Abstract
Objective:
The ultra-short course preseasonal allergy vaccine, containing the adjuvant monophosphoryl lipid A (MPL), is effective in treating allergic symptoms; however, the efficacy in controlling asthmatics symptoms has not been fully demonstrated. We aimed at evaluating whether the ultra-short preseasonal course of immunotherapy contributes to asthma control.
Methods:
Four subcutaneous injections of the active product (Pollinex Quattro) were administered, before the pollen season, to 20 Parietaria-sensitive mild, untreated asthmatics (M/F: 12/8; age: 38 ± 14 years). After the screening visit (visit 1), asthma control was assessed by the Asthma Control Test (ACT) immediately before the first (visit 2) and immediately after the last (visit 5) injections, as well as during the pollen season (visit 6). Bronchial and alveolar exhaled nitric oxide (NO) concentrations were also measured. Nine Parietaria-sensitive mild asthmatics (M/F: 3/6; age: 40 ± 12 years) served as untreated controls.
Results:
The ACT remained constant during allergen exposure in specific immunotherapy (SIT)-treated asthmatics (visit 2: 22 ± 3.2; visit 5: 23 ± 2.8; visit 6: 22 ± 3.6; analysis of variance [ANOVA], p = 0.47), whereas it dropped during pollen exposure in controls (visit 2: 20 ± 2.5; visit 5: 21 ± 2.8; visit 6: 16 ± 5.7; ANOVA, p = 0.01). The forced expiratory NO (FENO) values significantly increased during pollen exposure in both groups; however, the alveolar NO concentrations remained stable in SIT-treated asthmatics (p = 0.11), whereas they doubled in controls (p = 0.01).
Conclusions:
The current findings show that the preseasonal vaccine adjuvated with MPL contributes to the maintenance of control of asthma during the pollen season.
Introduction
Bronchial asthma is one of the most common chronic diseases worldwide, characterized by an infiltrate of inflammatory cells such as lymphocytes, eosinophils and mast cells in the bronchial wall. Up to 80% of asthmatics also present with features of rhinitis, which triggers asthma attacks and worsens health-related quality of life (HR-QoL). To manage chronic respiratory allergies, international guidelines [Global Initiative for Asthma, 1995] recommend the avoidance of allergens and removal of triggers, together with the pharmacological therapy and specific immunotherapy (SIT). The latter is considered the only treatment capable of modifying the natural course of allergy, preventing (or delaying) the onset of asthma in patients with allergic rhinitis [Bousquet et al. 1998]. In addition, SIT has been demonstrated to improve upper and lower airway symptoms, and to reduce the use of medications [Holt et al. 1998].
To improve the efficacy of SIT, immunological adjuvants have been incorporated in the allergy vaccines. The monophosphoryl lipid A (MPL), a detoxified lipopolysaccharide component extracted and purified from Salmonella minnesota, appears to promote and increase the immunological response [Wheeler et al. 2001]. In a recent study from our group [Scichilone et al. 2011], a short course of preseasonal vaccine for Parietaria was demonstrated to reduce the concentration of 8-isoprostane, a marker of bronchial inflammation, as early as the fourth (last) administration, and to maintain low levels of inflammation during the pollen season. The preseasonal MPL vaccine has been shown to be effective in relieving nasal and ocular symptoms [Patel and Salapatek, 2006]; however, the efficacy of the adjuvated vaccine in controlling asthmatics symptoms has not been demonstrated.
In the current explorative study, we tested whether, in asthmatics with concomitant rhinitis, a preseasonal course of Parietaria MPL vaccine is effective in: (1) reducing the level of airway inflammation; and (2) maintaining optimal asthma control as well as symptom-related quality of life. To comply with the objectives of the study, we avoided any influence of corticosteroid treatment and smoking exposure on the explored outcomes.
Methods
Subjects
Individuals attending the Allergy Outpatient Clinic of the Division of Respiratory Diseases of the University of Palermo, Italy, who had previously received the diagnosis of asthma and concomitant rhinitis, were enrolled. To be eligible for the study, subjects had to be allergic only to Parietaria, with no previous history of any type of specific immunotherapy. Subjects with moderate or persistent asthma, those who were under regular anti-asthmatic treatment or unable to perform pulmonary function tests, and individuals with a family history of fatal asthma were excluded. In addition, current smokers were not allowed to participate to the study. Subjects were tested at least 4 weeks after their most recent upper respiratory infection. Skin prick tests were performed using the 10 most common regional commercial allergen extracts (Lofarma Laboratories, Milan, Italy): Alternaria tenuis, Parietaria, Olea europaea, Mix compositae, Mix graminacee, Cupressus sempervirens, Dermatophagoides pteronyssinus (DPT) and Dermatophagoides farina (DF), cat and dog danders. Histamine hydrochloride (10 mg/ml) and glycerol saline were used as positive and negative controls, respectively. The mean wheal size was recorded after 15 minutes and skin prick test was considered positive with a wheal size of 3 mm or greater. Eligible subjects who refused SIT were invited to participate as untreated controls. All study subjects were allowed to use symptomatic treatment for upper and lower respiratory symptoms as required. The study was performed in accordance with the Good Clinical Practice guidelines recommended by the International Conference on Harmonization of Technical Requirements and was approved by the Ethics Committee of University Hospital of Palermo, and all participants gave written informed consent before inclusion.
Study design
The study included two groups: group 1 consisted of subjects who underwent preseasonal SIT and group 2 included individuals who did not receive SIT. All subjects underwent the same functional, clinical, and biological assessments.
The study entailed six visits. At visit 1, which took place in the months of July and August 2010, all inclusion and exclusion criteria were checked and subjects were enrolled. The screening evaluation included a respiratory questionnaire for both upper and lower airways, allergy skin testing to a panel of 10 common aeroallergens, and lung functional assessment. The presence of features of rhinitis was also diagnosed according to the ARIA recommendations [ARIA, 2010]. During the months of September–October (prepollen season), each subject visited the Clinic on four separate occasions (visits 2–5). Group 1 received a subcutaneous injection of the active product with increasing strength; the first three 1.0 ml increasing strength injections were administered at 1-week intervals, and the last top-strength injection was administered at 6 weeks from the first injection. At the time of the first (visit 2) and the last (visit 5) injections, HR-QoL and the level of asthma control were assessed, and measurements of nitric oxide (NO) concentrations in the exhaled air were obtained. The asthmatics that served as untreated controls also underwent the same clinical, functional and biological assessments. Finally, all subjects (groups 1 and 2) returned to the Clinic during the peak of the pollen season (May 2011) to undergo the same investigations (visit 6). The pollen count was provided by the Italian Society of Aerobiology, which weekly updates the pollen concentrations in the air by region. The pollen count in Sicily in May 2011 for Urticaceae was between 20 and 69.9 grains per cubic meter of air, which is considered high. The protocol of the study is described in Figure 1.

Schematic of the protocol of the study.
SIT characteristics
The ultra-short course preseasonal allergy vaccine (Allergy Therapeutics, UK, Ltd, Worthing, UK), containing appropriate allergoids with the MPL, was employed in the current study. In detail, the vaccine consists of purified extract of Parietaria judaica pollen allergen (par j1: 16.64 µg/ml), which is modified with glutaraldehyde. The MPL adjuvant is adsorbed to the L-tyrosine at 50 µg/ml in all vaccine dose levels.
Clinical assessment
Asthma control test
Asthma control was assessed by using a self-administered questionnaire (Asthma Control Test [ACT]). The ACT is a validated five-item instrument, each item rated on a five-point scale [Schatz et al. 2007], providing a total score that allows us to discriminate between ‘well controlled’ (total score: 25), ‘partially controlled’ (total score: >20 and <25), and ‘not controlled’ (total score: <20). The questions address symptoms occurring within the 4 weeks preceding the evaluation.
Rhinasthma
The HRQoL was evaluated by the Rhinasthma questionnaire [Baiardini et al. 2003], which consists of 30 items. The questionnaire explores three domains: quality of life related to upper airways (UA), lower airways (LA), and respiratory allergy impact (RAI). This analysis leads to a composite score, namely the global summary (GS) score, which indicates the overall impact of the disease. Answers to the Rhinasthma items are converted into a score from 0 to 100: higher scores correspond to worse QoL related to respiratory symptoms.
Functional assessment: spirometry
Functional assessment included conventional spirometry. Forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC), as well as forced expiratory airflows at different lung volumes (FEF25, FEF50, FEF75 and FEF25-75 of FVC), were recorded. All spirometric measurements were obtained from a computerized water-sealed spirometer (Biomedin, Padua, Italy), which allowed compliance with criteria for acceptability and reproducibility online. Acceptability and reproducibility of FEV1 and FVC were evaluated based on the recommendations of the European Respiratory Society [Miller et al. 2005].
Biological assessment: measurement of exhaled NO
The measurement of the fraction of exhaled NO is a noninvasive method, simple and reproducible, which reflects the degree of inflammation in the airways. The exhaled NO was obtained by means of a chemiluminescence analyzer (FENO Hypa, Medisoft), which detects the light emitted by the photochemical reaction between NO and ozone generated by the tool. The amount of light emitted is proportional to the concentration of NO, in parts per billion (ppb). Subjects were in sitting position during the entire procedure. Each individual was asked to take a deep inspiration from room air and, immediately after, to forcefully exhale through the mouthpiece that was connected to the online analyzer. To determine whether subjects maintained a constant flow rate during exhalation, a continuous monitoring of expiratory flow rate by graphic display on the monitor screen was used. All samples were acquired in the morning between 09:00 and 12:00. Subjects performed two acceptable and reproducible maneuvers at expiratory airflow of 50 ml/s, and the mean of the two observations was used for analysis. In addition, measurements were conducted at expiratory airflow rates of 100, 150 and 350 ml/s. This allowed us to estimate the concentration of NO coming from the peripheral lung (concentration of alveolar NO [CANO]). CANO is described by the slope of the plotting of the expiration rates of NO against the exhalation airflow.
Statistical analysis
The data obtained are presented as mean ± standard deviation (SD). The analysis of variance (ANOVA) was used for repeated measures. A p-value less than or equal to 0.05 was considered statistically significant. The statistical package we employed was StatView 5.0.1 (Abacus Concept, Berkeley, CA).
Results
The study was conducted on a total of 29 asthmatics: group 1 consisted of 20 subjects (M/F: 12/8, age 38.2 ± 14 years) and group 2 comprised 9 age-matched subjects (M/F: 3/6, age 40 ± 12 years). Lung function was within the normal range (FEV1% predicted, for group 1: 103 ± 15%; for group 2: 93 ± 21%). The two groups did not differ in terms of baseline lung function characteristics, level of asthma control and HR-QoL (Tables 1 and 2 at the time of recruitment). In addition, the levels of exhaled NO concentrations were similar between the two groups (group 1 versus group 2; forced expiratory NO [FENO]: 42.3 ± 23 ppb versus 54.6 ± 31 ppb, p = 0.24; CANO: 5.1 ± 2.9 ppb versus 5.1 ± 3.2 ppb, p = 0.95). All subjects in groups 1 and 2 completed the protocol and none of them reported adverse events associated with the SIT.
Lung function characteristics of the study subjects at visit 1.
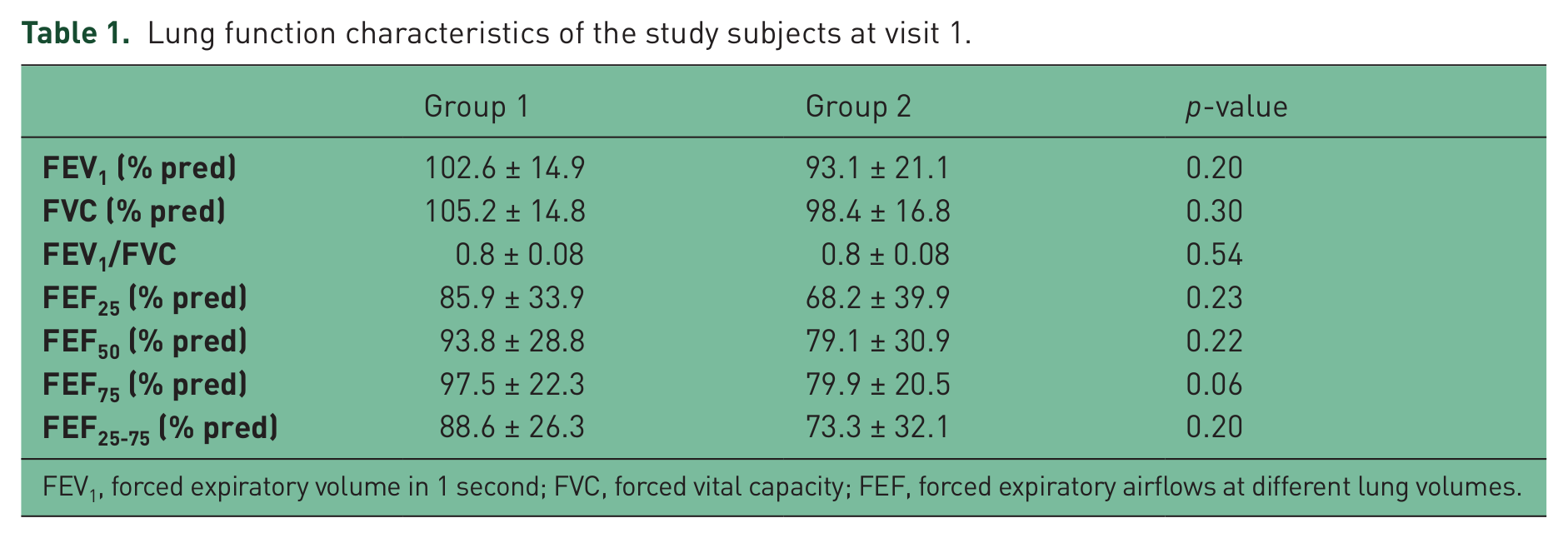
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; FEF, forced expiratory airflows at different lung volumes.
Level of asthma control (ACT) and health-related quality of life (Rhinasthma) in the two study groups at visit 1. UA, LA and RAI are the three domains of the Rhinasthma questionnaire.

ACT, asthma control test; UA, upper airways; LA, lower airways; RAI, respiratory allergy impact; GS: global summary score.
Lung function
Lung function did not change during the study periods in both groups (ANOVA for repeated measures: p > 0.05 for both groups). In group 1, FEV1% predicted was 103 ± 15% at visit 2, 104 ± 15% at visit 5 and 105 ± 14% at visit 6. In group 2, FEV1% predicted was 93 ± 21% at visit 2, 97 ± 22% at visit 5 and 97 ± 24% at visit 6.
ACT
The level of asthma control, as assessed by the ACT, remained constant during the vaccine administrations and did not decrease during allergen exposure in group 1 (visit 2: 22 ± 3.2; visit 5: 23 ± 2.8; visit 6: 22 ± 3.6; ANOVA, p = 0.47). Conversely, the ACT scores significantly dropped during pollen exposure in SIT-untreated subjects (group II, visit 2: 20 ± 2.5; visit 5: 21 ± 2.8; visit 6: 16 ± 5.7; ANOVA, p = 0.01) (Figure 2).
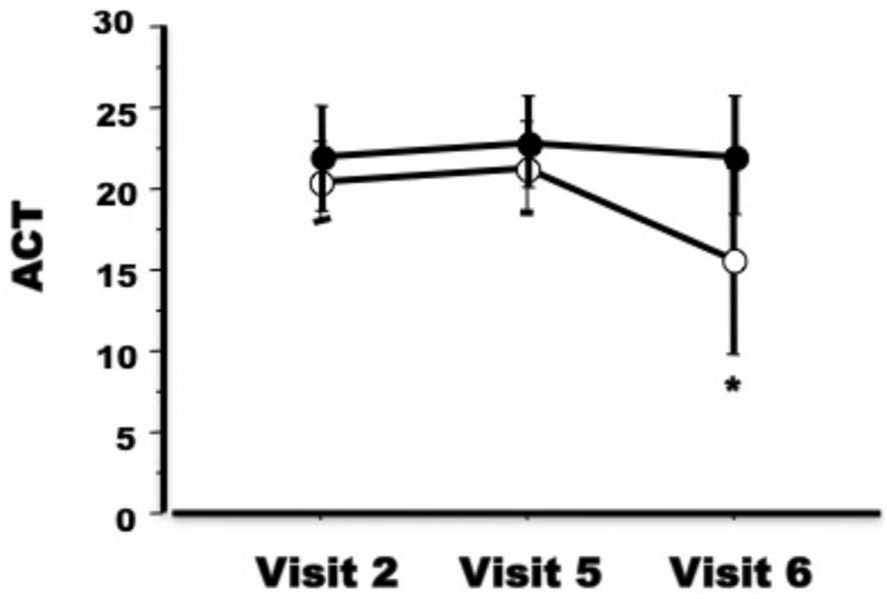
Asthma Control Test (ACT) score in specific immunotherapy (SIT)-treated (closed circles) and SIT-untreated (open circles) groups. *p = 0.01 analysis of variance (ANOVA) for repeated measures. For further explanation of visits 2, 5 and 6 see the Methods section.
Rhinasthma
The overall impact of the respiratory diseases on patients’ quality of life did not change significantly during pollen exposure either in treated or untreated subjects. Indeed, in group 1 the Rhinasthma global score was 14 ± 11 at visit 2, 10 ± 9 at visit 5, and 18 ± 14 at visit 6 (ANOVA, p = 0.08). In group 2, the Rhinasthma global score was 22 ± 15 at visit 2, 21 ± 15 at visit 5, and 32 ± 18 at visit 6 (p = 0.11). Similar trends were obtained for the Rhinasthma partial scores in both groups (data not shown).
Exhaled nitric oxide
Figure 3(A) shows the changes in exhaled NO concentrations for both groups during the study visits. The FENO values significantly increased during pollen exposure in both groups; however, the increase was more impressive in SIT-untreated asthmatics (group 2), in whom FENO concentrations doubled (visit 6 versus visit 2, mean difference: 72 ppb). In particular, in group 1 FENO was 42 ± 23 ppb at visit 2, 34 ± 16 ppb at visit 5, and 63 ± 35 ppb at visit 6 (p = 0.03), whereas in group 2 FENO was 56 ± 37 ppb at visit 2, 100 ± 69 ppb at visit 5, and 128 ± 87 ppb at visit 6 (p = 0.009). Interestingly, the alveolar NO concentrations remained stable in group 1 throughout the study (5.1 ± 2.9 ppb at visit 2, 4.1 ± 2.6 ppb at visit 5, and 5.7 ± 3.6 ppb at visit 6; p = 0.11). On the other hand, the CANO doubled during the pollen season with respect to the preseason values in group 2 (5.0 ± 3.2 ppb at visit 2, 5.4 ± 3.4 ppb at visit 5, and 12.9 ± 9.8 ppb at visit 6 (p = 0.01) (Figure 3(B)).

(A) FENO (forced expiratory nitric oxide) and (B) CANO (concentration of alveolar nitric oxide) concentrations in the expired air in specific immunotherapy (SIT)-treated (closed circles) and SIT-untreated (open circles) groups. *p = 0.009; #p = 0.03, §p = 0.01, analysis of variance (ANOVA) for repeated measures. For further explanation of visits 2, 5 and 6 see the Methods section.
Discussion
The findings of the present study clearly show that, in a selected population of mild asthmatics, an ultra-short course of preseasonal immunotherapy contributes to maintain control of asthma symptoms during pollen season. The current observations suggest that the clinical benefit provided by the vaccine could be mediated by anti-inflammatory effects, which mainly occurred at the level of peripheral airways, as indicated by the unchanged alveolar NO concentrations during pollen season in the treated subjects. The finding of preserved HR-QoL both in treated and untreated subjects advocates for larger studies specifically aiming at assessing the role of preseasonal SIT on quality of life.
The preseason vaccine used in this study consists of natural allergens chemically modified with glutaraldehyde and adsorbed on L-tyrosine, to ensure a slow release of active ingredient, to reduce the possibility of having immediate and potentially dangerous reactions, therefore resulting in greater desensitization and higher tolerability. In addition, the immune response is enhanced and directed by adjuvant MPL, which is derived from the cell wall lipopolysaccharide of Salmonella minnesota R595 (Gram negative), and chemically detoxified, thus promoting the Th1 response [Hopkins et al. 2001]. Puggioni and colleagues demonstrated in vitro that MPL promotes immune deviation of allergen-induced peripheral Th2-cell responses in favor of protective Th1 responses [Puggioni et al. 2005]. We recently showed that, in asthmatics, the ultra-short course of preseasonal vaccine for Parietaria with MPL exerts anti-inflammatory effects, as documented by the significant reduction in the levels of 8-isoprostane in the exhaled breath condensate [Scichilone et al. 2011], which appeared to be maintained during and after pollen season. It is logical to speculate that the addition of MPL would account for the anti-inflammatory effects that were observed in the current study. However, the lack of a control group receiving vaccine with no MPL does not allow us to draw definite conclusions.
The clinical benefit of preseasonal vaccine are based on recent observations and mostly related to rhinocongiuntivitis symptoms [Drachenberg et al. 2001]. A significant effect of ultra-short course of immunotherapy to grass over placebo in relieving rhinocongiuntivitis allergic symptoms was shown in the largest controlled study of allergen-specific immunotherapy conducted to date [DuBuske et al. 2011]. Subjects receiving the four injections of MPL vaccine achieved a significant 13.4% benefit during the peak of the pollen exposure. Data regarding the beneficial effect on asthmatic symptoms are scarce and can be extrapolated by the observational study of Musarra and colleagues [Musarra et al. 2010], who followed patients with asthma allergic to Parietaria and subjected to MPL SIT. The authors found that, after 5 years of discontinuing the vaccine, symptoms of asthma were still low, whereas they doubled in the control (untreated) group. The lack of a placebo-controlled group in the current study suggests that the findings related to the clinical improvements should be interpreted with caution.
Taken together, these studies led us to investigate whether a short course of vaccine adjuvanted with MPL, already shown to be well tolerated and efficacious in allergic rhinitics, can also provide beneficial effect on lower airways symptoms. To this aim, we employed the ACT questionnaire, which has been largely used in clinical trials attempting to optimize the level of asthma treatment. The results of the current study demonstrate that asthma control can be maintained by preseasonal immunotherapy. In this regard, it should be noted that our study population consisted of asthmatics with mild forms of the disease, and therefore our findings cannot be extrapolated and applied to subjects with more severe asthma.
Interestingly, the clinical benefit paralleled the anti-inflammatory effect of SIT during pollen season, as demonstrated by the lack of increased markers of inflammation in the expired air. Evaluation of NO in exhaled air has been proposed as a valuable clinical tool for asthma diagnosis and monitoring. A relationship between exhaled NO and clinical signs and symptoms of asthma has been shown in different studies [Alving et al. 1993; Mahut and Delclaux, 2011; Caudri and de Jongste, 2010; Tseliou et al. 2010; Perez-de-Llano et al. 2010; Sandrini et al. 2010]. Recently, Papakosta and colleagues performed a study on asthmatic patients in order to evaluate the relationship between ACT and the degree of airway inflammation, by means of exhaled NO concentrations, before and after treatment with inhaled corticosteroids [Papakosta et al. 2011]. The study demonstrated an improvement in lung function and ACT scores and a parallel decrease in NO levels in the expired air. With regard to the effect of SIT, Cevit and colleagues conducted a study of asthmatic children allergic to dust mites and subjected to immunotherapy subcutaneously [Cevit et al. 2007]. The study showed a reduction in plasma levels of NO after 1 year, possibly reflecting a reduction in systemic allergic reaction. In patients with house dust mite asthma, an incremental schedule of autologous autovaccine with Escherichia coli showed a significant effect on bronchial inflammation as expressed by a reduced increase of exhaled NO [Rose et al. 2011]. It is not surprising that changes in markers of airway inflammation were associated with improvements in asthma control, rather than in lung function. Indeed, noninvasive assessment of inflammation has been used to identify patients with uncontrolled asthma [Profita et al. 2006] and at risk of exacerbation [Gagliardo et al. 2009]. On the other hand, lung function was in the normal range; therefore, any significant correlation with indices of airway inflammation would be unlikely.
In recent years, the contribution of the alveolar component of NO levels has been investigated [van Veen et al. 2006; Paraskakis et al. 2006; Kelly, 2010]. This observation emphasizes the role of alterations in the peripheral bronchial district on the quality of life of patients with asthma and rhinitis, which is impaired to varying degrees in the absence of traditional physiological or biological functional alterations. As also inferred in several studies, the peripheral airways in asthmatics are the site of an inflammatory infiltrate that is not observed in nonasthmatic controls, and, within asthmatic lungs, is more intense than in the central airways [Hamid et al. 1997]. In a study conducted in consecutive asthmatic children [Puckett et al. 2010], increased levels of CANO were related to poor asthma control independent of lung function, atopic status, or use of inhaled corticosteroids. The finding that CANO increases in uncontrolled asthmatics supports the observations that the site of inflammation might change with the disease severity, becoming more prevalent and clinically important in the periphery of the airways. In this context, the effect of SIT on alveolar NO levels is intriguing. The most plausible explanation for this observation is that the anti-inflammatory contribution of SIT, which is administered systemically, occurs primarily in the periphery of the lung. In this scenario, it is unclear whether the anti-inflammatory effect is specifically due to the properties of the adjuvant that is incorporated in the vaccine. Additional studies, specifically designed to investigate these mechanisms, are required.
We did not document any adverse effect related to the administration of the vaccine. The safety and tolerability of short-term specific immunotherapy with pollen allergoids adjuvanted by MPL has been largely demonstrated [Baldrick et al. 2002a, 2002b; Rosewich et al. 2010]. In a prospective open study [Rosewich et al. 2010], a large cohort of juvenile patients with rhinitis, conjunctivitis and/or asthma received four preseasonal injections with pollen allergoids formulated with MPL over a minimum of 3 weeks. Response to treatment was assessed as good or very good in the majority of patients. The safety of the ultra-short term SIT with pollen allergoids adjuvanted with MPL was recently confirmed in an Italian survey, which was conducted prospectively for a 3-year period on a total of 510 adult individuals with seasonal rhinitis and/or asthma [Crivellaro et al. 2011].
The study has some limitations. We acknowledge that the small number of subjects may have influenced the outcomes. For example, the lack of difference between groups with respect to parameters reflecting quality of life could be simply due to this factor. However, the study was exploratory in nature, and was intended as preliminary to larger studies with the attempt to select those biological markers and clinical variables that could be employed when assessing the effect of preseasonal immunotherapy.
In conclusion, we have shown for the first time that an ultra-short course of preseasonal immunotherapy provides clinical benefit in mild asthmatics, by maintaining symptom control during pollen season. Our findings suggest that the effect of SIT on asthma control could be mediated by anti-inflammatory effects on peripheral airways. Further studies are needed to specifically explore the mechanism behind these observations.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflict of interest.
