Abstract
Extracellular vesicles (EVs), which are nanosized membranous structures released by diverse cell types, serve as crucial mediators of intercellular communication. Recent evidence has highlighted the dynamic transfer of various biological components, including proteins, lipids, mRNAs, non-coding RNAs, miRNAs, and DNA, via EVs. Immuno-stimulated cells actively release EVs that play a pivotal role in regulating the innate immune system. This study comprehensively reviews the current scientific findings, shedding light on the intricate biological roles of EVs in regulating innate immune cells and the overall immune system. This discussion encompasses diverse pathophysiological conditions and provides valuable insights into the multifaceted contributions of EVs to innate immune responses.
Introduction
Extracellular vesicles (EVs) are a diverse group of lipid bilayer-enclosed secretory vesicles naturally released by various cell types. 1 This classification includes exosomes, microvesicles, and apoptotic bodies, each distinguished by a unique mechanisms of generation. 1 Figure 1(a) illustrates exosome formation initiated by the endocytosis of the plasma membrane, which leads to the creation of multivesicular bodies (MVBs) from early endosomes. Subsequent fusion of MVBs with the plasma membrane results in the release of small vesicles termed exosomes.2,3 Microvesicles are generated through budding of the plasma membrane and typically exhibit larger dimensions than exosomes.4,5 Apoptotic bodies, the largest of the three EV subtypes, emerge during cell apoptosis.6,7
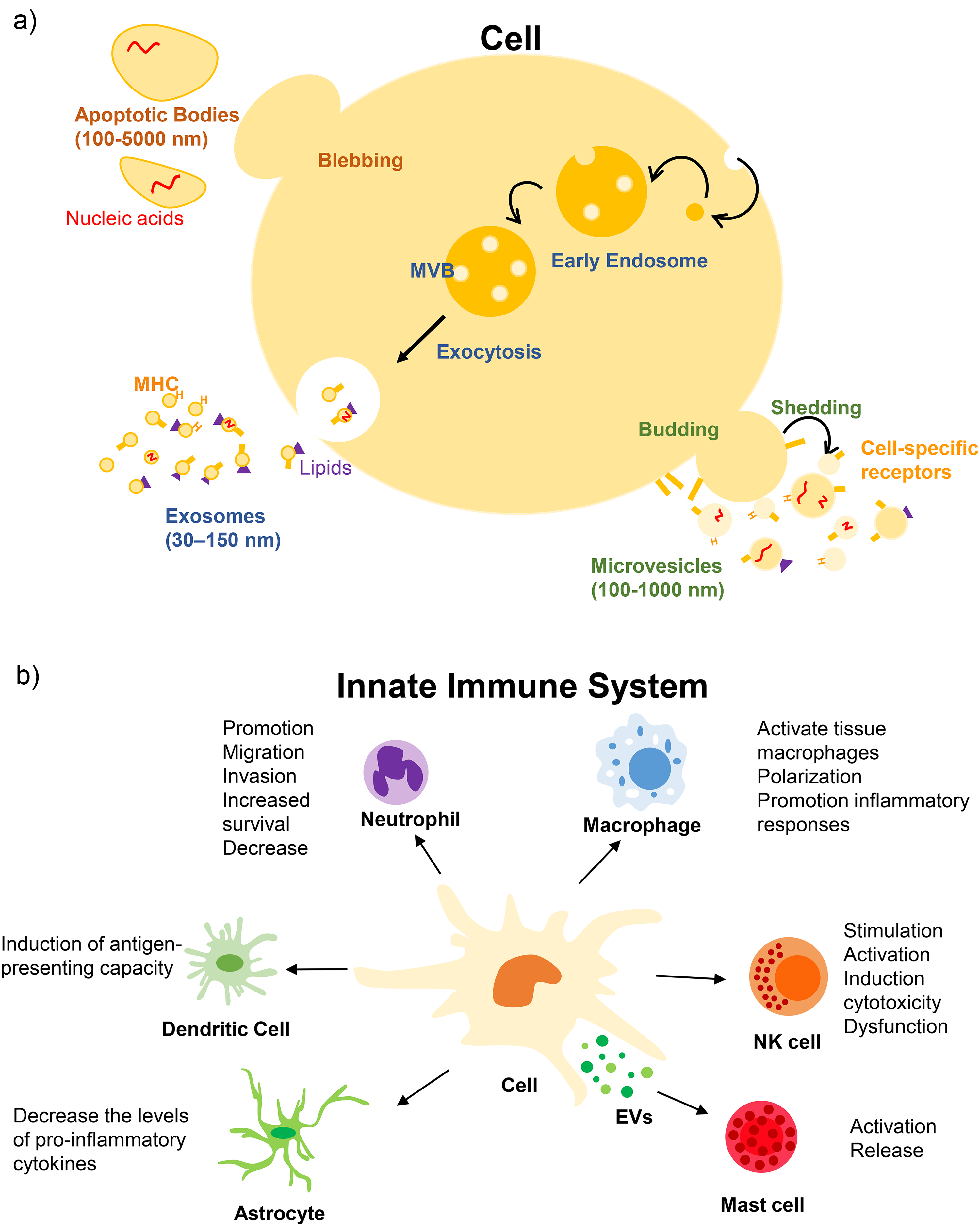
Extracellular vesicles. (a) Generation mechanism of extracellular vesicles in mammalian cells. (b) Biological roles of extracellular vesicles in innate immune system.
Irrespective of their subtype, EVs function as carriers of various components including proteins, lipids, mRNAs, non-coding RNAs, miRNAs, and DNA, facilitating intercellular communication under diverse pathophysiological conditions. 8 Recent studies have underscored the critical regulatory role of EVs in innate immune cells and the broader innate immune system.9–12 Activated innate immune cells considerably contribute to the generation of EVs, initiating a cascade of immune network responses.13–22 This work offers a comprehensive overview of the current understanding of the biological functions of EVs released by innate immune cells, shedding light on their intricate roles in intercellular communication within the context of the innate immune system.
Neutrophils and EVs
Numerous studies have provided insights into the intricate relationship between EVs and neutrophils, highlighting their pivotal roles in various physiological and pathological processes. Kuravi et al. investigated the effects of platelet-derived EVs on neutrophil adhesion to endothelial cells. Their findings revealed that platelet-derived EVs activate neutrophils and considerably enhance their interactions with endothelial cells. This suggests that platelet-derived EVs can fuel neutrophil-mediated tissue inflammation. 13 Activated platelets have been implicated in the generation of neutrophil-derived EVs. These EVs exert regulatory effects on endothelial cell expression, specifically by influencing ICAM-1 levels and modulating neutrophil extravasation. 23 Singhto et al. extended these observations by demonstrating that EVs produced by macrophages exposed to calcium oxalate play a crucial role in stimulating interleukin-8 (IL-8) production in renal cells. In turn, this facilitates neutrophil migration and invasion, revealing a potential link between EVs and renal inflammatory processes. 24
Moreover, investigations into the interplay between mesenchymal stem cell (MSC)-derived EVs and neutrophil function have yielded intriguing results. Mahmoudi et al. showed that MSC-EVs considerably increased the survival of neutrophils in patients with severe congenital neutropenia, positioning MSC-EVs as a promising therapeutic candidate for combating serious infections associated with congenital neutropenia. 14 However, contrasting findings indicated that MSC-EVs reduced the number of neutrophils in patients with chronic granulomatous disease. Nevertheless, these EVs upregulate the inflammatory activity of neutrophils, thereby underlining the complex and context-dependent effects of MSC-EVs on neutrophil function. 25 Wang et al. explored the role of MSC-EVs in polymorphonuclear leukocyte (PMN)-associated neuronal ischemia. Their investigations revealed that MSC-EVs play a crucial role in reducing the infiltration of leukocytes, including PMNs, into the brain. This suggests a potential therapeutic avenue for utilizing MSC-EVs to prevent PMN-associated neuronal ischemia. 26
Leal et al. 27 investigated the relationship between tumor-derived EVs and neutrophil extracellular trap (NET) formation. Their study demonstrated that tumor-derived EVs actively contribute to the formation of NETs in mice exposed to granulocyte colony-stimulating factor, suggesting a collaborative role between tumor-derived EVs and neutrophils in the development of cancer-associated thrombosis.
Additionally, EVs produced by the stimulated neutrophils demonstrated functional activity. Vargas et al. investigated the effect of neutrophil-derived EVs on the proliferation of airway smooth muscles (ASM) in patients with asthma. Their results indicated that EVs actively generated by lipopolysaccharide (LPS)-stimulated neutrophils play a role in promoting ASM proliferation. This sheds light on the potential involvement of neutrophil-derived EVs in the airway remodeling observed in patients with asthma. 28
The immunomodulatory role of neutrophils is contingent on the composition of the released EVs, thereby dictating whether they exert suppressive or promotional effects on innate immune responses. For instance, Zhu et al. highlighted that EVs released from PMNs upon stimulation by the pro-inflammatory cytokine TNF-α enhance the activation of M1 macrophages. 29 Furthermore, Jiao et al. demonstrated that miR-30d-5p-enriched EVs from neutrophils contribute to sepsis-related acute lung injury by inducing M1 macrophage polarization. 30
In contrast, neutrophil-derived EVs have demonstrated substantial anti-inflammatory effects.31,32 Zhang et al. reported that EVs originating from neutrophils elicit tumor cell apoptosis by delivering cytotoxic proteins and activating the caspase signaling pathway. 33
Collectively, these studies underscore the multifaceted interactions between EVs and neutrophils and provide valuable insights into their roles in inflammation, immune responses, and disease pathogenesis. Further exploration of these intricate relationships holds promise for the development of novel therapeutic strategies targeting EV-mediated regulation of neutrophil function in various clinical contexts.
Natural killer cells and EVs
The intricate interplay between natural killer (NK) cells and EVs reveals a complex landscape of immune modulation and functional crosstalk. Several studies have suggested that NK cell-derived EVs exert cytotoxic effects on target cells and modulate immune response.34–39 For example, Lugini et al. highlighted the dynamic nature of NK cell-derived EVs by demonstrating that activated NK cells release EVs expressing killer proteins, such as Fas ligands and perforin molecules, along with typical protein markers of NK cells. This suggests a crucial role for NK cell-derived EVs in immune surveillance and homeostasis. 16 Moreover, EVs from activated NK cells induce apoptosis in target cells by delivering cytotoxic proteins including perforin, granzyme A, granzyme B, and granulysin. 35 NK cell-derived EVs constitute a vital component of immune cell functionality and facilitate immune responses. These EVs have been shown to transport an array of cytotoxic proteins and nucleic acids originating from NK cells, thus exhibiting therapeutic potential for treating diseases. 40
In addition, Sokolov et al. reported that EVs derived from NK cells influence trophoblast cells by promoting their migration while simultaneously inhibiting their proliferation through the phosphorylation of STAT3 at Ser727. 41 Moreover, EVs from IL-1β-stimulated NK cells impact endothelial cells by reducing proliferation, migration, activating caspases, and altering marker expression. 34
Furthermore, NK cell-derived EVs not only carry cytotoxic proteins and cytokines but also contain miRNAs. In a novel perspective, NK cell-derived EVs were found to alleviate depression-like symptoms by carrying miR-207, which targets and regulates the function of astrocytes, offering a potential avenue for exploring the interplay between the immune system and mental health. 42 Wang et al. demonstrated that EVs with miR-1249-3p cargo derived from NK cells could reduce inflammation and insulin resistance via the SKOR1 signaling pathway in a mouse model. 43
Additionally, NK cell-derived miRNAs-enriched EVs (miR-10b-5p, miR-92a-3p, and miR-155-5p) promote the downregulation of Th1 cell IFN-γ and IL-2 production. 44 NK cell-derived EVs alleviate TGF-β-dependent immune suppression. 45 Additionally, EVs derived from NK cells inhibit tumor growth and counteract the TGF-β-dependent immune escape mechanism. 47
Numerous studies have underscored that EVs derived from NK cells encapsulating miRNAs manifest anti-tumor efficacy by inhibiting cancer cell proliferation.46,47 Additionally, Neviani et al. has demonstrated that EVs released from NK cells can inhibit the immune escape of cancer cells. 48 EVs derived from NK cells, particularly EVs containing IL-15 and IL-21 released upon NK cell stimulation, have been observed to augment cytotoxic activity. 49 Similarly, in a study conducted by Kim et al. IL−15/21- bearing EVs derived from NK cells were found to enhance crucial cytotoxic molecule activation in the apoptotic pathway, thereby improving anti-tumor effects in hepatocellular carcinoma cells. 50
While EVs released by normal cells play essential roles in intercellular communication, tumor cell-derived EVs exhibit immunosuppressive properties, shaping the tumor microenvironment and influencing peripheral tolerance in cancer patients. 51 Liu et al. uncovered a pivotal role for tumor cell-derived EVs in promoting tumor growth by inhibiting the IL-2-mediated activation of NK cells. 52 Furthermore, Huyan et al. illustrated that EVs harboring miR-221-5p and miR-186-5p released from cancer cells inhibited NK cell function by impeding the stability of mRNAs associated with DAP10, CD96, and perforin genes. 53 The diverse functions of EVs were underscored by Shoae-Hassani et al. in their investigation of neuroblastoma-and NK cell-derived EVs. Their findings indicated that neuroblastoma-derived EVs act as tumor promoters by creating a supportive niche for tumor growth. In contrast, naïve NK cells exposed to EVs from NK cells previously co-cultured with neuroblastoma cells displayed considerably enhanced cytotoxicity against neuroblastoma cells. 54 Further insights into the immunotherapeutic landscape revealed that EV-circUHRF1, which is predominantly secreted by hepatocellular carcinoma (HCC) cells, contributes to immunosuppression by inducing NK cell dysfunction, potentially conferring resistance to anti-PD1 immunotherapy in HCC patients. 55
The multifaceted involvement of NK cell-derived EVs in diverse physiological processes underscores their potential as therapeutic targets and highlights the need for further exploration to unravel the intricate mechanisms governing their functions.
Dendritic cells and EVs: orchestrating immune responses
Dendritic cells (DCs), recognized as pivotal antigen-presenting cells, are subject to intricate modulation by EVs, unveiling a realm of immune regulatory functions. In a noteworthy study, EVs derived from mast cells were found to activate immature DCs, inducing the upregulation of Major Histocompatibility Complex (MHC) class II, CD80, CD86, and CD40. This cascade of events not only highlights the stimulatory role of EVs in DCs activation but also emphasizes their involvement in the initiation of adaptive immunity orchestrated by DCs. 17
Tumor derived EVs originating from tumor cells are implicated in the suppression and evasion of immune responses. 56 Expanding on this theme, a separate investigation revealed that EVs derived from TS/A tumor cells exerted control over DCs maturation. These tumor-derived EVs induced the accumulation of myeloid precursors in the spleen by targeting CD11b, concurrently suppressing the differentiation of myeloid precursors into DCs. This dual impact suggests a nuanced regulatory role of tumor-derived EVs in the intricate process of DCs maturation. 57
In addition, Salimu et al. demonstrated that prostate cancer-derived EVs can suppress the function of DCs by attenuating the induction of CD73 on DCs through the action of EVs containing prostaglandin E2. 58 Likewise, Gassmann et al. emphasized that EVs released by Ewing sarcoma may induce both local and systemic pro-inflammatory conditions, by inhibiting CD4+ and CD8+ T cell proliferation and suppressing IFN-γ release, while simultaneously promoting the secretion of IL-10 and IL-6. Furthermore, these EVs contribute to the suppression of adaptive immunity by impairing the differentiation and function of antigen-presenting cells, such as DCs. 59
Dengue virus (DENV), which is disseminated through Aedes spp. mosquitoes, is one of the most important arboviral infections globally. 60 Martins et al. highlighted that DENV can alter the composition of EVs released from primary DCs, favoring conditions conducive to viral propagation. 61 Izquierdo-Serrano et al. demonstrated that EVs derived from Listeria monocytogenes infected DCs elicited an anti-pathogenic state, thereby enhancing responsiveness to subsequent bacterial infections with L. monocytogenes in immature recipient DCs. 62 Interestingly, both in vitro and in vivo studies have revealed that Porphyromonas gingivalis, a prominent oral pathogen, induces DCs senescence. This effect was mediated by paracrine actions facilitated by EVs derived from DCs co-cultured with P. gingivalis.63,64 Moreover, an in vivo study demonstrated that EVs released from macrophages infected with Mycobacterium bovis and M. tuberculosis (BCG) induced the activation and maturation of DCs derived from mouse bone marrow. 65
Additionally, EVs derived from mesenchymal stromal cells influence DCs function, thereby affecting immune responses. Reis et al. demonstrated that miR-21-5p enriched in EVs released from mesenchymal stromal cells modulates DCs function and migration. Their study revealed that transfection of DCs with miR-21-5p mimics considerably reduced DCs migration toward the CCR7 ligand CCL21, along with a tendency toward decreased CCR7 protein expression and reduced production of pro-inflammatory cytokines. 66
DCs contribute to immune regulation not only through direct antigen presentation but also by releasing EVs that carry immune-modulating molecules. These EVs have been shown to express biologically functional MHC and T-cell co-stimulatory molecules, enabling them to directly activate adaptive immune responses. 67 Building on this concept, Gao et al. demonstrated that DCs-derived EVs containing TNF-α play a critical role in activating the endothelial NF-κB signaling pathway, which contributes to vascular inflammation and the progression of atherosclerosis. 18 Furthermore, these EVs have been found to express functional IL-15 receptor alpha, which promotes NK cell proliferation and enhances IFN-γ secretion. 68
In addition to their role in immune activation, DC-derived EVs have gained attention for their potential to counteract tumor immune evasion. Studies have shown that these EVs can induce caspase-dependent apoptosis in tumor cells and enhance NK cell responses, thereby strengthening anti-tumor immunity.69–74
In summary, DC-derived EVs are indispensable immune regulators that play a dual role in modulating adaptive immunity and influencing the intricate balance of immune responses in health and disease. Further exploration of these interactions holds promise for the identification of novel therapeutic targets and strategies for the treatment of immune-related disorders.
Macrophages and EVs: orchestrating inflammatory responses
Compelling evidence continues to reveal the pivotal role of EVs as indispensable regulators of macrophage-mediated inflammatory responses. Lee et al. demonstrated dynamic alterations in EVs containing miRNA repertoires throughout the progression of acute lung injury, underscoring the intricate interplay between EVs and pulmonary inflammation.4,75 Furthermore, their findings emphasized the effective modulation of alveolar macrophage phenotypes by EV-miRNAs, reinforcing the crucial role of pulmonary EVs in shaping the trajectory of lung inflammation.75–77
The involvement of macrophage-derived EVs in diverse immunopathological processes has been extensively explored. These EVs are predominant in bronchoalveolar lavage fluid during infection-induced acute lung injury. 19 Ye et al. provided insights into the pro-inflammatory nature of macrophage-derived EVs, demonstrating their role as major contributors to neutrophil recruitment and activation. 20 Additionally, Jiao et al. uncovered the impact of EVs produced by macrophages under hemorrhagic shock, highlighting their ability to induce reactive oxygen species (ROS) production within polymorphonuclear leukocytes (PMNs) and subsequent PMN necroptosis. 78 Moreover, EVs derived from LPS-induced inflammatory macrophages impair ion channel proteins in pulmonary epithelial cells, thereby promoting epithelial cell dysfunction and damage. 79
Remarkably, the biological activities of macrophage-derived EVs appear to depend on the polarization status of macrophages. An in vivo study highlighted that EVs released from M1 macrophages have been documented to serve as carriers for the chemotherapeutic agent paclitaxel, facilitating its targeted delivery to tumor tissues and resulting in an augmentation of therapeutic efficacy in tumor chemotherapy. 80 Additionally, M1 macrophages suppress trophoblast migration and invasion in vitro by releasing EVs containing miRNAs. 81 Interestingly, Jing et al. pointed out that EVs derived from trophoblasts could be transferred into macrophages to induce M1 polarization through the IκBα-mediated NF-κB pathway. Furthermore, M1 macrophages induced by EVs from trophoblasts were found to suppress the proliferation, migration, and apoptosis of trophoblasts via TNF-α. 82
Wang et al. showed that circUbe3a in M2 macrophage-derived small EVs promotes cardiac fibroblast proliferation, migration, and phenotypic transformation by sponging miR-138 5p and translationally repressing RhoC. Their study demonstrated the potential of EVs and circUbe3a to exacerbate myocardial fibrosis following myocardial infarction. 83 Furthermore, another study revealed that miR-378a-3p contained in EVs derived from M2 macrophage- reduces cardiomyocyte pyroptosis by inhibiting human antigen receptor expression and translocation to the cytoplasm, leading to the destabilization of NLRP3 and blocking the activation of the NLRP3/Caspase-1/GSDMD pathways after myocardial infarction. 84
Moreover, Wang et al. revealed that bone fracture healing in individuals with diabetes is considerably delayed due to an imbalance in the osteoimmune microenvironment, which is characterized by an abnormal increase in M1 macrophages and a decrease in M2 macrophages. Additionally, they found that M2-EVs have the potential to promote the transformation of macrophages from M1 to M2 by targeting the PI3 K/AKT pathway. This suggests a promising therapeutic approach for improving the diabetic osteoimmune microenvironment and provides a new perspective and potential treatment strategy for delayed bone repair in individuals with diabetes. 85
In addition, Gao et al. assessed the activity of EVs derived from RBC in a sepsis model and found that EVs exacerbated the inflammatory response in both serum and lung tissue by increasing the production of pro-inflammatory factors such as TNF-α, IL-6, and IL-1β. 86 This leads to the reduced survival of mice with sepsis. Furthermore, transfusion of macrophages pretreated with red blood cell-derived EVs into mice worsened systemic inflammation. In vitro experiments have confirmed the pro-inflammatory properties of EVs, as they elevate cytokine levels in lipopolysaccharide-stimulated macrophages and promoted macrophage polarization to a pro-inflammatory phenotype. RBC-derived EVs play a significant role in promoting macrophage polarization to a pro-inflammatory phenotype during sepsis. The study demonstrates that these EVs enhance macrophage cytokine production by activating the TLR4-MyD88-NF-κB-MAPK signaling pathway, which ultimately aggravates the inflammatory response. These findings suggest that the inflammatory effects of RBC-derived EVs are critical for understanding transfusion-related immunomodulation, and offer potential therapeutic avenues for patients undergoing blood transfusions, particularly in the context of inflammation and sepsis. 86
In addition, EVs derived from the human umbilical cord (hUC-EVs) and arsenic trioxide have been shown to be effective in treating acute graft-versus-host diseases through immunomodulation. The study revealed that hUC-EVs loaded with arsenic trioxide promote the transition of M1 to M2 type macrophages via the mTOR autophagy pathway. 87
This dichotomy in macrophage polarization extends to the effects of EVs on cancer cell behavior. Studies have revealed that EVs derived from M2 macrophages considerably promote cancer cell migration and invasion both in vitro and in vivo.88–91 Guan et al. revealed that exosomes derived from M2 macrophages accelerate the migration and invasion of lung adenocarcinoma (LUAD) cells. Specifically, miR-1911-5p contained within EVs secreted by M2 macrophages enhances LUAD cell migration and invasion by targeting the CELF2 protein, leading to the downregulation of ZBTB4. The inhibition of ZBTB4 promotes LUAD progression. 91 In addition to their role in cancer progression, macrophage-derived EVs have also been shown to modulate inflammation and pain signaling by transferring functional proteins and RNAs to recipient cells, suggesting their broader relevance in inflammatory conditions. 92
Conversely, EVs derived from tumors play a pivotal role in orchestrating the polarization of macrophages within the tumor microenvironment, thereby promoting an immunosuppressive environment favorable to tumor progression.93–95 For instance, tumor-derived EVs enriched with miR-19b-3p have been shown to facilitate M2 macrophage polarization in lung adenocarcinoma, which in turn promotes tumor metastasis by enhancing the secretion of oncogenic EVs carrying LINC00273 and activating the Hippo signaling pathway. 95 Additionally, tumor-derived EVs can influence macrophage metabolic programming and cytokine secretion profiles, further supporting tumor growth and immune evasion.93,94
Numerous studies have revealed that EVs derived from stem cells can regulate the function and proliferation of macrophages in various diseases.96–100 For instance, Li et al. suggested that human umbilical cord mesenchymal stem cell-derived EVs have potential as a therapeutic approach for osteoarthritis by promoting M2 macrophage polarization, anti-inflammatory cytokine expression, and modulating the PI3K-Akt signaling pathway. 97
Furthermore, it has been elucidated that mesenchymal stromal cells possess the capacity to regulate macrophages. Hwang et al. emphasized that mesenchymal stromal cells induce anti-inflammatory effects by promoting the differentiation and maturation of M2 macrophages. 101
Additionally, bacteria-derived EVs can induce macrophage polarization and modulate macrophage function. Bhatnagar et al. demonstrated that macrophages infected with M. avium stimulate uninfected macrophages through EV production, showing diverse inflammatory responses orchestrated by biologically active M1 macrophage-derived EVs in infectious diseases. 102 Kim et al. demonstrated that EVs derived from Lactobacillus plantarum facilitate anti-inflammatory M2 macrophage polarization. Furthermore, their findings underscored the therapeutic potential of EVs released from L. plantarum in mitigating inflammation-associated responses linked to M1 macrophage activation. 103
In summary, the intricate interplay between macrophages and EVs has emerged as a central player in the orchestration of inflammatory responses. The diverse functions of macrophage-derived EVs, which are influenced by their polarization status, offer new avenues for understanding and potentially manipulating immune and inflammatory processes in health and disease. Further exploration of these interactions holds promise for the advancement of diagnostic and therapeutic strategies against inflammatory diseases.
Antigen presenting cells and EVs: bridging innate and adaptive immunity
EVs released by antigen-presenting cells (APCs), such as DCs and macrophages, carry surface MHC class I and class II molecules and therefore can potentially stimulate CD8 and CD4 T cells, respectively,.104,105 However, the ability of peptide-MHC (pMHC) -containing EVs derived from APCs to activate T cells depends on their concentration and the presence of co-stimulatory molecules on the EV surface. 106 Furthermore, the quality of specific immune cell activation is influenced by the antigen presentation mechanisms employed. EVs contribute to antigen presentation via three main mechanisms.9,107,108 First, in direct antigen presentation, EVs derived from professional APCs display MHC-peptide complexes, along with co-stimulatory and adhesion molecules, directly engaging with T cells to activate an immune response (Figure 2(a)).109,110 Second, in semi-direct antigen presentation (also known as the cross-dressing process), EVs bind to the surface of DCs. These EVs remain on the DCs membrane, allowing their pMHC complexes to be directly presented to T cells, while co-stimulatory signals are provided by DCs. 109 This process enhances immune synapse formation by increasing the density of pMHC complexes, thereby facilitating more efficient T cell activation (Figure 2(b)).111,112 Finally, in indirect antigen presentation (cross-presentation), EVs transfer their antigenic peptides to MHC molecules of recipient APCs. The recipient APCs load these peptides onto their own MHC molecules and present them to T cells, initiating a specific immune response (Figure 2(c)). 113 Studies have highlighted the significant potential of EVs in vaccination and tumor immunity, emphasizing their critical role in cross-presentation, which is essential for immune responses against tumors and infections. In tumor immunity, migratory DCs release EVs that transfer antigens to lymph node-resident DCs, thereby enhancing antigen presentation. As a result, EVs carrying pMHC-I complexes from these resident DCs primed and activated the naïve CD8+ T cells. These findings underscore the potential of EVs in immunotherapy, opening new avenues for cancer and viral treatments through targeted antigen delivery.114,115

Mechanisms of antigen presentation by EVs. (a) Direct presentation: EVs display pre-loaded MHC-peptide complexes to directly activate T cells. (b) Semi-direct presentation: EVs bind to the surface of DCs and provide co-stimulatory signals when DCs present their MHC-peptide complexes. (c) Indirect presentation: EVs transfer their antigenic peptides to the MHC molecules of recipient APCs, recipient APCs present these complexes to T cells.
Mast cells and EVs: unraveling the intricacies of immune regulation
Although the biological relationship between mast cells and EVs remains a mystery, the current research sheds light on this enigmatic topic, unraveling the intricate interplay between these two entities. In a noteworthy study, mast cell-derived EVs emerged as potent modulators of immune responses. These EVs were found to promote the mitogenic activity of T and B lymphocytes both in vitro and in vivo, revealing their crucial role in the immune regulatory system. 21 Mast cell-derived EVs exhibit the capacity to evoke diverse immune responses from various immune cell types. Specifically, Vukman et al. highlighted that EVs derived from mast cell induce TNF-α expression from other mast cells. 116 Remarkably, a recent study revealed compelling insights into the potential exacerbation of eosinophilic allergic inflammation. This exacerbation appears to be linked to the activation of human group 2 innate lymphoid cells induced by miR103a-3p derived from mast cells. These findings suggest that the activation of human group 2 innate lymphoid cells by mast cell derived miR103a-3p may play a pivotal role in intensifying eosinophilic allergic inflammation. 117
Moreover, Zou et al. have underscored that EVs originating from mast cells play a pivotal role in exacerbating oxidative stress and eliciting inflammatory responses in asthmatic mice, a phenomenon mediated by the DDAH1/Wnt/β-catenin signaling axis. In the present study, elevated expression of miR-21 in asthmatic mice was correlated with reduced levels of antioxidant enzymes and an increase in inflammatory cell counts, effects that were successfully mitigated upon inhibition of miR-21. 118 This study elucidated that miR-21 specifically targets DDAH1, orchestrating the modulation of the Wnt/β-catenin signaling pathway. Concurrent downregulation of DDAH1 compromised the efficacy of the miR-21 inhibitor. Consequently, the researchers posit that miR-21, released from mast cell-derived EVs, instigates oxidative stress and inflammation in asthmatic mice through the intricate orchestration of the DDAH1/Wnt/β-catenin signaling axis. 118
Expanding the spectrum of mast cell-derived EV functions, another study revealed their ability to upregulate the expression of plasminogen activator inhibitor type-1 in endothelial cells. This finding suggests a potential role of mast cell-derived EVs in procoagulant states associated with endothelial dysfunction syndromes. 22
To explore the multifaceted roles of mast cell-derived EVs, researchers have investigated their impact on airway epithelial cells and the epithelial to mesenchymal transition (EMT). 119 Yin et al. conducted an in-depth investigation into the influence of EVs on the transition from an epithelial to a mesenchymal state. This in vitro study systematically explored the effects of mast cell-derived EVs on epithelial A549 cells by employing analyses at the RNA and protein levels along with phosphorylation events using a phosphorylated protein microarray. 119 The results of this investigation shed light on the intricate communication between mast cells and airway epithelial cells, particularly in the context of EMT, and provide valuable insights into the mechanisms involved in airway remodeling. These findings have implications for respiratory conditions such as asthma and chronic obstructive pulmonary disease.
Furthermore, emphasizing the multifaceted roles of mast cell-derived EVs, another study demonstrated their capacity to stimulate the antigen-presenting ability of immature DCs. This stimulation was evidenced by the upregulation of MHC class II, CD80, CD86, and CD40 expression. Additionally, this study highlighted the efficient uptake of EV-containing antigens by DCs, shedding light on the indispensable role of mast cell-derived EVs in facilitating collaborative interactions between mast cells and DCs within the innate immune system. 17
A recent study revealed that EVs from tonsil-derived mesenchymal stem cells (T-MSCs) possess the ability to modulate gene expression in mast cells. Notably, stimulation of Toll-like receptors (TLRs) induces alterations in both the size and quantity of released EVs particles. Intriguingly, EVs from T-MSCs primed with TLRs induced a reduced number of transcriptomic changes in mast cells compared with unprimed EVs. This phenomenon influences a more discerning spectrum of genes, highlighting the specificity of the impact of TLR-primed T-MSC EVs on mast cell gene expression. 120
Numerous studies have shown that EVs originating from cancer cells elicit mast cell activation.121–123 In a study by Shefler et al. EVs derived from non-small-cell lung cancer promoted lung angiogenesis. 122 These EVs potentially activate mast cells within the lung tumor microenvironment, leading to the release of various cytokines and chemokines. The cumulative effect of this interplay suggests a mechanism by which non-small cell lung cancer-derived EVs promote angiogenesis in the lungs. 122
Additionally, the reciprocal interaction between mast cells and cell membranes of lung tumor cells induces mast cell activation, resulting in the release of miRNAs encapsulated within EVs. This intricate intercellular communication underscores the dynamic involvement of mast cells in the tumor microenvironment. The miRNAs released within EVs may play a role in modulating signaling pathways, contributing to the complex regulatory processes associated with mast cell-tumor cell interactions in the context of lung tumors. 124
In summary, emerging evidence suggests that mast cell-derived EVs play a pivotal role in modulating immune responses, influencing immune cell activity, and contributing to an intricate network of interactions within the immune system. Further exploration of the biological intricacies of mast cell-EV interactions holds promise for uncovering novel insights into immune regulation and potential therapeutic avenues for immune-related disorders.
Conclusions: unveiling the potential of EVs in innate immunity
This review highlights the pivotal role of EVs as key regulators of innate immunity, emphasizing their capacity to mediate intercellular communication and modulate immune responses. The dynamic release of EVs by innate immune cells in response to external stimuli underscores their intricate involvement in immune system regulation, as illustrated in Figure 1(b) and detailed in Tables 1 and 2. Furthermore, the identification of critical EV-associated components relevant to innate immune disorders positions these vesicles as promising candidates for diagnostic and therapeutic applications in a range of immune-related conditions.
Biological functions of extracellular vesicles (EVs) derived from innate immune cells.

Biological functions of extracellular vesicles (EVs) from other cell types on the innate immune cells.

Despite their great potential, EVs face several significant challenges that must be addressed to fully realize their clinical applications. These challenges include developing high-efficiency methods for isolating EVs, improving the delivery of therapeutic cargo, ensuring the stability of EVs in the bloodstream, accurately targeting specific tissues, and achieving effective delivery of therapeutic cargo inside cells. Overcoming these obstacles is crucial for harnessing the full therapeutic potential of EVs and expanding their use in disease prevention, diagnosis, and treatment. Continued research is essential to resolve these issues and unlock new opportunities for developing innovative therapies across a wide range of diseases.
In conclusion, this review provides a comprehensive synthesis of existing knowledge on the biological functions of EVs within the innate immune system, offering a robust foundation for future investigations. By addressing existing knowledge gaps and exploring innovative strategies, this review aspires to catalyze advancements in the field. Continued research into EV-based diagnostics and therapeutics holds great promise for transforming our understanding of innate immunity and fostering the development of novel clinical interventions.
Footnotes
Acknowledgement
During the preparation of this work, the authors used ChatGPT to enhance language and readability. Following its use, the content was thoroughly reviewed and edited with input from native speakers. The authors take full responsibility for the final version of the manuscript.
Author contributions
B.J. and H.L. developed the outline of the study; B.J., J.P. prepared the original draft; and H.L. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Global LAMP Program of the National Research Foundation of Korea (NRF), grant (No. RS2024-00444460), and the Korea Basic Science Institute (KBSI), grant (No. 2023R1A6C101B022), funded by the Ministry of Education. Additional support was provided by the KRIBB Research Initiative Program, grant (No. KGM5322523), and the Mid-career Faculty Research Support Grant at Changwon National University in 2024.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
