Abstract

Introduction
Isopropyl alcohol (IPA) is a chemical commonly used in a variety of household and commercial applications. Clinical data on the effects of acute IPA toxicity in humans are well described. However, data on the effects of chronic IPA exposure on the human respiratory system is lacking. The following is a case report of a 63-year-old woman presenting with bilateral lung infiltrates, hemoptysis, and respiratory failure in the setting of chronic IPA inhalation.
Case report
This patient is a 63-year-old woman transferred from an outside hospital with complaints of hemoptysis, acute respiratory failure, and bilateral pulmonary infiltrates on chest imaging. Pulmonary consultation was obtained.
The patient had no significant past medical history or previously known pulmonary conditions. She was admitted to an outside hospital 7 days prior to transfer complaining of 1 week of increasing dyspnea and hemoptysis. She denied fever, chills, chest pain, nausea, or vomiting. She denied sick contacts, recent travel, or exposure to tuberculosis. At presentation to the other facility her oxygen saturation was 88% on room air, blood pressure was 121/72 mmHg, heart rate was 88 beats/min, temperature was 98ºF and was noted to be in respiratory distress. Chest imaging revealed bilateral infiltrates (Figure 1), and she was started on broad-spectrum antibiotics (vancomycin, azithromycin, and ceftriaxone) for a presumed pulmonary infection; however, on hospital day 3 the patient’s respiratory status deteriorated with worsening hypoxemia and increasing oxygen requirement, and she was intubated for impending respiratory failure. Echocardiogram obtained at that time showed a left ventricular ejection fraction of more than 70%, no valvular or wall motion abnormalities, and a small pericardial effusion. Pulmonary artery pressure was unable to be estimated. An autoimmune process was suspected: but serum studies including antinuclear antibodies, rheumatoid factor, and antineutrophil cytoplasmic antibodies were negative. Due to an unclear etiology, she underwent right video-assisted thoracotomy (VATS) and right lung biopsy. VATS revealed significant and diffuse pleural adhesions involving the entire pleural cavity with the entire lung adhered to the pleural wall. Bronchoscopy performed at the same time showed unremarkable mucosa with some bloody secretions throughout the subsegmental bronchi. Ceftriaxone was changed to meropenem, and both vancomycin and azithromycin were discontinued as the respiratory cultures grew rare Pantoea species that were sensitive to meropenem. No acid fast bacillus, fungal, or yeast elements were seen on smear. No adenovirus, influenza A or B, parainfluenza virus, or respiratory syncytial virus were isolated. After the procedure, the patient developed intermittent a-v block mobitz type I that was thought to be due to hypoxemia and was managed conservatively. A right heart catheterization showed a pulmonary artery pressure of 38/23 mmHg with a pulmonary capillary wedge pressure of 22 mmHg.

Chest X-ray at initial presentation in a patient with chronic isopropyl alcohol inhalation injury. Chest X-ray 2 days postadmission to the outside hospital showed extensive bilateral lung infiltrates predominantly involving the lower lobes.
The patient was transferred for a higher level of care to our institution. Physical examination at presentation showed an obese female (body surface area = 1.86 m2), sedated, mechanically ventilated (day 6) via a 7-mm oral endotracheal tube, afebrile, heart rate was 57 beats/min, blood pressure was 156/70 mmHg, orally intubated with assist control volume targeted ventilation with a rate of 12 breaths/min, tidal volume of 500 ml, positive end-expiratory pressure (PEEP) of 10 cm H2O with FIO2 of 60% and oxygen saturation of 98%. Arterial blood gases showed a pH of 7.41, PCO2 of 42 mmHg, PO2 of 85 mmHg, and oxygen saturation of 96%. A right-sided chest tube and a right subclavian central line were in place. Her chest auscultation revealed bilateral breath sounds, bibasilar crackles with no wheezing. Abdominal examination was benign and bilateral peripheral edema was noted. Laboratory examination showed hemoglobin of 8.9 g/dl (12–16 g/dl), white blood cells were 11.7 k/μl (4.5–11.0 k/μl), platelets were 159 k/μl (150–400 k/μl), international normalized ratio (INR) was 1.4, creatinine of 0.9 mg/dl (0.5–1.5 mg/dl), sodium was 145 mEq/l (135–148 mEq/l), potassium was 4.9 mEq/l (3.5–5.0 mEq/l), HCO3 was 29 mEq/l, and an anion gap of 11. Amylase was 38 U/l (30–115 U/l), and lipase was 64 U/l (23–300 U/l). Lactic acid level was 1.4 mmol/l (0.5–2.2 mmol/l). Liver function tests were within normal limits except for lactate dehydrogenase (LDH) which was 757 U/l (300–600 U/l). Reticulocyte count was 3.6% (0.5–2.1%), haptoglobin was 132 mg/dl (31–197 mg/dl). Iron was 61 µg/dl (40–160 µg/dl), iron binding capacity was 290 (260–460 µg/dl), percentage saturation was 21%, ferritin was 197 ng/ml (38–384 ng/ml). Vitamin B12 and folate were normal. Serum osmolality was not measured. Repeat collagen vascular work-up showed antinuclear antibody was not detected, double-stranded DNA was not detected, rheumatoid factor <20 IU/ml, antineutrophil cytoplasmic antibody <20, Jo-1 antibody was 0, scleroderma antibody was 0, histone antibody was 0, cardiolipin IgG and IgM were within normal limits (WNL), antiglomerular basement membrane antibody (IgG) was negative (<1.0), anti-SSA and anti-SSB were WNL, and complement levels were normal. HIV was negative. Sedimentation rate was 18 mm/h (0–20 mm/h), and C-reactive protein was 0.52 mg/dl (0.0–1.0 mg/dl). Chest X-ray and computed tomography of the chest corroborated extensive infiltrates throughout both lungs (Figures 1, 2, and 3). A small pneumothorax was seen on her chest imaging post-VATS procedure and was conservatively managed. After 48 hours of having no significant air leak or chest tube drainage, her chest tube was removed. Broad spectrum antibiotics were continued. Repeat cultures remained negative.
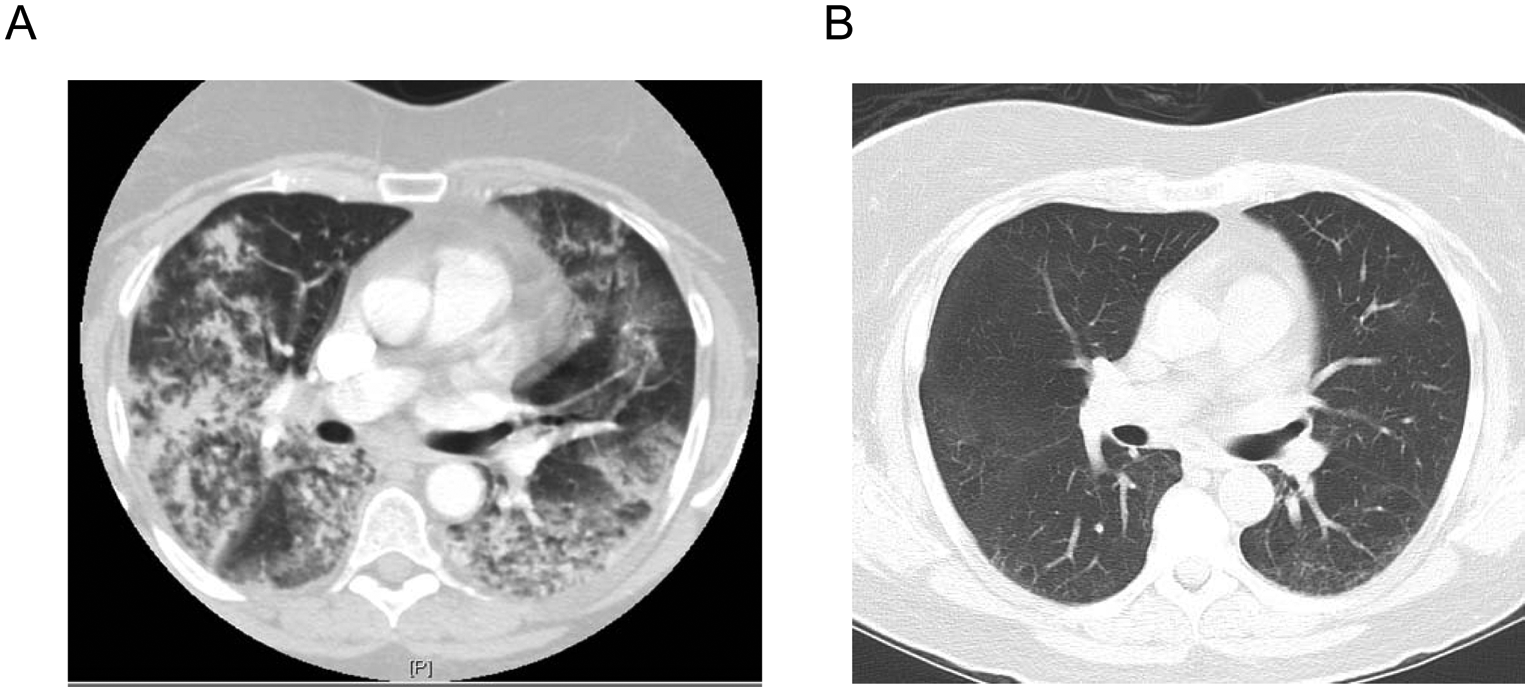
Computed tomography (CT) of the chest at presentation and at follow up in a patient with chronic isopropyl alcohol inhalation injury.

Computed tomography (CT) of the chest at the level of lower lobes.
Reevaluation of her lung biopsy revealed an airway centric pattern of fibrosis and chronic inflammation with scattered lymphoid aggregates (Figure 4). Subpleural fibrosis and intraalveolar hemosiderin-laden macrophages were observed. Histology of her lung biopsy showed obliteration and remodeling of smaller bronchioles with associated prominent peribronchiolar metaplasia (lambertosis). Chronic inflammation with lymphoid aggregation was seen in many of the airways with mature fibrosis with elastosis within interlobular septa and pleura. Some increased pigment-laden macrophages were also noted. These findings were suggestive of chronic inhalation injury and careful history revealed that the patient had been chronically abusing IPA by inhalation multiple times daily for the last 3 years. Typically, the patient would soak a cloth with IPA, place it to her nose/mouth, and inhale it repeatedly.

Lung histology in a patient with chronic isopropyl alcohol inhalation injury following chronic 2-propranol inhalation.
Given her history of chronic IPA inhalation, biopsy findings, and hemoptysis, intravenous solumedrol (40 mg every 6 hours) was started. On day 9 posttransfer, her respiratory status had improved and she was successfully extubated and steroids were continued. She continued to improve; and by hospital day 21 was weaned to nasal cannula (2 l/min) and was discharged to a rehabilitation facility on steroid taper. At 4-week follow up, patient was breathing comfortably on room air with maintenance of oxygen saturation above 90% during a six-minute-walk test. A repeat computed tomography of chest at 5-month follow up showed significant improvement in lung infiltrates and ground glass opacities with the presence of residual subpleural ground glass opacities (Figures 2 and 3).
Discussion
IPA is a common chemical used in a variety of household and commercial applications with frequent human exposure. Although acute effects of IPA inhalation injury have been studied, the effects of chronic subacute exposure is not well known. To the best of the authors’ knowledge, the above case represents the first published case of the histological and clinical presentation of prolonged IPA inhalation injury in a human.
In vivo studies show that IPA is rapidly absorbed through the lung parenchyma into systemic circulation with an absorption efficiency of 85–99% [Earnestgard et al. 2003; Laham et al. 1980; Jammalamadaka and Raissi, 2010]. Systemically, IPA follows first-order kinetics with a half-life of 2.5–4.2 hours. The majority of IPA is metabolized by the hepatic alcohol dehydrogenase to acetone, which has a longer half-life, while a smaller portion is excreted unchanged by the kidneys or lungs. Symptoms of acute IPA intoxication involve central nervous system (CNS) symptoms and include slurred speech, headache, dizziness, confusion, muscle incoordination, and even coma. Gastrointestinal symptoms include nausea, vomiting, and hematemesis. Laboratory evaluation generally yields a high osmolar gap. Serum acetone levels will be elevated and IPA levels can be measured directly if gas chromatography is available. Serum IPA levels and osmolar gap were not measured in our patient as she was transferred after 1 week of her initial presentation and clinically did not show signs of acute ingestion, rather an inhalation injury. Acute IPA intoxication is thought to occur secondary to either direct toxicity from IPA itself or its metabolite acetone, but controversy exist to which of these substances may be the primary agent causing toxicity [Chan et al. 1993].
In vivo rat studies of acute IPA inhalation show severe irritation of mucous membranes with subsequent development of ataxia, prostration, and narcosis in a dose–time-dependent manner with rapidly increasing systemic levels of IPA and its metabolite acetone [Laham et al. 1980]. Histological evaluation of these animals after acute exposure showed extensive pneumonitis and lung edema. In addition, edema throughout the CNS and foamy vacuolization of liver cells was described [Laham et al. 1980]. Human data on toxic inhalation of IPA show cerebral edema and CNS toxicity [Vicas and Beck, 1993]. Presentation of acute IPA inhalation injury reported to be similar to the ingestion injury likely secondary to the rapid/efficient absorption of IPA through the lung parenchyma [Vicas and Beck, 1993]. Furthermore, human studies show that women may be more susceptible to the toxic effects of inhaled IPA [Earnestgard et al. 2003].
The histologic sequelae of chronic IPA inhalation is not well known. Health hazard reports of chronic inhalational exposure reported symptoms of difficulty concentrating, dizziness, cough, and chest pain [Dhillon and von Burg, 1995]. The patient in the above case was chronically abusing IPA by inhalation over a 3-year period to soothe her nerves and help her sleep. Of note is that the patient’s respiratory cultures grew a Pantoea species. Pantoea, a genus of the Enterobacteriales family, has been known to be associated with both organic dust toxic syndrome as well as routine exposure linked to extrinsic allergic alveolitis (hypersensitivity pneumonitis) in chronically exposed agricultural workers [Milanowski et al. 1998]. However, we felt that our patient’s histological findings were more consistent with a chronic inhalational exposure rather than hypersensitivity pneumonitis for two reasons. First, pathology in hypersensitivity pneumonitis typically shows lymphocytic, macrophage, plasma cell infiltration in the alveolar wall and interstitium with noncaseating granulomas whereas in our patient no granulomas were identified and lymphocytic infiltration was predominantly in a peribronchiolar pattern. Second, our patient had no identifiable risk factors for chronic Pantoea exposure was able to return to previous habitat and was weaned off steroids without recurrence of these symptoms. One would expect that if chronic Pantoea-associated extrinsic allergic alveolitis was the culprit then the patient’s symptoms would recur with steroids taper and on return to her previous environment. It is not clear whether the chronic IPA inhalation predisposed her to Pantoea infection.
In the above case, we chose to treat our patient with steroid therapy. Little data is available regarding the role of steroids in the treatment of either acute or chronic inhalation injury. Systemic steroids have previously been suggested as a possible treatment of acute inhalation injury from smoke inhalation and burn injuries [Beal et al. 1968; Greenhalgh, 2009]. However, its role in both acute chemical inhalation injury and chronic inhalation exposure is not well known. In our patient, histological examination of the lungs was supportive of a chronic toxic exposure with lymphocytic infiltration in airway-centric distribution; therefore, steroids were a reasonable choice of therapy. Further studies on the true efficacy of steroids in both acute and chronic inhalation injury are warranted. An interesting finding was the presence of extensive pleural adhesions noted at the time of lung biopsy. The pathophysiology of these pleural adhesions in relation to her chronic IPA inhalation is not clear.
Conclusions
IPA inhalation exposure in the acute setting shows rapid systemic absorption and appearance of its metabolite acetone. Chronic IPA exposure is less well understood. The above case may represent the histological outcome of chronic IPA inhalation injury in an adult, most notable for chronic airway-centric chronic inflammation and fibrosis. In this case, the patient was successfully treated with steroid therapy and follow-up imaging showed almost complete resolution of chest imaging abnormalities. The required time course for the resolution versus persistence of these imaging and histological abnormalities in the setting of a previous chronic exposure is unclear. IPA inhalation injury should be considered in the setting of acute respiratory failure and hemoptysis.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
