for postpartum bleeding from 13 studies including 725 pregnancies. 58 Postpartum hemorrhages (PPHs) were reported in 260 out of 725 (35.9%) patients. Major bleeding (⩾1000 ml) occurred in 14 (1.9%) patients, clinically relevant non-major bleeding (⩾500–1000 ml) in 41 (5.7%) patients and minor bleeds (<500 ml) in 205 (28.3%) patients. However, a retrospective cohort study of pregnant women reported postpartum hemorrhage (>500 ml) in 18% of LMWH users and in 22% in those who did not use LMWH [blood pressure 0.8; 95% CI 0.5–1.4]. 59 The reported incidence of postpartum bleeding without anticoagulant use ranges between 4% and 22% of all pregnancies.59–61 Therefore, we suspect that pregnancy-related bleeding in women using anticoagulants is frequently underreported. 35
Definitions of PPH vary and there is no consistent definition. The World Health Organization (WHO) and the RCOG uses a definition of more than 500 ml of blood loss from the genital tract within 24 h of delivery, whereas RCOG discerns between major PPH (>1000 ml) and minor PPH (500–1000 ml).62,63 In addition, commonly used classification criteria for anticoagulant-related bleeding events in pregnancy and postpartum period may be not adequate. Therefore, a recent systematic review published also of behalf of the ISTH proposed a new definition and classification for bleeding events related to the use of anticoagulants during pregnancy and the postpartum period. 64 This classification is shown in Tables 3 and 4.
Proposed classification for antepartum and secondary postpartum (24 h to 6 wk after delivery) periods.

Red, major bleeding; orange, clinically relevant non-major bleeding; green, minor bleeding.
Early pregnancy loss before the 13th gestational week (first trimester).
Placenta previa requiring delivery.
Defined as any overt, actionable sign of hemorrhage (vaginal and non-vaginal) that does not fit the criteria of major bleeding (including spontaneous subcutaneous hematoma >25 cm2 or >100 cm2 if provoked).
Adapted from Tardy et al. 64
CRNMB, Clinically relevant non major bleeding.
Proposed classification for primary postpartum (first 24 h of delivery) period.

Red, major bleeding; orange, clinically relevant non-major bleeding; green, minor bleeding.
In case of hemorrhage of non-gynecological origin, refer to Table 3.
Adapted from Tardy et al. 64
CRNMB, Clinically relevant non major bleeding.
In case spontaneous delivery is being chosen, women must be instructed to immediately stop anticoagulant therapy upon the first signs of labor, such as contractions or broken membranes. With a planned delivery or elective cesarean section, the last injection before delivery with LMWH in a therapeutic dose should be 24 h, whereas a 12-h window is sufficient in case of a prophylactic dose of LMWH. Considering the risk of spinal epidural hematomas, these time intervals also apply when neuraxial analgesia and anesthesia is given, and are in line with current guidelines. An alternative for pain relief could be remifentanil during delivery if neuraxial analgesia is not available. 65 If the time interval is too short and an emergency cesarean section occurs, patients will have to undergo general anesthesia. Preliminary results on 587 women who participated in the Highlow study [ClinicalTrials.gov identifier: NCT001828697] showed that in only 2.4% of women the time interval was too short to receive neuraxial anesthesia in patients using prophylactic-dose LMWH and in 6.0% of patients using intermediate-dose LMWH. 66
Women in whom acute VTE is diagnosed up to 1 month before expected delivery have a high risk of recurrent or extension of VTE if anticoagulation is interrupted. Therefore we consider an induced delivery, with the intention that the time without anticoagulation is a short as possible. Women with a diagnosis of proximal DVT or PE 2 weeks before expected delivery have the highest risk for recurrent or extension of VTE; they may be considered for changing LMWH to therapeutic intravenous UFH which should be discontinued 4 h before the expected time of delivery or when obtaining neuraxial analgesia.
Postpartum management
Resumption of LMWH after delivery is advised after 12–24 h for prophylactic dose, and 24 h postpartum when restarting higher doses of LMWH, provided vaginal blood loss is normal. Because the risk of VTE remains increased, anticoagulant treatment for acute PE should be continued until 6 weeks postpartum and with a minimum duration of 3 months.7,57 LMWH, danaparoid and Fondaparinux can be used safely during breastfeeding. VKAs are also compatible with breastfeeding as only minute amounts of VKAs were found in breastmilk. 38 Continuing LMWH until 6 weeks postpartum is the most practical option for most women already using LMWH. A switch can be made to oral VKAs instead of subcutaneous LMWH in the postpartum period based on patient preference. 7 VKAs should be started at least 1 day after restart of LMWH in order to avoid double anticoagulation immediately after delivery. LMWH can be stopped when the international normalized ratio (INR) is >2.0. DOACs should be avoided in women who want to breastfeed. 52
Case continued
After 3 years, our patient consulted us again in order to get contemporary preconceptional advice. With her history of pregnancy-related VTE, we advised her to use antepartum and postpartum prophylaxis. After 1 year she became pregnant and participated in the Highlow study [ClinicalTrials.gov identifier: NCT001828697], an international, multicenter, randomized controlled trial, comparing two doses of LMWH (low and intermediate dose) for prevention of recurrent VTE. She was randomized to intermediate dose and delivered a healthy boy after 39 weeks of gestational age with 200 cc blood loss and continued LMWH until 6 weeks after delivery.
Prevention of pregnancy-related VTE
Prevention of recurrent VTE
Women with a history of VTE are at risk of recurrence during pregnancy and in the postpartum period. Without thromboprophylaxis, the absolute risk of recurrent VTE is estimated to be between 2% and 10%.67–70 The risk of recurrent VTE during pregnancy depends on factors that were present during the first VTE. Women with a first episode of VTE that was associated with a major risk factor only (major trauma, surgery, plaster cast immobilization) are at low risk (2%) for recurrence during pregnancy without prophylaxis. Therefore only postpartum prophylaxis for 6 weeks is suggested.7,38,71 In all other women with a history of VTE, that is, unprovoked VTE, recurrent VTE, VTE associated with a minor risk factor (travel, minor trauma), or VTE in the presence of a hormonal risk factor, antepartum and postpartum thrombosis prophylaxis is suggested. 7 The recently published ASH guidelines regard a VTE risk of 3% antepartum and 1% in the postpartum period to justify prophylaxis with LMWH. 7
The optimal dose of LMWH for prophylaxis of recurrent pregnancy-related VTE is unknown. As of today only two very small randomized controlled trials (n = 16 and n = 40), with limitations in their methodological quality, were published.72,73 Based on limited evidence the American College of Chest Physicians (ACCP) 2012 guidelines recommend use of either a low prophylactic dose or an intermediate dose (half of therapeutic) with no preference for one dose over the other for prevention of pregnancy-related VTE recurrence. 38 Interestingly, the more recent American ASH VTE pregnancy guideline suggests against an intermediate dose for prevention of pregnancy-related recurrence of VTE, based on the same limited evidence. 7 Currently, the Highlow study [ClinicalTrials.gov identifier: NCT01828697], an international, multicenter, randomized controlled trial, compares two doses of LMWH (low versus intermediate) to prevent pregnancy-related recurrent VTE. 74 As of today, more than 965 patients have been enrolled and results are expected in 2022.
Prevention of first pregnancy-related VTE
Primary prevention should be considered in women at increased risk for VTE, most notably women with thrombophilia. The risk of a first pregnancy-related VTE in women with thrombophilia strongly depends on the type of thrombophilia and the presence of a (first degree) family history of VTE. 7 An extensive overview of the relative and absolute risks of VTE in patients with inherited thrombophilia is provided in more detail previously 35 and is beyond the scope of this review.
Long-term outcomes of pregnancy-related VTE
Evidence on the association between a pregnancy-related PE and subsequent chronic thromboembolic pulmonary hypertension (CTPH) is lacking. A large prospective cohort study observed a CTPH incidence of 3.8% after an acute first episode of PE. In the subgroup of patients with a transient risk factor, including pregnancy, no increased risk for CTPH was observed. However no specific numbers of pregnancy were reported. 75 Post-thrombotic syndrome (PTS) is a common long-term complication of pregnancy-related VTE, especially pregnancy-related DVT.76,77 In a cohort study that followed 204 women with a history of pregnancy-related DVT in the lower limb for 3–16 years, the prevalence of PTS was 42%. Interestingly, of women with a pregnancy-related PE, 24% reported PTS. 78
Preconceptional management of women using anticoagulants
Use of DOACs outside of pregnancy should preconceptionally be converted to VKA or LMWH. 79 Evidence that DOACs actually lead to embryopathy 38 is scarce, but it cannot be excluded either. 53 When unplanned pregnancy occurs while on DOAC treatment, DOAC should be immediately discontinued and switched to therapeutic dose of LMWH. An international registry on DOAC exposure during pregnancy coordinated by the ISTH is ongoing. 54 In women using VKA, a change to therapeutic LMWH is also advised at the time of a positive pregnancy test, with the addition of vitamin K for a few days. 53 Women with mechanical heart valves are a high-risk group in whom VKA treatment continuation may be considered, but is beyond the scope of this review. We reviewed this in more detail previously. 35
Conclusion
Management of pregnancy-related VTE is challenging and data are limited. Guidelines vary and base their recommendations on low quality evidence. We conclude that the pregnancy-adapted YEARS diagnostic algorithm is well tolerated and the most efficient diagnostic algorithm for pregnant women with suspected PE. LMWH is the first-choice anticoagulant treatment in pregnancy and should be continued until 6 weeks postpartum and with a minimum of 3 months. Thrombolysis and the use of IVC filters are not routinely recommended in pregnancy as there is less experience and evidence but could be considered in selected cases. Neuraxial analgesia can be given in most patients, provided time windows are respected. Management of delivery needs a multidisciplinary approach in order to decide to the most ideal delivery plan.
Supplemental Material
Author_Response_1 – Supplemental material for Contemporary best practice in the management of pulmonary embolism during pregnancy
Supplemental material, Author_Response_1 for Contemporary best practice in the management of pulmonary embolism during pregnancy by Hanke M. G. Wiegers and Saskia Middeldorp in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v.1 – Supplemental material for Contemporary best practice in the management of pulmonary embolism during pregnancy
Supplemental material, Reviewer_1_v.1 for Contemporary best practice in the management of pulmonary embolism during pregnancy by Hanke M. G. Wiegers and Saskia Middeldorp in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.1 – Supplemental material for Contemporary best practice in the management of pulmonary embolism during pregnancy
Supplemental material, Reviewer_2_v.1 for Contemporary best practice in the management of pulmonary embolism during pregnancy by Hanke M. G. Wiegers and Saskia Middeldorp in Therapeutic Advances in Respiratory Disease
Footnotes
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Hanke Wiegers and Saskia Middeldorp are investigators of the Highlow study, a randomized controlled trial comparing low-dose LMWH with intermediate-dose LMWH in pregnant women with a history of VTE [ClinicalTrials.gov identifier: NCT01828697]. Saskia Middeldorp reports grants and fees paid to her institution from GSK, BMS/Pfizer, Aspen, Daiichi Sankyo, Bayer, Boehringer Ingelheim, Sanofi, and Portola.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


