Abstract
The objective of this study was to investigate the usefulness of soluble triggering receptor expressed on myeloid cells-1 (sTREM-1) in predicting short-term therapeutic response to methotrexate (MTX) in rheumatoid arthritis (RA). Patients with active RA, with Disease Activity Score-28 joints (DAS-28) >3.2, starting oral MTX, were included. We measured at baseline, 3 and 6 mo: DAS-28, Health Assessment Questionnaire–Disability Index (HAQ-DI), patient’s perception of disease severity, morning stiffness and pain, as well as modifications in sTREM-1 levels. A reduction in DAS-28 > 1.2 at 3 or 6 mo was considered adequate response. A significant decrease in DAS-28 was observed at 3 and 6 mo. HAQ-DI also decreased at 3 and 6 mo. No significant changes were observed in sTREM-1 levels at 3 or 6 mo. Using as cut-off a baseline value of sTREM-1 levels > 390 pg/ml, we obtained low values of sensitivity (61.5%), specificity (59.3%), positive predictive value (59.3%) and negative predictive value (61.5%) for adequate response to MTX at 3 mo. We found no clinical value of sTREM-1 levels in predicting therapeutic response to MTX in RA. Further studies should evaluate if sTREM-1 levels are predictive for other outcomes, including higher structural damage or good response to biologics.
Introduction
Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease characterized by chronic inflammation of synovial tissue, leading to the destruction of cartilage and bone in the joints. Methotrexate (MTX) is considered as a cornerstone of drugs employed as first-line treatment to control disease activity in RA. The MTX rate of efficacy to achieve a therapeutic success rate as monotherapy is around 35%. 1 However, rate of therapeutic response in order to achieve significant clinical response (defined as achieving with the treatment a disease remission or at least low-disease activity), is only around 50%. 2 There is a high proportion of patients who do not exert a significant clinical response, and to date there is not a single biomarker used to predict failure to MTX at the onset of therapy. 3 Seeking new biomarkers that may aid in a clinical decision for prescribing MTX remains an important issue in the research agenda. Within this context, triggering receptor expressed on myeloid cells-1 (TREM-1) is a transmembrane receptor belonging to the immunoglobulins superfamily that is expressed on myeloid cells, including neutrophils, monocytes and macrophages. 4 TREM-1 acts as an amplifier of the inflammatory response up-regulated by several stimuli such as pro-inflammatory cytokines and toll-like receptor (TLR) ligands. 5 TREM-1 has a membrane domain binding to DAP12, which is an immunoreceptor tyrosine-based activation motif-containing adaptor molecule. TREM-1 triggers the processes for secretion of pro-inflammatory cytokines, and other molecules potentiating the inflammatory response, including TNF-α, IL-8, granulocyte macrophage colony-stimulating factor, monocyte chemotactic protein 1 and myeloperoxidase.5,6 TREM-1 mRNA is overexpressed in synovial samples obtained from patients with RA. 7 In these patients with RA there is an increase of the number of TREM-1-positive cells in the synovium, which can be demonstrated by immunohistochemistry techniques. 7 More recently, some cross-sectional studies have evaluated the relation of soluble TREM-1 (sTREM-1) with some clinical characteristics in RA.7,8 These studies performed in patients with RA observed that high levels of sTREM-1 are related to disease activity, C-reactive protein levels (CRP), elevated erythrocyte sedimentation rate (ESR), and an increase in TNF-α levels.7,8 However, these studies are limited to evaluating whether sTREM-1 levels have a potential utility as a biomarker to predict the therapeutic response to synthetic disease-modifying antirheumatic drugs (synthetic DMARDs). In order to evaluate this important issue, follow-up studies are required. Although, to the best of our knowledge, to date there is only a cohort study performed, with the aim of evaluating whether there is an association between serum sTREM-1 and clinical response in early RA treated with DMARDs. 9 Molad et al. found that high sTREM-1 levels in RA predict a high Disease Activity Score assessing 28 joints (DAS)-28 after onset of therapy in patients who were DMARD naïve. 9 However, a limitation of this study was the presence of a wide heterogeneity in therapeutic schemas, making interpretation of the study results difficult. To date, there is a lack of information about the value of high sTREM-1 levels in identifying differences in therapeutic response to specific synthetic DMARDs. Therefore, we investigated the usefulness of serum levels of sTREM-1 for predicting short-term therapeutic response to MTX in patients with RA.
Materials and methods
Study design
This was a prospective cohort study. This study was conducted according to the recommendations described by the 64th Declaration of Helsinki and was in accordance with the ethical standards of the Ethics and Research Board of Hospital General Regional 110 del Instituto Mexicano del Seguro Social (approval code: R-2012-1303-48). Informed consent was obtained from all individual participants included in the study.
Study development
This observational cohort study included Mexican patients with RA who were consecutively enrolled at an outpatient rheumatology clinic of a secondary-care hospital [Hospital General Regional 110, Instituto Mexicano del Seguro Social (IMSS)] in Guadalajara, Mexico. All patients were included if they met the following criteria: (1) 1987 American College of Rheumatology criteria for RA; (2) were ≥18 yr of age; (3) had moderate or severe active disease using the operative definition of a DAS-28 joint disease >3.2l and (4) had not received MTX for at least ≥6 mo. Patients with prednisone (or equivalent) were included if the doses were <10 mg/d. The prednisone doses were prescribed at the last consult prior to starting this cohort; the rheumatologist kept the prednisone doses stable before the study onset and during the follow-up. Another inclusion criterion was no prescription of concomitant treatment with other synthetic DMARDs, including sulfasalazine, antimalarials or leflunomide at the start of the study. Patients with RA with a history of taking any of these DMARDs were allowed to participate only if they had at least a period of 3 mo without taking these drugs before the study onset.
Some reasons for not taking these synthetic DMARDs for 3 or more mo before starting this study were: (1) relative or absolute contraindications to receive these other DMARDs; (2) a lack of therapeutic adherence; (3) to have a history of moderate or severe side effects; and (4) inefficacy to these drugs. Patients receiving non-steroidal anti-inflammatory drugs and/or analgesics, such as paracetamol or tramadol, were allowed to participate.
We excluded patients with any of the following conditions: (1) pregnancy or nursing; (2) active infection at the time of study inclusion; (3) history of receiving treatment with cyclophosphamide or azathioprine; and (4) patients receiving prednisone or equivalent ≥10 mg/d. Additionally, we also excluded the following: (1) patients who were candidates to receive combined therapy with synthetic or biologic DMARD according to their rheumatologists; (2) chronic infections such as hepatitis B or C and HIV; (3) patients with overlap syndrome (considered to be an autoimmune connective tissue disease in which the same patient might present with clinical features of two or more diseases, mainly RHUPUS disease where patients have simultaneous features of systemic lupus erythematosus and RA); (4) patients with transaminases more than twofold the normal values; (5) pulmonary fibrosis; (6) serum creatinine >1.2 mg/dl; and (7) any other contraindications for MTX.
Clinical assessment at baseline and follow-up
At baseline, all patients were interviewed during the first visit prior to the prescription of MTX by trained researchers using a structured questionnaire to assess sociodemographic and clinical variables. These researchers also assessed disease activity using the DAS-28 index. Briefly, DAS-28 is a validated modified disease activity score of 28 joints that combines four single response measures into a global continuous score of RA disease activity. The components of DAS-28 are: (1) a 28 tender joint count, (2) 28 swollen joint count; (3) the results of ESR or CRP taken at time of the clinical evaluation; and (4) a patient’s perception of his/her general health described on a visual analogue scale (VAS) from 0 to 100. For this study, we chose the value of ESR for computation of DAS-28. We also assessed impairment in functioning using the Health Assessment Questionnaire–Disability Index (HAQ-DI), and patient perception of pain, morning stiffness and severity of disease activity (using VAS from 0 to 100). During the first visit, a peripheral blood sample was taken to measure the sTREM-1 levels and other laboratory determinations in order to classify patients into two groups: (1) high serum sTREM-1 levels; and (2) low sTREM-1 levels (Figure 1). Every patient was scheduled for a second and third visit at 3 and 6 mo. During follow-up, we assessed changes in DAS-28, HAQ-DI and VAS results for pain, morning stiffness,and severity of disease activity, as well as sTREM-1 levels.
Study flow chart.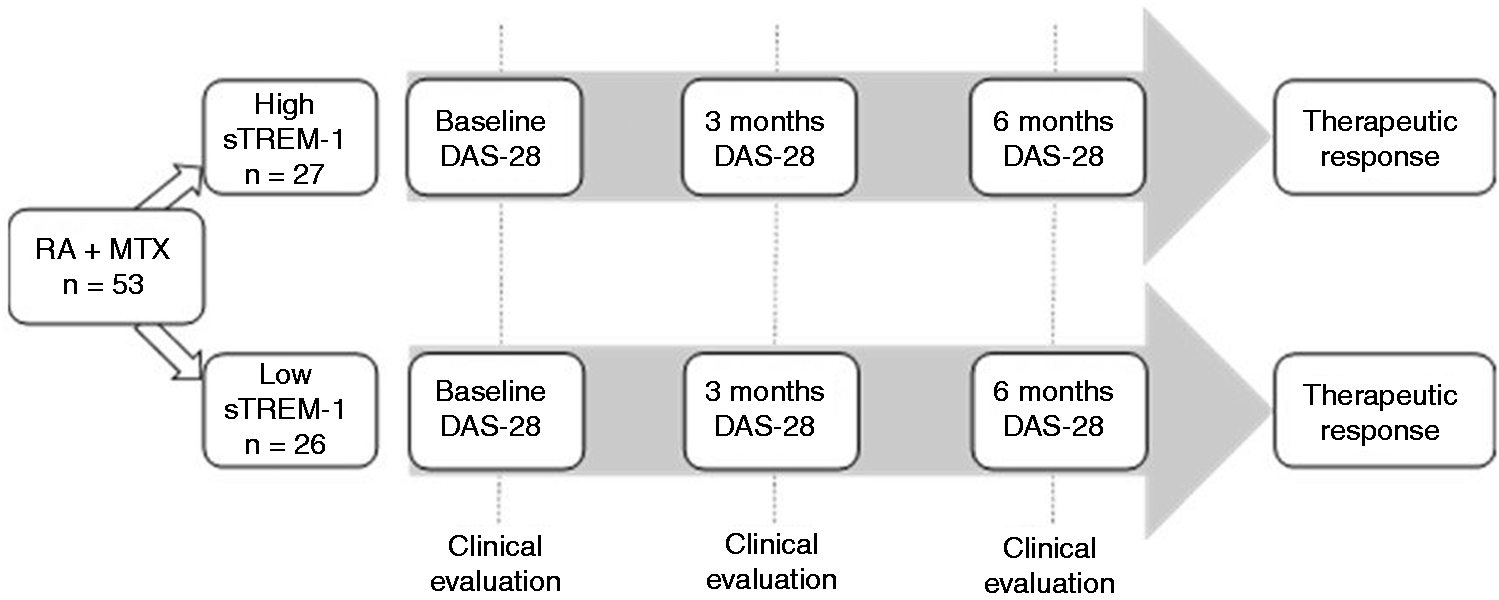
sTREM-1 levels determinations in serum
Blood samples were obtained from all patients at baseline (prior to MTX onset), and during visits at 3 and 6 mo in order to determine serum sTREM-1 levels expressed in pg/ml, which were quantified using ELISA (CUSABIO; Biotech Co. Wuhan, China). This ELISA kit, pre-coated with a specific sTREM-1 Ab, has a detection range of 7.8–500 pg/ml recombinant and natural human sTREM-1 with no significant cross-reactivity of interference, sensitivity of <1.95 pg/ml, intra-assay precision of <8% and inter-assay precision of <10%. We also included determination of CRP and rheumatoid factor (RF) levels (by nephelometry) and ESR by Westergreen.
Primary response
Primary response was defined as a decrease of DAS-28 > 1.2 points. During follow-up, these measures of response to the treatment were evaluated at 3 and 6 mo.
Other secondary response variables
Improvement in secondary variables was identified as significant decrease in HAQ-DI, improvement in patient’s perception of the severity for pain, and morning stiffness, and physician’s perception of the severity for disease activity (VAS 0–100).
Statistical analysis
We hypothesized that sTREM-1 levels might predict the therapeutic response to MTX. Therefore, we classified the therapeutic response in two groups: (1) responders (decrease in DAS-28 > 1.2 from the baseline score); and (2) non-responders (patients with RA that did not achieve this decrease in DAS-28). Categorical variables are expressed as frequencies (%) and continuous variables expressed as medians and ranges. Baseline comparisons between responders and non-responders were done for continuous variables with the Mann–Whitney U-test, and for comparisons between categorical variables we used the χ 2 (or Fisher’s exact test when required). Correlations between serum sTREM-1 levels, with other continuous variables (DAS-28, ESR, CRP concentrations and RF titers) were computed by Spearman’s correlation coefficient (rs). Comparison of the modifications of the values of continuous variables (DAS-28, HAQ-DI and sTREM-1 levels) at baseline vs. 3 and 6 mo was computed by Wilcoxon rank-sum tests. We performed a receiver operating characteristic (ROC) analysis in order to identify the cut-off point of sTREM-1 baseline levels that were more efficient for discriminating between responders and non-responders at 3 mo. To do this, we used as gold standard the presence of therapeutic response based on decrease on DAS28 > 1.2 at 3 mo and a graph of sensitivity, and 1-specificity was run using as diagnostic test the baseline values of sTREM-1.
Statistical significance was set at P < 0.05. All analyses were performed with SPSS v. 23 statistical software (IBM, Armonk, NY, USA).
Results
A total of 148 patients with RA were screened. Of these, 95 patients were excluded for the following reasons: active infection (n = 26); antecedents of intolerance to MTX (n = 12); receiving, at the time of study onset, a dose of prednisone ≥10 mg/d (n = 37); and receiving, at the time of study onset, a combination of synthetic DMARD (n = 20).
Fifty-three patients with RA were included in the study after the prescription of MTX (baseline) and were followed to 6 mo. At 3 mo, all the patients included remained under treatment with MTX, but at 6 mo, only 35 patients remained in the study. Eighteen patients dropped out of the study because they declined to continue in the cohort.
Clinical characteristics of the patients included in the cohort.
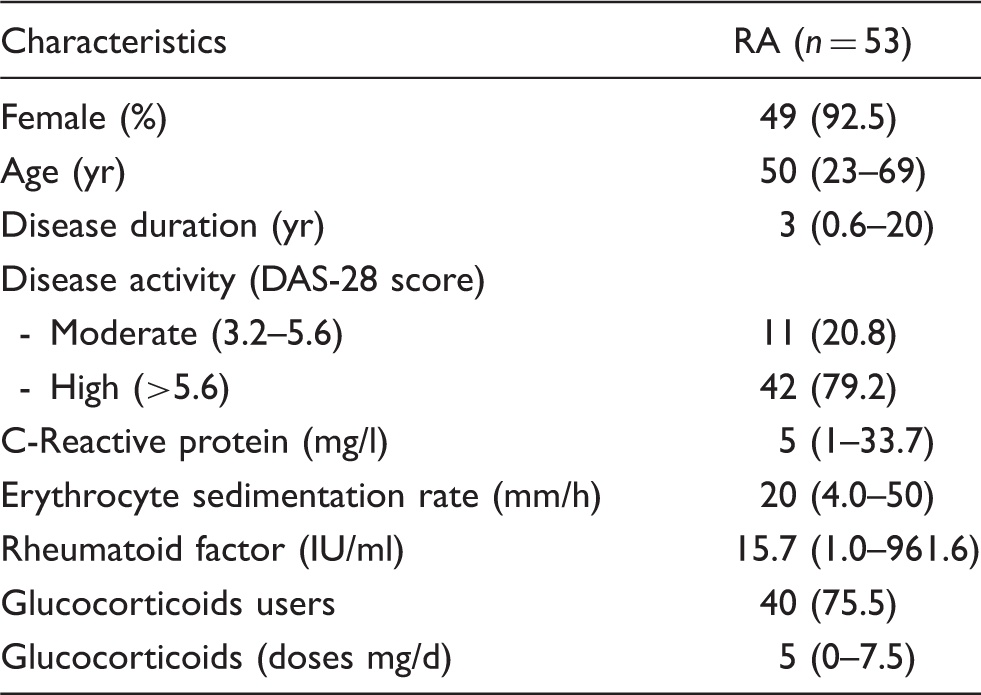
Quantitative variables are expressed at median (range), qualitative variables expressed on n (%).
Comparison of changes in clinical variables and sTREM-1 levels from baseline to 3 and 6 mo.
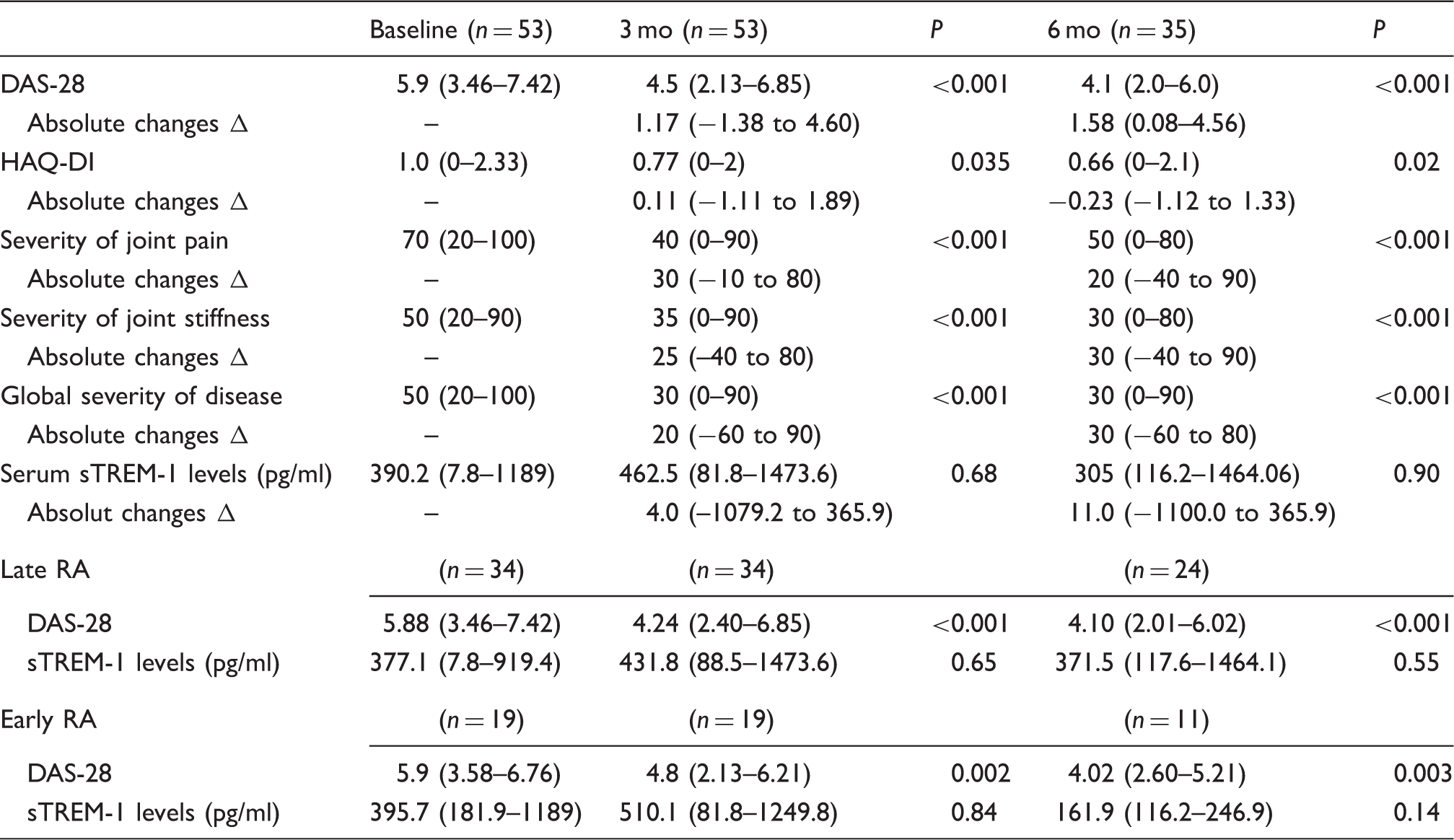
Severity of joint pain, joint stiffness, and global severity of disease were based on VAS from 0 to 100, with 100 indicating highest severity. Late RA≥2 yr; early RA < 2 yr; absolute changes Δ correspond to the difference between 3 mo and baseline, and 6 mo and baseline. Comparison of medians between follow-up and baseline was computed using Wilcoxon Rank-sum test.
Comparison of clinical and laboratory characteristics between patients who achieved therapeutic response at 3 and 6 mo (decrease in DAS-28 > 1.2) vs. patients who did not achieve this response.
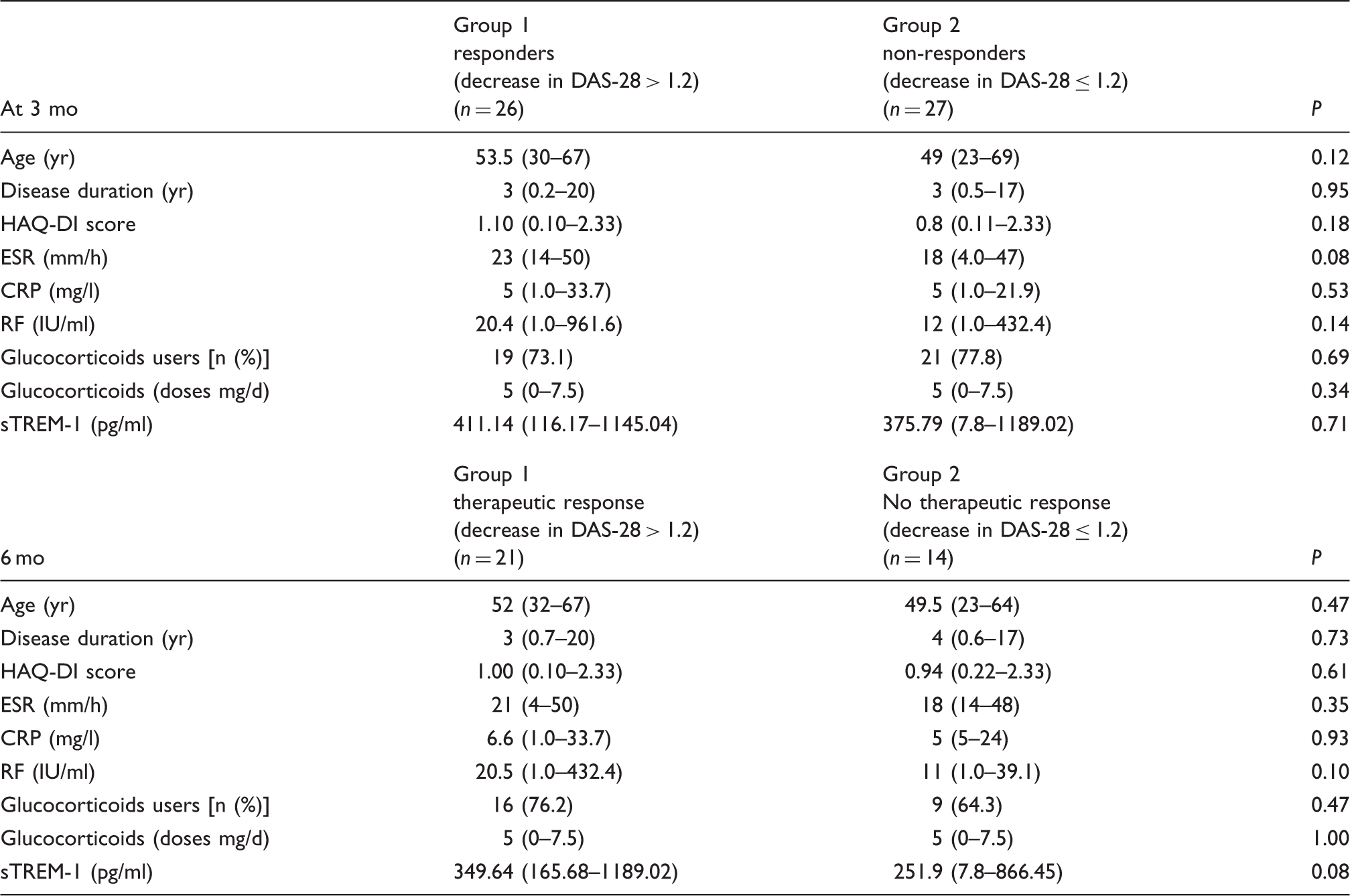
Quantitative variables are expressed in median (ranges). Comparison of medians with Mann–Whitney U test. Responders were considered if they achieved a decrease in DAS-28 > 1.2 regarding to the baseline values. Otherwise, patients were classified as non-responders. Comparison of medians between follow-up and baseline was computed using Wilcoxon rank-sum test.
We performed a ROC analysis in order to find the values of sTREM-1 that were more efficient to discriminate between responders and non-responders at 3 mo. After testing different cut-off points, a value of >390 pg/ml was considered the most efficient with a sensitivity of 55% and specificity of 52% (data not shown). This value of 390 pg/ml also corresponds with the median values for sTREM-1 of patients with RA included in this cohort.
Sensitivity, specificity and predictive values of high levels of sTREM-1 (>390 pg/ml) for predicting therapeutic response (decrease in DAS-28 > 1.2 at 3 and 6 mo with respect to the baseline values).
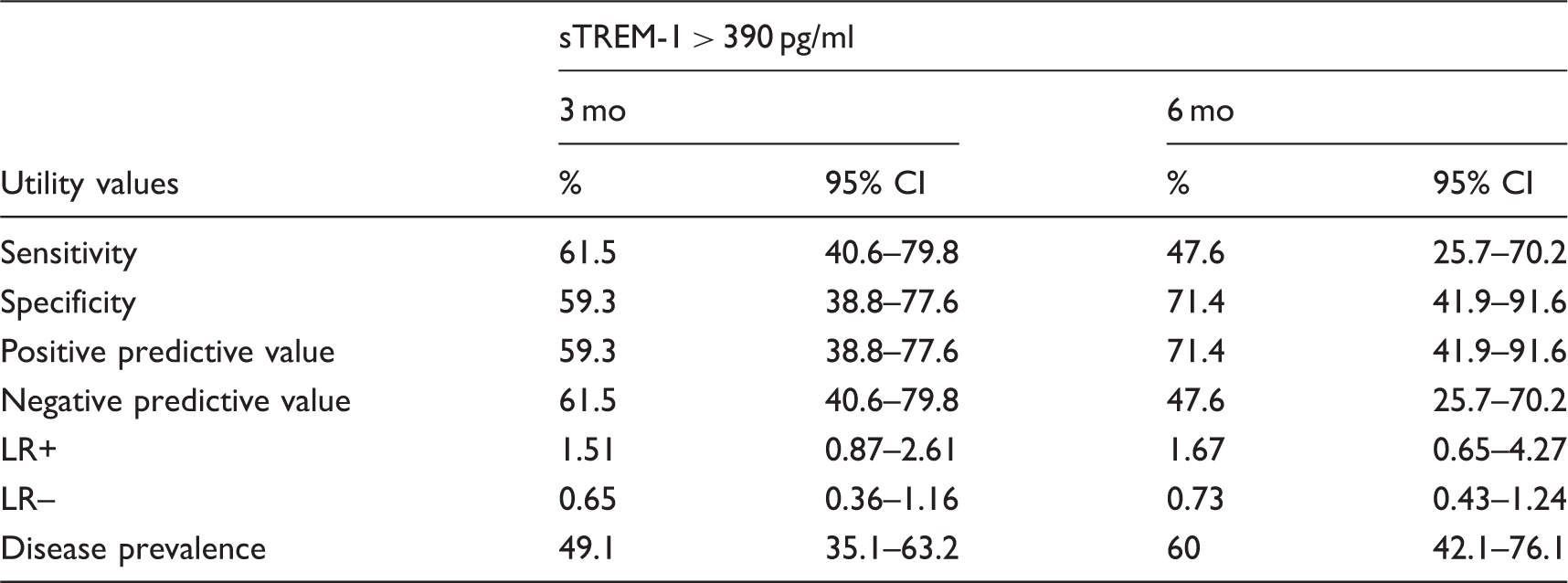
LR: Likelihood ratio.
Discussion
Our results showed that sTREM-1 levels at baseline do not predict therapeutic response to MTX in patients with established RA. Although patients receiving MTX had a significant decrease in DAS-28, as well as an improvement in functioning at 3 and 6 mo, these changes were not followed by modifications in sTREM-1 levels.
TREM-1 is a member of the superfamily of immunoglobulins, expressed as transmembrane receptor on myeloid cells-1. 10 TREM-1 acts as an amplifier of the inflammatory response and induces the secretion TNF-α, IL-8 and others pro-inflammatory cytokines. 6 After proteolytic cleavage of TREM-1, the main fragment is released into the circulation as sTREM-1. This soluble form acts as a decoy receptor, precluding TREM-1 activation by its ligands. Therefore, sTREM-1 acts as a competitor by the ligands of TREM-1, leading to a decrease of the cascade of pro-inflammatory mechanisms attributed to this binding. 11
Different studies have observed an increase in plasma sTREM-1 levels in RA.7–9 Choi et al. observed that sTREM-1 levels are associated with disease activity, higher levels of acute-phase reactants and increase in TNF-α. 8 Molad et al., 9 in a prospective cohort study, reported that higher sTREM-1 levels are associated with an increase in titers of anti-CCP Abs. However, Molad et al. observed differences in serum sTREM-1 due to other variables such as disease duration or treatments. 9 Molad et al. described that higher levels of sTREM-1 are observed in patients with early RA who never have been under treatment with DMARD vs. those with established RA under treatment with DMARDs. 9 These authors also identified that in early RA the treatment with synthetic or biologic DMARDS decreases sTREM-1 levels. 9 Nevertheless, some questions arise from these interesting results, the most relevant of which is was the modification of sTREM-1 levels observed influenced by the presence of potential confounders? One of the most important confounders introduced in the Molad et al. study is the differences in the mechanism of action and effects of synthetic and biologic DMARDs. 9 Biological agents have substantial differences in their effects on cytokines and there is a wide variation of effects of synthetic DMARDs on inflammation. The cohort of patients with early RA in the study by Molad et al. were treated with a wide variability of biological and synthetic DMARDs: a third of their patients received moderate doses of prednisone, 63% MTX, and antimalarials were prescribed to approximately half of their patients. 9 However, all patients with established RA that were included in that study received MTX, but half of them had combined therapy with other synthetic DMARD, including antimalarials, sulfasalazine or leflunomide; moreover, 55% of patients received biologics (including anti-TNF-α agents, anti-CD20 or anti-IL-6). This wide variability of drugs prescribed to these patients and mechanisms of action might influence the results on therapeutic response. Consequently, it is still necessary to demonstrate the effects of specific DMARDs on serum sTREM-1 levels. We tested the hypothesis that MTX might decrease not only serum sTREM-1 levels, but also that those sTREM-1 levels might be associated with the clinical response in DAS-28. MTX is a very interesting synthetic DMARD to be evaluated for studies of the value of clinical biomarkers not only because it is currently the most prescribed drug for controlling RA,12,13 but also because of its anti-inflammatory effects on immune response of cells, including T lymphocytes, myeloid cells (e.g. monocytes and granulocytes), and their effects related to inhibition of IL-8, IL-6 and leukotriene B4 secretion. 14 TREM-1 is a receptor expressed on myeloid cells, and its expression is up-regulated by several pro-inflammatory molecules. 6 After TREM-1 activation there is a proteolytic cleavage during the inflammatory process producing increased levels of sTREM-1. 15 High sTREM-1 levels have been observed in a series of inflammatory disorders such as with inflammatory bowel disease and ankylosing spondylitis and RA disorders.16,17 Therefore, it is reasonable to speculate that some of effects of MTX on myeloid cells might produce changes in sTREM-1 levels accomplishing the therapeutic response to this DMARD. However, high sTREM-1 levels reflect myeloid cell activation and these levels have been associated with disease activity, increase in acute-phase reactants and high TNF-α levels. 8
Nevertheless, in contrary to our main hypothesis, we did not observe any meaningful changes in sTREM-1 levels during MTX treatment. These findings, which contrast with previous findings described by other authors, 9 might be attributable to other factors such as that two-thirds of our patients had late RA. Additionally, we have other differences that can account for our results, one of which is that in contrary to the previous report evaluating sTREM-1 levels in a prospective cohort, 9 we used stricter criteria for the follow-up: we chose to use fixed times for each follow-up visit in this cohort, a strategy that increased the validity of our results. Additionally, in order to avoid the potential effect of confounders, our patients were not receiving other DMARDs during the follow-up, making evaluation of the effects of the treatment with MTX more reliable. Therefore, we consider that these strengths increase the validity of our conclusion that MTX does not modify the sTREM-1 levels in established RA, even if patients achieve a good therapeutic response with this DMARD.
Choi et al. observed that patients with a DAS-28 > 3.2 have higher plasma sTREM-1 levels, suggesting that this molecule contributes to the inflammatory process observed in RA. 8 Unfortunately, we were unable to confirm this finding because we included only patients with active RA (DAS-28 > 3.2) in order to evaluate if sTREM-1 levels might predict a therapeutic response. In contrast with the observations made by Molad et al. in patients with early RA where sTREM-1 levels were found to be significantly decreased because of the treatment, 9 we did not identify a decrease of sTREM-1 levels in the smaller sample of our patients with early RA (n = 19) under treatment with MTX. There are many possible explanations for these discrepancies in findings: firstly, Molad et al. did not provide information about the length of time to the second visit where they evaluated serum sTREM-1 levels9—variable times of follow-up are well-recognized confounders. Instead, our study was able to provide fixed times for the follow-up evaluations (measurements of sTREM-1 were performed at baseline, 3 mo and 6 mo after the start of MTX therapy). These fixed times for the clinical assessment and determination of sTREM-1 levels might decrease the measurement bias inherent to differences in times of follow-up. Secondly, Molad et al. also included a diverse range of treatments. 9 Moreover, most of their patients with established RA had two or more synthetic or biological DMARDs associated with MTX; these differences in therapeutic schemas contribute to difficulty in interpreting whether sTREM-1 levels decrease with some drugs or they are unaffected by others.
A confounder that might affect the sTREM-1 levels are high doses of corticosteroids. Patients in the study by Molad et al. had mean doses of prednisone of 20 mg/d, 9 given that this drug is a potential confounder that might affect the sTREM-1 levels. We instead excluded patients who received prednisone ≥10 mg/d. No correlation was observed between treatment with low-doses of glucocorticoid and sTREM-1 levels. Hence, low doses of glucocorticoids do not act as confounders regarding sTREM-1 levels.
Therefore, our study suggests that, based on a decrease in DAS-28 > 1.2 in discordance with sTREM-1 levels, there is no relation between high levels of sTREM-1 and differences in therapeutic response.
In our study, no correlation was found between serum sTREM-1 levels and ESR, CRP and RF, although sTREM-1 can be considered as an independent molecule of these biomarkers.
It is without doubt that seeking biomarkers of therapeutic response in late RA is a relevant issue. MTX is a synthetic DMARD used as first-line agent and is more frequently prescribed as monotherapy in newly diagnosed RA. However, there are some circumstances where MTX continues to be prescribed as monotherapy in late RA. In developing and developed countries there are studies demonstrating a delay of several years to first visit to a rheumatologist for the following reasons: (1) the interval from the time a patient suffers the first symptom and seeking attention from primary care physicians (PCP); and (2) the interval between the first consultation with the PCP and the referral to the rheumatologist.18,19 This problem increases with the lack of agreement over diagnosis between the PCP and rheumatologists where patients with RA are erroneously diagnosed with other diseases. 20 In consequence, many patients with established RA initiating MTX might have a delay of several years before referral to a rheumatologist after the onset of the first RA-related symptom. We have previously reported that a subgroup of patients with RA had a mean of delay of treatment onset with a synthetic DMARD of 2–9 yr. 21 Another important reason for prescription of monotherapy with MTX in late RA is related to the high rates of MTX suspension related to lack of adherence or moderate/severe side effects. Lack of adherence to MTX reported by some authors ranges from 10% to 50% in the first 9 mo after the prescription of this DMARD. 21 In their study, Thurah et al. observed that at 5 yr, 95% of patients prescribed MTX had gaps longer that 90 d without receiving MTX. 22 The persistence of prescription to MTX as monotherapy in late RA has been described as 50% at 5 yr and 30% at 10 yr. 23 Therefore, in the clinical context, it is a common practice to re-introduce the prescription of this DMARD. Consequently, the identification of potential biomarkers predicting therapeutic response for this DMARD could be useful for clinicians.
Our study has several limitations. First, we were unable to compare our sTREM-1 levels between patients with active vs. inactive RA. These data should be used to identify the value of sTREM-1 as a marker of disease activity. Second, we could only provide the absence of changes in sTREM-1 values exclusively in patients treated with MTX. Other studies evaluating different synthetic or biologic DMARDs should be analyzed.
We conclude that there are no changes in sTREM-1 levels in patients with late RA using MTX; moreover, these levels are not useful to predict therapeutic response in patients with late RA treated with this DMARD. In consequence, sTREM-1 levels are not useful as a clinical tool with which to discriminate differences in probability of therapeutic response in these patients.
Future studies utilizing other synthetic DMARDs or biological agents should be performed in order to identify whether sTREM-1 levels may change with these therapies or whether these levels can be used as a parameter to be associated with therapeutic response.
Footnotes
Acknowledgements
The authors wish to thank to IMSS Foundation (Fundacion IMSS, A.C.) for their support of this research. The authors also thank Maggie Brunner, M.A., for editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the ‘Fondo de Investigación en Salud’ del Instituto Mexicano del Seguro Social (Grant: FIS/IMSS/PROT/G12/1136). Dr Gonzalez-Lopez holds a Fundacion IMSS, A.C research scholarship (Beca de Excelencia en Investigación 2016 por la Fundación IMSS, A. C.)
