Abstract
The present study was conducted to determine whether avian reovirus (ARV) activates the phosphatidylinositol 3-kinase-dependent Akt (PI3K/Akt) pathway according to the PXXP or YXXXM motifs of σA and σNS proteins. Gene splicing by overlap extension PCR was used to change the PXXP or YXXXM motifs of the σA and σNS genes. Plasmid constructs that contain mutant σA and σNS genes were generated and transfected into Vero cells, and the expression levels of the corresponding genes were quantified according to immunofluorescence and Western blot analyses. The Akt phosphorylation (P-Akt) profile of the transfected Vero cells was examined by flow cytometry and Western blot. The results showed that the σA and σNS genes were expressed in the Vero cells, and P-Akt expression in the σA mutant groups (amino acids 110–114 and 114–117) was markedly decreased. The results indicated that the σA protein of ARV activates the PI3K/Akt pathway via the PXXP motif. The results of this study reveal the mechanisms by which ARV manipulates the cellular signal transduction pathways, which may provide new ideas for novel drug targets.
Introduction
Avian reovirus (ARV) is one of the most important avian viruses, causing clinical diseases in poultry worldwide and resulting in severe economic losses.1,2 ARV-affected flocks commonly suffer viral arthritis or tenosynovitis, runting-stunting syndrome (RSS), enteric disease, immunosuppression and malabsorption syndrome.2–5 In the early stages of ARV infection, ARV activates the phosphatidylinositol 3-kinase-dependent Akt (PI3K/Akt) signalling axis in Vero cells 6 – a pathway that is associated with cell survival, proliferation, migration, differentiation and apoptosis.7,8
PXXP or YXXXM/YXXM motifs are present in a number of viral and cellular proteins involved in PI3K signaling and form extended helices that bind to SH domains on the p85 subunit of PI3K.9–15 Therefore, as our previous research review 16 showed that various amino acid sequence have PXXP/YXXXM/YXXM motifs, σA, σNS, μA, μB and μNS of ARV were speculated to be involved in PI3K signalling. The results showed that σA and σNS-expressing cells had higher P-Akt levels than the pcAGEN-expressing cells and that in the cells expressing other proteins (i.e. μA, μB and μNS), pretreatment with the PI3K inhibitor LY294002 inhibited Akt phosphorylation in σA- and σNS-expressing cells. 16 These results indicate that the σA and σNS proteins can activate the PI3K/Akt pathway.
Mutant the PXXP motif of NS1 protein from Influenza A virus or mutant the YXXM motif of envelope protein of avian leukosis virus results in loss of PI3K/Akt pathway activation.15,17 According to amino acid sequence analysis of the σA and σNS genes, the PXXP and YXXXM motifs are conserved between different ARV strains. The aim of the current study was to determine whether the σA and σNS proteins affect the activation of the PI3K/Akt pathway in vivo via the PXXP or YXXXM motifs. To accomplish this objective, we mutated the PXXP or YXXXM motifs of σA and σNS genes. Plasmid constructs containing mutant σA and σNS genes were generated and transfected into Vero cells, and the expression levels of the σA and σNS genes were quantified according to immunofluorescence and Western blot analysis. The Akt phosphorylation (P-Akt) profiles of the transfected Vero cells were examined by flow cytometry and Western blot analysis.
Materials and methods
Plasmids and primers
The plasmids σA-pcAGEN and σNS-pcAGEN were generated by our lab.
16
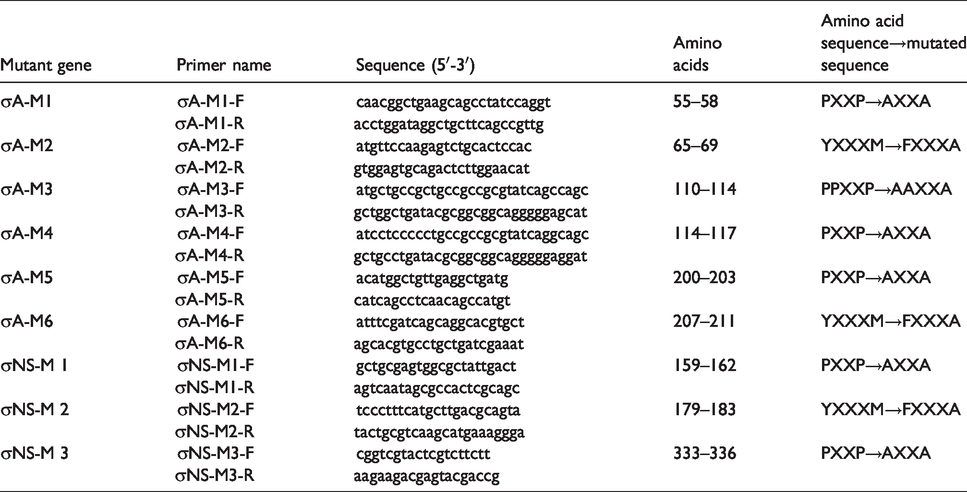
Nine pairs of primers were designed to mutate the PXXP or YXXXM motifs of the σA and σNS genes (Table 1). The primer sequences are presented in Table 1. The red colours represent the mutant bases. The complete σA gene (1248 bp) was amplified using the primers σA-F (5′-gatgatctcgag
Primer sequences.
RNA extraction and RT-PCR amplification of the mutant σA and σNS genes
Genomic RNA was extracted from 200 µl of ARV using an EasyPure viral DNA/RNA kit (Transgen, Beijing, PR China) according to the protocol suggested by the manufacturer.
RT-PCR was performed using an RNA LA PCR kit (Takara). Gene splicing by overlap extension PCR was used to mutate σA and σNS genes. To amplify the σA-M1 gene (σA gene mutant 1), the PCR protocol consisted of three rounds of PCR amplification. The first PCR amplification used σA-F and σA-M1-R primers, with σA-pcAGEN as the template; the second PCR amplification used σA-M1-F and σA-R primers, with σA-pcAGEN as the template. The PCR products of these two amplifications were obtained via gel extraction. The third PCR amplification used σA-F and σA-R primers with the gel extraction purification product of the first and second PCR used as the template. The PCR product was then obtained via gel extraction.
We used the same method to amplify the other mutant genes: σA-M2, σA-M3, σA-M4, σA-M5, σA-M6, σNS-M1, σNS-M2 and σNS-M3.
Recombinant plasmid construction
The mutant σA and σNS gene products were cloned into pMD18-T cloning vectors (Takara) according to the manufacturer’s instructions. The constructed recombinant plasmids were designated σA-M1-pMD18T, σA-M2-pMD18T, σA-M3-pMD18T, σA-M4-pMD18T, σA-M5-pMD18T, σA-M6-pMD18T, σNS-M1-pMD18T, σNS-M2-pMD18T and σNS-M3-pMD18T. The plasmids were digested with XhoI and NotI enzymes (Takara) and were then ligated into the corresponding sites of pcAGEN expression vectors before being transformed into competent Escherichia coli cells (DH5α). Positive colonies, which were designated σA-M1-pcAGEN, σA-M2-pcAGEN, σA-M3-pMD18T, σA-M4-pcAGEN, σA-M5-pcAGEN, σA-M6-pcAGEN, σNS-M1-pcAGEN, σNS-M2-pcAGEN and σNS-M3-pcAGEN, were identified by PCR and double digestion and sequenced by Invitrogen (Guangzhou, PR China).
Expression of σA and σNS proteins
Plasmids (pcAGEN, σA-pcAGEN, σNS-pcAGEN, σA-M1-pcAGEN, σA-M2-pcAGEN, σA-M3-pMD18T, σA-M4-pcAGEN, σA-M5-pcAGEN, σA-M6-pcAGEN, σNS-M1-pcAGEN, σNS-M2-pcAGEN and σNS-M3-pcAGEN) were extracted using a plasmid mini kit (Omega Bio-Tek, Norcross, GA). Vero cells were seeded onto 6- or 24-well cell culture plates and were then transfected with 2.5 or 0.5 µg of the appropriate expression vectors using Lipofectamine® 3000 transfection reagent (Invitrogen) according to the manufacturer’s instructions.
Immunofluorescence and Western blot analysis to quantify protein expression
After the cells were seeded onto 24-well plates and allowed to adhere, Vero cells were transfected with various plasmids for 6 h and were then washed three times with PBS. The cells were then fixed with cold methanol for 10 min at room temperature, washed three times with PBS and blocked for 1 h in 10% normal goat serum (Abcam, Cambridge, UK) in PBS containing 0.5% Triton X-100 (Sigma–Aldrich, St Louis, MO). The cells were then incubated with primary Abs against ARV 18 overnight at 4°C. Following three 5-min washes with PBST, the cells were incubated with fluorescently labelled secondary Abs (Alexa Fluor; Abcam) for 60 min at room temperature.
Vero cells seeded onto six-well plates were harvested, and their cell lysates were used for Western blot analysis to detect σA and σNS protein expression, as described by Xie et al. 18 Proteins were visualized using an enhanced chemiluminescence reagent (Bio-Rad, Hercules, CA) and were detected using a Bio-Rad ChemiDoc MP Imaging System.
Flow cytometry and Western blot analysis of P-Akt expression
For flow cytometry and Western blot analyses of P-Akt expression, Vero cells seeded onto six-well plates were harvested following transfection with the corresponding plasmids for 6 h. The cells were then subjected to flow cytometry and Western blot analysis to detect P-Akt expression.
For flow cytometry analysis, the transfected cells were detached from the culture plates via incubation with Accutase (Sigma–Aldrich) for 5 min. Then, the cells were washed with PBS and fixed and permeabilized by incubation with Cytofix/Cytoperm solution (BD Biosciences, Franklin Lakes, NJ) at 4°C for 20 min. Next, the cells were washed twice with FACS buffer (0.5% BSA, 0.01% sodium azide in DPBS) and then incubated at 4°C for 30 min with a rabbit Ab (Cell Signaling Technology, Danvers, MA) against P-Akt. After this incubation, the cells were washed twice with FACS buffer and then incubated at 4°C for 30 min with goat anti-rabbit IgG conjugated to Alexa Fluor® 488 (Abcam). After staining, the cells were washed twice with FACS buffer and re-suspended in FACS buffer for analysis. The samples were then analysed via flow cytometry (Beckman Coulter, Brea, CA).
Vero cells seeded onto six-well plates were harvested, and their cell lysates were subjected to Western blot analysis to detect P-Akt expression, as described by Wang et al. 19
All flow cytometry and Western blot analyses were repeated three times.
Results
Recombinant plasmid construction
The recombinant plasmids σA-M1-pcAGEN, σA-M2-pcAGEN, σA-M3-pMD18T, σA-M4-pcAGEN, σA-M5-pcAGEN, σA-M6-pcAGEN, σNS-M1-pcAGEN, σNS-M2-pcAGEN and σNS-M3-pcAGEN were first subjected to PCR amplification (Figure 1) and double digestion (Figure 2). Then, DNA sequencing was performed to ensure that the recombinant plasmids contained intact mutant σA and σNS genes, validating that the recombinant plasmids were successfully constructed.
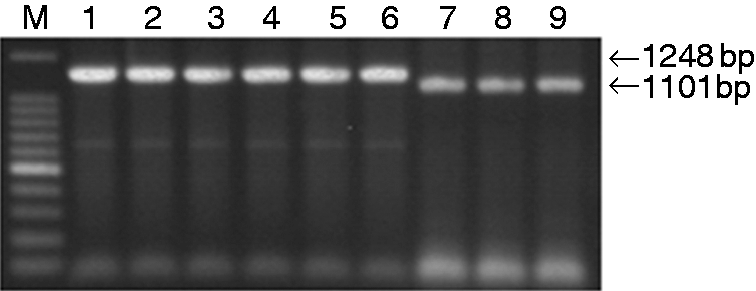
Identification of the recombinant plasmids by PCR. Lane M: 100-bp DNA ladder; Lane 1: σA-M1-pcAGEN; Lane 2: σA-M2-pcAGEN; Lane 3: σA-M3-pcAGEN; Lane 4: σA-M4-pcAGEN; Lane 5: σA-M5-pcAGEN; Lane 6: σA-M6-pcAGEN; Lane 7: σNS-M1-pcAGEN; Lane 8: σNS-M2-pcAGEN; Lane 9: σNS-M3-pcAGEN.
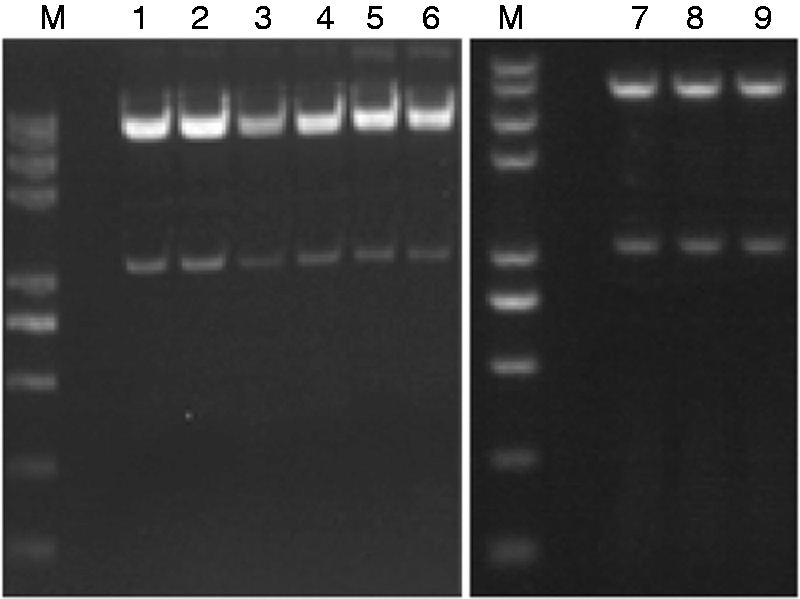
Identification of the recombinant plasmids by double digests. Lane M: Trans2K Plus II DNA Marker; Lane 1: σA-M1-pcAGEN; Lane 2: σA-M2-pcAGEN; Lane 3: σA-M3-pcAGEN; Lane 4: σA-M4-pcAGEN; Lane 5: σA-M5-pcAGEN; Lane 6: σA-M6-pcAGEN; Lane 7: σNS-M1-pcAGEN; Lane 8: σNS-M2-pcAGEN; Lane 9: σNS-M3-pcAGEN.
σA and σNS protein expression
As shown by the immunofluorescence and Western blot analyses, the σA and σNS proteins were expressed in the Vero cells 6 h after transfection with the corresponding plasmids (σA-pcAGEN, σA-M1-pcAGEN, σA-M2-pcAGEN, σA-M3-pMD18T, σA-M4-pcAGEN, σA-M5-pcAGEN, σA-M6-pcAGEN, σNS-pcAGEN, σNS-M1-pcAGEN, σNS-M2-pcAGEN and σNS-M3-pcAGEN; Figures 3 and 4). Cells that were not transfected cells and transfected with pcAGEN served as negative controls cells (Figure 3a and b and Figure 4, lanes 1 and 2).
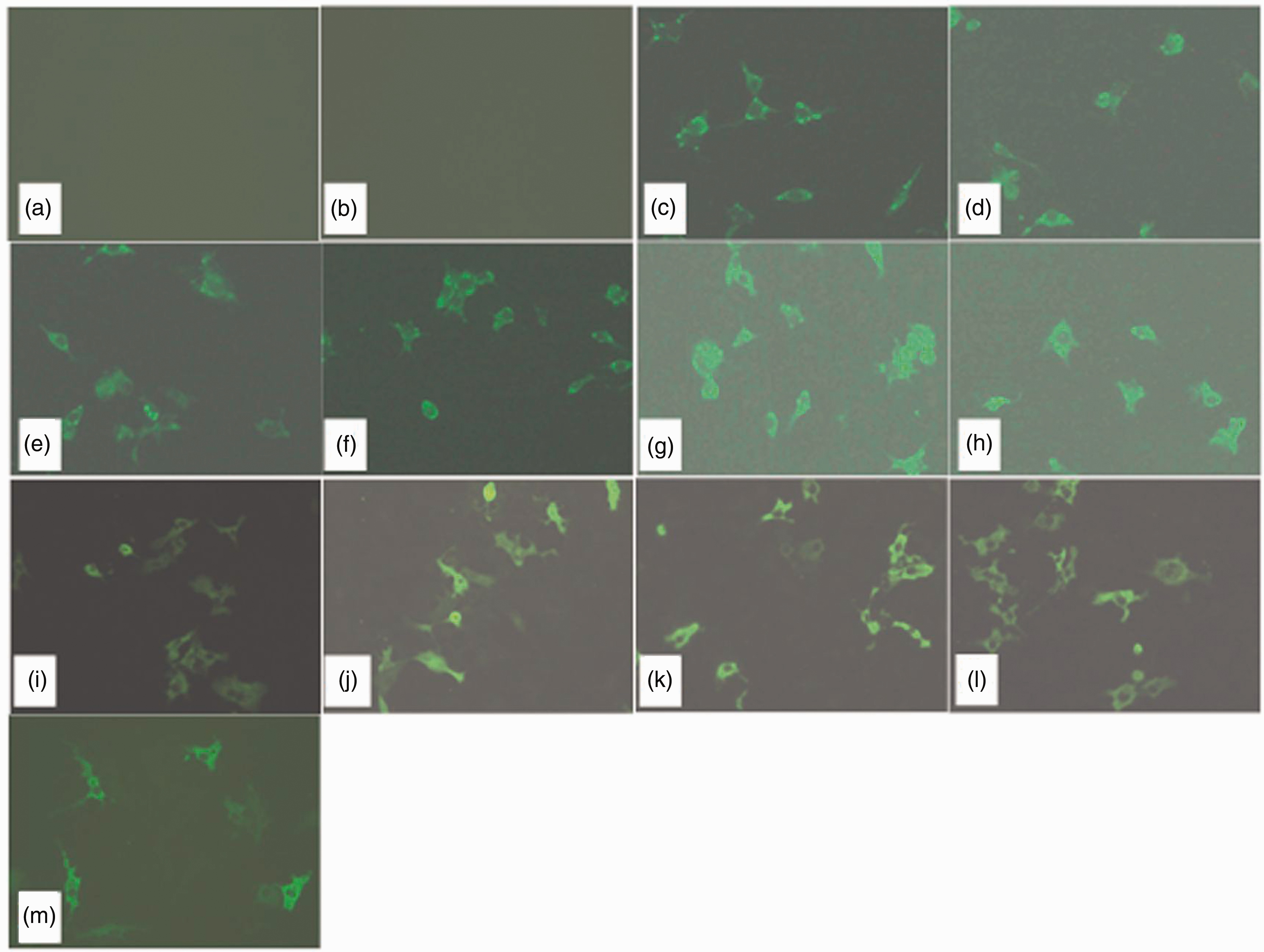
IFA analysis of the expression of recombinant plasmids in transfected Vero cells. (a) Negative control, (b) pcAGEN, (c) σA-M1-pcAGEN, (d) σA-M2-pcAGEN, (e) σA-M3-pcAGEN, (f) σA-M4-pcAGEN, (g) σA-M5-pcAGEN, (h) σA-M6-pcAGEN, (i) σA-pcAGEN, (j) σNS-M1-pcAGEN, (k) σNS-M2-pcAGEN, (l) σNS-M3-pcAGEN, (m) σNS-pcAGEN.
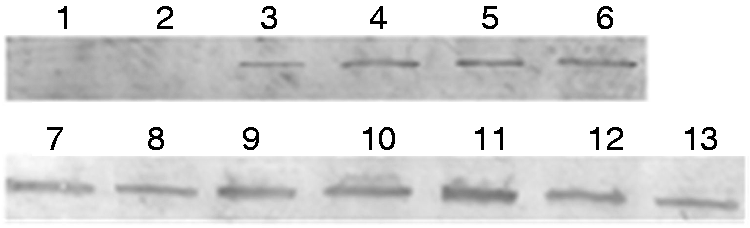
Western blot analysis of gene expression. Lane 1: Negative control; Lane 2: pcAGEN; Lane 3: σNS-M1-pcAGEN; Lane 4: σNS-M2-pcAGEN; Lane 5: σNS-M3-pcAGEN; Lane 6: σNS-pcAGEN; Lane 7: σA-M1-pcAGEN; Lane 8: σA-M2-pcAGEN; Lane 9: σA-M3-pcAGEN; Lane 10: σA-M4-pcAGEN; Lane 11: σA-M5-pcAGEN; Lane 12: σA-M6-pcAGEN; Lane 13: σA-pcAGEN.
σA and σNS proteins activate the PI3K/Akt signalling pathway
Figure 5 shows that σA-M3- and σA-M4- expressing cells had the lowest levels of P-Akt compared to σA-pcAGEN, σA-M1-pcAGEN, σA-M2-pcAGEN, σA-M5-pcAGEN and σA-M6-pcAGEN. However, these cells had the same levels of P-Akt as negative control and pcAGEN-expressing cells. Figure 6 shows the same results as Figure 4. After mutation at amino acids 110–114 (PPXXP→AAXXA) and 114–117 (PXXP→AXXA), the σA gene lost the capacity to increase P-Akt expression in Vero cells.
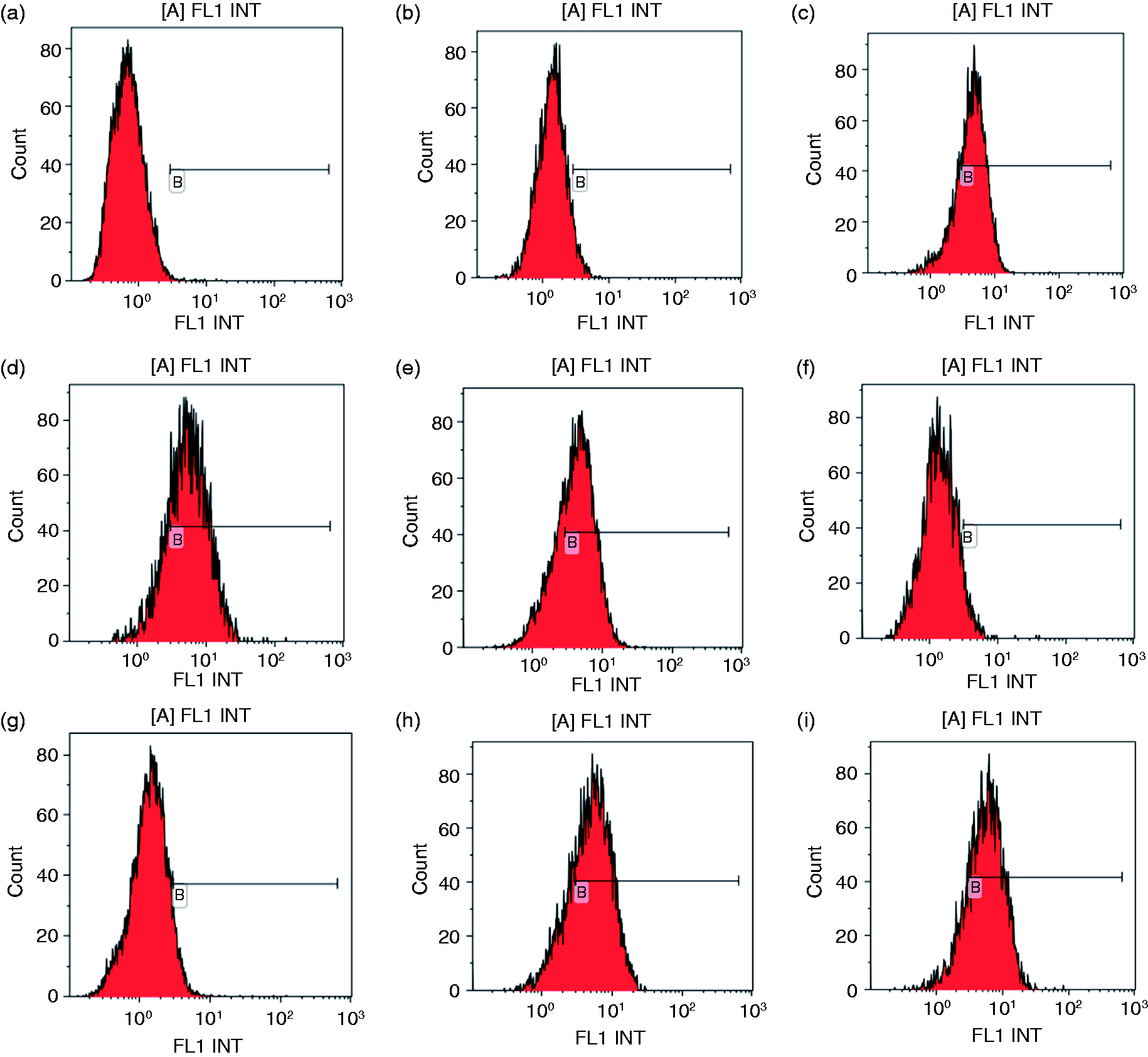
Flow cytometry analysis of P-Akt expression levels. (a) Negative control, (b) pcAGEN, (c) σA-pcAGEN, (d) σA-M1-pcAGEN, (e) σA-M2-pcAGEN, (f) σA-M3-pcAGEN, (g) σA-M4-pcAGEN, (h) σA-M5-pcAGEN, (i) σA-M6-pcAGEN.
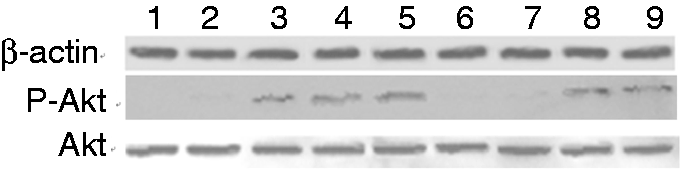
Western blot analysis of P-Akt expression levels. Lane 1: Negative control; Lane 2: pcAGEN; Lane 3: σA-pcAGEN; Lane 4: σA-M1-pcAGEN; Lane 5: σA-M2-pcAGEN; Lane 6: σA-M3-pcAGEN; Lane 7: σA-M4-pcAGEN; Lane 8: σA-M5-pcAGEN; Lane 9: σA-M6-pcAGEN.
The σNS-M1-, σNS-M2- and σNS-M3-expressing cells had similar P-Akt levels compared to σNS-pcAGEN, but these cells had increased levels of P-Akt expression compared to negative control cells and pcAGEN-expressing cells (data not shown). The mutations at amino acids 159–162 (PXXP→AXXA), 179–183(YXXXM→FXXXA) and 333–336 (PXXP→AXXA) did not affect the capacity of the σNS gene to increase P-Akt expression in Vero cells.
Discussion
The PI3K/Akt pathway is involved in numerous cellular processes, such as cell proliferation, differentiation and survival.7,8 PI3K is activated by the binding of autophosphorylated tyrosine kinase receptors or non-receptor tyrosine kinases to the SH2 and SH3 domains of its regulatory/adaptor subunit p85. 19 This binding is mediated by PXXP and YXXXM/YXXM motifs and activates the PI3K/Akt pathway.9–15 As reported by Shin, 15 the Influenza A virus NS1 protein activates the PI3K/Akt pathway by direct interaction with the p85 subunit of PI3K (SH2-binding motif YXXXM and SH3-binding motif PXXP).
The S2-encoded protein σA, a component of the inner core shell, displays anti-IFN activity by preventing the activation of the dsRNA-dependent protein kinase PKR; this activity is likely linked to its capacity to bind and sequester dsRNA.20,21 The non-structural protein σNS is encoded by the ARV S4 genome segment. As a non-structural RNA-binding protein that accumulates in viral factories of ARV-infected cells, σNS is a likely candidate for playing key roles in RNA packaging and replication. 22
Our previous works showed that the σA and σNS proteins of ARV activate the PI3K/Akt pathway. 16 According to amino acid sequence analysis of the σA and σNS genes, σA contains four PXXP and two YXXXM motifs, and σNS contains one YXXXM and two PXXP motifs. Mutation of the PXXP motif of NS1 protein from influenza A virus or mutation of the YXXM motif of envelope protein from avian leukosis virus will result in loss of PI3K/Akt pathway activation.15,17 We speculate that the mutant PXXP and YXXXM motifs for the σA and σNS genes of ARV may also affect the activation of the PI3K/Akt pathway.
Gene splicing by overlap extension PCR was used to change the PXXP or YXXXM motifs of the σA and σNS genes and to construct the mutant σA and σNS recombinant plasmids. The mutant σA and σNS recombinant plasmids were transfected into Vero cells. The P-Akt profile of the transfected Vero cells was examined by flow cytometry and Western blot analysis.
In this study, after amino acids 110–114 (PPXXP→AAXXA) and 114–117 (PXXP→AXXA) were mutated, the σA gene lost its capacity to improve P-Akt expression in Vero cells, and the σA protein of ARV activated the PI3K/Akt pathway via the PXXP motif. PXXP motif of the σA protein of ARV is involved in PI3K signalling, and the mutant PXXP motif of σA affects PI3K/Akt pathway activation. These results are similar to those of a previous study. 16 However, whether this activation was a result of the σA protein binding to the p85 subunit via interactions with PXXP motifs, as occurs in other viruses,11,15 requires further study.
We also demonstrated that the mutations at amino acids 159–162 (PXXP→AXXA), 179–183 (YXXXM→FXXXA) and 333–336 (PXXP→AXXA) did not affect the capacity of the σNS gene to increase P-Akt expression in Vero cells. The σNS protein may activate the PI3K/Akt pathway via more than one motif, other motifs or σNS interaction with the P85 subunit according to the middle protein. These hypotheses must be further studied.
Moreover, the relationship between the p110 catalytic subunit of PI3K and the PXXP or YXXXM/YXXM motifs of the σA/σNS genes will be further studied, and these research results will be published in the future.
Although the exact mechanisms by which PI3K/Akt regulates ARV replication and other biological functions of PI3K/Akt in virus infection remain uncharacterized, our study reveals the mechanism underlying PI3K/Akt activation and adds a novel aspect to the functions of the σA and σNS proteins of ARV.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: this study was funded by the National Natural Science Foundation of China (nos. 31660715 and 31160512), the Guangxi Science and Technology Bureau Program (AA17204057 and AD17195083), the Guangxi BaGui Scholars Program Foundation (2019-79) and the National Ten-Thousand Talents Program of China (W02060083).
