Abstract
Rheumatoid arthritis (RA) is a chronic inflammatory disease that primarily affects the synovial joint linings, resulting in progressive disability, increased mortality, and considerable economic costs. Early treatment with disease-modifying antirheumatic medications (DMARDs) can significantly improve the overall outlook for people with RA. Contemporary pharmaceutical interventions, encompassing standard, biological, and emerging small molecule disease- modifying anti-rheumatic medications continue to be the cornerstone of RA management, with substantial advancements made in the pursuit of achieving remission from the disease and preventing joint deformities. Nevertheless, a substantial segment of individuals with RA do not experience a satisfactory response to existing treatments, underscoring the pressing need for novel therapeutic options. Biologic DMARDs are among the therapy choices. Non-tumor necrosis factor inhibitors (Non-TNFi) such as abatacept, rituximab, tocilizumab, and sarilumab are examples, as are anti-tumor necrosis factor (TNF) medications such as infliximab, adalimumab, etanercept, golimumab, and certolizumab pegol. More recent biomarkers have emerged and showed usefulness in the early detection of RA. These biomarkers, often referred to simply as “biomarkers”, are quantifiable indicators of normal or pathologic processes, and they can also gauge treatment response. The assessment of RA treatment response typically combines patient-reported outcomes, physical evaluations, and laboratory findings, as there isn’t a single biomarker that has proven sufficient for measuring disease activity. This review explores the usage of biologic DMARDs as a therapeutic approach for RA, as well as the biomarkers typically used for RA early diagnosis, prognosis prediction, and disease activity evaluation.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune condition with an unknown cause, displaying a higher prevalence in females compared to males. Women make up approximately 70–80% of RA cases, and it is most commonly seen in older individuals. The emergence of RA is thought to result from an intricate interaction of elements, encompassing genetic, environmental, and immunological factors. 1 As per the World Health Organization, roughly 1% of the global population is affected by RA.2,3 In developed countries, the incidence of new cases falls within the range of 5 to 50 per 100 000 adults. Methods such as the Clinical Disease Assessment Index (CDAI) and the Disease Activity Assessment Index (SDAI) have typically been used to assess the activity of RA. 4 To attain clinical remission and effectively control RA's activity, rheumatologists must maintain vigilant and precise monitoring, allowing them to make necessary adjustments to treatment plans.
RA is frequently treated with disease-modifying antirheumatic medications (DMARDs), which include biological DMARDs (bDMARDs), targeted synthetic DMARDs (tsDMARDs), and conventional synthetic DMARDs (csDMARDs). 5 Commonly employed treatments for RA and other inflammatory joint conditions include corticosteroids, DMARDs like methotrexate (MTX), leflunomide, sulfasalazine, and hydroxychloroquine, as well as biological response modifiers (BRMs) such as abatacept, adalimumab, anakinra, etanercept, infliximab, and rituximab. 1
Clinical manifestations of rheumatoid arthritis
Clinical signs of RA encompass arthralgia, swelling, erythema, and can even restrict joint mobility. Timely diagnosis is regarded as the principal benchmark for achieving the best possible results.6,7 Significant reductions in the quality of life can be averted by identifying and treating the deterioration in musculoskeletal health and the onset of RA during its initial stages. There is a need for a cost-effective, minimally invasive biochemical test that can ideally detect and differentiates various common types of arthritis at an initial stage. Early biochemical tests for diagnosing established RA have traditionally relied on measuring rheumatoid factor (RF), which, in its present form, has demonstrated a sensitivity of 63% and a specificity of 94% for advanced or established disease. 8 Early diagnosis, on the other hand, remains a substantial issue because it mainly relies on medical information gathered from the individual's medical history, supported by physical examination, blood tests, and imaging assessments. Notably, several factors come into play, including the patient's knowledge of RA, their readiness to seek medical guidance, the duration between symptom onset and the initiation of suitable management, and the investigative proficiency of the doctor – all of which significantly impact the management and prognosis of RA. When RA is not effectively managed or reaches a severe stage, the risk of extra-articular manifestations increases. Pleuritis, pericarditis, rheumatoid nodules, keratitis, small vessel vasculitis, and a variety of nonspecific extra-articular symptoms are examples of these. 9
Current therapeutic options
While there is no cure for RA, attempts are being undertaken to accelerate the diagnosis and reach a state of low disease activity as soon as possible. The widespread use of pharmaceutical treatments, like corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs), has demonstrated their effectiveness in lowering stiffness and discomfort. It is critical to remember that these treatments have minimal effect on slowing the progression of the disease. 10 Given that DMARDs have been demonstrated to be quite effective in lowering disease activity and significantly reducing or deferring joint abnormalities, there has been a surge in interest in their efficacy during the last 20 years. 11 New small molecules, biological DMARDs, and conventional synthetic medications are the three main types of therapeutic choices. Traditional DMARDs like auranofin, minocycline, azathioprine, and cyclosporine are rarely used in current therapy. JAK inhibitors (Olumiant), RANKL antibodies (Pralia), IL-6 receptor antibodies (Kevzara), anti-CD20 antibodies (Truxima, Rixathon), and TNF inhibitors (Amjevita, Renflexis, Erelzi, Cyltezo, Imradl) are examples of biological DMARDs.
Despite the proliferation of new medication and management strategies, long-term overall sickness reduction remains elusive for many patients, underlining the need for cutting-edge therapeutic options. As a result, finding reliable diagnostic and prognostic markers of the condition is critical so that clinicians may determine the appropriate first course of treatment. 12 This review presents an up-to-date assessment of recent research on bDMARDs, known biomarkers for RA diagnosis, new prognostic predictors, and the possibility of innovative pharmaceutical therapeutics to improve therapy protocols.
Biological DMARDS (bDMARDs)
Biological DMARDs (bDMARDs) are a class of drugs designed to directly target the molecules or molecular pathways involved in the inflammatory processes of rheumatoid arthritis (RA). Several biological DMARDs have demonstrated clinical and radiological success in treatments (Table 1). TNF- was the primary target of the first generation of bDMARDs, but subsequent medications have been intended to target B cells (CD-20), IL-6, and CD28 antibodies. 13
Biological DMARDs for RA.

TNF-α inhibitor (TNFi)
TNF-α is an inflammatory cytokine produced by activated monocytes, macrophages, and T lymphocytes. TNF-α functions through two types of receptors, TNF receptors 1 and 2, which have different species specificities and TNF-α binding affinities. TNF-α and its receptors work together to activate the key signaling pathways such as the NF-κB system, RANKL signaling, the extracellular signal-regulated kinase (ERK) pathway, the tumor progression locus 2 (TPL2) pathway, and proapoptotic signaling. TNF-α levels that are elevated in inflammatory musculoskeletal disorders have led to the theory that TNF-α contributes to local bone loss in these conditions. 22 TNF has been linked to a variety of activities, including endothelial cell activation, angiogenesis promotion, and modulation of fibroblasts, keratinocytes, enterocytes, chondrocytes, and osteoclasts, among other cell types. TNF-α antagonists may potentially improve the vascular stiffness in RA patients, according to existing data. 23 Many RA patients who were previously unable to work were able to do so after receiving anti-TNF therapy. 24 Low et al. (2017) discovered that TNFi can minimize the likelihood of a heart attack in RA patients taking sDMARD therapy. Information about the effectiveness and potential side effects of TNFi has been obtained during the last 15 years, mostly through registries established at the regional and national levels following the arrival of these treatments on the market. According to the latest research, TNFi is the best bDMARD therapy for RA patients who do not respond well to or are unable to handle standard sDMARD drugs. 25 Despite differences in biochemical and pharmacological properties, there are no discernible clinical differences in terms of efficacy or safety among the five currently marketed TNFi. Anti-TNF medication does not appear to raise the incidence of major bacterial infections in a large group of RA patients when compared to MTX therapy. 26 As a result, practical variables such as dosing schedule or manner, as well as more general cost considerations, have a substantial impact on the selection of a TNFi. Many recently produced biosimilar medications have already been approved by regulatory agencies. A biosimilar, which is a biologic pharmaceutical product that is identical to the original medication, is typically developed by a separate company. 27
Infliximab (IFX), the first tumor necrosis factor inhibitor (TNFi) used to treat RA, is a chimeric monoclonal antibody. This antibody combines a mouse idiotype with a human antibody architecture. IFX effectively decreases TNF-'s biological effects by binding to all TNF-forms. It has a long history of safety, and it is administered intravenously. 28 Individuals receiving IFX therapy for RA experienced significant reductions in adhesion molecules as well as levels of MCP-1, IL-1, IL-6, and IL-8. There is also evidence that the synovial lining is becoming thinner. 29 IFX biosimilars, including IFXdyyb, SB2, CT-P13, BOW015, NI-071, PF-06438179/GP1111, STI-002, and ABP 710, have received regulatory approval in various countries. 13 IFX has been linked to a number of side effects, including an increased risk of severe infections, reactivation of hepatitis B or tuberculosis, and an increased risk of lymphoma and other cancers. Adalimumab (Ada) is a monoclonal antibody that specifically targets TNF. It has a low toxicity profile and is provided every two weeks by subcutaneous injections. 30 More than half of RA patients using Ada therapy have anti-Ada antibodies (AAA). The TNF-binding region of Ada is the principal target of these extremely specific AAA responses, which reduce the clinical efficacy and nullify its therapeutic benefit. 31 Ada is known as an effective antirheumatic medication that can produce remission and stop radiographic progression. Furthermore, research has indicated that combining MTX and Ada in a treatment regimen is more effective than doing so independently. As Cohen et al., (2017) 32 reviewed a number of Ada biosimilars, including ABP 501, Adfrar, and ZRC-3197, have been licensed in several countries. Ada has a number of undesirable side effects, including an increased risk of heart failure, allergic skin reactions, and the reappearance of dormant infections. Etanercept, on the other hand, is a recombinant protein created by combining two soluble, naturally existing human 75-kDa TNF receptors. It is administered twice a week via subcutaneous injection, and its toxicity profile is comparable to that of IFX and Ada. 33 Etanercept has consistently demonstrated the efficacy in slowing the radiographic progression and preserving joint function in both elderly and younger RA patients. 34
According to a published study, 50% to 75% of people who take etanercept achieve clinical remission. Examples of authorized etanercept biosimilars are SB4 and GP2015. 35 The human IgG1 kappa monoclonal antibody golimumab targets human TNF-α in both its soluble and transmembrane bioactive forms. It is given once a month via subcutaneous injection. Short-term bad consequences include serious infections, cancer, TB, and mortality do not appear to have changed. It has a reasonable level of safety. Additional long-term monitoring studies are required to assess its safety further. 36 In comparison to the 50-milligram dose, the 100-milligram dose showed modestly higher rates of severe infections, demyelinating events, and cancers. 37 Two golimumab biosimilars, BOW100 and ONS-3035, are currently in the preliminary stage of development. 38 A human anti-TNF antibody's Fab fragment, certolizumab pegol, efficiently suppresses both membrane-bound and soluble TNF. It is covalently bonded with polyethylene glycol. This medication is administered via subcutaneous injection every two weeks and is generally well tolerated. Preclinical testing for PF-688, a biosimilar to certolizumab pegol, is now underway. 38 In roughly 2% of cases, users of certolizumab pegol experience significant adverse effects. 39 It's worth noting that clinical trials for onercept and lenercept for TNFi failed. TNF inhibitors, on the other hand, have fundamentally changed how RA is treated and are now a key component of disease management. Healthcare workers in charge of patient care should have a basic awareness of the risks. More research is needed, however, to investigate the viability of rationally produced dominant negative TNF variants in TNF signaling suppression. 40
Antibodies that target B-Cell depletion and inhibition
Rituximab is a chimeric monoclonal antibody that targets B cells to express CD20. It has an effect on these cells at various stages of development, from early pre-B cells to mature B cells, excluding plasma cells that have already undergone terminal differentiation. 41 Rituximab, by targeting CD20, can destroy certain subsets of B lymphocytes by a variety of mechanisms, including complement-dependent cytotoxicity, apoptosis induction, and growth inhibition. B lymphocytes contribute to the inflammatory cascade by presenting antigens and generating pro-inflammatory cytokines like TNF-, VEGF, MCP, macrophage migration inhibitory factor, RF, and ACPA autoantibodies. 42 Rituximab is thought to affect CD4 + cells, resulting in a significant decrease in T-cells in people with RA. 43 The combination of rituximab and MTX significantly slowed the course of joint deterioration in RA patients who had previously failed to respond to TNFi treatment. 44 Several nations’ regulatory bodies have approved rituximab biosimilars, including BCD-020, Maball, and MabTas. Infections, late-onset neutropenia, mucocutaneous reactions, and hypogammaglobulinemia have all been documented as side effects of Rituximab therapy. Furthermore, Rituximab therapy has been related to progressive multifocal leukoencephalopathy in a small number of instances. 45
Belimumab, a monoclonal antibody is designed particularly to target the B lymphocyte stimulator (BLyS). It exhibits a high tendency for binding with soluble human BLyS, decreasing the protein's biological activity. In RA patients, RF levels are linked to BLyS levels, which are raised in both the blood and synovial fluid. The BLyS mechanism of action is critical for B cell survival, and blocking it can cause autoimmune B-cell clones to die. 46 Nonetheless, Belimumab failed to show promise in RA phase II clinical trials. The efficacy of three promising alternative CD-20-targeting antibodies including obinutuzumab, ibritumomab, and ocaratuzumab must be studied further in clinical trials. It is suggested that focusing on suppressing B-cell modulatory cytokines may be more successful than reducing B cell populations more deeply. 47
Therapies directed at T-Cell
T-cell co-stimulation is controlled by the soluble fusion protein abatacept, which is entirely for human. It is made up of a modified Fc region of human IgG1 and an extracellular domain of human CTLA-4. 48 T cells enter the synovial joint, generating pro-inflammatory cytokines such as IL-17 and interferons, causing bone and cartilage degeneration. T cells require a co-stimulatory signal after antigen identification in order to fully activate. Abatacept inhibits the CD80/CD86 interaction in the same way that CTLA4 does, and has a higher affinity for these molecules than CD28. 49 Abatacept, unlike many other biologic medications, does not directly inhibit inflammatory proteins. It instead attaches to their surfaces, preventing cellular communication. This medication is prescribed for people who haven't responded well to one or more DMARDs and is available in both infusion and injectable forms. When taken with a TNF-blocker, abatacept may raise the risk of serious infections. 50 Abatacept may cause headaches, colds, sore throats, nausea, and an increased risk of infection. T-cell-specific drugs, such as ciclosporin, anti-CD4 antibodies, anti-CD5 antibodies, or alemtuzumab, have not consistently exhibited beneficial clinical effects. The involvement of T cells and their subtypes must be studied further. Clinical trials are currently underway for a variety of other T-cell therapies, including Sirukumab, Olokizumab, Clazakizumab, and ALX-0061. 47
Inhibition of IL-6
Tocilizumab (TCZ), a humanized monoclonal antibody, is designed to specifically target the IL-6 receptor found on cell surfaces throughout the body. 51 IL-6 is produced by a variety of cell types, including T cells, B cells, monocytes, fibroblasts, endothelial cells, and synovial cells. Both the soluble IL-6 receptor (sIL-6R) and the membrane-bound IL-6 receptor (mIL-6R or CD 126) are activated by IL-6. IL-6 can exacerbate bone loss, promote pannus development by upregulating vascular endothelial growth factor (EGF) expression, accelerate bone resorption by increasing osteoclastogenesis, and make leukocytes more susceptible to oxidative stress in the context of RA pathogenesis.52,53 Tocilizumab (TCZ), which is available in subcutaneous and intravenous forms, has a very minimal likelihood of causing an immunological reaction. 54 In TCZ-treated individuals, life-threatening infections do not appear to be related with lower neutrophil counts. 35 Sirukumab, a human monoclonal antibody with strong affinity for IL-6, demonstrated favorable results and an expected safety profile in a phase 3 clinical trial. 55 Because it focuses on cytokine inhibition rather than cytokine receptor inhibition, this provides a unique opportunity to examine the function of cytokine inhibition in RA. Headaches, elevated blood pressure, nasopharyngitis, and upper respiratory tract infections are the most commonly reported side effects in clinical trials. Several IL-6 inhibitors are now being studied in clinical trials, including clazakizumab, olokizumab, sarilumab, ALX-0061, and MEDI5117. The findings of these trials showed that anti-IL-6 medicines could be exceptionally effective treatments.56,57
IL-1 inhibition
The immunological and pro-inflammatory properties of the cytokine IL-1 are well established. IL-1RI and IL-1RII are two membrane-bound IL-1 receptors that look like immunoglobulins. Unlike IL-1RI, which begins signaling at the cell surface, IL-1RII acts as a decoy receptor, binding to and obstructing IL-1. It is possible for the two circulating IL-1 receptors to bind to IL-1 and change its accessibility. 58 Anakinra (rHuIL-1ra), an injectable recombinant IL-1 receptor antagonist, is not glycosylated. It is distinct from native human protein in that it has an extra N-terminal methionine added, given once a day. Anakinra inhibits IL-1 and IL-1 activity by engaging with the IL-1 receptor. However, it does have certain downsides, such as the need for regular injections and possibly painful injection sites. It can be taken alone or in combination with DMARDs, albeit anti-TNF medications should not be utilized in this combination. Infections, allergies to the upper respiratory tract, and gastrointestinal issues are just a few of the potential side effects that must be closely monitored. It's worth noting that anakinra patients with RA had better cardiac contractility three hours after a single dose. 59 Consequently, those with heart failure and/or severe or resistant pericardial disease may think about Anakinra as a potential therapy option. 60 More research is needed to investigate the benefits of suppressing IL-1 in this patient population. Several interleukin cytokines and their receptors have also been explored as potential therapeutic targets. A phase III study of the IL-17 inhibitor Secukinumab, for example, revealed advantages in RA patients who had not reacted adequately to TNF inhibitors. 61 In a randomized phase II study, stekinumab and MTX were combined, but there were no favorable effects on IL-12/23 suppression. As a result, clinical trials are being conducted to assess the efficacy of medications targeting IL-7, IL-15, IL-18, IL-21, IL-32, and IL-33. 62
Contributing element to osteoclast differentiation
Human monoclonal antibodies of the IgG2 type called denosumab (DMab) prevent bone resorption. To do so, it binds to and inhibits RANKL, a crucial cytokine essential for osteoclastogenesis and bone resorption. 63 Memory B cells have also been discovered to produce RANKL, which has been linked to bone deterioration in RA. Furthermore, by encouraging the proliferation of Treg cells, RANKL has been identified as a critical element in the establishment of immunological tolerance. This implies that RANKL antagonists may influence how the immune system is regulated. 64 The combination of synovial cell overgrowth, activated immune cells, and cytokines promotes TNF and RANKL-driven osteoclast formation. The fact that RA patients suffer from both localized and systemic bone loss has led researchers to explore if inhibiting RANKL would be an effective method to limit pathological bone resorption. 65 There is evidence that individuals with RA who have progressive bone erosions should consider combining denosumab with DMARDs. 66 DMab was shown to be effective in lowering both localized and systemic bone loss associated with RA in two phase II trials and one randomized observational trial. Phase III trials are required to determine the best dosage and assess its efficacy to prevent bone degeneration. Low blood calcium and phosphate levels, numbness, cellulitis, and muscle cramps are all possible side effects. Overall, DMab has a bright future in the treatment of RA. 63 A phase IIb research has found a significant response to mavrilimumab, a new monoclonal antibody that precisely targets the GM-CSF receptor alpha, revealed a novel therapeutic approach. 67
Diagnostic biomarkers of RA
Numerous researches have been undertaken to discover the predictive and prognostic indicators for RA, and the results showed a diverse range of prospective markers. It's important to realize that not all of these markers have been thoroughly validated. These markers can be classified into several kinds based on their purpose and placement, as shown in Figure 1. It is important to highlight that the focus of this study is mostly on biochemical indicators, with a comprehensive discussion of clinical and imaging biomarkers being ignored due to the scope of this analysis.

Classification scheme for RA markers.
Biochemical markers
Clinical signs and indications have traditionally been used to diagnose RA. Unfortunately, early diagnosis of RA was challenging, frequently leading to postponed confirmation and severe joint damage. The increased demand for exceptionally sensitive and specific laboratory testing in the early stages of RA allows for earlier detection and therapy choices. Researchers looked into a variety of biomarkers in various cellular compartments, focusing on analytes associated with autoimmune processes, inflammation, and bone and cartilage metabolism. The goal is to identify biomarkers with clinical utility for diagnosing RA. 12
Genetic markers
HLA-DR B1
In the published literature, there are extensive analyses of genetic markers in RA. 68 Although, at least 12 genetic loci have been linked to RA susceptibility, it is important to recognize that each of these loci has a small impact. Furthermore, as the similar variants are ubiquitous in the general population, these genetic changes, even when considered collectively, cannot be utilized to diagnose the disorder (Table 1). Only two of these genetic loci have compelling evidence tying them to illness severity or prognosis. Variations in the human leukocyte antigen (HLA)-DRB1 gene were initially linked to RA. SE alleles, in particular, are associated with more severe illness symptoms and a higher likelihood of developing RA. 69 SE alleles, all encode the same amino acid pattern. Furthermore, the existence of anti-cyclic citrullinated peptide (anti-CCP) antibodies, which are used to identify anti-citrullinated protein antibodies (ACPA), is associated with genotypes. Individuals who have both anti-CCP antibodies and SE alleles frequently have faster joint deterioration as the disease progresses.70,71 Testing for the presence of SE alleles is frequently removed from clinical practice since anti-CCP antibodies are easier and less expensive to detect. Furthermore, the shared epitope (SE) is not required for the link between polymorphisms in the TRAF1/C5 susceptibility gene and erosive changes in RA to occur, according to two investigations.72,73 However, it's crucial to realize that possessing susceptibility variants carries a relatively low risk, typically about 1.5 and does not confer any clinical advantage. The PADI4 gene has been widely investigated in ethnic groups ranging from Asian to European to North American. Its varying impact on RA susceptibility in these countries is still being debated. The gene is intriguing for these reasons.74,75 A convincing study suggests that the PADI4 gene influences, how anti-CCPs affect the clinical characteristics of RA, and the PADI4 genotype appears to influence joint degeneration. 76 Despite its importance in Asian populations, the PADI4 genotype did not show significant relevance as a risk factor for RA in the most exhaustive investigation to date. 74 More research is required to determine PADI4's role as a RA marker.
Although the combination of HLA-DRB1 and anti-CCP antibodies remains the most precise genetic marker for predicting a more severe course of RA, several additional genetic loci are emerging, and some of these are expected to play a critical role in predicting RA outcomes. Despite the fact that many serological markers are being investigated for diagnostic or prognostic reasons in RA therapy, genetic indicators have the benefit of being stable over time. Here are some further details.
Genomic and proteomic biomarkers
HLA-DR B1
The potential use of genetic and proteomic indicators as clinical decision-making tools in RA is now being studied. Each technique has advantages and disadvantages. The HLA-DR shared epitope is a known risk factor for the development of anti-CCP antibody-positive RA, especially when paired with smoking, making it a useful predictor of RA susceptibility. 77 Genome-wide association studies are constantly uncovering new genetic indications connected to RA risk. Less is known, however, about the utility of genetic biomarkers in predicting disease progression or therapy efficacy. Gene-expression patterns, on the other hand, show promise as prognostic biomarkers in RA, albeit preliminary findings must be verified. 78 Finding biomarkers based on transcriptional or genotypic profiling, however, has considerable limitations. There is typically minimal link between gene transcript levels and the proteins they encode, and changes in genomic sequence are not always accompanied by changes in gene expression or protein function. Furthermore, posttranslational modifications are not taken into account in gene transcript profiling, which can be crucial in situations like RA where the synthesis of proteins is impaired. 79 These concerns have prompted many researchers to focus their efforts on identifying proteomic biomarkers, despite the fact that this field of study has its own set of obstacles, such as biased screening and technological challenges. 80
TRAF1/C5
Variations in the HLA-DRB1 area have been linked to RA susceptibility, including shared epitope alleles (SE) associated with disease severity. In addition to the major histocompatibility complex (MHC), there is strong evidence associating RA susceptibility to loci such as PTPN22, TRAF1/C5, and TNFAIP3. Surprisingly, the TRAF1/C5 region has been associated to non-cardiovascular death as well as susceptibility and severity of RA in certain groups. 81
Cui et al., in their study reported that the examination of 31 risk allele variants revealed a dominant allele (G) at the rs10919563 locus within the PTPRC gene, a well-established predictor of RA risk, responds better to anti-TNF medication. This link was significantly stronger in patients who had antibodies to the citrullinated peptide (ACPA) and/or RF. 82 The multi cohort study found no significant associations between treatment response and the remaining thirty risk alleles connected to RA. It should be emphasized that the study did not look into any possible links between the reaction and HLA-DRB1. According to BRAGGSS research in the UK, the response to anti-TNF medication was not connected with HLA-DRB1 and PTPN22 variants. However, another UK-based study validated the link between PTPRC mutations and response across the entire cohort, despite the fact that it was not significant only in the ACPA positive group. 83
Autoantibodies
Rheumatoid factor
RF antibodies are antibodies that specifically target the Fc region of human IgG immunoglobulin. 84 They are a distinguishing feature of RA, and hospital studies have revealed that 60–80% of those with RA have them. The American Rheumatism Association (ARA) incorporated RF into its laboratory criteria in 1987. 85 RF, on the other hand, is not limited to RA; it can be found in various rheumatic disorders, chronic inflammatory syndromes, infections, and even in healthy people. 86 Rheumatoid factor's sensitivity in identifying RA ranges from 19% to 53%, while its specificity ranges from 91.7% to 98.6%. 87 Numerous studies have found a link between RF and radiographic outcomes, and RF is routinely used to diagnose and predict the outcomes in RA. 88 The disease stage influences how well it predicts the outcomes. 89 As a result, RF positive is related with higher disease activity and the formation of erosions in the early stages of RA. However, as the condition advances, its accuracy as a predictor of future joint development deteriorates. 90 The existence of the RF test indicates an increased risk of developing RA, making it a helpful diagnostic marker. 91
Anti-critrullinated protein/peptide antibodies
Citrulline, a modified version of the amino acid arginine, is one of the peptides or proteins identified and targeted by anti-citrullinated protein/peptide antibodies (ACPA). Citrullinated antigens can appear in a variety of structural forms.
Antibodies against cyclic citrullinated peptide
Antibodies that target cyclic citrullinated peptide (anti-CCP) may offer greater predictability of disease development in people with RA. Anti-CCP antibodies have been connected to joint degeneration and have been found to be predictive of RA in many investigations in recent years.91,92 Anti-CCP antibodies, which are more specific than RF, can be discovered years before the start of RA symptoms.92,93 Sensitivity ranges for reported diagnosis ranged from 39% to 94%, with specificities ranging from 81% to 100%. 94 Anti-CCP antibodies were approved by the European League Against Rheumatism (EULAR) in 2007 for the early identification of RA. 95 The American College of Rheumatology (ACR) has revised the RA categorization criteria. Anti-CCP positive rheumatoid arthritis (RA) is more commonly associated with localized joint destruction, especially when combined with HLA-DRB1 alleles having the same epitope. HLA-DR3, on the other hand, has been linked to anti-CCP negative RA.96,97 RF test is particularly efficient in distinguishing patients with RA from those with other rheumatic disorders due to its high specificity.
Antiperinuclear factor/antikeratin antibodies
Antikeratin antibodies (AKA) and antiperinuclear factor (APF) have both demonstrated the RF specificity for the diagnosis of RA. 98 APF is found in 49 to 91% of RA patients, with a sensitivity range of 73 to 99%. Antikeratin antibodies are also seen in RA patients, with positive results occurring in 36 to 59% of cases. 99 APF and AKA levels stay stable independent of disease duration. These antibodies can appear as early as the first stages of RA.98,100 As a result, APF and AKA may aid in the early identification of RA, allowing for prompt intervention and drug delivery. These antibodies exhibit correlations with the presence of RF, the activity and severity of RA, and with each other. 98 Additional research has shown the utility of assessing anti-CCP, AKA, APF, and certain RF isotypes for predicting early structural damage in the illness course. 101 The best RA diagnostic marker among AKA, anti-CCP, and APF remains unknown. According to a published study, AKA and anti-CCP are both more effective than APF in RA diagnosis. As a result, clinicians may choose AKA or anti-CCP testing to aid with the diagnosis of RA. 102 The complexity and accessibility of APF testing, which is not commonly performed as a regular practice, provide a significant barrier.
Anti-mutated citrullinated vimentin
MCV (mutated citrullinated vimentin), often known as Sa, is an important auto-antigen found in synovial tissue. In the diagnosis of RA, anti-Sa antibodies have a sensitivity of 20% to 40% and a specificity of 98%. Furthermore, anti-Sa antibodies strongly predict major joint issues and extra-articular RA symptoms. 103 A novel anti-MCV ELISA test was recently released, representing a step forward in RA diagnosis. According to Bang et al., this test has the same specificity and sensitivity as anti-CCP antibodies. A published study evaluated the baseline antibodies against MCV, CCP types 2 and 3 (both IgG isotype), and 3.1 (both IgG and IgA isotype) in 210 early RA patients over a two-year period. The Larsen score was used to assess disease activity at baseline and monthly during 24 months using radiographs of the hands and feet. Anti-MCV antibodies were associated with more severe illness, as seen by higher DAS28, ESR, and IgG levels, as compared to anti-CCP2, CCP3, and CCP3.1 antibodies. 104 Another study looked at anti-MCV antibody levels at the start of treatment, one year later, and two years later in 162 individuals with early arthritis. When the indicated limit of 20 U/mL was used, the results showed that anti-MCV antibodies exhibited 92.3% specificity and 59.3% sensitivity. Patients with positive anti-MCV outcomes had higher Sharp-van der Heijde scores, higher ESR levels, and higher CRP levels at all assessment time points. 105 According to the published studies, anti-MCV antibodies may be a more sensitive and specific alternative to anti-CCP testing in diagnosing RA.106,107
Antip68
Antibodies against the stress protein immunoglobulin heavy-chain binding protein (anti-BIP or antip68) are found in more than 60% of RA patients. These antibodies have also been found in experimental arthritis animal models. 108 Furthermore, BIP levels in the rheumatoid joint are raised in sera from people in the early and pre-disease stages of the disease. 109 These data showed that BIP may be a significant auto-antigen in RA, but more study is needed to assess its usefulness as a biomarker. Anti-CCP antibodies and RF are two extensively utilized biomarkers in the diagnosis and prognosis of RA. Anti-MCV antibodies have been proposed as an alternative due to their high specificity, which often outperforms anti-CCP antibody testing; nevertheless, it is unclear whether their benefits are significant enough to warrant routine clinical testing.
Inflammatory markers
Erythrocyte Sedimentation Rate (ESR)
According to several studies, increased ESR during early RA may be an indication of long-term radiological progression. 95 ESR, along with CRP, is one of the most commonly utilized laboratory tests for evaluating the disease severity. ESR values usually reflect disease activity in the weeks preceding the appearance of symptoms. However, it's important to realize that several confounding factors might alter ESR findings, including age, gender, fibrinogen levels, rheumatoid factor (RF), high levels of gamma globulins, and anemia. 110 Nonetheless, ESR readings have been included into a risk prediction model aimed at identifying persons at risk of rapid radiographic progression (RRP). This model can predict if joint deterioration in RA patients would progress over time. 111
C-reactive protein
C-reactive protein (CRP), an acute-phase blood test that is extremely sensitive, has been associated with radiographic disease progression, medication response, and clinical disease activity in RA patients. 99 CRP, like ESR, is included in a variety of disease activity grading schemes. CRP, on the other hand, is generated by the liver in direct reaction to the impact of inflammatory cytokines on hepatic function, making it a more specific biomarker for measuring RA disease activity. 112 CRP, as opposed to ESR, is a more accurate predictor of transient fluctuations in disease activity. 113 Serum CRP level is connected with joint damage in addition to reflecting disease activity. 114 CRP was established as the most reliable indication of radiographic damage in a cohort of individuals with early inflammatory polyarthritis whose outcomes were studied from the start over a five-year period. 115 Furthermore, CRP testing is the preferred biomarker for assessing disease activity since it is simple, accessible, and inexpensive.
Calprotectin
Calprotectin, a key protein found in leukocytes, has been demonstrated to correlate with both clinical assessments and laboratory results in inflammatory disorders. 116 Calprotectin concentrations were shown to be elevated in the synovial fluid of people with RA. 117 Furthermore, in one study, calprotectin levels and joint injury measures, as well as laboratory and clinical markers of inflammation, were found to be substantially and statistically significantly associated. 118 Calprotectin was discovered to be an independent predictor of both clinical and radiographic joint deterioration after a ten-year study. This suggests that it could be useful as a biomarker for erosive illness in people with RA. 119
Serum amyloid-associated (SAA) protein
SAA protein, like CRP, is a precursor for Amyloid A and has been linked to a number of clinical indications in inflammatory diseases. However, SAA levels are typically elevated and vary greatly between individuals.120,121 According to a study, when RA patients’ synovial tissue fibroblasts are exposed to SAA, they create MMPs. 122 As a result, SAA may have a major impact on the breakdown of the extracellular matrix in RA patients’ joint tissues. It is important to note that there is a significant link between SAA levels and CRP levels in the sera of RA patients. This correlation does not exist in healthy people, implying that SAA measures may not provide any further information. 122 In a recent study, researchers discovered that using SAA in a RA diagnostic model improved its sensitivity when compared to using only anti-CCP. However, as a result of this combination, the model's specificity was reduced. 123
Other inflammatory markers, such as E-selectin, 124 Thioredoxin, 125 intracellular adhesion molecule (I-CAM)1 126 and vascular cell adhesion molecule (V-CAM)1.124,127 These markers have been connected to RA disease activity, but there isn't much evidence that investigating them can disclose much more than what can be learnt by tracking CRP and ESR levels. As a result, the two biomarkers most often employed in clinical practice to assess RA are still ESR and CRP.
Cytokines/inhibitors/adipocytokines
In RA, pro- and anti-inflammatory cytokines are out of balance, resulting in chronic inflammation. Pro-inflammatory cytokines are normally low, but in RA, they become persistently increased, resulting in protracted inflammation. The abnormal activation of critical inflammatory cytokines and their receptors is thought to be important in the pathophysiology of RA. Macrophages and fibroblasts in the synovium of RA patients produce a wide range of cytokines, including IL-1, IL-6, IL-15, IL-18, tumor necrosis factor (TNF), and granulocyte-macrophage colony-stimulating factor (GM-CSF), among others. In RA, cytokines such as IL-1, IL-1, IL-6, M-CSF, IL-15, IL-17, and TNF- promote the growth, survival, and activity of osteoclasts, resulting in bone loss. 128 IL-6 is the most common cytokine found in the blood serum and synovial fluid (SF) of RA patients, and research has found a link between IL-6 levels and disease activity in these body fluids. However, the findings on the association between IL-6 levels and joint injury are conflicting; some studies detect a relationship, while others find none. Although some studies imply a link, others do not, there is conflicting evidence about whether IL-6 levels are associated with joint injury. 129 According to certain research, the progression of joint deterioration in people with rheumatoid arthritis (RA) has not been definitively linked to IL-6 levels. 130 It's also important to realize that baseline IL-6 concentrations can vary greatly between people, frequently by a factor of 100, and can rise with exercise. 131 The combination of anti-CCP and IL-6 provided the strongest discriminatory capacity for diagnosing established RA in a multivariate investigation covering all RA biomarkers. However, its utility as a biomarker in predicting disease outcomes remains unknown. According to a published study, higher levels of soluble TNF receptor (sTNFR)-II, an inflammatory marker that reflects TNF levels, can appear up to 12 years before the onset of RA. As a result, sTNFRII may be a valuable biomarker for predicting the beginning of future illnesses. 132 An in-depth examination of 16 cytokines and cytokine-related indicators found that seven of these analytes IL-1, IL-1 receptor antagonist (Ra), IL-4, IL-10, TNF-, and sTNFRI showed significant increases in levels as early as five years before the RA diagnosis. 133 In a separate investigation, 23 cytokines and chemokines were discovered in the synovial fluid of people with early synovitis. The findings revealed that the composition of synovial fluid was changed, if only briefly, in those who went on to develop RA. Three months following the onset of symptoms, patients with early arthritis who did not proceed to RA had significantly higher levels of cytokines such as IL-2, IL-4, IL-13, IL-17, IL-15, basic fibroblast growth factor (bFGF), and EGF. 134 This distinguishing feature was no longer evident in established RA. Other cytokines that can inhibit osteoclastogenesis include IL-4, interferon (IFN), IL-13, and IL-18. While inhibitory cytokines such as TGF- and IL-1Ra, as well as the suppressor of cytokine signaling-3 (SOCS3), are produced in the RA synovium, their levels are insufficient to ameliorate synovitis. The importance of IL-17 production in the development of RA joint lesions is well recognized, as IL-17 can stimulate the production of pro-inflammatory cytokines (such as IL-1 and TNF-), upregulate RANKL, and increase the activity of MMPs, all of which contribute to matrix degradation and bone resorption. 135 Elevated levels of IL-17A have been seen in the synovium and synovial fluid of RA patients. 136 In addition, recent clinical research found a link between synovial IL-17A expression and joint damage. 137 Adipocytokines such as leptin, visfatin, resistin, and adiponectin have been studied in both healthy and RA patients. 138 Adipocytokine concentrations were discovered to be elevated in RA patients and may affect the degree of radiographic joint deterioration. Radiographic joint damage was associated with reduced leptin levels and greater visfatin levels.
Various chemokines, including monocyte chemoattractant protein (MCP)-1, macrophage inflammatory protein (MIP)-1, RANTES, MCP-4, pulmonary and activation-related chemokine (PARC), MIP-3, growth-related oncogene (GRO), IL-8, monocyte induced by IFN- (Mig), interferon-inducible protein (IP)-10, and stromal cell-derived factor (SDF)-1, have been identified in the synovial tissue (ST) and synovial fluid (SF) of RA patients. 139 TGF, CXCL13, MIP-3, and monocyte colony-stimulating factor (M-CSF) were revealed to have the capacity to predict the level of disease activity in rheumatoid arthritis in a recent study. 140 These chemokines promote leukocyte recruitment and retention in the joints, which aids in the progression of RA. It's vital to remember that many chemokines have additional biological functions, such as releasing inflammatory mediators, promoting cell proliferation, and assisting in angiogenesis. As a result, the role of cytokines and associated molecules in the pathogenesis of RA is significant. More research is needed in the future to acquire a better understanding of their involvement in the disease and their potential as RA biomarkers.
CRP and ESR remain the gold standard indicators for assessing inflammation in clinical practice. However, due to the intricate structure of the cytokine network and their volatility, it is still uncertain if these cytokine markers are suitable as biomarkers in a clinical setting. To study the possibility of more advanced models in forecasting results for RA patients, larger collections of cytokines, chemokines, and adipokines are needed.
Immunological markers
T lymphocytes, specifically CD4+ T cells are abundant in the synovium of people with RA. The underlying cause of the chronic inflammation found in established RA is the defective interactions among T cells, macrophages, and fibroblasts within an aberrant milieu. 141 Treg cells come in a variety of forms including thymus-derived CD4 + CD25+ cells. These Treg cells are critical for maintaining self-tolerance and preventing the advancement of autoimmune diseases. 142 Treg cells are further subdivided into subgroups, one of which being T helper 3 (Th3) cells, which are required for immunological tolerance. 143 In RA, these cells may operate incorrectly or ineffectively. 144 According to published studies, inflamed joints have larger amounts of CD4+ CD25+ Tregs, and these intra-articular Tregs have stronger suppressive capacities than their peripheral counterparts. 145 Even while CD4+ CD25+ Tregs are growing more suppressor cells, RA remains uncontrolled. The authors believe that effector cells in RA may be less vulnerable to the suppressive actions of CD4+ CD25+ Tregs. Several studies have been conducted to compare the CD4+ CD25+ Tregs observed in RA patients to those seen in other kinds of inflammatory arthritis.145,146 Furthermore, people with RA and other kinds of inflammatory arthritis have higher levels of CD4+ CD25+ Tregs in their synovial fluid. The length or severity of the disease do not appear to have a significant impact on this enrichment. Furthermore, it has been demonstrated that synovial fluid CD4+ CD25+ Tregs can limit the cytokine production and cell proliferation. Despite the fact that a number of studies have given consistent results, the potential that Tregs could function as markers of the condition or prognosis of a disease is still being debated 146 as augmented 145 or reduced. 19 The frequencies of CD4+ CD25+ Tregs in synovial fluid differ from those found in healthy people.
Discussion
The quest for novel biomarkers with significant therapeutic applications and their significance in rheumatoid arthritis (RA) remain a hot research area. In addition to well-established markers like RF and anti-CCP, the addition of anti-MCV has proved helpful in the early-stage RA diagnosis. Many potential biomarkers have been extensively studied in recent years for prognostic and predictive purposes (as given in Table 2) and diagnosis (as given in Table 3) of RA. It should be noted that inflammatory markers (CRP and ESR) and autoantibody assays (RF and anti-CCP) are now the only tests commonly employed in clinical practice.
Biomarkers outcome in rheumatoid arthritis.
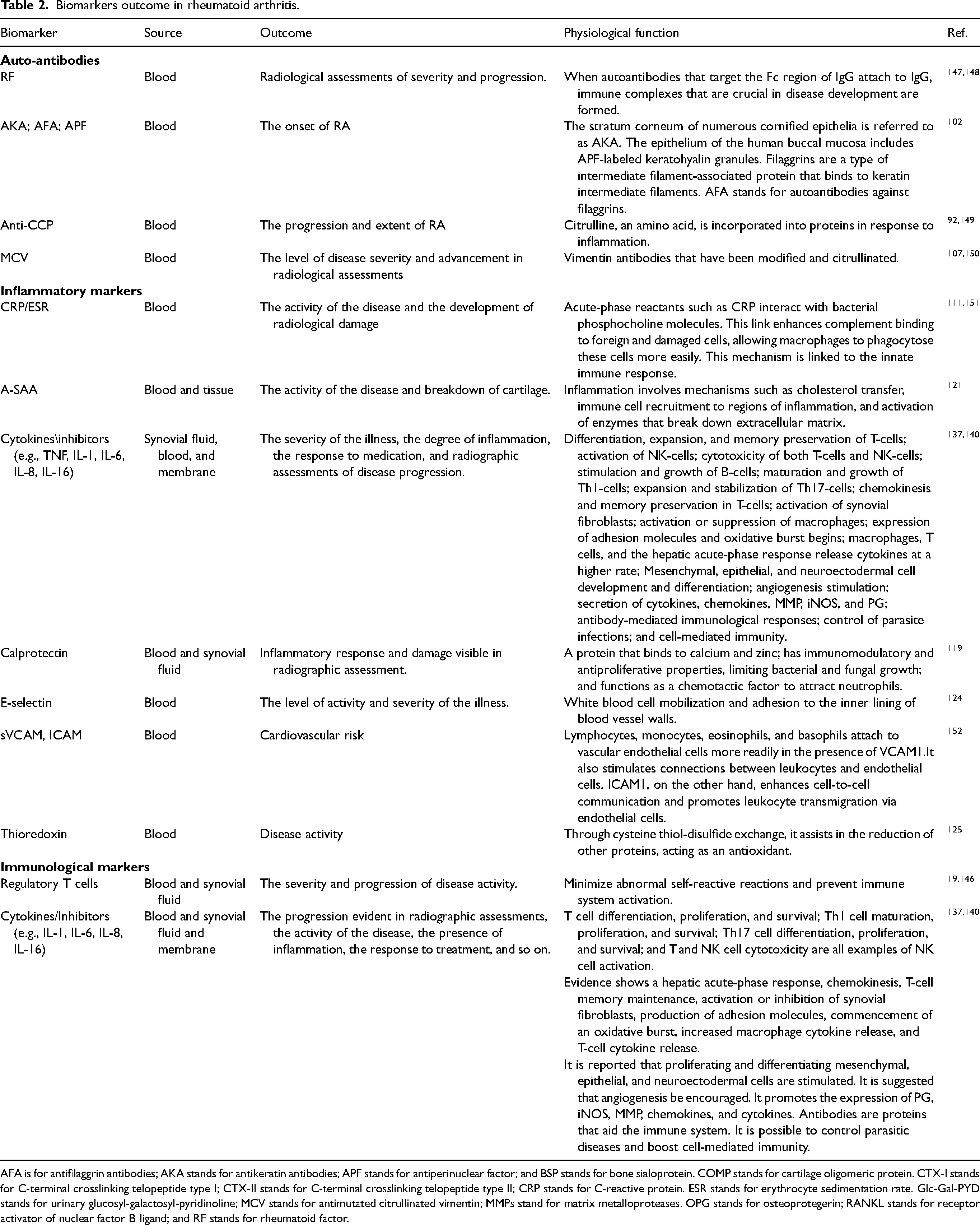
AFA is for antifilaggrin antibodies; AKA stands for antikeratin antibodies; APF stands for antiperinuclear factor; and BSP stands for bone sialoprotein. COMP stands for cartilage oligomeric protein. CTX-I stands for C-terminal crosslinking telopeptide type I; CTX-II stands for C-terminal crosslinking telopeptide type II; CRP stands for C-reactive protein. ESR stands for erythrocyte sedimentation rate. Glc-Gal-PYD stands for urinary glucosyl-galactosyl-pyridinoline; MCV stands for antimutated citrullinated vimentin; MMPs stand for matrix metalloproteases. OPG stands for osteoprotegerin; RANKL stands for receptor activator of nuclear factor B ligand; and RF stands for rheumatoid factor.
Biomarkers in rheumatoid arthritis vs. other diseases.

Data presented as median (25th percentile, 75th percentile) or n (%).
Antikeratin antibodies (AKA); antifilaggrin antibodies (AFA); antiperinuclear factor (APF); ankylosing spondylitis (AS); BSP: bone sialoprotein. OA: osteoarthritis; PMR: polymyalgia rheumatica; PsA: psoriatic arthritis; ReA: reactive arthritis; RF: rheumatoid factor; SLE: systemic lupus erythematosus; UA: undifferentiated arthritis.
The anti-CCP2 antibody test is still the most accurate way to forecast how joint erosion will progress, even if it may not have high sensitivity in the early stages of the illness. Numerous research and patient groups have revealed associations between the progression of the disease and a variety of different biomarkers in urine or serum samples, including COMP, GlcGal-Pyd, MMP3, CTX-I, CTX-II, and C2C (Table 2). Currently, research is focused on identifying novel biomarkers with therapeutic potential and understanding their significance in relation to RA. In addition to well-established markers like RF and anti-CCP, the addition of anti-MCV has proved effective in the early-stage RA diagnosis. In recent years, numerous possible biomarkers for prognosis (Table 2) and diagnosis (Table 3) have been intensively studied. It is unclear to what extent these markers correlate or provide additional information beyond what autoantibodies and inflammatory markers do. However, it appears prudent to explore them as a group of biomarkers, as this technique may yield more detailed data than the primarily individual tests conducted so far. Prospective cohort studies allow us to uncover new and improved biomarkers, which improves our ability to foresee and recognize sickness. As a result, it is possible to discriminate between people who are expected to have a milder course of the disease and those who are likely to have a more aggressive or rapidly developing variety, protecting the latter group from potentially deadly medicines. Prospective cohort studies also allow you to track the progression of the disease in each individual case and compare the impact of different treatments over time. A distinct biomarker signature that may be used to predict and track a range of key outcomes for patients may be produced by creating complicated models that incorporate a variety of biomarkers and clinical indicators. As a result, it may be easier to tailor treatment plans for groups of patients with similar biomarker profiles, capturing the essence of customized medicine.
Biomarkers are critical for directing clinical and treatment strategies at all phases of rheumatoid arthritis. They perform a variety of critical functions, such as predicting disease onset in at-risk individuals, improving diagnostic precision by bridging serological gaps, providing prognostic insights critical for treatment decisions, assessing treatment responses and outcomes, and making it easier to track disease activity and progression. RF and ACPA are used as biomarkers to identify those who have the condition or are at risk of getting it. These markers shed light on the disease by anticipating the extent of bone erosions, the duration of the sickness, and the likelihood of developing rheumatoid arthritis.
Conclusions
There is no single therapy that works consistently well for all RA patients, and different people respond differently to different drugs. In this case, looking into certain biomarkers could be incredibly useful in discovering patients and ensuring the sensible therapeutic use of more expensive medications. As a result, based on the illness stage, this research contributes to the selection of the most appropriate therapeutic strategies for RA patients. It also provides the framework for future studies aiming at identifying biomarkers linked with different stages of RA.
Footnotes
Author contributions
Muhammad Riaz and Ghulam Rasool contributed in the conceptualization, designing and writing the manuscript draft of this review study. Ruhamah Youaf and Hina Fatima contributed in the literature search, visualization and drafting the manuscript. Naveed Munir and Hasan Ejaz contributed in the visualization, validation and critical reviewing of the manuscript draft. All authors contributed in this review study and approved the manuscript draft for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
