Abstract
Chronic airway infection with
Keywords
Background
Cystic fibrosis (CF), an autosomal recessive genetic disorder, is caused by mutations in the CF transmembrane conductance regulator (
An estimated 70,000 patients worldwide have CF,
5
including 28,983 patients in the US.
6
The majority of these patients develop respiratory failure due to progressive lung disease caused by chronic bacterial infection and concomitant airway inflammation.
7
Several antibiotics administered
Inhaled antibiotics approved in Europe and the US.

CF, cystic fibrosis; EMA, European Medicines Agency; EU, European Union; FDA, US Food and Drug Administration; FEV1, forced expiratory volume in 1 second; US, United States.
The efficacy and safety of tobramycin inhalation solution (TIS) are well established in patients with CF aged ⩾6 years.29,30 Therefore, the US treatment guidelines strongly recommend chronic use of inhaled tobramycin in patients with CF who have moderate-to-severe lung disease with persistent
Nebulized antibiotics and associated challenges in patients with cystic fibrosis
Nebulized antibiotics have been established as effective treatment options for chronic
Tobramycin inhalation powder
TIP, an innovative drug–device combination, was developed with an aim to overcome the challenges associated with nebulized antibiotics, to minimize treatment burden, and to improve treatment adherence in patients with CF. TIP was developed using PulmoSphere™ (Novartis Pharma AG, Basel, Switzerland) technology (Figure 1).35,41

(a) Scanning electron microscopic image of typical micronized drug particles, TIP particles, and TIP particle (close up) and (b) T-326 Inhaler for use with TIP (TOBI® Podhaler™) (images included with permission from Geller
TIP is manufactured
Studies have shown that TIP improves intrapulmonary deposition efficiency and shorter administration time, when compared with nebulized tobramycin (TIS).42,43 In addition, inhalation
A phase I pharmacokinetic study of TIP in patients with CF reported a mean administration time of 4.9 minutes with the T-326 Inhaler as compared to 15.8 minutes with nebulizers, excluding the nebulizer cleaning and disinfection time.
41
Similarly, in a multicenter, randomized, open-label, phase III trial (EAGER), the mean administration time was significantly lower for TIP as compared with TIS (5.6
This comprehensive review, based on data from both pivotal clinical trials and real-world studies describes the efficacy, safety, and additional benefits (convenience, adherence, quality of life, and minimal device contamination) associated with the use of TIP in CF patients with chronic

Overview of phase III and IV studies and evaluated endpoint.
Pharmacokinetics
The pharmacokinetic profile of TIP was evaluated in different clinical studies. In a multicenter, open-label, active-controlled, single-dose escalation, phase I study in CF patients aged ⩾6 years with FEV1 ⩾40% predicted, administration of TIP 112 mg (4 × 28 mg) capsules resulted in similar systemic exposure to that of TIS at the standard dose of 300 mg/5 ml.
41
Furthermore, serum tobramycin concentrations were assessed in the EVOLVE study
44
and both serum and sputum tobramycin concentrations were assessed in the EDIT and EAGER studies.43,45 Blood and sputum samples were collected between 0–6 and 0–2 hours postdose, respectively; such samples were also collected predose. Tobramycin was analyzed at a central laboratory. In the EVOLVE study, there was no evidence of serum tobramycin accumulation with successive cycles of TIP 112 mg (tobramycin peak levels: cycle 1, 1.99 ± 0.59 μg/ml and cycle 2, 1.64 ± 0.96 μg/ml; tobramycin trough levels: cycle 1, 0.29 ± 0.27 μg/ml and cycle 2, 0.38 ± 0.44 μg/ml).
44
In the EDIT study, the mean peak and trough serum concentrations of tobramycin after 28 days of treatment were 1.48 and 0.41 μg/ml, respectively, and the mean maximum sputum concentrations of tobramycin were 1140 and 1739 μg/g at days 1 and 29, respectively.
45
Importantly, systemic levels were low, relative to those associated with toxicity with intravenous tobramycin (10–12 μg/ml).35,46 Moreover, the EAGER study showed that serum tobramycin concentrations were similar for TIP and TIS, and that sputum tobramycin concentrations were generally greater for TIP 30 minutes postdose on day 28 of the third cycle of treatment (mean ± SD: TIP, 1979 ± 2770 μg/g; TIS, 1074 ± 1182 μg/g). Of note, serum-to-sputum tobramycin concentrations were comparable in both the EDIT and EAGER studies (data on file). In the EAGER study, the majority (>91%) of TIP patients had
Clinical and microbiologic efficacy in phase III studies
The phase III clinical trials reviewed in this article primarily focused on the efficacy and safety of a drug–device combination of TIP (tobramycin 112 mg delivered
Summary of key phase III studies.
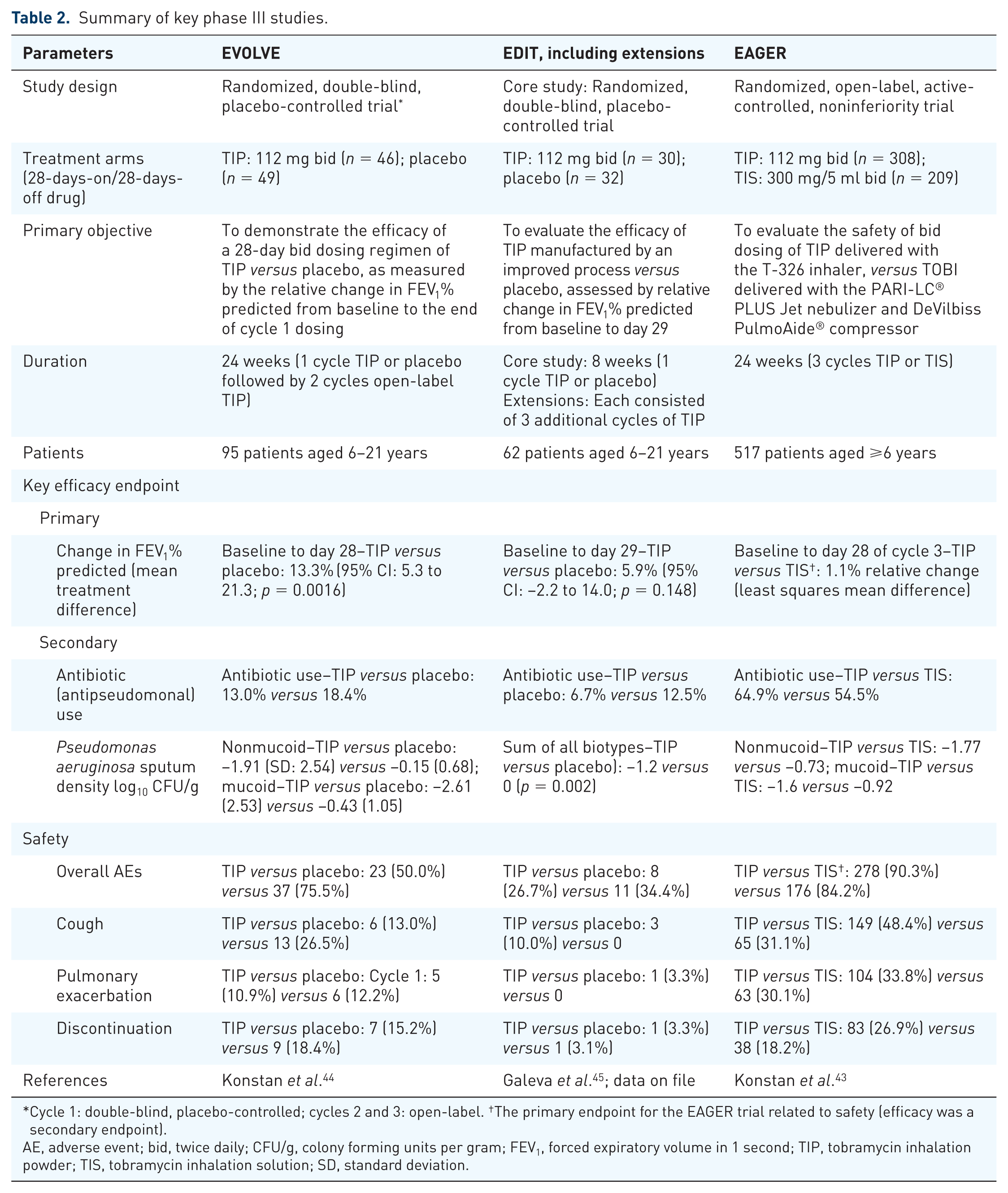
Cycle 1: double-blind, placebo-controlled; cycles 2 and 3: open-label. †The primary endpoint for the EAGER trial related to safety (efficacy was a secondary endpoint).
AE, adverse event; bid, twice daily; CFU/g, colony forming units per gram; FEV1, forced expiratory volume in 1 second; TIP, tobramycin inhalation powder; TIS, tobramycin inhalation solution; SD, standard deviation.
In the EVOLVE study, TIP showed an improvement in forced expiratory volume in 1-second percent predicted (FEV1 % predicted)
The EAGER study enrolled patients with prior exposure to inhaled antipseudomonal antibiotics. The increases in FEV1% predicted from baseline to day 28 of cycle 3 were similar between the two arms. The results demonstrated similar efficacy and safety profiles for TIP and TIS over a 6-month study period with a significantly reduced administration time for TIP
Microbiologic endpoints were included to evaluate the efficacy of TIP and TIS in all three trials, with sampling of oropharyngeal swabs and sputum following consistent methodology and cultures done at the same central laboratory. In the EVOLVE and EDIT studies,
Phase IV studies
Several studies (ETOILES, 2403, FREE, FR01, BR01, and GB01) have been conducted to collect real-world data to investigate whether the features of TIP translate into real benefits when used in routine clinical practice as demonstrated in previous studies.37,50 The results of these real-world studies (ETOILES, 2403, FREE, FR01, and GB01) showed that TIP treatment was associated with lung function benefits and suppression of
Summary of key phase IV studies.
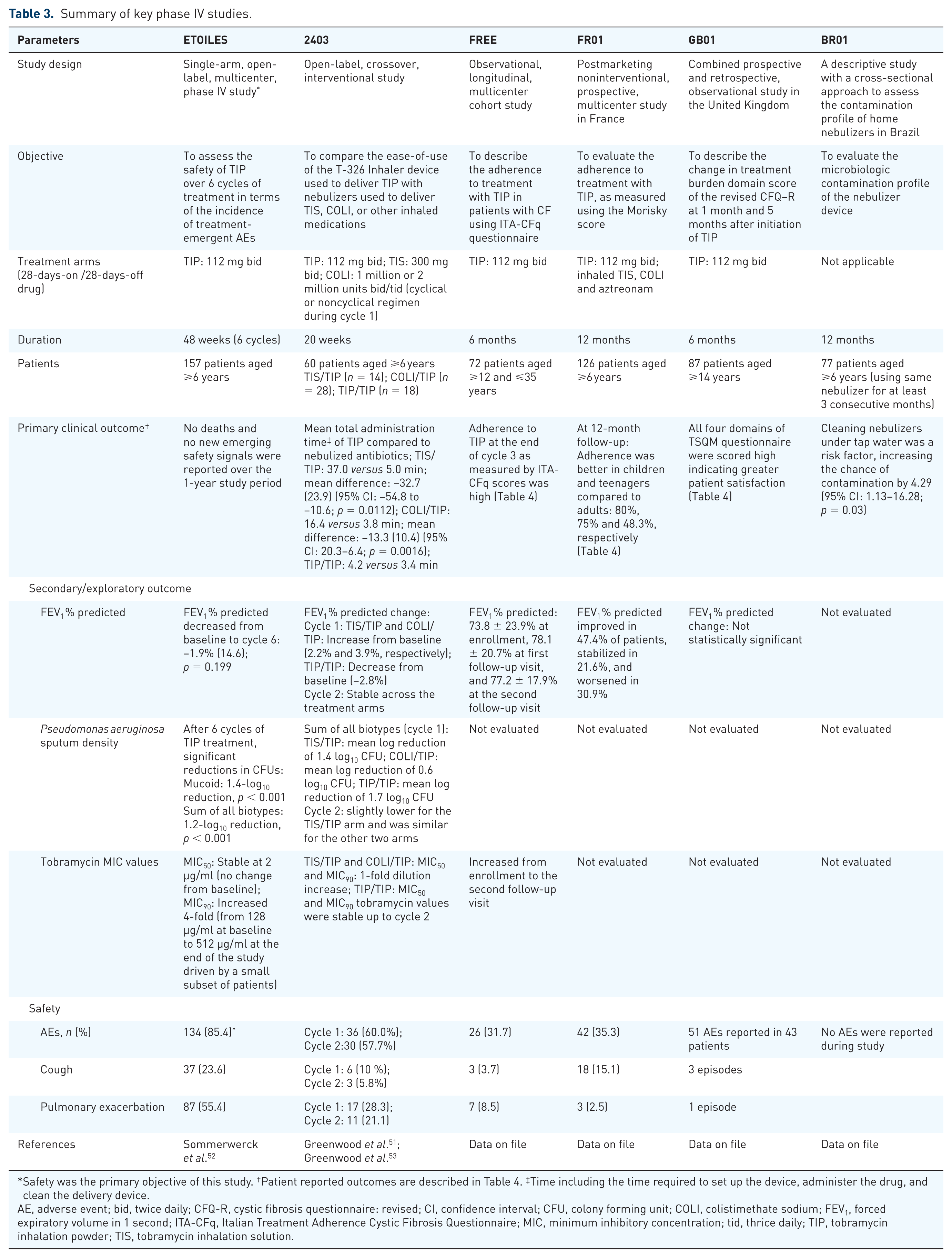
Safety was the primary objective of this study. †Patient reported outcomes are described in Table 4. ‡Time including the time required to set up the device, administer the drug, and clean the delivery device.
AE, adverse event; bid, twice daily; CFQ-R, cystic fibrosis questionnaire: revised; CI, confidence interval; CFU, colony forming unit; COLI, colistimethate sodium; FEV1, forced expiratory volume in 1 second; ITA-CFq, Italian Treatment Adherence Cystic Fibrosis Questionnaire; MIC, minimum inhibitory concentration; tid, thrice daily; TIP, tobramycin inhalation powder; TIS, tobramycin inhalation solution.
Summary of patient-reported outcomes assessed in key phase III * and IV † studies.

Konstan
AEs, adverse events; CFQ-R, cystic fibrosis questionnaire: revised; CI, confidence interval; COLI, colistimethate sodium; ITA-CFq, Italian Treatment Adherence Cystic Fibrosis Questionnaire; QoL, quality of life; SE, standard error; SD, standard deviation; TIP, tobramycin inhalation powder; TIS, tobramycin inhalation solution; TSQM, treatment satisfaction questionnaire for medication.
Patient-reported outcomes
Patient satisfaction, convenience and adherence were evaluated in phase III (EAGER) and phase IV (2403, FREE, FR01, and GB01) studies using different questionnaires such as the modified-treatment satisfaction questionnaire for medication (TSQM),54,55 ACCEPTance, 56 the revised cystic fibrosis questionnaire (CFQ-R), 57 the Italian CFQ (ITA-CFq), and the Morisky scale. 58 EAGER, 2403, FREE, and GB01 studies utilized the TSQM questionnaire for evaluating patients reported outcomes (effectiveness, satisfaction, convenience, and side effects). ACCEPTance and patient preference questionnaires were used in the 2403 study. Furthermore, CFQ-R and ITA-CFq were used in the GB01 and FREE studies, respectively (Table 4). The TSQM scores for the treatment satisfaction domain were significantly higher for TIP with regard to effectiveness, convenience, and global satisfaction in EAGER (Table 4). Similarly, the 2403 study showed high scores for TSQM in cycle 1, which were either sustained or further improved in cycle 2 for the majority of domains, indicating greater treatment satisfaction in patients receiving TIP over TIS and COLI. 51 In the GB01 study, increases were seen in the scores for all four domains of TSQM (data on file).
A real-world study by Harrison and colleagues showed that the proportion of participants reporting ‘excellent adherence’ was increased by twofold after switching from TIS to TIP (43–83%). 37 Another real-world study showed that the majority of patients expressed satisfaction with TIP administration time (100%), time to clean (97.1%), portability (97.1%), and ease of use (94.3%). Overall, the patient preference for TIP was based on shorter administration time, convenience, and ease of use. 59 In summary, results from various real-world studies are in line with the phase III clinical trial data that suggest improved patient adherence with TIP as compared with TIS. 43 The detailed patient reported outcomes from various studies are presented in Table 4.
Inhalation-device contamination
Various studies have investigated the role of home nebulizers as a source of contamination in patients with CF.39,40,60,61 An open-label, crossover, interventional phase IV study (2403) analyzed the contamination profile of both nebulizers and the T-326 inhaler, while a descriptive study, BR01, analyzed the contamination profile of nebulizers with regard to methods of cleaning to minimize contamination. In the 2403 study, microbial contamination of the nebulizers was assessed at the start and end of the first treatment period, second treatment period, and at the discontinuation visit if applicable. For patients on TIP, the T-326 Inhaler used in the last week of TIP treatment was cultured. Device samples were obtained from four locations on the nebulizer (mouthpiece, reservoir cup, filter, and tubing) and from one location on the T-326 Inhaler (mouthpiece). A central laboratory performed all device cultures, as well as sputum cultures from patients. The results of this study showed that the T-326 Inhaler used to deliver TIP was much less frequently contaminated than the nebulizers, thus potentially reducing the sources of pathogenic bacteria in patients with CF. 53 In the BR01 study, microbial contamination of the nebulizers was assessed using samples taken from the mouthpiece and the reservoir cup, and cultures were performed at a central laboratory. The latter study concluded that cleaning nebulizers with tap water increased the chance of contamination by 4.29 fold (Table 5). Assessment of contamination of various parts of the nebulizer showed that the frequency of contamination was 60.8% in the mouthpiece and 62.2% in the cup, which was consistent with the reported pattern of contamination profile of nebulizers.39,60
Summary of key phase IV studies device contamination results.

CI, confidence interval; COLI, colistimethate sodium; OR, odds ratio; TIP, tobramycin inhalation powder; TIS, tobramycin inhalation solution.
Safety and tolerability
Inhaled tobramycin preparations have the advantage of minimal systemic exposure, and hence have a different adverse event (AE) profile compared with parenteral formulations.
62
The phase III (EVOLVE, EDIT, and EAGER) and phase IV (ETOILES, 2403, FREE, FR01, and GB01) studies suggested that there were no unexpected safety signals with TIP treatment. In the EVOLVE study, the incidence of AEs reported among TIP-treated patients was lower (50.0%) when compared with placebo-treated patients (75.5%) in cycle 1.
44
The incidence of lung disorders (preferred term mainly for pulmonary exacerbations) was comparable in cycle 1; however, the frequency appeared to be higher for any given cycle in the TIP arm compared with the placebo arm (Table 2). The incidence rates of overall AEs were generally higher in the placebo arm
In the ETOILES study (
The data from phase IV studies showed that TIP was well tolerated, and safety findings from real-world studies are consistent with results of the phase III studies.
In general, the most common AEs in patients receiving TIP were cough and pulmonary exacerbations.43,45 Cough was the most common AE in the TIP groups in all three studies, EVOLVE, EDIT, and EAGER. However, there were no treatment discontinuations due to this AE (Table 2).44,45
Postinhalation cough is reported as a common side effect associated with both wet- and dry-powder inhalation in patients with CF in various clinical studies.43,44,52 The reason for the relatively high incidences of cough, dysphonia, and dysgeusia could be the delivery of a relatively high powder load or deposition of tobramycin to the posterior pharynx causing irritation, which decreases with time.
43
Therefore, ETOILES used a specific case-report form to record and characterize postinhalation events (including cough), capturing time of onset and duration. In this study, 78 patients reported postinhalation cough, which was highest in cycle 1 (31.4%) and subsequently decreased during cycles 4–6 (21–22%). In most cases, the postinhalation cough was generally of short duration (<4 min) and decreased over time, with no action required, possibly due to patients becoming more experienced with the administration of TIP.
52
Furthermore, there is growing evidence that proper inhalation techniques may result in minimization of postinhalation cough for ‘high-dose’ dry powder products like TIP. Reduction in postinhalation cough was observed for inhaled drugs when a higher resistance DPI with a lower PIFR was used. The flow rate independence in total lung dose
Common measures to minimize postinhalation cough utilized during clinical trials were drinking water, less forceful inhalation, and correction of inhalation technique. 52 Additional cough-mitigation strategies include: (1) avoid pressing button more than once; (2) tilt head back slightly during inhalation; and (3) inhalation with a single, slow, and deep breath to minimize cough.
Conclusion
Nebulized antibiotics have significantly contributed to increasing the life expectancy in CF patients with chronic airway infection; however, the high treatment burden and nebulizer contamination are major concerns. TIP administered
Footnotes
Acknowledgements
The authors acknowledge Anupama Tamta (Novartis Healthcare Pvt. Ltd., Hyderabad, India) for providing medical writing assistance for this manuscript.
Funding
The study was sponsored by Novartis Pharma AG.
Conflict of interest statement
KH and LD are full-time employees of Novartis Pharmaceuticals Corporation.
