Abstract
Introduction
Severe exacerbations in patients with chronic obstructive pulmonary disease (COPD) and/or emphysema are a major cause of hospitalization and can accelerate the loss of lung function. As a consequence, patients’ quality of life is negatively affected and there is increased medical expenditure [Arnedillo, 2010]. A COPD exacerbation can be life-threatening, and most often follows an infection that drives neutrophilic inflammation and proteinase release [Silverman et al. 1989]. COPD exacerbations occur commonly in patients with a deficiency of the protein alpha-1 antitrypsin (AAT) that inactivates proteolytic enzymes in pulmonary tissue [Kohnlein and Welte, 2008]. The predominant normal genotype encoding for AAT is MM (PiMM) while more than 95% of subjects with severe AAT deficiency (i.e. AAT serum levels below 11 μmol or 50 mg/dl) are homozygous ZZ (PiZZ) [de Serres, 2002].
Augmentation therapy, also called replacement therapy, with AAT concentrates is focused on correcting the deficiency state of patients, thus restoring enzymatic control over human neutrophil elastase [Gadek et al. 1981], and therefore preventing or slowing further destruction of lung tissue [Seersholm et al. 2000; The Alpha-1-Antitrypsin Deficiency Registry Study Group, 1998]. Chronic augmentation therapy has been shown to effectively raise serum and lung levels of AAT in deficient subjects [Barker et al. 1994; Gadek et al. 1981; Soy et al. 2006; Tirado-Conde et al. 2008; Wewers et al. 1987] and is well tolerated [Hubbard et al. 1988; Vidal et al. 2010]. Furthermore, augmentation therapy has been reported to lower the likelihood of admission to hospital due to severe exacerbations [Dirksen et al. 2009].
All of these findings have contributed to the growing body of evidence in support of the benefits of AAT augmentation therapy. As a consequence, appropriately powered placebo-controlled clinical trials are becoming difficult to carry out due to ethical reasons. Therefore, the need for retrospective studies to confirm the outcomes of augmentation therapy is clear. In this study, the protective effect of chronic AAT augmentation therapy on the reduction of exacerbations, as well as its effects on hospitalization costs, were retrospectively assessed in Spanish AAT-deficient patients.
Materials and methods
Patients
Patients treated by individual physicians were identified in the pneumology departments of 41 hospitals in Spain. Individuals fulfilling the following selection criteria were included in the study: (1) both men and women older than 18 years of age; (2) diagnosed with severe AAT congenital deficiency (i.e. PI*ZZ genotypes and combinations of Z, rare and null alleles expressing AAT serum concentrations <11 μmol or 50 mg/dl); (3) who had been receiving continuous augmentation therapy with Trypsone® (Grifols) or Prolastin® (Talecris) during a minimum of 18 months before being included in the study; and (4) with available medical records of 18 months before starting augmentation therapy.
All subjects provided written informed consent to participate in the study with the exception of those deceased, whose data were maintained under confidentiality according to the Spanish Law on the protection of existing data (Ley Orgánica 15/1999 de Protección de Datos).
Study design and objectives
In this multicentre, observational, retrospective study, the medical records of identified patients were manually examined by investigators. Sociodemographic, clinical and study variables were recorded in an electronic Case Report Form (eCRF).
The study was conducted in accordance with the Declaration of Helsinki of the 18th World Medical Assembly, Good Clinical Practice Guidelines and was subjected to the Spanish Medicines Agency rules for communication in pharmacovigilance of medications for human use (Agencia Española del Medicamento, Circular 15/2002). The study protocol was approved by the Institutional Review Board (IRB) of all centres to ensure ethical research practices.
The main objective of the study was to assess whether augmentation therapy with AAT concentrate in patients with AAT deficiency was associated with a reduction of both the number of exacerbations and the percentage of patients experiencing exacerbations. This was explored by comparing the 18 months before and the months after starting augmentation therapy. Two populations of patients were considered: (1) those who had already suffered at least one exacerbation in the 18 months previous to the starting of augmentation therapy; and (2) those who had not. Two types of exacerbations were considered: severe and nonsevere (mild).
Exacerbation was defined as a worsening of the basal condition of the patient, with a change in dyspnea, coughing and/or basal expectoration of the patient beyond the daily variability, which required a change of the patient’s COPD medical regimen for optimizing inhalation therapy to achieve maximal bronchodilator and anti-inflammatory effect [Global Initiative for Chronic Obstructive Lung Disease (GOLD), 2010; Sociedad Española de Neumología y Cirugía Torácica - SEPAR-ALAT, 2009]. A severe exacerbation was defined as one that required hospital admission [Burge and Wedzicha, 2003; Rodriguez-Roisin, 2000].
Secondary objectives were: first, to assess whether augmentation therapy is associated with improvement of respiratory function in patients with AAT deficiency; second, to assess whether augmentation therapy is safe; and, third, to assess whether augmentation therapy results in a reduction in hospitalization costs as a result of a decrease in severe exacerbations.
Studied variables and assessments
The number of exacerbations per patient, both severe and mild, was the primary variable. Changes in the number of exacerbations and in the percentage of patients experiencing exacerbations in the pre-augmentation and postaugmentation periods were measured and analysed statistically.
Collected demographic and medical history data included age, sex, weight, height, body mass index (BMI), smoking habits, occupational exposure to toxins, comorbid conditions, concomitant medications for COPD, and augmentation therapy data (dates, doses, changes). Laboratory data included phenotype, genotype if available, lung function parameters (forced expiratory volume in one second [FEV1], forced vital capacity [FVC], FEV1/FVC, carbon monoxide transfer function [TLCO], carbon monoxide transfer coefficient [KCO]), arterial blood gas measurements, pulse oximetry, bilirubin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), glucose, albumin, prothrombin time, and serum AAT level.
Evolution of patients’ lung function parameters in the 3-year span of the study was compared with that of healthy subjects of the same demographic characteristics (age, sex, height and weight) following the calculations provided by the Spanish Society of Pneumology and Thoracic Surgery (Sociedad Española de Neumología y Cirugía Torácica [SEPAR]) for the normal values of Spanish population [Sanchis et al. 1989].
Data on adverse events (AEs) during the study period were also collected.
Cost assessment estimates were based on the requirement for inpatient hospital care. Factors affecting the total cost included the location of care within the hospital (ICU, ward), the cost of 1 day in the ICU, cost of 1 day in a ward, and the clinical testing performed. It was assumed that, in case of exacerbation, half of patients received their initial care in a primary care setting, and the other half went to a hospital emergency unit [Martínez Sanz and Pérez Maroto, 2004]. It was further assumed that spirometry, arterial blood gas tests, chest radiography, electrocardiograms, and blood analysis were carried out. Basal costs were estimated according to a previous economic study [Martínez Sanz and Pérez Maroto, 2004] using a correction update to the annual increase in prices in Spain [Instituto Nacional de Estadística, 2010].
Statistical analysis
Sample size was determined based on the primary endpoint (number of severe exacerbations in the 18 months before and after the start of augmentation therapy). Accepting an alpha risk of 5% and a beta risk of 20% in a two-tailed assessment, 71 subjects were required to detect a difference equal to or exceeding one additional severe exacerbation assuming a standard deviation of 3 severe exacerbations (common to both 18-month periods, before and after the start of augmentation therapy). Sample size estimation was based on the Student’s t-test for independent random sampling. Estimation was corrected for Wilcoxon paired samples analysis (assuming that Wilcoxon asymptotic efficiency, relative to the t-test, never falls below 0.95) [Siegel and Castellan, 2000]. Therefore, it took a minimum of 75 patients. However, we recruited all patients that accomplished selection criteria at the study sites (127 patients), to avoid selection bias and to increase the power of the analysis.
To analyse the effect of possible confounding factors (those associated with changes in the physician’s clinical practice and those related to the evolution of the patient status), a multivariate logistic regression analysis was used. Possible confounding factors were preselected by means of the Spearman nonparametric correlation (r > 0.1 for those variables associated with the treatment effect) and McNemar test (p < 0.2 between before and after starting treatment for those variables associated with the presence of treatment). Preselected variables were included in a logistic model with an intrasubject factor (presence of treatment). Confounding was identified as a change in intrasubject effect odds ratio (OR) greater than 10% as compared with the full model [Maldonado and Greenland, 1993]. To avoid selection bias, patients without exacerbations in the first study period were included in the analysis.
Continuous variables are represented as mean and standard deviation (SD) unless otherwise indicated to be standard error of the mean (SEM) or 95% confidence interval (CI). Where appropriate, minimum and maximum are shown. For categorical variables, absolute and relative frequency in percentage are presented. Chi-square test or Fisher’s exact test were used to compare proportions. The Student’s t-test and analysis of variance (ANOVA) were used for comparison of means in the case of normality, or the Mann–Whitney–Wilcoxon and Kruskal–Wallis tests in the case of nonnormality. Pearson’s correlation coefficient was used to analyse the correlation between two quantitative variables, or the Spearman correlation coefficient in the case of nonnormality.
The statistical packages SPSS 17.0, STATA 9.1 and R 2.9.1 were used for statistical analyses, excepting sample size calculations for which the software GRANMO 5.2 [Marrugat et al. 1998] was used.
Results
Population characteristics
A total of 127 patients were included in the study, and initiated augmentation therapy between June 1990 and October 2008. The flow of patients through the study and data available for analysis are shown in Figure 1. Of the 127 patients, 75 of them (59.1%) presented with at least one exacerbation in the period prior to treatment and 35 (27.6%) experienced at least one severe exacerbation.

Flow of patients and data available for analysis through the study (AAT, alpha-1-antitrypsin; AT, augmentation therapy)
Main baseline sociodemographic and clinical characteristics of patients are shown in Table 1. There were no statistically significant differences in the sociodemographic characteristics between the total population and the population of patients experiencing exacerbations. The percentage of comorbidities was found to be consistently higher in patients experiencing exacerbations, although this difference was not statistically significant.
Main baseline sociodemographic and clinical characteristics of patients.

SD, standard deviation; BMI, body mass index
Augmentation therapy profiles
In the total population, 68 patients (53.5%) received augmentation therapy with Prolastin® and 59 (46.5%) received Trypsone®. The distribution was very similar in the population with exacerbations: 42 (56%) and 33 (44%) patients, respectively.
In the total population, eight patients (6.3%) were administered augmentation therapy weekly, 22 patients (17.3%) biweekly and 97 patients (76.4%) every 3 weeks. The average AAT concentrate dose administered was 60.7 ± 3.8 mg/kg/week. In the population with exacerbations, regimens were similar and average dose was almost identical (60.6 ± 3.3 mg/kg/week). There were no differences in treatment pattern between the patients treated with Prolastin® and those treated with Trypsone®.
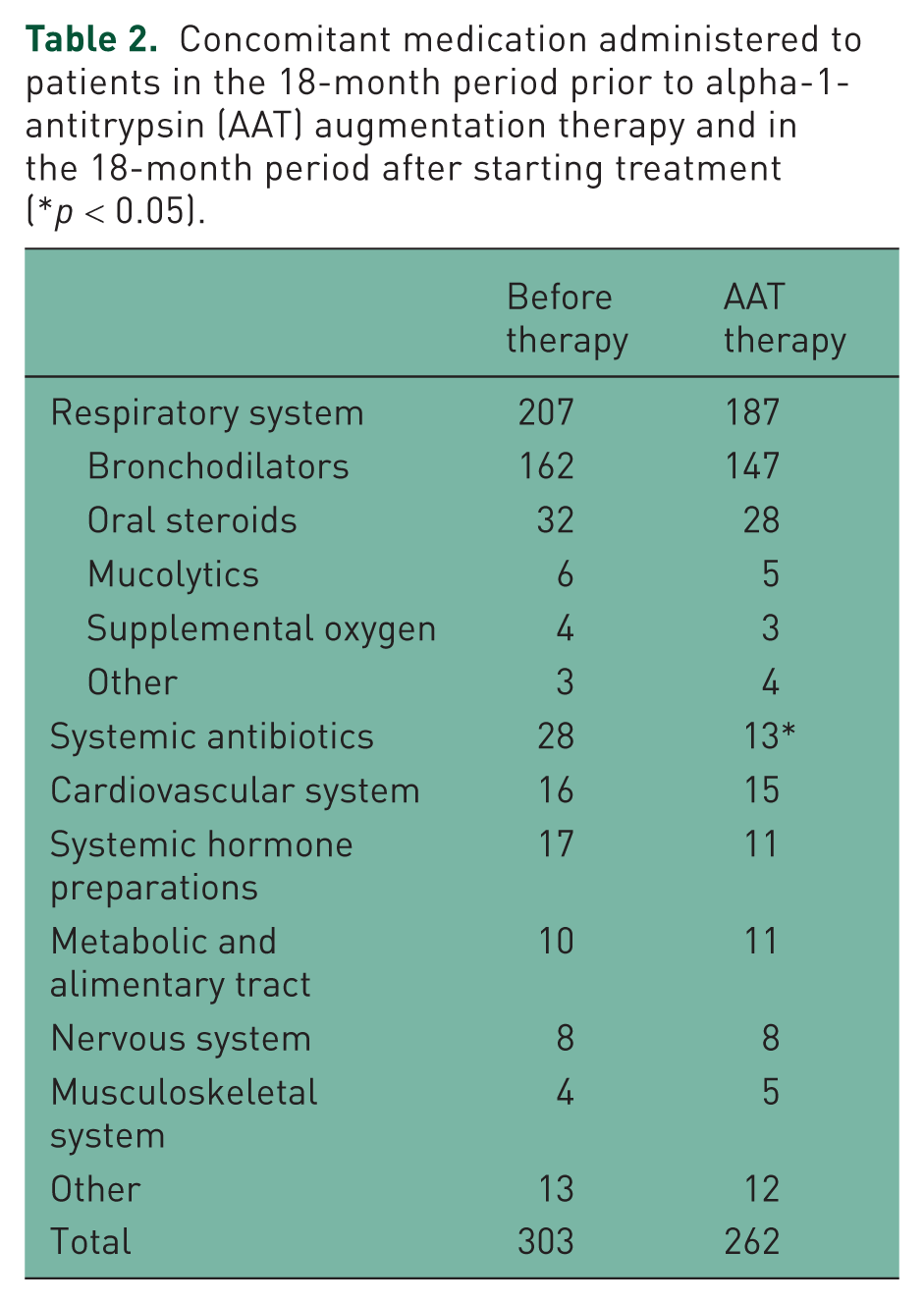
The most commonly administered baseline medications (n = 303) were respiratory treatments (68.3%) and systemic antibiotics (9.2%). Concomitant medications were in general less frequently administered in the 18-month period while on AAT treatment, although the reduction was statistically significant only in systemic antibiotics (n = 13, 31.7% versus n = 28, 68.3%; p < 0.05). Full results of concomitant medications are shown in Table 2.
Concomitant medication administered to patients in the 18-month period prior to alpha-1-antitrypsin (AAT) augmentation therapy and in the 18-month period after starting treatment (*p < 0.05).
Incidence of exacerbations
The mean number of exacerbations per patient in the total patient population was reduced from 1.2 ± 1.6 in the 18 months prior to augmentation therapy with AAT to 1.0 ± 2.2 in the 18 months after starting treatment (p < 0.01), and from 2.0 ± 1.6 to 1.4 ± 2.7 (p < 0.01) in the population with exacerbations. The number of severe exacerbations, although also reduced, did not change significantly. These data are summarized in Figure 2.

Effect of alpha-1-antitrypsin (AAT) augmentation therapy on patients’ number of exacerbations observed in the18-month period prior to therapy (AAT) augmentation therapy and in the 18-month period after starting treatment. Results are presented as mean ± SEM (*p < 0.01).
By considering the difference between means, augmentation therapy was associated with a reduction of 2.4 exacerbations (95% CI 0.7–5.6) for every 10 treated patients in the total population (meaning 2.8 patients to be treated to prevent one exacerbation over 1 year), and with 6.3 exacerbations (95% CI 1.2–11.4) for every 10 treated patients in patients with previous exacerbations (meaning 1.1 patients to be treated to prevent one exacerbation over 1 year).
When the percentage of patients experiencing exacerbations was calculated, the value was significantly reduced in the total patient population after starting augmentation therapy (59.1% before treatment versus 44.1% during treatment; p < 0.005) whereas in patients with severe exacerbations the difference was not statistically significant (46.7% versus 55.4%, respectively).
In the total patient population, 43.7% of patients improved (had fewer exacerbations) after starting treatment versus 21.4% of patients worsened (had more exacerbations) (p < 0.01). When only severe exacerbations were considered, 42.9% of patients improved versus 12.0% worsened (p < 0.001). In the population with exacerbations in the pretreatment period, 42.9% of patients improved with regard to severe exacerbations versus 15.0% of patients worsened (p < 0.001). These results are summarized in Figure 3.
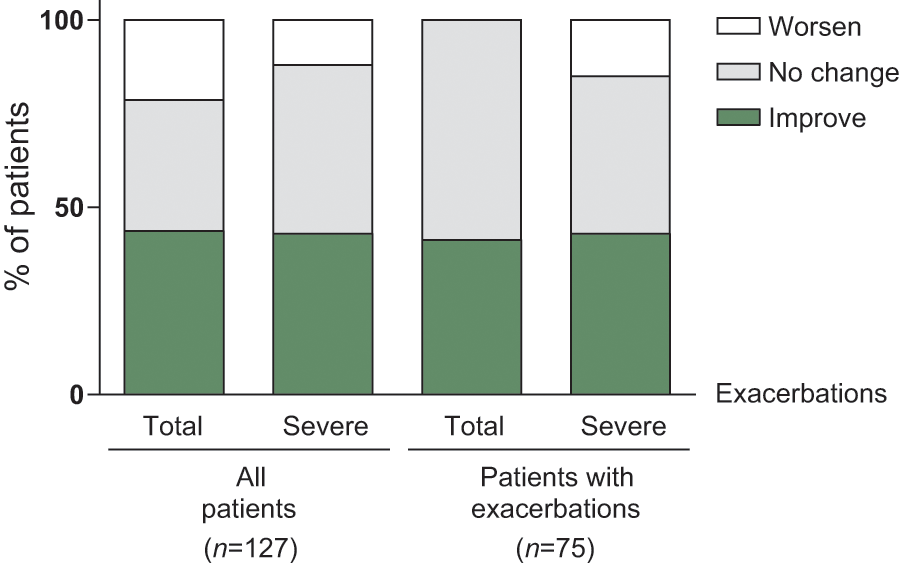
Percentages of patients who had fewer (improve), more (worsen) or unchanged exacerbations in the 18-month period after starting treatment compared with the 18-month period prior to AAT augmentation therapy.
Multivariate analysis to elucidate the real effect of treatment on the incidence of exacerbations revealed that there were fewer recorded values in the analytical variables (lung function and laboratory) in the period after starting treatment (n = 104 observations, 52 patients × 2 periods) as compared with the pre-augmentation period (n = 254 observations, 127 patients × 2 periods). Intrasubject effect OR with or without imputation of missing values were 2.41 (95% CI 1.38–4.23) and 2.15 (95% CI 1.11–4.14), respectively. The difference was 10.8%, slightly above the 10% limit, meaning that missing values probably caused a small bias in the sense that the observed effect of the AAT treatment was less than the actual effect. Modelling with the imputed values suggests that untreated patients would be expected to be at 1.4- to 4.2-fold more risk of exacerbation than treated patients (p < 0.05).
Lung function
In all studied parameters, average values were lower after the 18-month period of treatment than in the 18-month period prior to treatment (see Table 3). Differences were statistically significant in FEV1 for the total patient population (1.25 l versus 1.19 l; p < 0.05) and FEV1/FVC for the population with exacerbations (45.2 versus 42.2; p < 0.005). However, the decrease of lung function parameters between the first and second periods in the study population was similar to that which can be observed in the normal healthy Spanish population, adjusted for similar age, sex, height and weight, in the same time period (data not shown).
Lung function parameters observed in patients in the 18-month period before AAT augmentation therapy and in the 18-month period after starting treatment. Results are presented as mean ± SD (*p < 0.05; **p < 0.005)

AAT, alpha-1-antitrypsin; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; TLCO, carbon monoxide transfer function.
A further analysis in the total patient population with regard to the change in lung function variables that took place in the first period, compared with change that took place in the second period, revealed that the rate of decline was more marked in the period prior to treatment, being statistically significant for FEV1 (difference of means between first and last analysis in the 18-month period prior to treatment versus the 18-month period of treatment was 0.10 ± 0.09 l versus 0.03 ± 0.03 l; n = 21–36; p < 0.05).
Laboratory parameters
As could be expected, serum AAT levels were higher in the augmentation therapy period with respect to the period prior to treatment: 60.5 ± 52.5 mg/dl in the total population and 61.1 ± 57.1 mg/dl in patients experiencing exacerbations versus 22.4 ± 10.1 mg/dl and 23.2 ± 9.6 mg/dl, respectively (p < 0.001). There were no differences between periods and populations in most of the other studied laboratory parameters (pulse oximetry, bilirubin, ALT, AST, glucose, albumin, prothrombin time, pCO2 and pH). Conversely, pO2 in the first period was 68.7 ± 12.7 mmHg in the total population and 67.6 ± 14.4 mmHg in patients experiencing exacerbations, while in the second period values were, respectively, 65.6 ± 8.5 mmHg and 65.2 ± 7.4 mmHg, slightly but significantly lower (p < 0.01).
Safety
A total of 16 AEs, other than exacerbations, were registered in 11 patients during the study: two of them (in one patient) in the period prior to treatment and 14 in the treatment period. Table 4 summarizes the distribution of adverse events between periods and relevance. Five AEs were serious: the one that took place in the first period was an exacerbation of infectious bronchiectasis. The other four were one massive pulmonary thromboembolism, one myeloid leukaemia, one acute myocardial infarction and one haemorrhagic infarction. None of them was considered to be related to the treatment. There were 11 nonserious AEs. The majority of them were considered to be related to the treatment and were mild/moderate cutaneous or sensitive manifestations such as chills, facial redness, sensation of cold, mild oedema/pruritus, cutaneous exantema, fever and anxiety. Those nonserious AEs considered unrelated to the treatment were obstructive sleep apnoea, urinary infection, hypertension and pneumonia.
Adverse events observed in patients, distributed according to period (18-month period before alpha-1-antitrypsin (AAT) augmentation therapy and in the 18-month period after starting treatment), relevance and their relation to treatment.

Cost assessments
The average number of days of admission to the floor wards for the total population was 3.9 before treatment and 3.0 in the treatment period, while in the population with exacerbations the values were 6.7 and 4.6 days, respectively. There were no admissions to the ICU. Following the calculation parameters established in the methods, the cost of 1 day of care for patients admitted to the ward was estimated as €409.33 and as €200.64 if admission was not required. Using these parameters, the costs before and after starting treatment are summarized in Figure 4. After 18 months of AAT augmentation therapy, hospitalization costs savings per patient were €416.76 per subject in the total patient population (a reduction of 24%, approximately) and €907.05 in the population with exacerbations (a reduction of 31%, approximately), both statistically significant (p < 0.05 and p < 0.001, respectively).

Total cost per patient in relation to the number of days by admission and associated expenditures due to exacerbations. Comparison of the 18-month period before alpha-1-antitrypsin augmentation therapy and the 18-month period after treatment. Results are presented as mean ± SEM (*p < 0.05; **p < 0.001).
Discussion
Augmentation therapy with AAT concentrates is becoming widely accepted as an effective, safe and well-tolerated treatment for AAT deficient patients with COPD and pulmonary emphysema [Barker et al. 1994; Gadek et al. 1981; Hubbard et al. 1988; Soy et al. 2006; Tirado-Conde et al. 2008; Vidal et al. 2010; Wewers et al. 1987]. Results of this retrospective study confirm the effectiveness of chronic augmentation therapy in the reduction of the incidence of severe exacerbations, with the consequence of less hospital admission-derived costs and less administration of concomitant medications.
The clinical benefit of augmentation therapy with AAT was called into question in a 2010 review [Gotzsche and Johansen, 2010], on the basis of evidence from two available clinical studies [Dirksen et al. 1999, 2009]. Nevertheless, the review was later alleged to be flawed and poorly designed [The Alpha-1 Foundation, 2011]. Overall results of our study, focused on exacerbation frequency, are in the line of those supportive of the efficacy of AAT augmentation therapy.
The retrospective nature of our study as well as the absence of a control group (the same subjects, evaluated before and after starting treatment, were their own control) could be considered limitations. However, both study populations (total population and population with exacerbations) presented identical sociodemographic and therapeutic characteristics. This allowed appropriate statistical comparison. Moreover, the action of possible sources of bias and confounding factors on the real effect of augmentation therapy on the incidence of exacerbations was taken into account. The less frequent measurement of analytical variables observed during the treatment period is consistent with the fact that patients in better health tend to have less data collected in their medical records. In addition, it is likely that, after starting augmentation therapy, patients would have enhanced alertness to exacerbations, and hence a trend to register more minor exacerbations in comparison with the period prior to augmentation therapy. Therefore, the conclusions of the study would be reinforced because the decrease of the incidence of exacerbations that was observed after starting AAT therapy would be even more marked.
Average dose and regimen (~60 mg/kg/week) were in accordance to the recommendations of the Spanish Registry of Patients with Alpha-1-antitrypsin Deficiency [Miravitlles et al. 1998]. Most patients were administered infusions on an every 3 weeks regimen, a common practice in Spain which has been described to be feasible to achieve adequate trough total AAT [Soy et al. 2006], while increasing patient’s comfort. In fact, serum AAT levels in the posttreatment period were successfully corrected to >60 mg/dl.
Our results showed that AAT treatment was associated with both a significant reduction in the number of exacerbations in the two populations of the study and in the percentage of patients experiencing exacerbations in the total population. Moreover, the percentage of patients in whom the incidence of exacerbations and severe exacerbations improved was significantly higher than the percentage of patients in whom the incidence did not improve. In contrast, in a randomized, double-blind, placebo-controlled study, exacerbation frequency was found unaltered by treatment in patients with AAT deficiency, while a reduction in exacerbation severity was observed [Dirksen et al. 2009]. However, the authors pointed out that the number of patients included (n = 77) was possibly too small to draw any conclusion on the treatment effect on the rate of exacerbations. Actually, moderate and severe exacerbations occur frequently in subjects with AAT deficiency receiving augmentation therapy and are associated with worse health status [Campos et al. 2009; Needham and Stockley, 2005]. The progression of pulmonary function impairment in AAT-deficient patients [Kohnlein and Welte, 2008] can hinder the analysis of treatment effect in the absence of placebo controls.
In our study, administration of concomitant medications directed to the respiratory system was slightly reduced after starting augmentation therapy. The reduction was significant for use of systemic antibiotics, probably due to the fewer exacerbations after AAT treatment. This would be in accordance with preliminary data reported [Berger et al. 2004], and reinforce the finding that augmentation therapy was associated with a reduction in exacerbations.
Analysis of pulmonary parameters, particularly FEV1 and FEV1 / FVC, confirmed a decline of pulmonary function during the study in spite of treatment, consistent with previous studies [Piitulainen and Eriksson, 1999]. Interestingly, once corrected, the decline was similar to that found in the normal healthy population, caused by the normal evolution of the respiratory function associated with age. This probably indicates that AAT augmentation therapy is able to sustain evolution of lung function of AAT-deficient patients in a profile equivalent to that of normal population. In a recent meta-analysis, small but consistent beneficial effects of augmentation therapy on the decline of FEV1 were found [Chapman et al. 2009].
Safety assessment showed a low number of adverse events, most of them nonserious or not related to treatment. This is in accordance with the previously reported safety profile and tolerability of AAT concentrates [Hubbard et al. 1988; Vidal et al. 2010].
Hospital cost analysis indicated that, in the 18-month period after augmentation therapy, days of hospitalization were fewer in both the total population of patients and in the population with exacerbations. This resulted in hospitalization costs being significantly reduced to about three-quarters to two-thirds of the corresponding costs in the 18-month period before treatment. Nevertheless, the impact of these positive results in a cost-effectiveness assessment of AAT augmentation therapy should be confirmed in more in-depth, long-term, pharmacoeconomic-focused analyses. Other AAT-deficiency-associated parameters beyond the incidence of exacerbations, such early onset of emphysema [Needham and Stockley, 2005] and lung transplantation [Cassivi et al. 2002] should be considered from a cost-effectiveness point of view. These conditions have a significant impact on the patient functional status, and may lead to earlier retirement, thus incurring high indirect costs.
In summary, the results of this study show that AAT augmentation therapy is safe and was associated with a reduction in both severe and mild exacerbations in patients diagnosed with AAT deficiency. Although treatment is not a cure for the disease, it can slow down the progression of pulmonary function to a degree similar to that of the normal population. Moreover, by reducing the hospitalization days, AAT augmentation therapy can help to improve quality of life in these patients while reducing hospitalization-derived costs.
Footnotes
Acknowledgements
The authors are grateful to all colleagues and researchers from the centres that participated in the rEXA study: Dr Carmen Montero Martínez (Hospital Juan Canalejo); Dr Esther Rodríguez and Dr Rafael Vidal (Hospital Vall d’Hebrón); Dr Gema Tirado-Conde (Hospital Clínic i Provincial de Barcelona); Dr Ignacio García-Talavera (Hospital Nuestra Señora de la Candelaria); Dr Miriam Calle Rubio (Hospital Clínico San Carlos); Dr Francisco Javier Domínguez del Valle (Hospital Navarra); Dr Ana Bustamante (Hospital Sierrallana); Dr Adolfo Baloira Villar (Complejo Hospitalario de Pontevedra); Dr Maria José Mejuto Martín (Hospital Arquitecto Marcide); Dr Santiago Ramos Rodriguez (Hospital Central de Asturias); Dr Olga Rajas (Hospital de la Princesa); Dr Enrique Alonso Mallo (Hospital Rio Carrión); Dr Pilar Berlinches (Hospital Santa María Rosell); Dr Pedro Marcos Velásquez (Complejo Hospitalario Ourense); Dr Xavier Aguilar (Hospital Joan XXIII ); Dr Francisco Casas Maldonado (Hospital Clínico Santa Cecilia); Dr Javier García (Hospital Nuestra Señora del Prado); Dr Hortensia Cantos (Hospital Valle del Nalón); Dr Bautista Galdiz Iturri (Hospital de Cruces); Dr Vicente Roig Figueroa (Hospital Clínico Universitario de Valladolid); Dr Ibrahim Hamad (Hospital Gutiérrez Ortega); Dr Isabel Parra Parra and Dr Francisco José Ruíz López (Hospital Virgen Arrixaca); Dr María Dolores Corbacho (Hospital Povisa); Dr Luís Pérez de Llano (Hospital Xeral-Calde); Dr Jose Antonio Gutiérrez Lara (Hospital Infanta Cristina); Dr Jose Maria Hernández (Hospital La Palma); Dr Andrés Sánchez Antuña (Hospital San Agustín); Dr Margarita Marín and Dr Maria José Bueso (Hospital General de Castellón); Dr Javier Jareño (Hospital Militar Gómez Ulla); Dr Paco Carrión (Hospital Clínico Universitario de Valencia); Dr Luis Valdés Cuadrado (Hospital de Conxo); Dr Luis Mateo Caballero (Hospital Mérida); Dr Sergio Cadenas (Hospital Virgen Blanca de León); Dr Juan Ortiz de Caracha (Hospital del Bierzo); Dr Miguel A. Ponce González (Hospital Dr Negrín); Dr Jaume Sauleda (Hospital Son Dureta); and Dr Cristina García Vadillo (Hospital Puerto Real). Writing assistance for the preparation of this manuscript was provided under the direction of the authors by Jordi Bozzo, PhD at Grifols. Editorial assistance was provided by Martin Kenig at PAREXEL, and Mark Forshag, MD and Sandra Nieto at Grifols.
This study was sponsored by Grifols.
The authors declare no conflicts of interest in preparing this article.
