Abstract
Neisseria meningitidis infections in sub-Saharan Africa usually present with distinct symptoms of meningitis but very rarely as fulminant septicemia when reaching hospitals. In Europe, development of persistent meningococcal shock and multiple organ failure occurs in up to 30% of patients and is associated with a bacterial load of >106/ml plasma or serum. We have prospectively studied 27 Ethiopian patients with meningococcal infection as diagnosed and quantified with real-time PCR in the cerebrospinal fluid (CSF) and serum. All presented with symptoms of meningitis and none with fulminant septicemia. The median N. meningitidis copy number (NmDNA) in serum was < 3.5 × 103/ml, never exceeded 1.8 × 105/ml, and was always 10–1000 times higher in CSF than in serum. The levels of LPS in CSF as determined by the limulus amebocyte lysate assay were positively correlated to NmDNA copy number (r = 0.45, P = 0.030), levels of IL-1 receptor antagonist, (r = 0.46, P = 0.017), and matrix metallopeptidase-9 (MMP-9; r = 0.009). We also compared the inflammatory profiles of 19 mediators in CSF of the 26 meningococcal patients (2 died and 2 had immediate severe sequelae) with 16 patients with Streptococcus pneumoniae meningitis (3 died and 3 with immediate severe sequelae). Of 19 inflammatory mediators tested, 9 were significantly higher in patients with pneumococcal meningitis and possibly linked to worse outcome.
Keywords
Introduction
Bacterial meningitis is an important cause of morbidity and mortality in sub-Saharan Africa. Neisseria meningitidis, mainly capsular group A, has, until recently, caused recurrent epidemics of meningitis in this area, including Ethiopia. 1 Streptococcus pneumoniae is the second most prevalent pathogen and causes even higher mortality and morbidity than N. meningitidis in the same region.1–3 A recent study of bacterial meningitis patients in three university hospitals in Ethiopia from February 2012 to June 2013 found that, among 46 patients with real-time PCR (RT-PCR) confirmed etiology, N. meningitidis was the most prevalent cause (59%) followed by S. pneumoniae (39%). Merely 2% had meningitis caused by Haemophilus influenzae. 1
The clinical presentations of N. meningitidis infections in sub-Saharan Africa appear to differ from the disease spectrum observed in industrialized countries on hospital admission. Meningitis, characterized by fever, headache, neck and back rigidity and impaired cerebral function, is the dominant clinical presentation in sub-Saharan Africa. The development of fulminant meningococcal septicemia with persistent septic shock, multiple organ failure and large hemorrhagic skin lesions, which are easy recognizable by health care personnel, appears to be very uncommon in this region of Africa. In Europe, as many as 30% of patients contracting invasive meningococcal infection may develop septic shock usually combined with renal and pulmonary failure and extensive skin hemorrhages.4,5 Half of patients with septic shock also exhibit clinical signs of meningitis;4,5 however, coagulopathy and multiple organ failure are less pronounced, with lower mortality than fulminant meningococcal septicemia with minimal pleocytosis (< 100 × 106 leukocytes per liter cerebrospinal fluid (CSF)) and lack of distinct signs and symptoms of meningitis.4,5 The causes explaining the different clinical presentations in sub-Saharan Africa versus Europe are not obvious. European studies in the last 30 yr have documented that the different clinical pictures can be explained by compartmentalized growth of the meningococci.4,6–9 In patients developing distinct meningitis without compromised circulation, the growth of meningococci occurs primarily in the subarachnoid space, with 10 to 1000-fold higher concentrations of LPS, N. meningitidis DNA (NmDNA) and inflammatory mediators in the cerebrospinal fluid than in the blood.4,6–9 In patients developing fulminant meningococcal septicemia with persistent septic shock, the bacterial proliferation occurs primarily in blood and microcirculation in the extra-cerebral organs, leading to much higher concentrations of NmDNA, LPS and inflammatory mediators than detected in the subarachnoid space.4,6–9 The difference in clinical presentation between meningococcal meningitis and fulminant septicemia is caused primarily by a much higher growth velocity of the meningococci in the blood and extra-cerebral vessels in patients with fulminant septicemia.4–9 Meningococcal LPS is the most potent trigger of the innate immune system via CD14, TLR4 and myeloid differential factor 2 (MD2).4,6–9 One aim of this study was to quantify the growth of meningococci in the circulation and CSF by using RT-PCR of NmDNA and determine the copy number per milliliter. We hypothesized that the copy number of NmDNA would be below 106/ml serum since the patients did not develop the septic shock, extensive skin hemorrhages, renal and pulmonary impairment associated with NmDNA copy numbers in plasma or serum levels ranging from 106 to 108/ml. 4
Meningitis caused by S. pneumoniae has a higher case fatality rate and causes more sequelae in patients in sub-Saharan Africa and in industrialized countries than meningococcal meningitis.2,3,10,11 The biological basis explaining this difference has not been fully elucidated. One hypothesis is that the inflammatory responses to the Gram-negative N. meningitidis and the Gram-positive S. pneumoniae in the subarachnoid space are quantitatively different, and that this is reflected in the outcome. Few studies have performed head-to-head comparison of the cytokine profiles in CSF of the two pathogens. Examination of CSF from patients with meningococcal or pneumococcal meningitis living in Brazil and Burkina Faso, respectively, suggests that the profile of cytokine and chemokine differs, possibly explaining the difference in outcome.12,13
A second aim of the present study was to compare the profile of inflammatory mediators in CSF in 26 patients with N. meningitidis meningitis and 16 patients with S. pneumoniae meningitis all diagnosed by RT-PCR. We quantified the levels of 18 different cytokines, chemokines and matrix metallopeptidase 9 (MMP-9) in CSF and compared the results of the two groups. In addition, we evaluated the quantitative relationship between LPS and NmDNA and the inflammatory mediators in patients with meningococcal meningitis.
Materials and methods
Patients and samples
Patients admitted to three referral teaching university hospitals in Gondar, Hawassa and Addis Ababa in Ethiopia during February 2012–June 2013 were recruited to this study, as previously described. 1 Among the 139 patients with clinical signs of meningitis and turbid CSF having their CSF tested by RT-PCR for presence of DNA from N. meningitidis, S. pneumoniae, or H. influenzae, sufficient CSF volumes for testing of inflammatory mediators were available from 43 patients (27 with meningococcal meningitis and 16 with pneumococcal meningitis). None of the patients presented at hospital admission with typical signs and symptoms of meningitis combined with massive skin hemorrhages, persistent shock or dwindling urine production. All patients or parents of patients received clinical study information sheets and gave written informed consent. A total of 27 meningococcal patients had LPS and MMP-9, 26 had 18 different cytokines and chemokines, and 23 had the copy number of N. meningitidis analyzed in CSF. NmDNA in serum was quantified in 23 of the 27 patients. CSFs from the 16 patients with pneumococcal meningitis were assayed for the same 18 different cytokines, chemokines and MMP-9 as the meningococcal meningitis patients.
All patients had a completed case report form comprising personal data, clinical information on admission and the immediate outcome, i.e. death or survival and immediate severe sequelae, such as deafness or neurological impairment observed during the hospital stay. Long-term sequelae were not studied. The study protocol was reviewed and approved by the National Research Ethics Review Committee of Ethiopia (3-10/6/5-04) and Regional Ethics Committee in Norway (2011/825b). 1
The CSFs and sera were stored at –20°C immediately after collection at the local clinical laboratory, during transportation to Armauer Hansen Research Institute (AHRI) in Addis Abeba. The samples were transported to Norwegian Institute of Public Health by air on dry ice and, upon at arrival, were stored at –80°C until thawed for PCR analysis. The samples were transported frozen to Oslo University Hospital Ullevål, stored at –80°C and thawed for PCR, LPS and cytokine analyses.
Quantification of N. meningitidis DNA
The copy number of NmDNA in serum and CSF was determined using quantitative PCR (qPCR), as previously described,
14
except that the sequence-specific hybridization probes (0.3 µM per reaction) 5′-
Quantification of inflammatory mediators in CSF
The levels were quantified by Bio-Plex Pro Cytokine, Chemokine, and Growth Factor assay (Bio-Rad, Hercules, CA) for the following 18 cytokines and chemokines: IL-1β, IL-1 receptor antagonist (IL-1ra), IL-2, IL-4, IL-6, IL-8, IL-10, IL-12p70, IL-18, granulocyte colony stimulating factor (G-CSF), IFN-ϒ, IFN-ϒ-induced protein 10 (IP-10), monocyte chemoattractant protein-1 (MCP-1), macrophage inflammatory proteins 1α (MIP-1α) , macrophage inflammatory proteins 1β (MIP-1β), regulated on activation normal T cells expressed and secreted (RANTES), TNF-α and TNF-related apoptosis inducing ligand (TRAIL). MMP-9 was quantified by Bio-Plex Pro Human MMP Assay (Bio-Rad) according to the manufacturer’s instructions (Figure 1 and Table 1).
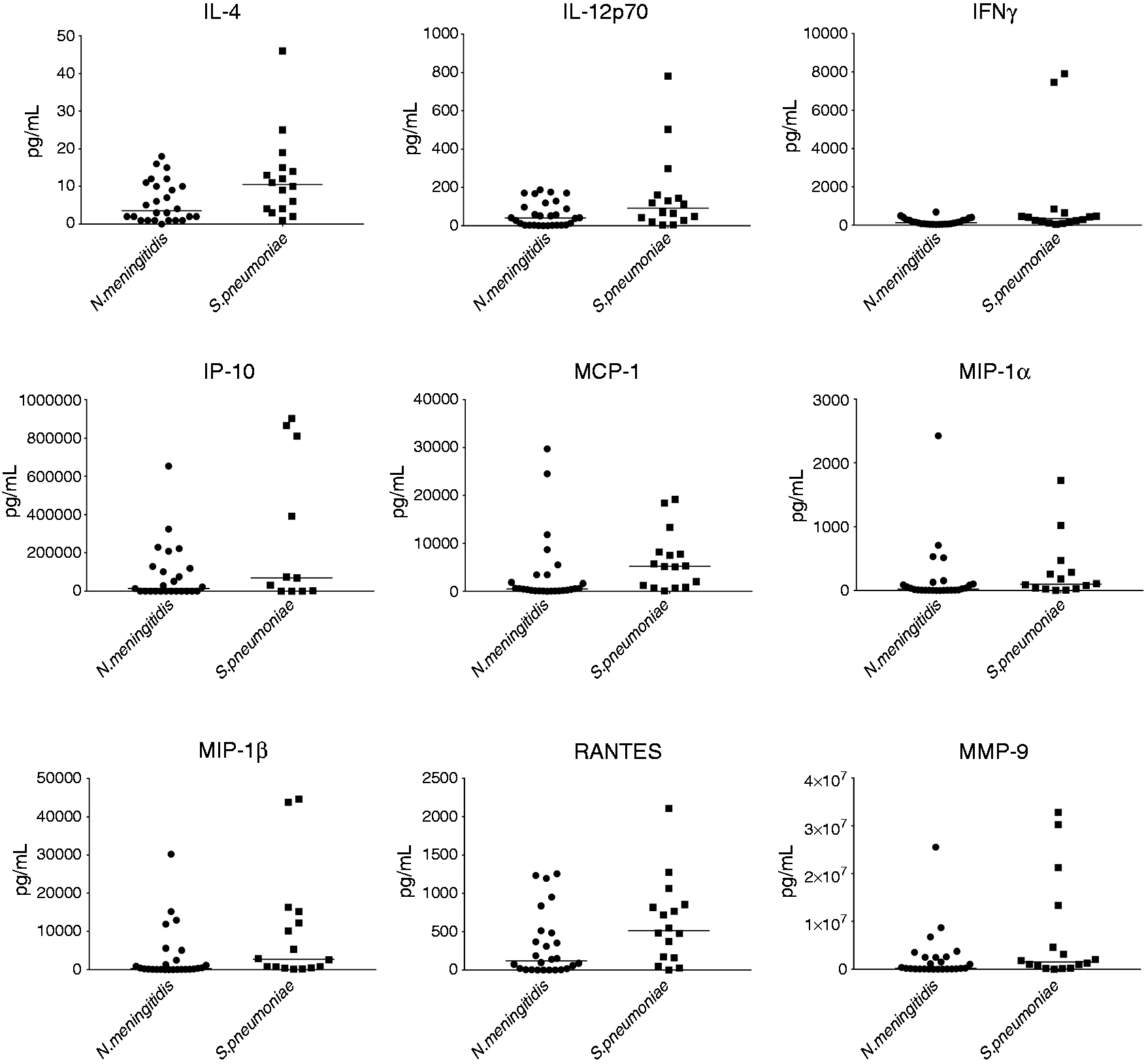
Scatterplot with median value of the concentrations (pg/mL) of inflammatory mediators in cerebrospinal fluids from Ethiopian meningococcal and pneumococcal patients with significant differences between the patient groups. IL-4: P = 0.0268, IL-12p70: P = 0.0260, IFN-ϒ: P = 0.0104, IP10: P = 0.0395, MCP-1: P = 0.0063, MIP-1α: P = 0.0198, MIP-1α: P = 0.0116, RANTES: P = 0.0395, MMP-9: P = 0.0486.
Concentrations (pg/ml) of inflammatory mediators in cerebrospinal fluids from Ethiopian meningococcal and pneumococcal patients, 2012–2013.
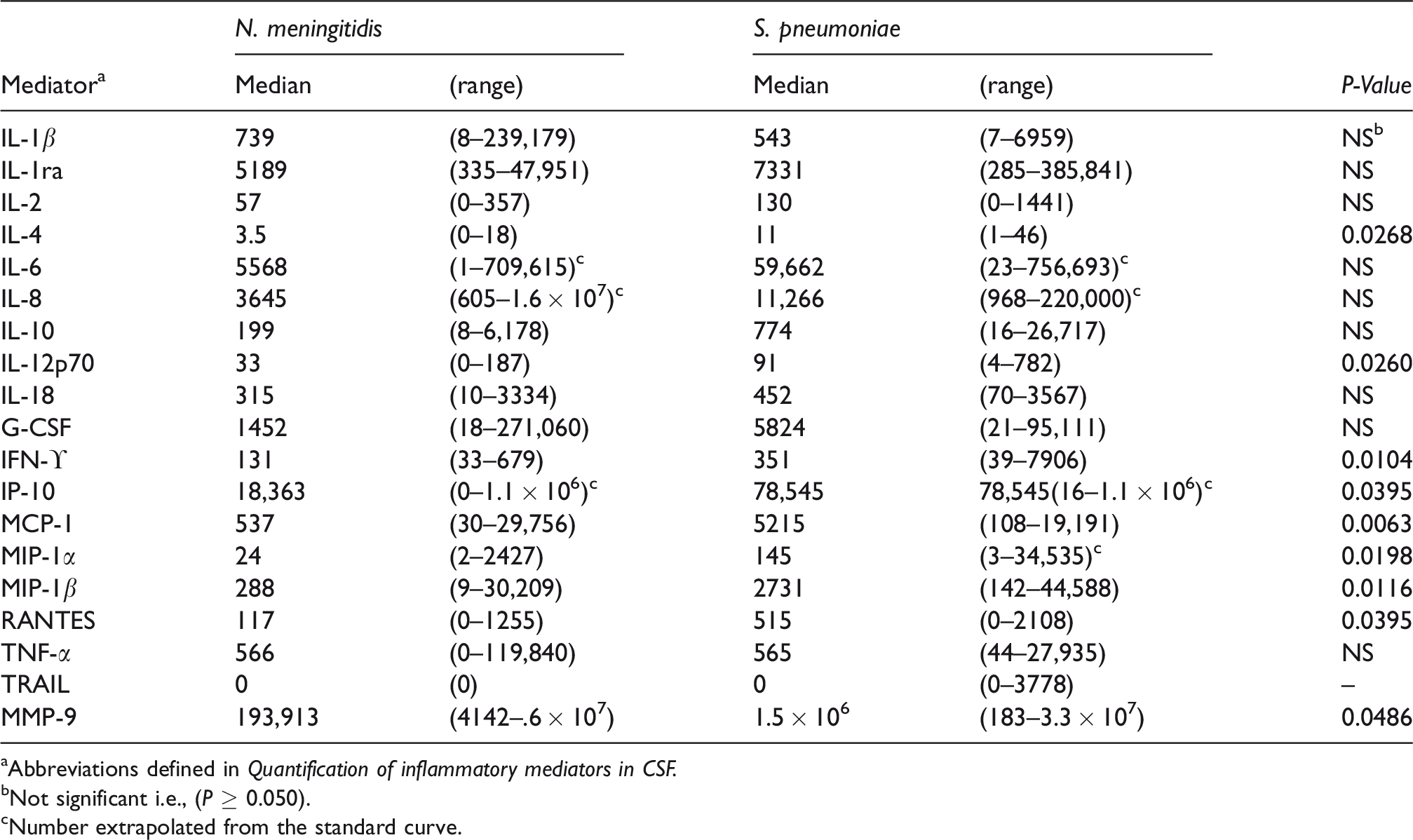
aAbbreviations defined in Quantification of inflammatory mediators in CSF.
bNot significant i.e., (P ≥ 0.050).
cNumber extrapolated from the standard curve.
Quantification of LPS activity in CSF
The levels of LPS were determined with Chromo-LAL® (Associates of Cape Cod, East Falmouth, MA) according to the manufacturer’s instructions and as described earlier. 14
Clinical definitions
Meningitis was diagnosed when the patients revealed neck and back rigidity and turbid CSF (≥1000 × 106 leukocytes/l) usually associated with fever, headache and impaired cerebral function. Fulminant meningococcal septicemia was diagnosed when patients presented with extensive skin hemorrhages (ecchymoses), septic shock (systolic blood pressure <100 mm Hg in patients 12 yr or older and 70 mm Hg in children below 12 yr old that would require fluid and vasoactive drug therapy in addition to antibiotics).4,6 Immediate recognizable sequelae comprised acute deafness or major impaired hearing, stroke, epileptic insults or other acute neurological or other impairment.
Statistical analysis
We assessed differences between the patient groups by using the Mann-Whitney’s test. Correlations were carried out using Spearman´s procedure. All calculations were performed using Graph Pad Prism version 3.0. (La Jolla, CA). The level of significance was set at 5% in all cases.
Results
Descriptive demographics of meningococcal and pneumococcal patients
All patients presented as meningitis. The median age and range (in parentheses) for 27 patients with meningococcal meningitis were 7 yr (5 d–35 yr) as compared with 10 yr (2 mo–78 yr) for 16 patients with pneumococcal meningitis. The age distribution between the groups was not significantly different. Among the N. meningitidis patients, 17 (63%) were males whereas 9 (56%) of the S. pneumoniae patients were males. The gender distribution was not significantly different between the two groups. In the N. meningitidis group, two (7%) died: a 4-yr-old boy and a 20-yr-old woman. In the S. pneumoniae group three (19%) died: a 4-mo-old girl, a 50-yr-old man and a 54-yr-old woman. Two patients, a 4-mo-old girl and a 5-yr-old boy (7%) with N. meningitidis developed immediately recognizable sequelae. Among those with S. pneumoniae meningitis, three (19%) had severe immediate sequelae (two boys of 4 and 5 mo, respectively, and a girl of 4 yr of age).
N. meningitidis DNA and LPS concentrations
A set of 23 paired CSF and serum samples from meningococcal meningitis patients as assayed to study the compartmentalization of the meningococci, i.e., the growth of the bacteria in CSF versus blood. The copy number of NmDNA in CSF was detectable in 22 (96%) of 23 tested CSF samples, with a median of 8.2 × 105 (range < 2.5 × 102–1.4 × 107) copies/ml. Among the 23 serum samples from the corresponding patients, the median NmDNA level was <3.5 × 103/ml (range < 3.5 × 103–1.8 × 105/ml). Only 6 (26%) of the 23 patients had detectable NmDNA (≥ 3.5 × 103/ml) in serum, with a median concentration of 2.1 × 104 (range 6.4 × 103–1.8 × 105) copies/ml. The difference in the levels of NmDNA in the two compartments (CSF and serum) was statistically significant (P = 0.008). In patients with paired samples from serum and CSF, the copy numbers of NmDNA were always higher in CSF than in serum, with the differences ranging from 10- to 1000-fold. The highest level in CSF was 1.4 × 107 copies/ml with a corresponding serum level of 1.0 × 105/ml. The highest serum level was 1.8 × 105/ml with a corresponding CSF level of 3.0 × 106/ml.
The median level of LPS in CSF of six patients with detectable NmDNA in serum was 679 (range 184–16,000) endotoxin units (EU)/ml. In 21 patients without detectable NmDNA copies (< 3.5 × 103/ml) in serum, the median CSF level of LPS was 100 (2 – 8055) EU/ml. The difference was significant (P = 0.01). In CSF samples, the LPS concentrations were significantly correlated only to NmDNA (r = 0.45, P = 0.03, n = 23), CSF levels of IL-1ra (r = 0.46, P = 0.02, n = 26) and MMP-9 (r = 0.50, P = 0.009, n = 27). Age did not correlate with LPS activity.
The numbers of NmDNA copies in CSF were positively correlated with IL-1β (r = 0.62, P = 0.002, n = 23) and MMP-9 (r = 0.46, P = 0.03, n = 23) and negatively correlated with IL-6 (r = –0.45, P = 0.03, n = 23). The NmDNA numbers in CSF were not significantly correlated to age. IL-18 was negatively correlated to age (r = –0.45, P = 0.02, n = 26).
Two meningococcal meningitis patients died and two had immediate observable sequelae. No significant difference was observed in the levels of LPS activity, NmDNA load and levels of cytokines, chemokines, MMP-9 in CSF, gender or age between these 4 patients and the 22 other surviving patients without immediate observable sequelae.
Comparison between meningococcal and pneumococcal meningitis patients
Significantly higher levels of IL-4, IL-12p70, INF-γ, IP-10, MCP-1, MIP-1α, MIP-1β, RANTES, and MMP-9 were found in CSF of 16 patients with S. pneumoniae meningitis as compared with 26 patients with N. meningitidis meningitis (Table 1). TRAIL was detectable in three out of 16 CSF samples from patients with pneumococcal meningitis as compared with none in the 26 patients with meningococcal meningitis. No statistically significant difference was found between patients with pneumococcal versus meningococcal meningitis for the following cytokines and chemokines: TNF-α, IL-1β, IL-1ra, IL-2, IL-6, IL-8, IL-10, IL-18, and G-CSF.
Inflammation markers and severity of disease among pneumococcal meningitis patients
We compared inflammatory markers S. pneumoniae patients who died (n = 3) or had immediate severe sequelae (n = 3) versus the 10 other patients who survived. Three deceased pneumococcal meningitis patients had significantly higher median levels of IL-1β (P = 0.04) and MMP-9 (P = 0.03) than the 13 survivors. There was no significant difference in concentration for any of the cytokines, chemokines or MMP-9 between the six patients whom either died or had immediately recognizable sequelae and the other 10 surviving patients who had no immediate severe sequelae. Since sequelae were only registered during admission to hospital, we do not know whether some of these 10 patients developed sequelae at a later stage.
Discussion
This study provides more exact information about the compartmentalized nature of N. meningitidis causing meningitis in Ethiopia. Furthermore, we confirm that the profile of inflammatory mediators in CSF differs between meningococcal and pneumococcal patients with meningitis as suggested by others.12,13 By extending the number of mediators studied, this difference becomes even clearer.
In 23 Ethiopian patients with meningococcal meningitis, the copy numbers of NmDNA were 10- to 1000-fold higher in CSF than in serum. After an initial bacteremic phase, meningococci penetrate the blood–brain barrier, utilizing type IV pili and various other surface-exposed bacterial adhesion molecules interacting with CD147 and other molecules on the endothelial cells.16,17 After gaining access to the subarachnoid space, the bacteria proliferate and reach higher levels in CSF than in the blood, inducing a more intense localized inflammation in the former.4,6–9,14,18 These patients have marked clinical symptoms of meningitis but no persistent septic shock. Among the Ethiopian meningococcal patients, only 6 (26%) of 23 had quantifiable meningococcal DNA (≥ 3.5 × 103/ml) in serum. These six patients also had significantly higher numbers of NmDNA copies and higher levels of LPS/ml in CSF than the 17 patients without detectable NmDNA in the circulation. One interpretation of this observation is that while meningococci proliferate to high levels in CSF, a certain fraction of bacteria translocate back into the circulation resulting in detectable bacterial load, i.e., levels above 3.5 × 103 meningococci/ml in serum. Another interpretation is that the genetic constitution of certain patients allows for more intense proliferation in both blood and CSF, although the proliferation in blood did not reach levels associated with shock, i.e., above 106 copies of N. meningitidis DNA per milliliter plasma or serum.4,14,19,20 In Norway, the same pattern as seen in this Ethiopian study has previously been observed. Only 9 of 28 (32%) Norwegian patients with distinct meningococcal meningitis without persistent shock had detectable NmDNA in heparin plasma with detection limit of 2.5 × 102/ml. 14
An interesting aspect concerning bacterial load in the circulation is that none of the patients with quantifiable copy number of meningococcal DNA reached 106 copies/ml. Previous European studies suggested that patients passing this level of meningococcemia are predisposed to development of persistent septic shock, renal and pulmonary failure combined with massive activation of the coagulation system.4,14,19,20 Unlike patients with clinically distinct meningitis, many patients with fulminant meningococcal septicemia with persistent shock and large hemorrhagic skin lesions, previously known as Waterhouse-Friderichsen syndrome, reveal levels of NmDNA copy numbers as high as 107 –108/ml plasma or serum.4,14,19,20 This high number of meningococci in the circulation is accompanied with LPS (endotoxin) levels in plasma or serum as high as 3800 EU/ml which induce a cytokine storm in the circulation and tissues of the large organs.4,6–9,14,15,18–25
Fulminant meningococcal septicemia with persistent shock and massive skin hemorrhages appears to be very uncommon among patients reaching hospitals in sub-Saharan Africa. 26 One likely explanation is that fulminant meningococcal septicemia develops so rapidly that patients living in rural areas die before they are transported to hospital. 27 However, patients living in cities—closer to the hospitals—should have increased chances of being discovered and receiving treatment. In a study from Kenyatta National Hospital in Nairobi, of 57 patients with meningococcal meningitis, 1 developed shock but multiple organ failure is not mentioned. 28 Use of prescribed or non-prescribed oral preadmission antibiotics with inhibiting effect on N. meningitidis in the circulation might influence the development of shock but have little impact on the development of meningitis. Among Ethiopian physicians, the experience is that the majority of patients presently have received antibiotics before they are admitted. In the 27 patients with meningococcal meningitis in this study 17 (63%) had not been treated with antibiotics before hospital admission, 7 were treated, and information was lacking for 3 patients.
Is meningococcal shock with organ failure really less common in Africa than in industrialized countries? 26 A search in PubMed using the key words of Waterhouse Friderichsen syndrome or fulminant meningococcal septicemia in the sub-Saharan region did not reveal any case description. None of our relatively small number of patients with laboratory confirmed N. meningitidis infection had levels of NmDNA copy numbers above 106/ml. None developed septic shock, multiorgan failure or extensive skin hemorrhages.
An alternative hypothesis to explain the rarity of fulminant meningococcal septicemia is that pre-existing Abs elicited by the microbial flora in the upper respiratory tract and gut may influence the clinical presentation. Such Abs, reacting with epitopes located in the meningococcal capsular polysaccharide or proteins and LPS in the outer membrane, may not be bactericidal but could still contain the very rapidly proliferation of N. meningitidis in the blood and capillaries of various organs.29,30 Africans growing up in the Europe do, however, develop fulminant meningococcemia although the incidence has not been established. 25
Could the difference in clinical presentation be explained by differences in serogroups infecting patients in sub-Saharan Africa, mainly serogroup A, versus patients in Europe where mainly serogroup B or C strains cause the infection? Observations from the serogroup A epidemics in Moscow in the 1980s and 1990s, however, indicate that serogroup A may cause fulminant meningococcal septicemia with lethal shock and high LPS levels in the blood in Europeans.31,32
Our results suggest that the levels of NmDNA copy numbers are significantly correlated with the biological activity of LPS in CSF. This is in accordance with European results but, to our knowledge, has not been previously studied in African patients. 14 The correlation coefficient (r = 0.45) in the present study was not as high as previously found (r = 0.96), but the number of patients in the present study was higher, i.e. 23 versus 9 in the earlier study. 14 However, we cannot exclude that LPS contamination may have occurred during collection and handling of some of the CSF samples, as judged by a marked difference between low level of NmDNA and high LPS activity. A series of previous studies suggest that N. meningitidis LPS is the most potent, but not the only, activator of the innate immune system.4,7–9,14,15,33–36
The relation between levels of biologically active LPS and various cytokines, chemokines and other inflammatory mediators is complex and time dependent. The cytokines TNF-α, IL-1β and IL-6 are locally produced in resident cells of the meninges and CNS as well as by leukocytes penetrating the blood brain barrier. In rabbit experimental meningitis models, TNF-α, IL-1β and IL-6 are elicited prior to the influx of leukocytes. 18 In our study, only IL-1β receptor antagonist and MMP-9 were significantly and positively correlated to the levels of LPS, while Waage et al. have previously documented that LPS activity was significantly correlated to TNF-α, but not to IL-1β or IL-6 in CSF. 18 The difference in assay methods, i.e. biological activity in cell assays versus Ab-based immunological tests and the time from initiation of the symptoms to lumbar puncture, which presumably is longer in Ethiopia than in Norway, clearly may influence the reported results.
The pattern of cytokines we observed in this study was highly variable between individual patients in the two patient groups (Table 1). IL-6, IL-8 and IP-10 were the cytokines with the largest variability. The variability of IL-6 has previously been observed among patients with bacterial meningitis. 37 Why IL-6 was negatively correlated to the copy numbers of NmDNA but not to LPS is not known and may be an arbitrary finding.
We chose the panel of inflammatory mediators in this study to verify the results from two previous studies comparing the inflammatory CSF response in meningococcal versus pneumococcal meningitis.12,13 In addition, we added several mediators that were relevant for subarachnoid inflammation elicited by LPS and other cell wall components, including those of Gram-positive bacteria and that could be determined with same assay system. MMP-9 was relevant given its previously documented prognostic value. 38 Patients in this study with S. pneumoniae meningitis had, as a group, significantly (7.7-fold) higher levels of MMP-9 in CSF than patients with meningococcal meningitis. MMP-9 disrupts the blood–brain barrier, possibly increasing brain edema and contributing in some cases to brain abscess formation, as observed in some patients with pneumococcal meningitis but very rarely in meningococcal meningitis. Furthermore, the median MMP-9 and IL1β levels were significantly higher in the three patients with lethal pneumococcal meningitis as compared with the 13 surviving patients. However, given the low numbers of diseased patients as compared with survivors, the result are associated with uncertainty. Age did not influence the levels of LPS nor copy number of meningococcal DNA in CSF. Age was significantly negatively correlated to IL-18 but not to any other cytokines or chemokines. An interesting observation was documented in the CSF of a 7-d-old boy who survived the initial illness (although the long term outcome is unknown) bearing the highest level of TNF-α, IL-6, IL-8 and the second highest level of IL-1β among 26 the meningococcal patients tested. Some neonates clearly have the capacity to mount a massive cytokine release in the subarachnoid space and still survive the developing brain edema. When comparing the levels of the N. meningitidis LPS and DNA or the 19 inflammatory mediators in the 4 patients who died or had immediate severe sequelae with the 22 other meningococcal patients, we did not observe a reproducible pattern that could explain the worse outcome. Our results suggest that the levels of cytokines in CSF do not have the same predictive value of the acute outcome as observed in serum or plasma for meningococcal patients. 22
Importantly, we found significantly higher levels of 9 of 19 inflammatory mediators in CSF in patients with pneumococcal as compared with meningococcal meningitis. Significantly higher levels of IFN-γ, MIP-1α and MMP-9 in CSF have previously been identified in pneumococcal versus meningococcal meningitis patients.12,13 This study adds MCP-1, MIP-1β, IL-4, IL-12p70, IP-10, RANTES and TRAIL as possible additional markers of increased inflammatory response in pneumococcal versus meningococcal meningitis. Pro-inflammatory mediators including TNF-α, IL-6, IL-8, 12p70, IL-18 and G-CSF and anti-inflammatory mediators including IL-10 and IL-1ra did not differ significantly between the two groups of meningitis in line with previous studies.12,13 If confirmed in a larger number of patients comprising both types of meningitis, the results may indicate that a different cytokine, chemokine and MMP-9 profile exists between these two major types of bacterial meningitis. The results may indicate where we should focus future research to unravel the underlying pathophysiology of pneumococcal meningitis to better understand the cause of the high case fatality rate. Future studies should also center on the bacterial load and outcome.39,40
Conclusion
In this study of 27 patients with meningococcal meningitis and 16 patients with pneumococcal meningitis admitted to three referral hospitals in Ethiopia during a 15-mo period, we found that N. meningitidis was compartmentalized primarily to the subarachnoid space. The numbers of meningococci were 10- to 1000-fold higher in CSF than in serum. The blood levels did not reach 106 bacteria/ml—bacterial concentrations that are associated with the development of persistent septic shock, multiple organ failure, severe coagulopathy and large skin hemorrhages. Patients with pneumococcal meningitis had, as a group, significantly higher levels in 9 out of 19 inflammatory mediators suggesting a different and more harmful inflammatory response as compared with patients with meningococcal meningitis.
Footnotes
Declarations
Ethical approval was obtained from the ethics committees of all the institutes participating in the current study, and ethics approval letters obtained from the institutes prior to commencing the study are available. Each patient that has participated in this study gave his or her informed consent/assent. Parents or guardians gave informed consent/assent for children under 18 yr, or for patients who were not in a condition to communicate, after being informed of all the pros and cons of the study through the accessing patient information form. Consent for publication was a matter discussed within the patient information form and all the patients who participated in the current study, as well as the participating institutes, agreed that the data generated from the study using the samples taken from their body would be analyzed and published in scientific journals.
Availability of data and material
All the data generated from the examination of the samples taken from the study participants are presented within the manuscript. Laboratory data for each patient can be obtained by the corresponding author (PB).
Acknowledgements
We thank the nurses and laboratory staff at Tikur Anbessa Specialized University Hospital, Gondar Medical University Hospital, and Hawassa University Hospital for their assistance during patient recruitment and inclusion. We thank Jan Oksnes, Anne Witsø and Inger Marie Saga for assistance with sample processing and real-time PCR; and Tsegaye Hailu and Legesse Negash for assistance with data management. Tsehaynesh Lema, Beyene Moges, Admasu Tenna, Fitsum Woldegebriel, Melaku Yidnekachew, Wondale Mekonnen, Arslan Ahmed and Einar Rosenqvist are thanked for their scientific and technical contributions to the project. The Regional Health Authority Helse Sørøst in Norway and Oslo University Hospital Ullevål financed the analyses performed at Oslo University Hospital including quantification of NmDNA, LPS, measurements of 18 cytokines and MMP-9. The PCR analyses were performed independently at the National Institute of Public Health and Oslo University Hospital Ullevål. Armauer Hansen Research Institute (AHRI) contributed to the work from its core funds provided by NORAD and SIDA. Wude Mihret and Berit Sletbakk Brusletto contributed equally to this manuscript. All authors read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Norwegian Research Council GLOBVAC grants 192477 to ER and 220829 to DAC, and Department of Clinical Chemistry, Oslo University Hospital, Ullevål.
