Abstract
The sensitivity of pigs to deoxynivalenol (DON) might be influenced by systemic inflammation (SI) which impacts liver. Besides following acute-phase proteins, our aim was to investigate both the hepatic fractional albumin (ALB) synthesis rate (FSR) and the ALB concentration as indicators of ALB metabolism in presence and absence of SI induced by LPS via pre- or post-hepatic venous route. Each infusion group was pre-conditioned either with a control diet (CON, 0.12 mg DON/kg diet) or with a DON-contaminated diet (DON, 4.59 mg DON/kg diet) for 4 wk. A depression of ALB FSR was observed 195 min after LPS challenge, independent of feeding group or LPS application route, which was not paralleled by a down-regulated ALB mRNA expression but by a reduced availability of free cysteine. The drop in ALB FSR only partly explained the plasma ALB concentrations which were more depressed in the DON-pre-exposed groups, suggesting that ALB levels are influenced by further mechanisms. The abundances of haptoglobin, C-reactive protein, serum amyloid A, pig major acute-phase protein, fibrinogen and LPS-binding protein mRNA were up-regulated upon LPS stimulation but not accompanied by increases in the plasma concentrations of these proteins, pointing at an imbalance between synthesis and consumption.
Keywords
Introduction
LPS are constituents of cell envelopes of most Gram-negative bacteria1–3 and belong to PAMP molecules. 4 As such, they are recognized by highly conserved surface receptors, most importantly by TLR4 and CD14, and particularly when LPS is bound to LPS-binding proteins (LBP). 5 , 6 TLR4/CD14 and co-receptor molecules are present on various cell types, notably on monocytes and macrophages, but also on hepatocytes. 7 Following binding, a signal transduction cascade is initiated, finally resulting in the expression of a number of inflammatory cytokines, such as TNF-α, driving the acute-phase response (APR). 2 ,8–11 Ultimately, this results in a modulation of tissue protein turnover, particularly in the liver and skeletal muscle. 12 Among others, the APR includes an up-regulation of those hepatic export proteins necessary for the innate immune response and that are termed positive acute-phase proteins (APP), for example serum amyloid A (SAA), pig major acute-phase protein (MAP), C-reactive protein (CRP), haptoglobin (HP) and others. In addition, other export proteins, i.e. the negative APP (e.g. albumin (ALB) and others) are down-regulated 11 , 13 , 14 in order to support the synthesis of positive APP by providing free Aa. In skeletal muscle, pro-inflammatory cytokines stimulate protein degradation which additionally provides Aa for the hepatic synthesis of positive APP. 15
Xenobiotics differ markedly in their biological effects and are always present in feed and food at varying levels. Deoxynivalenol (DON) is a mycotoxin formed by several species of the genus
Therefore, the aim of the present study was to examine both the fractional ALB synthesis rate and the ALB concentration as indicators of ALB metabolism in the presence and absence of SI. The latter was induced by infusion of LPS either via the pre-hepatic or post-hepatic route to elucidate the specific role of the liver in mediating a generalized SI. Moreover, to investigate the interactions between DON and LPS in more detail, each infusion group was conditioned either with a control diet containing a low background DON contamination (CON, 0.12 mg DON/kg diet) or with a DON-contaminated diet (DON, 4.59 mg DON/kg diet). To further characterize liver metabolism and APR under the described experimental conditions, indicators of liver protein turnover as well as hepatic mRNA- and protein abundances of further APP were determined.
Material and methods
Experimental design and procedures
The current investigations were part of a comprehensive experiment for studying the effects of DON in LPS-challenged pigs. Details of the experiment, including diet composition, housing conditions and surgical procedures were reported earlier. 29 , 30
Experiments and procedures were in accordance with the European Community regulations concerning the protection of experimental animals and the guidelines of the German Animal Welfare Act, and were approved by the ethical committee advising the Lower Saxony State Office for Consumer Protection and Food Safety (file number 33.4-42502-04-13/1274).
Briefly, a total of 41 barrows (German Landrace) with an initial mean body mass (BM) of 25.8 ± 3.7 kg were fed either a control diet (CON, 0.12 mg DON/kg diet) or a diet mainly contaminated with DON (DON, 4.59 mg DON/kg diet)for 4 wk starting at d 0.
At d 27 of the experiment, all animals were surgically equipped with indwelling systemic (jugular = post-hepatic) and portal (pre-hepatic) venous catheters. At d 29, pigs of the two feeding groups were randomly assigned to three infusion groups each with LPS (7.5 µg/kg BM dissolved in 0.9% NaCl,
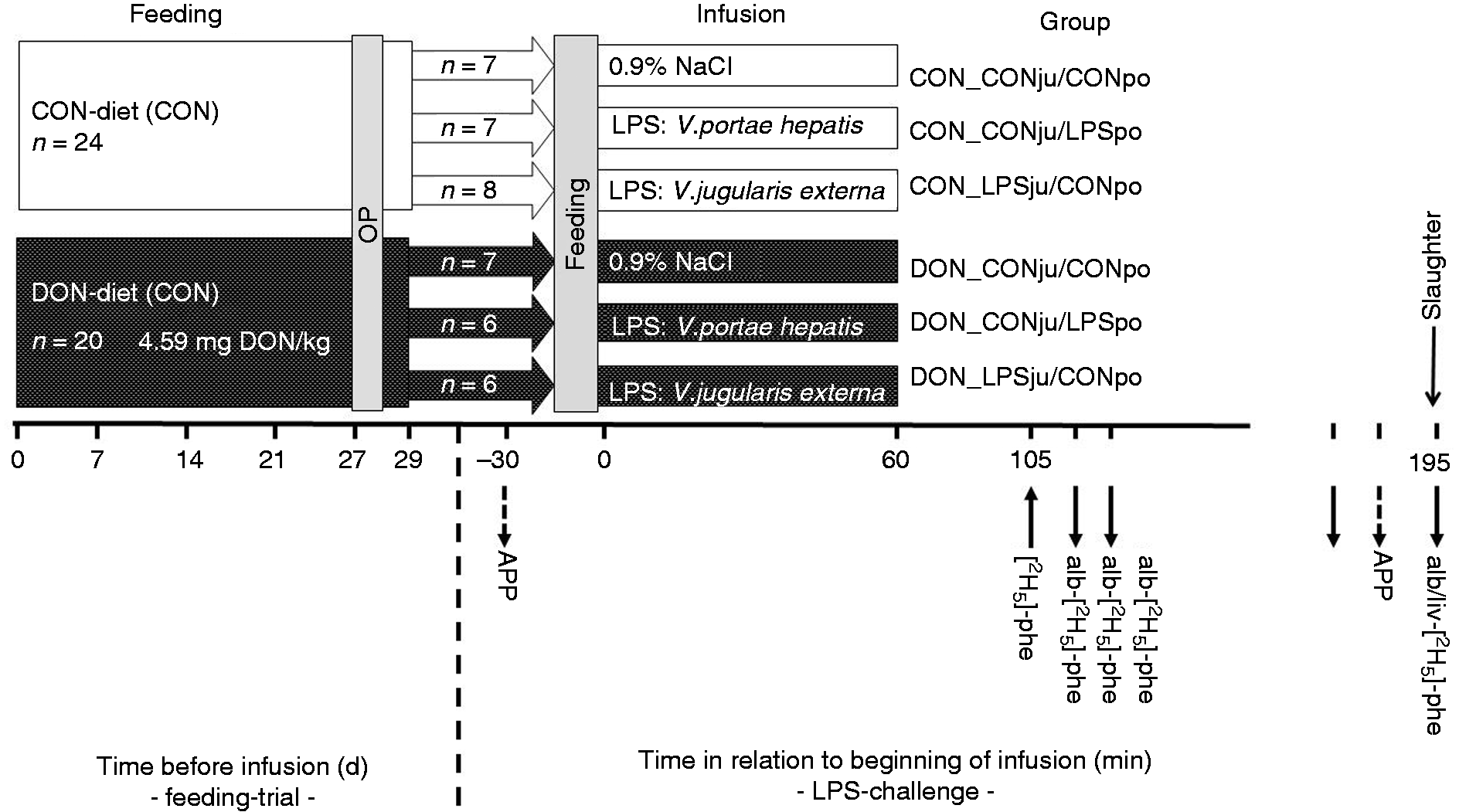
Experimental design: Pigs were fed either a control diet (CON) or a diet contaminated with mainly deoxynivalenol (DON) for 4 wk. At d 29 of experiment pigs of the two feeding groups were randomly assigned to three treatment groups each and were infused for 60 min with either a LPS solution, in jugular or portal vessels (LPSju or LPSpo, respectively), or with a saline solution (CONju and CONpo, respectively). Downstream arrows indicate time points of blood (APP, acute-phase proteins; ALB-[2H5]-Phe, samples for determination of Phe enrichment) and tissue sampling, while upstream arrow relates to the isotope tracer infusion – for further details see text.
Pigs were fed with 700 g feed per animal according to their feeding group at 15 min prior to start of the infusions. At -30 and 180 min relative to the start of the infusion blood samples for the analyses of APP were collected from the external jugular vein.
For quantifying the newly synthesized ALB and total liver protein, the flooding dose technique was applied as originally described for humans and experimental rodents 31 , 32 and later adjusted for pigs. 33
All pigs were injected with 5 ml/kg BM of a deuterated phenylalanine (Phe) solution (150 mMol/l, 29 molar percent excess (MPE) of l-[2H5] Phe; 125 mg total Phe/kg BM) prepared from l-Phe and l-[2H5]-Phe (Cambridge Isotope Laboratories Inc, Andover, MA, USA, 98% isotopic enrichment) at 105 min relative to the start of LPS/saline infusion into the internal jugular vein. Blood samples were collected at 30, 45, 60 and 90 min relative to Phe injection from the external jugular vein using Li-Heparin Monovette® (Sarstedt AG & Co., Germany). Blood samples were centrifuged (1915
Non-perfused liver tissue samples (
Synthesis of albumin and hepatic proteins
Determination of protein synthesis rates of ALB and total liver protein required the separation of free Phe from protein-bound Phe and involved an initial precipitation of plasma and tissue proteins using trichloroacetic acid and perchloric acid, respectively, as described in detail elsewhere.
21
,
23
Supernatants were used for further purification and analysis of tissue free Aa (see section
Acute-phase proteins
Haptoglobin was measured by ELISA as described earlier. 34 For Pig-MAP (pig major acute-phase protein, also known as ITIH4), a commercially available ELISA (ACUVET Biotech, Zaragoza, Spain) that was standardized to the European reference serum for APP (EU Concerted action QLK5-CT-1999-0153) was used. Serum amyloid A (SAA) was also measured by ELISA (Phase™, SAA Assay Multispecies Cat. No. TP-802, Tridelta, Kildare, Ireland) using the control material provided by this company (Cat. No. TP802-Con). For CRP, the Turbovet CRP pig assay, a particle enhanced turbidimetric immunoassay was used (ACUVET Biotech).
Expression of acute-phase protein genes
Frozen liver tissue was homogenized in liquid nitrogen using a 6770 Freezer/Mill® (SPEX SamplePrep LLC, New Jersey, USA) and from the resulting powder total RNA was isolated applying the NucleoSpin® RNA kit (Macherey-Nagel GmbH & Co. KG, Düren, Germany) according to the manufacturer’s protocol. Concentration and quality of the purified RNA were determined with a NanoDrop® ND-1000 (NanoDrop Technologies Inc., Wilmington, DE, USA) and RNA integrity was verified using agarose gel electrophoresis including 0.06% sodium hypochlorite (adapted from 35 ). One microgram of RNA was transcribed into cDNA with the qScript™ cDNA Synthesis Kit (Quanta Biosciences™, Inc., Gaithersburg, MD, USA) according to the manufacturer’s protocol. Gene-specific primer pairs (Table 1) were generated using Primer-BLAST 36 and obtained from Eurofins Genomics GmbH (Ebersberg, Germany). The generation of a single PCR product was evaluated by melting curve analysis, the product length was verified on 2% agarose gel electrophoresis and the PCR products were confirmed by sequencing. Primer efficiencies were calculated using a five-point dilution series of a cDNA sample mixture of the experiment. qPCR runs were conducted in duplicate 15 µl reactions on a CFX96™ Real-Time PCR System (Bio-Rad Laboratories, Hercules, CA, USA) using iTaq™ Universal SYBR® Green Supermix (Bio-Rad Laboratories, Hercules, CA, USA). The primers were added in the concentrations listed in Table 1 and the cDNA equivalent of 22.5 ng RNA was included. After an initial denaturation step of 30 s at 95°C, 40 cycles of 5 s denaturation at 95°C and 30 s of annealing and elongation at 60°C followed. The presence of a single PCR product was confirmed in each well with a melting curve from 60°C to 95°C in 0.5°C increments for 5 s each and every run was controlled by a no-template control in triplicate. Cq-Values of target and reference genes were obtained using CFX Manager™ Software 3.1 (Bio-Rad Laboratories, Hercules, CA, USA) and the evaluation was done with qbase+ (Version 3.1, Biogazelle, Zwijnaarde, Belgium). The optimal reference genes GAPDH, HPRT and TBP were identified with a geNorm analysis out of nine reference targets 37 and exhibited a mean reference target stability M value of 0.36. The normalized, relative expression levels were calculated using the geometric mean of the above-mentioned reference genes as normalization factor, relating the data to the geometric mean of all CON_CONjug_CONpo animals (ΔΔCq), and taking in consideration the primer pair-specific amplification efficiencies. 38
Characteristics of gene-specific primers used for qRTPCR.
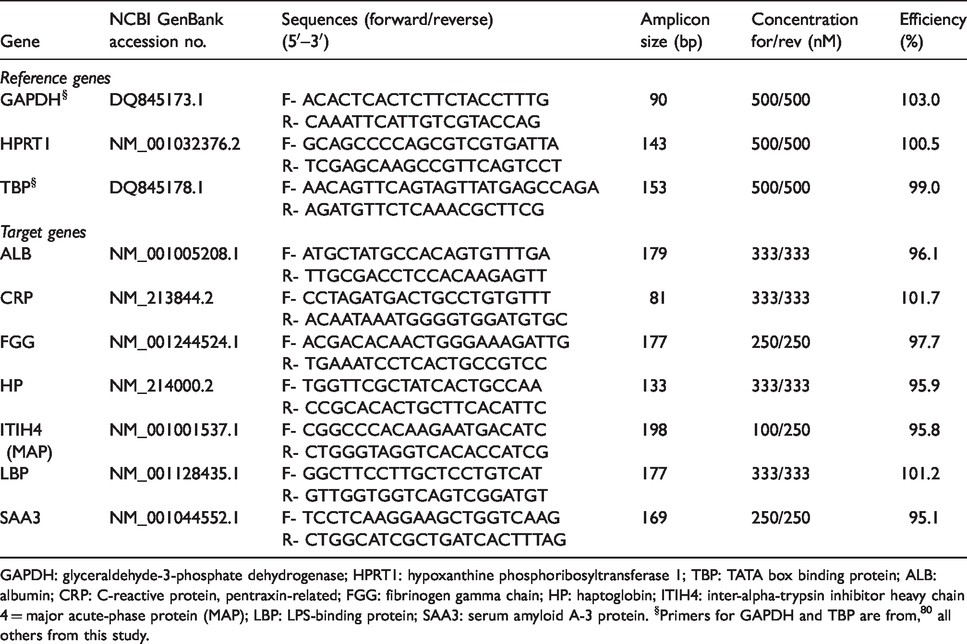
GAPDH: glyceraldehyde-3-phosphate dehydrogenase; HPRT1: hypoxanthine phosphoribosyltransferase 1; TBP: TATA box binding protein; ALB: albumin; CRP: C-reactive protein, pentraxin-related; FGG: fibrinogen gamma chain; HP: haptoglobin; ITIH4: inter-alpha-trypsin inhibitor heavy chain 4 = major acute-phase protein (MAP); LBP: LPS-binding protein; SAA3: serum amyloid A-3 protein. §Primers for GAPDH and TBP are from, 80 all others from this study.
Hepatic free and protein-bound amino acid composition
The combined supernatants of protein precipitation were neutralized with 2 M K2CO3 in the ratio 14:1 and diluted by factor 2 with ultrapure water.
About 5 mg of dried protein pellet was hydrolyzed with 2 ml 6 N HCl with ascorbic acid as antioxidant under N2 atmosphere at 110°C for 20 h. After evaporation to dryness under nitrogen stream at 60°C, hydrolyzed samples were re-dissolved in 0.1 N HCl and diluted by factor 50 with ultrapure water.
Aa concentrations were determined by HPLC using pre-column derivatization and fluorescence detection (1200 series, Agilent Technologies, Waldbronn, Germany). Briefly, Aa were derivatized with ortho-phthaldialdehyde/3-mercaptopropionic acid (OPA) and 9-fluorenylmethoxycarbonyl chloride (FMOC). The separation was carried out on a Hyperclone 5 µm reversed-phase C18 column (250 × 4 mm i.d.) protected by a 4 × 3 mm pre-column (both Phenomenex, Aschaffenburg, Germany) at 40°C. Fluorescence of Aa derivatives was monitored at excitation and emission wavelengths of 340 and 450 nm (OPA) or 266 nm and 305 nm (FMOC), respectively. Aa standard solution from Sigma-Aldrich (A9906, Munich, Germany) was used, and additional standards for Asn, Gln, Trp, Met sulfoxide and Met sulfone were prepared. Because of acid hydrolysis, Asn and Gln were converted to Asp and Glu and analysed as such. Cys and Trp could be not analysed. Met was calculated as the sum of Met, Met sulfoxide and Met sulfone.
Calculations and statistics
The fractional protein synthesis rate (FSR) of total liver proteins, expressed as percent per d, was calculated as follows:
32

Absolute total liver protein synthesis rate (ASR) was calculated by multiplying the absolute organ protein mass (organ mass multiplied by protein content) with the respective FSRmaximum divided by 100.
The analytically determined hepatic concentrations of protein, RNA, DNA were used to derive further parameters of protein metabolism such as RNA to protein ratio as indicator for protein synthesis capacity, protein to DNA ratio for cell size, ASR to RNA ratio, and ASR to DNA ratio for translational efficiency and DNA activity, respectively.
FSR of ALB was calculated according to Ballmer et al. 39 as the ratio between the time-related linear increase in MPE of ALB and the corresponding area under the MPE–time curve of the precursor pool (plasma free l-Phe) multiplied by 100. The ASR of ALB was estimated by multiplying the FSR of ALB with the intravascular ALB mass. The latter was determined by multiplying the measured plasma ALB concentration with the estimated plasma volume (Plasma volume = 1.06 + 0.037 · BM (kg)). 40 Regressions of l-[2H5]-Phe MPE in ALB on time were extrapolated to a zero-MPE to enable estimation of the time of first appearance of the label in the systemic circulation, which is equivalent to the hepatic ALB secretion time.
Data were evaluated by ANOVA with treatment group as fixed factor. Significant treatment effects (p < 0.05) were further assessed by the least significant difference test. The differences in APP concentrations between the time span from −30 min to 180 min relative to starting the infusions were not normally distributed and evaluated by the non-parametric Mann–Whitney
Results
Acute-phase proteins
The concentrations of CRP, MAP, SAA, HP and ALB were not different (
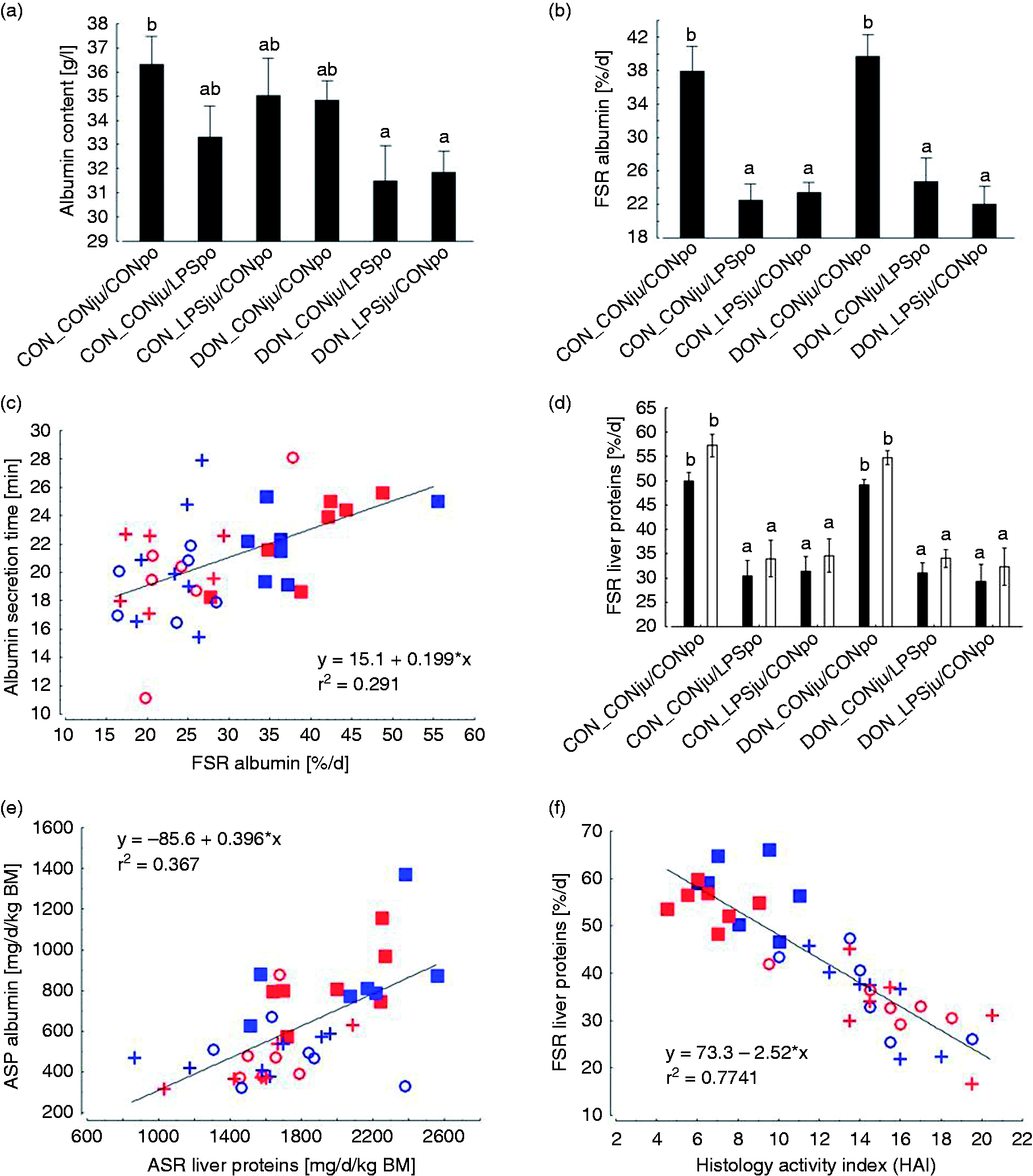
Plasma ALB content [A], fractional synthesis rate (FSR) of ALB [B], linear regression of ALB secretion time vs. ALB FSR [C], minimum (black bars) and maximum (white bars) liver protein FSR [D], linear regression of absolute synthesis rate (ASR) of ALB vs. liver protein ASR [E], and linear regression of hepatic protein FSR vs. liver histology index (data from Renner et al. 42 ) [F] of pigs fed either a control diet (CON) or a diet contaminated mainly with deoxynivalenol (DON) and following either jugular or portal LPS stimulation (LPSju or LPSpo, respectively; CONju and CONpo indicate placebo NaCl infusion). Columns indicate least square means (LSMEANS) and whiskers standard error of means. ab, LSMEANS with no common superscripts are significantly different (p < 0.05).
The relative changes in APP concentrations at 3 h, expressed as percentage of the initial levels at −30 min, were influenced by treatments for all measured APP, except for CRP (Figure 3). MAP levels decreased more pronouncedly after LPS infusion in DON-fed groups compared with both saline groups. Moreover, LPS infusion via the jugular vein resulted in a significantly steeper decrease when compared with portal vein LPS exposure. Such LPS effects on time-related MAP change were not observed in CON-fed groups. Similar to MAP alterations, the SAA concentrations decreased more pronouncedly in LPS-treated pigs fed DON-contaminated feed, although LPS infusion site did not induce an additional difference. A significant decrease in HP concentration was noticed in CON-fed groups exposed to LPS independently of infusion site when compared with the unexposed CON group. In DON-fed groups the LPS infusion via the jugular vein caused a significantly steeper decrease in HP concentration compared with the placebo DON-fed group, while portal LPS exposure caused a decrease in HP content comparable to both CON and DON-fed groups not exposed to LPS. Both DON-fed groups responded to LPS infusions with a decrease of ALB concentrations by approximately 20% compared with CON group not challenged with LPS. A similar decrease was noticed for the CON-fed group exposed to LPS via the jugular vein when compared with the non-LPS-infused CON and DON-fed groups.
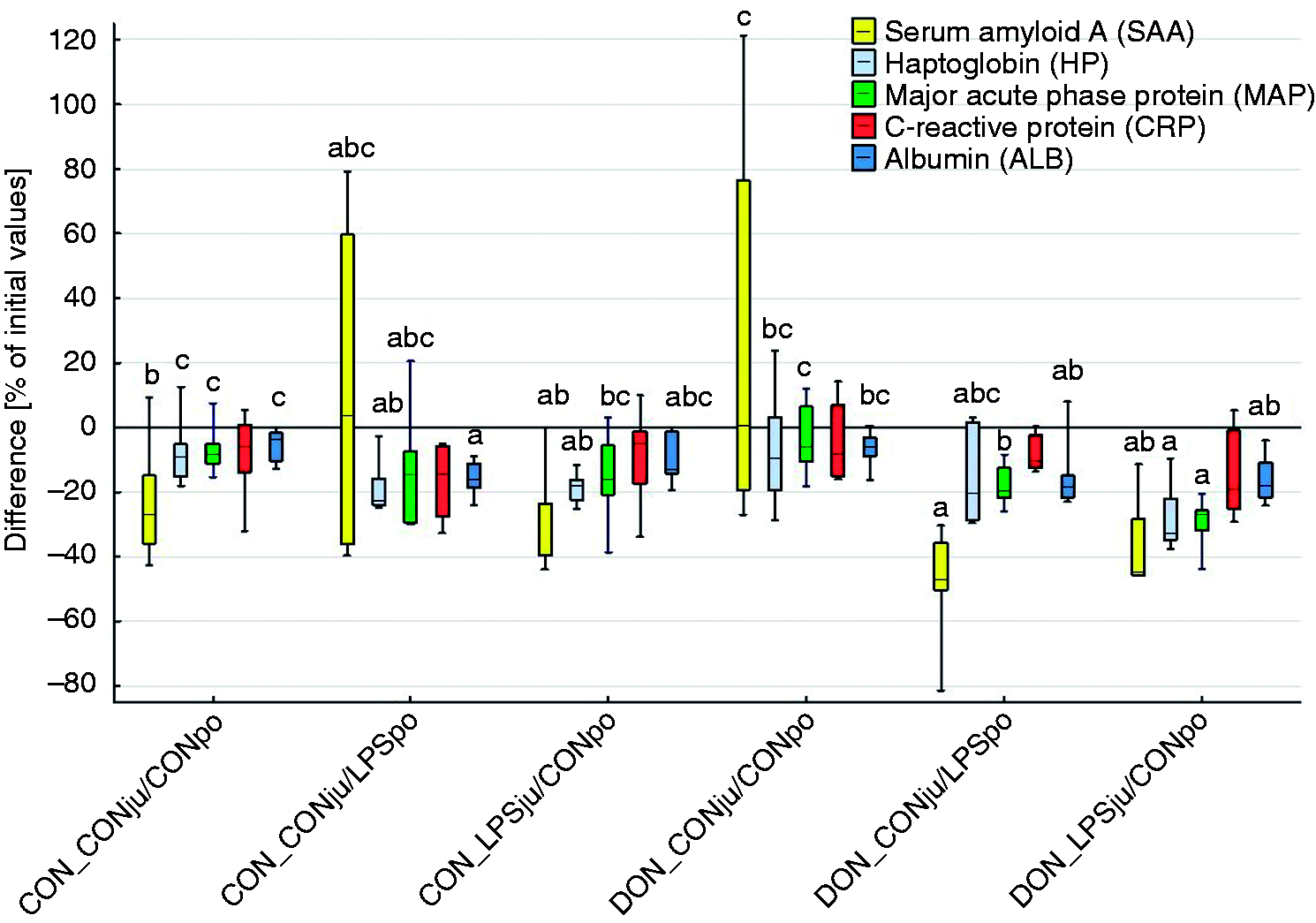
Change of acute-phase protein concentrations over time (-30 min to 180 min relative to infusion start) in heparinized plasma from
ALB synthesis and liver protein metabolism
The MPE of free l-[2H5]-Phe in blood was used as indicator for the precursor pool for ALB synthesis and decreased linearly over time (average
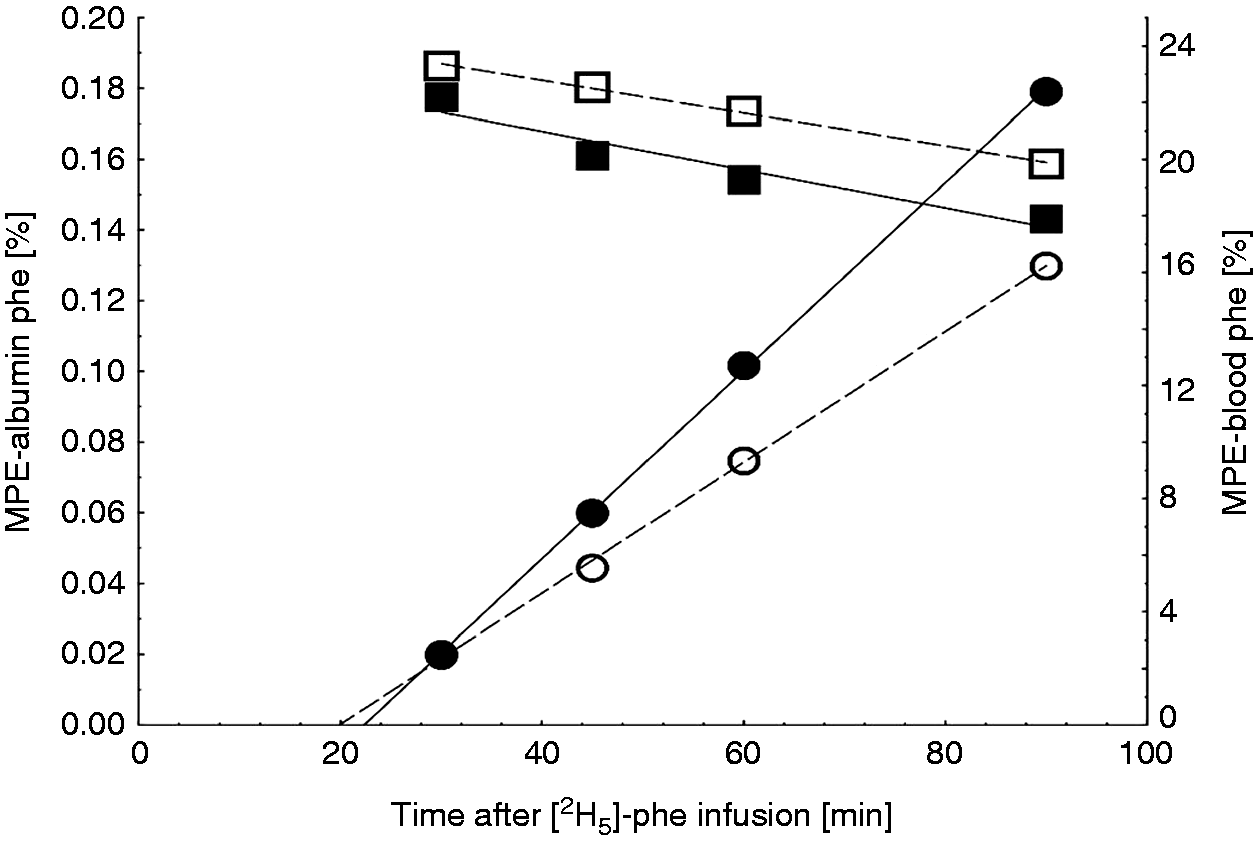
Time-dependent changes in the enrichment of Phe, expressed as molar percent excess (MPE) of l-[2H5]-Phe, in the precursor pool (free Phe in blood, squares) and bound in albumin (circles) exemplarily for 2 CON-fed pigs receiving either physiological saline (■, ●) or LPS treatment (□, ○) via the internal jugular vein. Regressions of MPE in albumin on time were extrapolated to a zero-MPE to enable estimation of the time of first appearance of the label in the systemic circulation which is equivalent to the hepatic albumin secretion time.
Feeding of DON-contaminated diets did not adversely affect ALB synthesis when infused with saline (Figure 2b), whereas LPS treatment induced a significant reduction in ALB synthesis rate irrespective of feeding regimen or LPS infusion site. Compared with placebo-infused CON- and DON-fed groups, the mean absolute decrease amounted to 16% units, which is equivalent to a relative reduction by 40%. The animal-individual ALB secretion time varied between approximately 11 and 28 min and was not influenced by treatments (Table 2, Figure 2c). ALB secretion time increased linearly with ALB FSR (Figure 2c). The slope of this regression suggested a mean increase in secretion time by 0.2 min for each increase in ALB synthesis rate by 1%.
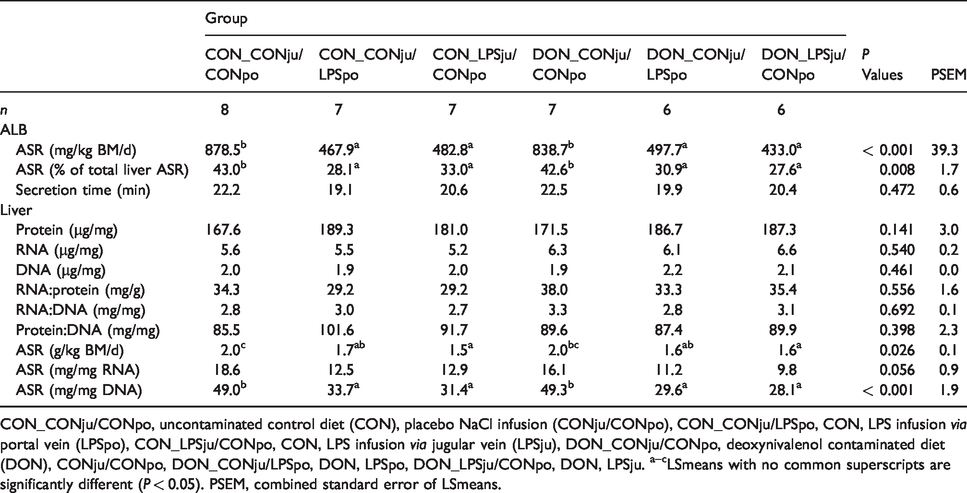
Absolute ALB and total liver protein synthesis rates (ASR) and further liver protein metabolism traits (Least square means, LSmeans).
CON_CONju/CONpo, uncontaminated control diet (CON), placebo NaCl infusion (CONju/CONpo), CON_CONju/LPSpo, CON, LPS infusion
The ASR of ALB, expressed in mg/kg BM and d, revealed similar significance relationships as described for fractional ALB synthesis rate. Thus, the LPS-infused groups synthesized on average approximately 388 mg ALB less than the groups not exposed to LPS (–45%; Table 2).
The FSR of total liver proteins was influenced by treatments in a similar fashion as described for ALB synthesis (Figure 2d). Thus, a mean relative decrease in FSR of 39% was noticed for LPS-infused groups compared with the placebo-infused groups irrespective of whether intracellular or plasma free Aa were considered as precursor pools (FSRminimum and FSRmaximum, respectively). In general, FSRminimum was systematically lower compared with FSRmaximum whereby absolute differences amounted to 6% and 3% for non-challenged and LPS-infused groups, respectively (data not shown).
As FSR of total liver proteins followed similar significance patterns as described for ALB FSR, the proportion of the ALB ASR of total liver ASR was influenced by treatments in a similar manner. Here, the mean proportion of ALB ASR was 13% (30% relative change) higher in saline-exposed groups compared with all four LPS-infused groups (Table 2). A general view of this parameter revealed a variation of the proportion of the ALB ASR of hepatic ASR between approximately 28 and 43%. The slope of the corresponding linear regression supports this magnitude and suggested a mean proportion of approximately 40% (Figure 2e).
Hepatic concentrations of total protein, DNA and RNA were not significantly influenced by treatments (Table 2). Also, protein synthesis capacity, expressed as the ratio between total RNA and protein, protein to DNA ratio as indicator for cell size and RNA to DNA ratio were not affected by treatments. ASR of total liver proteins reached approximately 2 g/kg BM/d in both groups not challenged with LPS, while lower rates (p < 0.05) were noticed in groups infused with LPS via the jugular vein independently of diet type. LPS application via the portal vein resulted in a less pronounced depression in ASR (Table 2). A trend for a decreased translational efficiency, i.e. the ASR to RNA ratio, was noticed for the LPS-infused groups. In contrast, the DNA activity, i.e. the ratio between ASR and DNA, revealed a clear picture as all LPS-infused groups synthesized significantly less total hepatic protein when compared with the unchallenged groups (–38% on average).
Renner et al. 42 could already demonstrate a significant increase in histopathological alterations in the same pigs as used in the present investigations. The severity of these lesions was evaluated together with the FSR of total hepatic proteins through linear regression. Each increase in severity of histopathological lesions by one score-point was associated to a mean decrease of FSR of total hepatic proteins by 2.5% as suggested by the corresponding linear regression (Figure 2f).
Hepatic free and protein-bound amino acid composition
The concentrations of cysteine and alpha-aminoadipic acid were lower in LPS-infused groups in the supernatants containing the free Aa with a similar trend for Asn (Figure 5). The remaining Aa and metabolites were not influenced by treatments (Supplemental Table 1). The concentration of cysteine in the hepatic free Aa precursor pool was linearly related to the FSR of ALB and hepatic total protein (Figure 6).
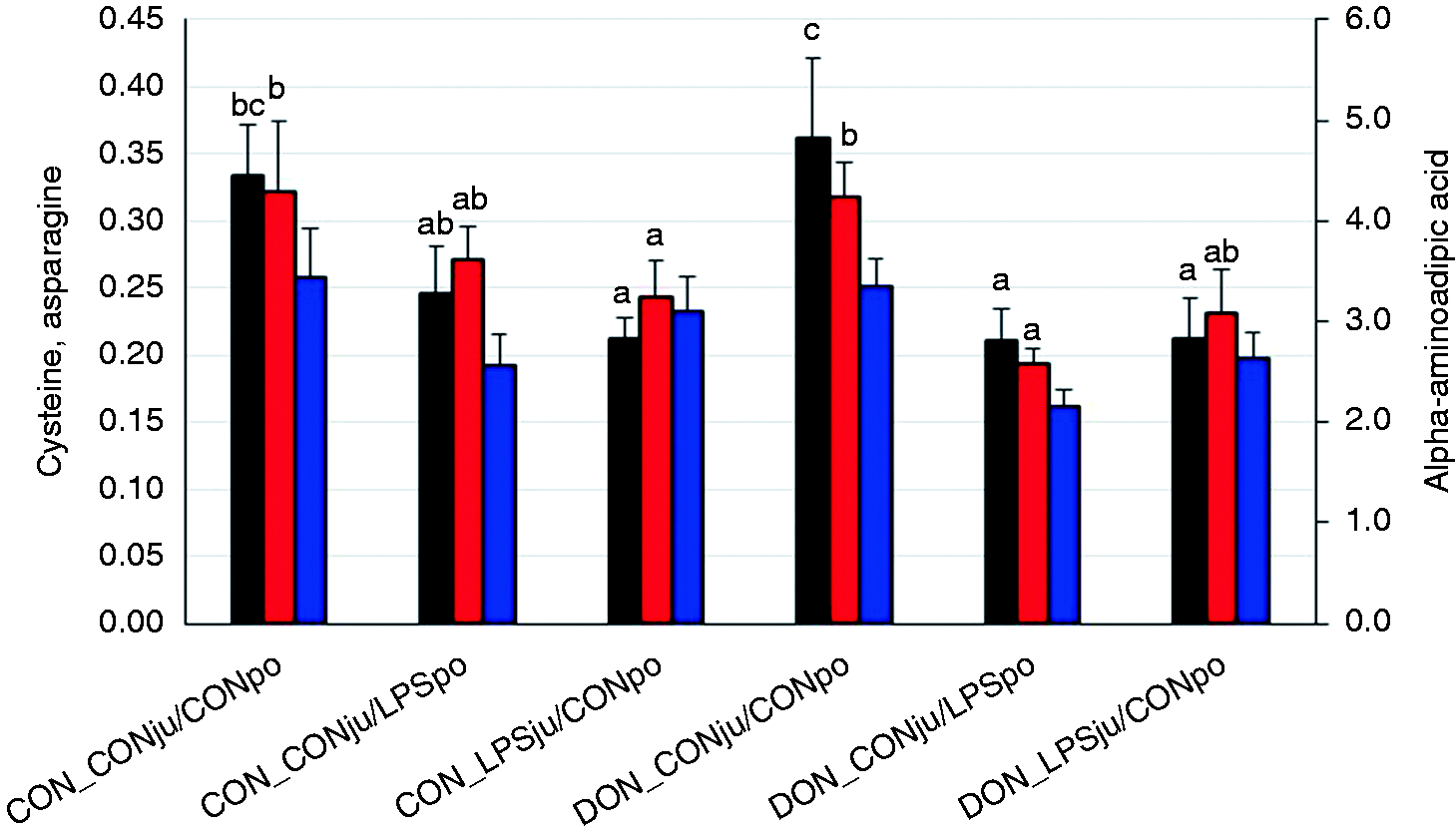
Aa concentrations in supernatants of liver homogenates (free amino acids, µMol/g) of pigs fed either a control diet (CON) or a diet mainly contaminated with deoxynivalenol (DON) and following either jugular or portal LPS stimulation (LPSju or LPSpo, respectively; CONju and CONpo indicate placebo NaCl infusion). Columns represent least square means (LSMEANS +SE). Different lower-case letters close to the upper whiskers indicate significant differences between treatment groups for individual amino acids (
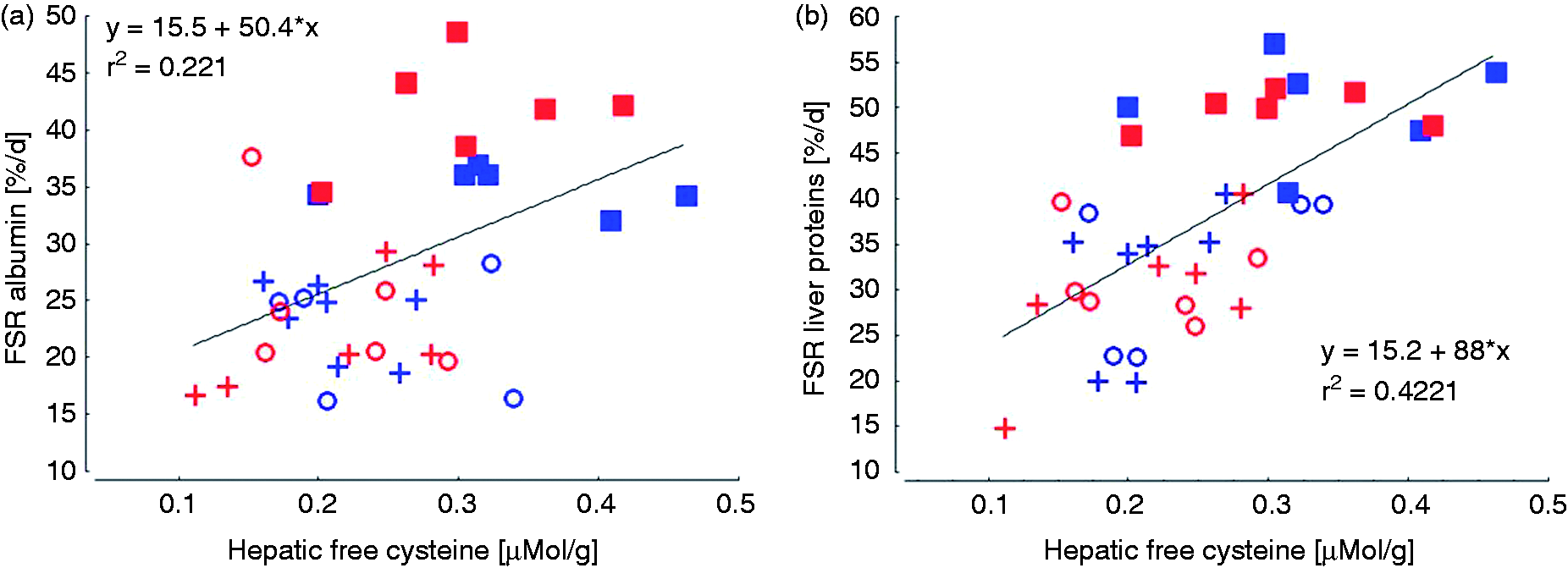
Linear regression of cystine concentration in the hepatic free Aa precursor pool on fractional albumin synthesis rate (FSR) [A] and on FSR of total liver proteins [B].
The concentrations of His, Asp, Ala, Val and Leu in the protein hydrolysate tended to be increased in LPS-exposed pigs compared with the non-exposed groups (P < 0.1, Figure 7). The concentrations of the other proteinogenic Aa remained unaffected by any of the treatments (Supplemental Table 2).
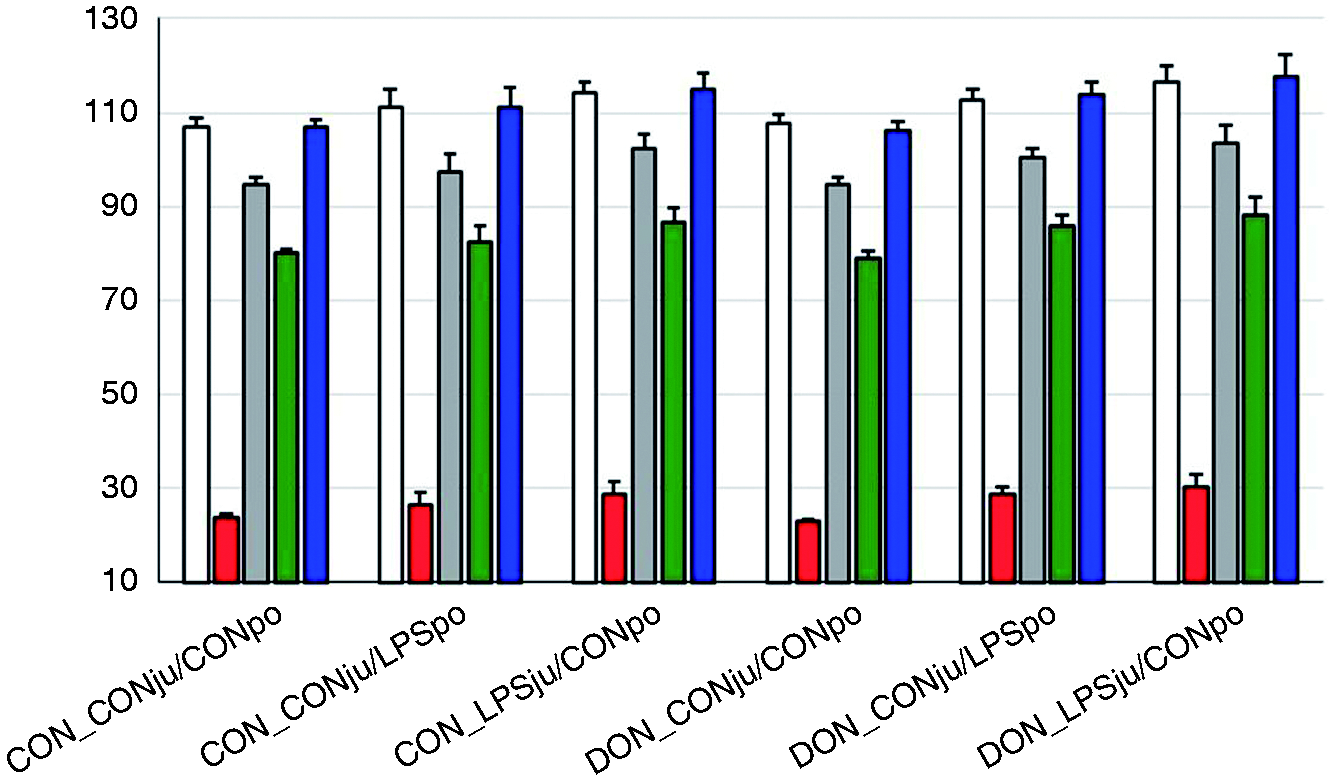
Aa concentrations in hydrolysates of total liver proteins (µMol/g) of pigs fed either a control diet (CON) or a diet mainly contaminated with deoxynivalenol (DON) and following either jugular or portal LPS stimulation (LPSju or LPSpo, respectively; CONju and CONpo indicate placebo NaCl infusion). Columns indicate least square means (LSMEANS+SE) and whiskers standard errors of LSMEANS. White: Asp (
Hepatic gene expression of APP
The mRNA abundance of the negative APP ALB was not influenced by any of the treatments (Table 3). In contrast, the mRNA expression of all measured positive APP was stimulated by LPS application independent of the infusion site, albeit the order of magnitude differed substantially (Table 3). The SAA3 mRNA abundance was much more induced compared with fibrinogen (Fib), HP, MAP, CRP, and LBP mRNA. Interestingly, the mRNA abundance of only SAA3 was significantly decreased in DON-fed pigs not challenged with LPS compared with their CON-fed counterparts (Table 3), while such effects were not observed for the other APP.
Relative mRNA abundance of hepatic acute-phase proteins determined at 195 min after LPS infusion start (Least square means, LSmeans)

CON_CONju/CONpo, uncontaminated control diet (CON), placebo NaCl infusion (CONju/CONpo), CON_CONju/LPSpo, CON, LPS infusion
Discussion
The liver plays a central role in metabolism and supports extra-hepatic tissues by providing nutrients and energy for metabolic purposes. 43 On the other hand, it is also an important secondary immunological organ and forms a second line of defence against Ags, xenobiotics like DON, and other substances which overcome the gastro-enteral mucosal barrier at the first pass. Second-pass delivery of these substances to the liver might result from peripheral sources. The defence mechanisms of the liver are directed to cope with those substances in a way that mediates either tolerance or that initiates a distinct immune response 8 depending on the type of substance, dosage, exposure duration and possibly the entry route to the liver. Thus, we hypothesized that a dietary pre-exposure to DON would modify the pre- or post-hepatic LPS-induced APR differently, with consequences for the ALB synthesis.
Significantly decreased FSR of ALB compared with the physiological saline treated control group was noticed under the conditions of an APR induced via a post-hepatic LPS challenge. 22 A parallel chronic oral or acute i.v. DON exposure did not modify the LPS-induced drop in ALB FSR. Thus, the LPS-triggered APR dominated the ALB synthesis depressing effect irrespective of a simultaneous DON exposure. 22 However, LPS might be increasingly transferred through an intestinal barrier compromised by DON as frequently discussed,44–46 targeting the liver in the first pass. The human liver harbours approximately 80% of all body macrophages and is patrolled additionally by blood monocytes, the two cell populations mainly contributing to LPS clearance and induction of APR and SI. 47 Consequently, we assumed that an LPS entry via the portal route would have consequences on the outcome of APR, including ALB synthesis, other than a peripheral (jugular) LPS entry. There were indeed some hints that the magnitude of SI was more pronounced in DON-fed pigs challenged with LPS via the jugular route compared with the hepatic portal route 29 although the SI pacemaker cytokine TNF-α kinetics did not reflect similarly directed effects but rather responded uniformly to LPS irrespective of LPS infusion site or DON exposure. 30 The extent of the SI-associated lactic acidosis was significantly more pronounced following jugular LPS administration, and was discussed as consequence of a less pronounced post-hepatic LPS clearance capacity. 29 A chronic metabolic acidosis induced by an ammonium chloride exposure in healthy male humans induced a decreased ALB FSR, an increased urinary nitrogen excretion and a negative nitrogen balance. 48 Acute metabolic acidosis in healthy human subjects revealed an unchanged ALB FSR, but a decreased muscle FSR, which explained the negative nitrogen balance observed under both chronic and acute metabolic acidotic conditions. 49 Taken together, both chronic and acute metabolic acidosis might affect muscle protein turnover and hepatic ALB synthesis independently of other SI-associated pathophysiological alterations. However, under the conditions of the present experiment, the more pronounced lactic acidosis in pigs exposed to LPS via the jugular route 29 did not exacerbate the SI-related decrease in ALB synthesis when compared with the portal application route.
Toxic effects of alcohol include distorted mitochondria and alteration of the organizational arrangement of the endoplasmic reticulum, ultimately resulting in a disruption of the protein and consequently ALB synthesis machinery. 50 Therefore, acute hepatocyte lesions induced either directly by toxins or secondarily by an exuberant innate immune response might reduce the hepatocellular integrity and thus capacity for ALB synthesis. Although functional capacity for respiration and Ca2+ accumulation of isolated liver mitochondria was not impaired by any of the treatments, the histopathological examination revealed significant lesions including haemorrhage and inflammation primarily. 42 Under the conditions of the present experiment the infiltration of the porcine liver by neutrophilic and eosinophilic granulocytes was observed across liver lobules, 42 suggesting a uniform spreading of inflammation and inflammatory mediators. A SI in rats induced by s.c. turpentine injection caused a patchy pattern of immuno-stained ALB in perfused rat livers which reflected the hepatic architecture of the acinus with positively stained hepatocytes around the central veins. This result was interpreted as an ALB synthesis breakdown in a zonal pattern. 51 An s.c. injection of rats with IL-1β was shown to induce SI and a depression of FSR of ALB. 52 However, the absence of immuno-stained ALB in periportal fields was discussed as a consequence of a zonal IL-1β gradient rather than resulting from the oxygen and nutrient gradient down to the central veins. 51 Based on the here established strong association between the histopathological cumulative score, largely determined by the uniform hepatic granulocyte infiltration, and the FSR of total liver proteins, it seems reasonable to conclude that functional impairment of the liver to synthesize ALB was associated to local inflammatory events.
ALB secretion time indicates the time required for synthesis and export of ALB by hepatocytes and does consequently provide further information on these processes as influenced by SI. The shortening in ALB secretion time observed with decreasing ALB FSR caused by SI might be interpreted as a consequence of an acute loss of hepatocellular integrity releasing labelled ALB molecules in the circulation, which could also contribute to an early depletion of hepatocytes from ALB. Further support for a compromised integrity of hepatocyte membranes comes from the increased peripheral activities of aspartate aminotransferase, γ-glutamyl-transferase and alkaline phosphatase. 42 Moreover, genes involved in protein export were down-regulated under the conditions of the SI, 53 further supporting the idea of a passive loss of hepatocellular proteins across damaged cell membranes.
ALB synthesis is regarded as highly sensitive to altered Aa levels in general and to Trp availability in the free-protein synthesis precursor pool in particular. 54 Moreover, Trp was discussed with regard to restoring ALB synthesis in alcohol induced depression of ALB synthesis. 54 , 55 Therefore, we assumed that the drop in ALB synthesis as determined in the present experiment upon LPS infusion could be due to an insufficient availability of free Trp and Aa usable for ALB synthesis. However, based on the Trp levels analysed in the hepatic free Aa precursor pool, we did not find indications of an LPS-induced limitation for this Aa. These results seem to contradict the increased mRNA expression of indolamine 2,3-dioxygenase (IDO) in LPS-treated pigs. 53 This ubiquitous enzyme catalyses the degradation of Trp to kynurenine and is mainly induced by pro-inflammatory stimuli. 56 , 57 In contrast, the liver-specific Trp 2,3-dioxygenase (TDO), which catalyses the same reaction, 58 was down-regulated possibly as an attempt to save Trp for the synthesis of APP. 53
In addition to the not well-understood specific Trp influence, other Aa effects arising from the Aa composition of ALB have be taken in consideration. In contrast to other APP such as CRP, Fib and HP, the ALB molecule contains much higher proportions of Cys. 15 , 59 , 60 Considering that ALB is a weak negative APP but accounts most for the total amount of all hepatic export proteins, a shortage in Cys availability might limit ALB synthesis. Out of the Aa analysed in the hepatic free Aa precursor pool, only Cys and the Lys degradation metabolite alpha-aminoadipic acid were significantly reduced upon LPS infusion, whereby the effects were more pronounced in the case of Cys. While a reduced α-aminoadipic acid content could be interpreted as a decreased Lys degradation to save this Aa for protein synthesis purposes, the LPS-induced reduction in Cys could indicate a certain limitation particularly for ALB synthesis. This conclusion is supported by the positive linear relationship between the Cys concentration in the free Aa precursor pool and the FSR of ALB and total hepatic proteins suggesting the FSR to be linearly dependent on the availability of free Cys. Interestingly, this linear relationship appeared to be even stronger for FSR of total proteins compared with ALB, which suggests that other hepatic proteins also rely on an adequate Cys availability, such as glutathione (GSH). One of the Aa of this tripeptide is Cys. GSH plays an important role in the enzymatic anti-oxidative defence system 61 and is rapidly up-regulated in various tissues of LPS-challenged mice. 62 Further information on the availability of protein synthesis precursor pool, i.e. free Aa, both for ALB and total hepatic protein synthesis can be gathered from the differences between the minimum and maximum FSR of total liver proteins. This difference was approximately 50% lower (6% vs. 3%) in LPS-challenged animals independently of LPS infusion site and feeding regimen, which might suggest that either more plasma-originating, or fewer hepatocellular free Aa were used or available for protein synthesis in order to meet the actual needs. This conclusion is also consistent with the idea of a partial mixing of both precursor pools due to hepatocellular lesions.
The reasons for the reduced availability of Cys might not only be based on a higher actual requirement but might also be caused by a reduced perfusion of hepatic sinusoids, ultimately resulting in a decreased nutrient delivery to the hepatocytes. SI is associated with a redistribution of intrahepatic blood flow due to the interactions between Kupffer cells, infiltrating leukocytes and sinusoidal endothelial cells resulting in a decreased perfusion and blood flow velocity in the liver sinusoids. 63 Together with the disseminated intravascular coagulation and the associated formation of micro-thrombi, the ischemia and organ damage are exacerbated. 64
In contrast to the Aa pattern of the precursor pool, the Aa composition of the protein hydrolysate did not provide hints at specific associations to ALB. However, in spite of the LPS-induced trend to increase the concentrations of His, Asp, Ala, Val and Leu, these effects appeared to reflect systematic LPS-induced alterations in hepatic proteins comprising both constitutive, in
Further support for the particular role of sulphur-containing Aa including Met and Cys for FSR of ALB comes from an experiment where an insufficient dietary supply with these Aa significantly reduced ALB FSR in LPS-challenged pigs. 65 In contrast, ALB FSR was not compromised in pigs challenged with LPS but adequately supplied with Met and Cys when compared with the non-challenged control group. 65
APR-Induced alterations in expression of ALB mRNA might also contribute to the observed marked decrease in FSR of ALB. However, we failed to detect any decrease in ALB mRNA expression due to any of the treatments 195 min after the initiation of the infusions. Others have examined the kinetic relationships between ALB mRNA expression and ALB synthesis in more detail. The ratio between murine hepatic ALB synthesis rate and mRNA levels remained relatively stable over time after turpentine induced APR. Based on these results it was concluded that APP and ALB synthesis adjustment during APR is not regulated by the rapidity of translation of individual mRNA molecules but by the absolute mRNA level. This, in turn, might be controlled by mRNA synthesis and/or degradation.
66
ALB mRNA levels were down-regulated to reach a nadir 36 h after turpentine injection. The decrease started at the first liver sampling point at 6 h p.i. Positive APP such as transferrin, fibrinogen and α1-acid glycoprotein peaked markedly in their mRNA abundance after 36 h, 24 h and 36 h, respectively, and were characterized by different half-lives. Similar findings were reported in turpentine-injected and
We assessed the ALB mRNA expression solely 195 min after the beginning of the LPS challenge. Therefore, this time span might have been too short to induce an effect on ALB mRNA expression. Consequently, the observed marked early drop in ALB FSR might have been caused by other mechanisms as discussed above.
In contrast to ALB mRNA, mRNA levels of all other APP were significantly up-regulated ∼3 h after starting LPS infusions, although the corresponding APP did not increase in systemic circulation within this period but rather showed significant decreases in some cases. This finding was somewhat unexpected, as most of them would be expected to be more or less induced in pigs as a result of an APR caused by several sterile and infectious stimuli.69–74 In discussing these time-dependent increases of APP with the decreases/no changes observed in the present experiment it needs to be considered that the first blood sample was taken 24 h after the initiation of the APR in most of the published porcine studies, whereas we specifically examined the first 3 h of the APR. Similarly to our study, Frank et al. 75 also failed to detect increases of CRP, HP or ceruloplasmin (Cp) after i.m. injection of 25 or 50 µg LPS/kg BM. Only for SAA was a significant increase noticed. However, APPs were measured only 48 h after the LPS challenge, which could also mean that kinetics of CRP, HP and Cp were different from that of SAA and that peak responses were possibly not recognized.
Only a few experiments were identified which reported APP concentrations for time spans markedly lower than 24 h after the challenge. An experimental infection of pigs with
The unchanged or even decreased levels of the analysed positive APP reported herein irrespective of an up-regulated mRNA expression might be the result of a time lag required for translation. However, based on the present and other results
20
,
22
suggesting that only approximately 20 min are required to synthesize and export both ALB and Fib, and that this time span might also apply for other hepatic export proteins, the translation step does not appear to be the reason for the decreased or unaltered levels of APP. Therefore, an assumed initial increase in synthesis of these APPs might have been insufficient to compensate the level of their consumption; in terms of protein turnover, the level of degradation/elimination and/or extravasation might have been higher than the level of their
We also tested the hypothesis that a dietary pre-exposure to DON would increase the transfer of intestinal-originating LPS through a DON-associated destruction of the mucosal barrier, as discussed above, whereby hepatic responsiveness to LPS and DON with consequences for hepatic ALB synthesis could be altered. However, no depressing effects on ALB and total liver protein FSR were found in the present experiment when looking at the consequences of DON exposure alone without an artificial LPS challenge. These results are in contrast to earlier findings where chronic oral DON exposure significantly depressed 20 or even stimulated ALB FSR. 22 While a depression of ALB FSR is to be expected based on the mode of action of DON, a stimulation of FSR is more difficult to discuss. 22 Although the latter two, and this experiment applied a restricted feeding regimen over 4–5 wk to avoid dietary effects following a DON-induced anorexia and used male castrated pigs comparable in age, diets contained different DON levels. While general effects of the particular experiments cannot be excluded, a DON dose-related effect on ALB FSR might be deduced. Thus, a depression, no-effect and stimulation of ALB FSR were associated with dietary DON concentrations of 5.7 mg/kg feed, 4.6 mg/kg and 3.1 mg/kg, respectively.
Besides the observation that DON alone failed to influence hepatic ALB and total protein synthesis, additional modifying effects of LPS transferred through a DON-compromised mucosal barrier can be excluded as pigs fed the DON-contaminated diet showed LPS levels both in hepatic portal and jugular vein comparable to those measured in the CON-fed group. 77 This finding is in line with earlier results demonstrating that a chronic DON exposure fails to influence mucosal tight junction proteins. 78 , 79
Conclusions
The marked depression of ALB FSR observed 195 min after initiating of an APR through a pre- or post-hepatic LPS challenge was not associated to a down-regulated ALB mRNA expression but by a reduced cysteine availability in the protein synthesis precursor pool. Dietary chronic pre-exposure with DON neither depressed ALB and total hepatic protein FSR in unchallenged pigs, nor gave rise to any interactive effects, with the LPS-induced APR suggesting that observed adverse effects on liver protein synthesis were dominated by the consequences of the SI.
In contrast, the mRNA expression of the positive APPs Fib, HP, MAP, CRP, LBP and SAA was significantly up-regulated due to LPS stimulation independently of feeding group and infusion site. This up-regulation was not accompanied by increases in plasma levels of the corresponding APPs, which could hint at an imbalance between synthesis and consumption.
Kinetic studies with frequent blood and liver sample collections covering a time span of several days are required to fully understand the regulation of APP in the course of a systemic inflammatory response in the pig.
Supplemental Material
sj-pdf-1-ini-10.1177_1753425920937778 - Supplemental material for Oral exposure of pigs to the mycotoxin deoxynivalenol does not modulate the hepatic albumin synthesis during a LPS-induced acute-phase reaction
Supplemental material, sj-pdf-1-ini-10.1177_1753425920937778 for Oral exposure of pigs to the mycotoxin deoxynivalenol does not modulate the hepatic albumin synthesis during a LPS-induced acute-phase reaction by Sven Dänicke, Erik Bannert, Tanja Tesch, Susanne Kersten, Jana Frahm, Susanne Bühler, Helga Sauerwein, Solvig Görs, Stefan Kahlert, Hermann-Josef Rothkötter, Cornelia C Metges and Jeannette Kluess in Innate Immunity
Footnotes
Acknowledgements
The authors would like to thank Nicola Mickenautsch for performing the isotope analyses.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported financially by Deutsche Forschungsgemeinschaft (DFG DA 558/1-4 and RO 743/3-3).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
