Abstract
A population pharmacokinetic analysis was performed to investigate the pharmacokinetics of moxifloxacin (400 mg) following a once-daily oral administration in 28 patients with respiratory tract infection disease. The maximum plasma concentration and the area under the plasma concentration–time curve were 3.97 µg/ml and 51.74 µg·h/ml, respectively; these values were nearly equivalent to those of healthy adult men. Two adverse drug reactions (nausea, vomiting) occurred, but both reactions were mild and nonserious and the patients recovered without treatment. The pharmacokinetic profile of moxifloxacin in Japanese patients with respiratory tract infection and an underlying disease should thus be considered safe and comparable with that in healthy adult men, and adjustment of dose may do not need for age, sex, body weight, or renal function.
Introduction
Acute respiratory tract infection is one of the most problematic infections induced by major causative bacteria, such as
High-dose penicillin antibiotics are the first choice for the treatment of bacterial pneumonia, but current increases in the isolation rates of bacteria showing resistance to penicillin antibiotics, such as penicillin-resistant
Moxifloxacin (MXF), a respiratory quinolone developed by Bayer Germany and Bayer Inter-national is the first fluoroquinolone antibacterial agent (FQ) with a once-daily oral administration of 400 mg to have the same dosage and administration regimen in Japan as in Europe and the US, based on the pharmacokinetics/pharmacodynamics (PK-PD) profile of the drug. The clinical efficacy and safety of this drug has been confirmed both within and outside of Japan [Welte et al. 2005; Fogarty et al. 2005; Anzueto et al. 2006; Torres et al. 2008; Ott et al. 2008; Kobayashi et al. 2005; Liu et al. 2014] based on its favorable antibacterial activity [Nishino and Otsuki, 2005; Inoue et al. 2005; Niki et al. 2011] and PK-PD profile [Jacobs, 2001; Onishi, 2005], and MXF is expected to be highly useful for the treatment of outpatients with 0–2 points CURB-65 of community-acquired pneumonia and secondary infections arising from chronic respiratory diseases.
Given that dose adjustments are not necessary for MXF, even for patients with impaired renal function [Stass, 2002; Mishina, 2010; Tanaka and Watanabe, 2005], and that MXF exhibits a relatively high antibacterial activity against anaerobic bacteria (compared with other FQs) [Tanaka and Watanabe, 2005], MXF is also highly useful for patients with recurrent respiratory tract infections, such as elderly patients with underlying diseases. While some reports on its pharmacokinetics in patients with infection have been published outside of Japan [Simon et al. 2003; Noreddin et al. 2007], the pharmacokinetics of MXF in Japanese patients with infection have not been sufficiently reported, and no report has been published so far concerning the measurement of drug concentrations in a population pharmacokinetic analysis of patients with respiratory tract infection and an underlying disease or complication.
Reported here are the results of our population pharmacokinetic analysis in Japanese patients with respiratory tract infection and an underlying disease or complications.
Patients and methods
Patients
Patients aged 20 years or older who visited the study site from 2008 through 2011, were diagnosed as having to acute exacerbation of COPD or respiratory infection in patients with previously diagnosed respiratory diseases or pneumonia, and voluntarily provided written consent were enrolled in this study. The following three inclusion criteria were applied to study subjects with a secondary infection in addition to a chronic respiratory infection:
body temperature above 37°C and presentation with clinical symptoms characteristic of respiratory infection, such as coughing, sputum, chest pain, and dyspnea;
white blood cell (WBC) count of 8000/mm3 or higher and a C-reactive protein (CRP) level of 0.7 mg/dl or higher; and
acute and new onset of an infiltration shadow on a chest X-ray image in addition to criteria 1 and 2 above for patients with pneumonia.
The subjects enrolled in this study were patients with an infection score of 0–2 using CURB-65 who had been previously treated with an antibacterial agent prior to the start of this study and whose symptoms had not improved despite treatment for 3 days or longer, in principle. Patient exclusion criteria for this study were serious cases with a CURB-65 score of more than three points or serious complications. Patients who cannot evaluate the effectiveness of the drug were also excluded. In addition, patients with impaired serious liver function, patients with the cerebrovascular disease, patients allergic to the quinolone medicine, patients with an extension of QTc, and pregnant patients were excluded from this study
Administration method
An oral dose of MXF (Avelox® Tablets, 400 mg; Bayer Yakuhin, Ltd) was consecutively administered once daily for 7 days, in principle.
For this study, patients less than 40 kg in weight and 65 years or older received treatment with the same dose as a physically unimpaired person. The treatment period was to be shortened when the treatment purpose was achieved or when discontinuation was unavoidable because of the onset of adverse drug reactions.
Contraindicated drugs and concomitant drugs/therapies
The use of other antibacterial agents that could influence the efficacy evaluation of MXF was prohibited during treatment with this drug. However, the use of oral antibiotic and long-term and small doses of macrolide antibacterial agents that had been continued from before the initiation of this study were permitted, provided that the dosage and administration schedule remained unchanged. The use of an antipyretic/analgesic agent was permitted only on an as-needed basis. The concomitant use of steroids (such as predonine) was permitted, provided that the dosage and administration schedule remained unchanged during the treatment period. When another drug was used, the drug name and duration of administration were recorded.
Observation items and timing
Plasma drug concentration
Blood specimens were collected at two or three time points (at approximately 1–2 hours, 10 hours, and 24 hours) after the third administration of the study drug. The specimens were centrifuged, and the plasma blood concentration of MXF was determined using high-performance liquid chromatography (HPLC). HPLC is a kind of column chromatography and is a method using a high-pressure liquid as a mobile phase.
A population pharmacokinetic analysis using a nonlinear mixed effect model was performed for the obtained plasma drug concentration data. NONMEM VI (version 2.0, FOCE INTER) was used for the analysis. A relative error model was used, after assuming that the individual variation (η) and the intra-individual variation (ε) would follow a log-normal distribution. Covariates were explored for age, weight (WT), creatinine clearance (CrCL), WBC count, CRP, aspartate transaminase (AST), alanine transaminase (ALT), sex, and severity of renal impairment. The level of significance for the likelihood ratio test was
Safety
The onset of adverse events was observed throughout the study period. A clinical laboratory test and chest X-ray examination were performed at baseline, 3 days after the initiation of treatment (whenever possible), and 7 days after the initiation of treatment or at treatment completion (discontinuation). When adverse events or abnormal variations in the clinical laboratory data were observed, the symptom, date of onset, severity, causal relationship with the drug, action and course, and outcome were investigated. The criteria for judging the seriousness of the adverse event and the causal relationship with MXF were determined prior to the initiation of this study, and these parameters were evaluated as ‘serious or nonserious’ and ‘related or not related’, respectively.
Results
Evaluated patients
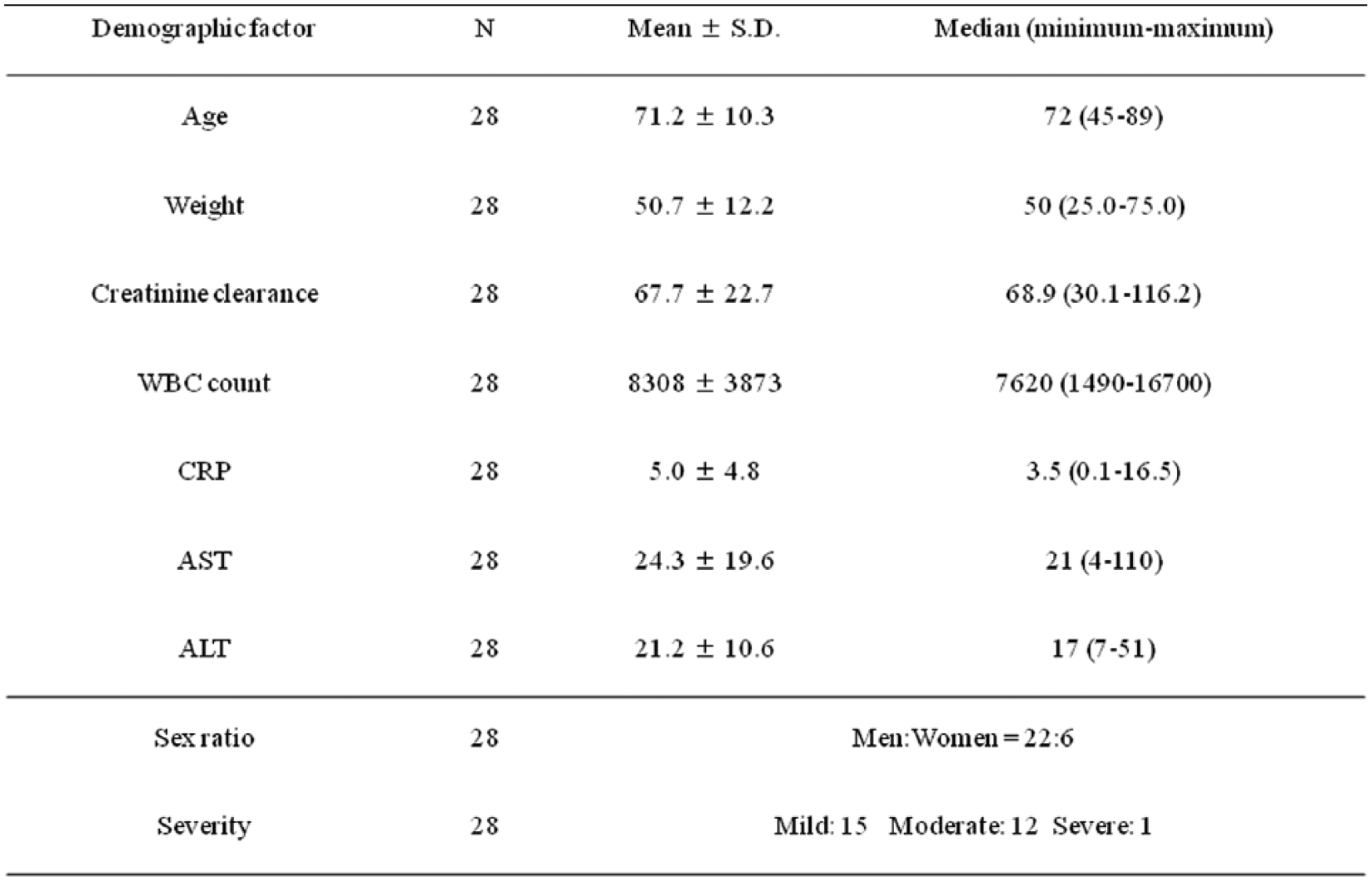
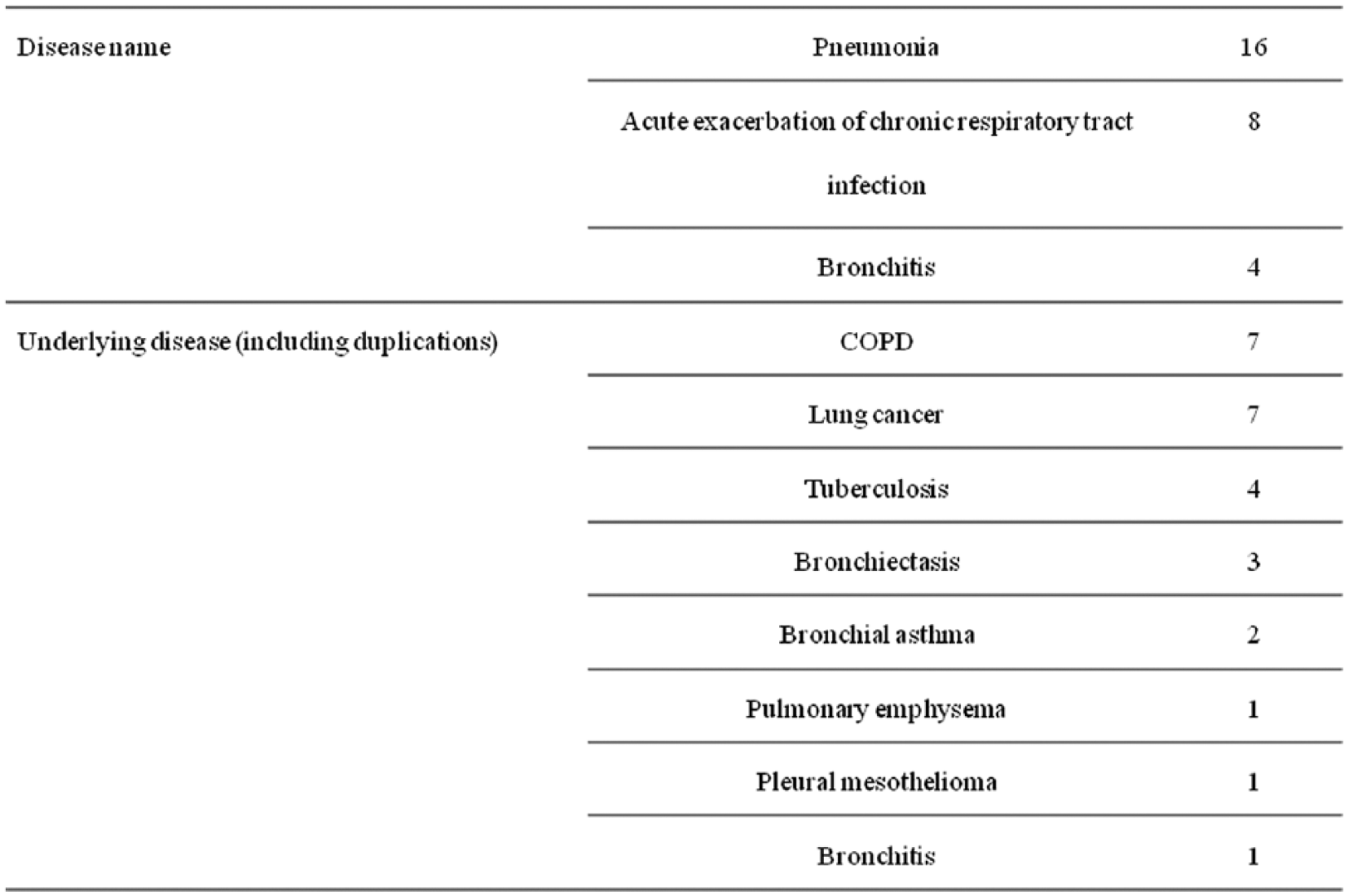
Of the 34 patients who were enrolled in this study and received MXF, 28 patients were included in the analyses of efficacy and pharmacokinetics after excluding six subjects for whom data regarding body weight, timing of the blood sampling, or CrCL were missing. The patient demographics of the 28 patients included in the pharmacokinetic analysis were as follows: 22 men and six women, mean age of 71.2 years, mean weight of 50.7 kg, and mean CrCL of 67.7 ml/min (Table 1). The diagnoses were pneumonia in 16 patients, exacerbation of the chronic respiratory tract infectious disease in eight patients, and bronchitis in four patients (Table 2). The underlying diseases were COPD in eight patients, lung cancer in seven patients, second infectious disease in tuberculosis aftereffects in four patients, bronchiectasis in three patients, bronchial asthma in two patients, and pleural mesothelioma, and bronchitis in one patient each (Table 2). The safety analysis included all of the patients who were treated with MXF (34 patients).
Demographic factors.

Disease names and underlying diseases.

Administration state
The mean MXF treatment period was 7.6 days (3–15 days), and 28 patients were treated for 7 days or longer.
Plasma pharmacokinetic parameters
Figure 1 shows the plasma drug concentration obtained from blood samples collected at 56 time points for the 28 patients who were included in the pharmacokinetic evaluation, and Tables 3 and 4 show the estimated pharmacokinetic parameters of the final pharmacokinetic model and the pharmacokinetic parameters of each patient, respectively. The maximum plasma concentration at up to 24 hours after administration (

Plasma concentration after the oral administration of moxifloxacin (400 mg) (left graph: linear scale, right graph: semilog scale). The open circles show the observed moxifloxacin concentrations at steady state after multiple doses of moxifloxacin (400 mg) administered once daily.
Pharmacokinetic parameters includes in final model.

Estimated pharmacokinetic parameters in each subject.


GOF (goodness of fit) plot of the final model. The mean estimates (PRED) and individual

Estimated changes in plasma drug concentration and measured values in each subject.
Safety
Adverse events consisting of nausea and vomiting and for which a causal relationship with MXF could not be denied (adverse drug reactions) occurred in one patient each, but both events were mild and nonserious and both patients recovered without treatment.
Discussion
The pharmacokinetic parameters obtained in this study were compared with the results of a phase I repeated oral-dose study of MXF in healthy Japanese adult subjects at steady state (
The mean age of the 28 subjects enrolled in this study was 70 years, and the underlying diseases included lung cancer in 7 patients, chronic obstructive pulmonary disease (COPD) in 7, tuberculosis in 4, and bronchial asthma in 2 (including some patients with more than one underlying disease). Clinically, FQs are frequently administered to elderly patients with relatively serious underlying diseases who develop recurrent infections. Given that MFLX, in particular, is expected to show a superior usefulness in patients with impaired renal function or aspiration pneumonia based on its drug profile [Stass, 2002; Hori, 2010; Mishina, 2010], compared with other FQs, MXF is likely to be frequently used in these patients. However, few studies have investigated the pharmacokinetic profile in patients with respiratory tract infections. Some reports have been published outside of Japan [Simon et al. 2003; Noreddin et al. 2007], but the measurement of drug concentrations in Japanese patients with infections in a population pharmacokinetic analysis has not been reported to date, and no study has been performed in Japan or elsewhere in patients with respiratory tract infections and an underlying disease.
The results of our population pharmacokinetic analysis showed that the plasma MXF concentration in Japanese elderly patients with respiratory tract infections and underlying diseases had a pharmacokinetic profile that was similar to that of healthy adult men.
MXF is therefore considered to be a useful therapeutic agent for respiratory tract infection that can be safely administered using a dosage and administration schedule of 400 mg once daily (which is the dame dosage and administration schedule used outside of Japan) for Japanese patients with infections and underlying diseases.
Footnotes
Acknowledgements
The authors would like to offer their special thanks to William Goldman for English revision.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that they have no competing interests.