Abstract
Our aim was to examine the effect of low-volume 0.9% NaCl adenosine, lidocaine and Mg2+ (ALM) ‘drip’ on early immune-inflammatory activation after a single laparotomy with no further manipulation. Male Sprague–Dawley rats were anesthetized and randomly assigned to one of the groups, baseline, 1 h infusion 0.9% NaCl ± ALM and metrics, 1 h infusion and 6-h metrics, and 6 h continuous infusion and metrics. Complete blood count, acid–base balance, systemic levels of IL-6 and IL-10, and coagulation status were measured. After 1 h, there was a disproportionate increase in circulating neutrophils between saline and ALM groups despite an identical 45% fall in lymphocytes. Disproportionate increases also occurred in platelet counts 1 h after surgery, and saline controls had increased respiratory alkalosis at 6 h with higher lactate. Systemic inflammation was also evident after 1 h in both groups (plasma IL-6 increase) and was amplified in saline-controls after 6 h. The ALM group increased anti-inflammatory cytokine IL-10. Surgery was not associated with acute coagulopathy; however, there were significant reductions in fibrinolysis. Following a single laparotomy, ALM infusion appeared to reduce stress-induced release of neutrophils and platelets into the circulation, and reduced acid–base disturbance. After 1 h, both groups had similar IL-6 levels, but ALM animals had increased IL-10, indicating improved inflammatory balance. The uncoupling of inflammation and coagulation activation but not fibrinolysis may offer a unique opportunity to investigate differential activation of innate immunity in response to sterile injury in this model.
Introduction
Surgical trauma is defined as an injury produced by or related to major surgery. World health authorities are increasingly aware of the need to improve the quality of surgical interventions and make surgery safer for the patient and more predicable for the surgeon.1–3 In a recent analysis of over 400,000 trauma patients in the UK, 4 the authors concluded that the surgical complications were largely physiologic, involving primary cardiac, renal, pulmonary, central nervous system and infectious events. 4 It has been estimated that up to 50% of perioperative adverse events may be preventable, 3 and many of these have been linked to the stress response to surgery. 5 The stress response is the body’s response to an injury, pain, hemorrhage, fluid loading, ischemia or infection, and operates at many levels, including activation of the sympathetic nervous–endocrine system [e.g. hypothalamic–pituitary–adrenal (HPA) axis], hematopoietic dysfunction, coagulopathy, immune-inflammatory activation and metabolic disturbances.3,6 From an evolutionary perspective, these innate responses have developed to promote healing, remove dead cells and protect the host from invading pathogens. However, under certain conditions, such as major surgery, collateral damage can occur from ‘overexpression’ of the immune and inflammatory systems, which normally are self-limiting and restorative. These complications can lead to secondary-hit events such as organ dysfunction and failure, immunosuppression and infection.
Over the past 8 years, we have been developing a small-volume resuscitation fluid therapy for acute hemorrhagic shock with and without traumatic brain injury, comprising 7.5% or 3% NaCl with adenosine, lidocaine and Mg2+ (ALM) bolus, and a normal saline ALM ‘drip’ for continuum-of-care prevention of secondary ‘hit’ complications. 7 The ALM bolus and/or infusion treatments have been shown in rat and pig models to protect against regional myocardial ischemia, lethal arrhythmias, cardiac arrest, hemorrhagic shock, coagulopathy, inflammation, endotoxemia, infection and sepsis.7–12 Adenosine, lidocaine and Mg2+ alone do not confer these benefits. 7 In the present study, we hypothesized that the low-volume ALM drip/infusion may improve physiologic status in the first few hours following a single laparotomy. The aim of the present study was to examine the effect of a front-line, low-volume 0.9% NaCl ALM ‘drip’ on the innate hematopoietic–immune response, inflammation and coagulopathy in a rat model of single laparotomy with no further manipulation and monitored up to 6 h.
Materials and methods
Animals and ethics
Male Sprague–Dawley rats (394 ± 8 g; n = 42) were obtained from James Cook University’s Breeding Colony, Townsville, Australia. All animals were housed in a 14 h–10 h light–dark cycle with free access to food and water ad libitum. Animals were anesthetized with 5% isoflurane in 100% oxygen, with 1.5 ± 0.1% isoflurane/100% O2 for maintenance throughout surgery. Animals received 5 mg/kg subcutaneous Carprieve (Provet, Townsville, Australia) in 1 ml 0.9% NaCl at the time of anesthesia induction for postoperative analgesia. Prior to anaesthesia recovery 2% lidocaine gel was applied topically to all incision sites. At study completion, animals were euthanized with 100 mg/kg thiopentone sodium (Thiobarb; Lyppard, Townsville, Australia). The study complies with the Guide for Care and Use of Laboratory Animals (NIH, 8th ed., 2011) and was approved by James Cook University Animal Ethics Committee (A2385). 13
Surgical protocol
Following anesthesia, sterile chronic catheters (Access Technologies, Skokie, IL, USA) were implanted in the left femoral vein and artery and connected to a vascular access harness (Instech, Plymouth Meeting, PA, USA), allowing for infusions and hemodynamic monitoring (Powerlab, ADInstruments, Bella Vista, Australia).
14
The animals remained anesthetized for a 20 min stabilization period prior to laparotomy. Baseline measurements of blood pressure parameters were recorded, and repeated at 1, 3 and 6 h (Figure 1). After the stabilization period, a 5-cm transverse laparotomy was performed, measured 2 cm below the xiphoid process. Immediately after the laparotomy was re-measured ensuring 5 cm opening of the abdominal wall, and the muscle and fascia was closed with running braided silk suture followed by a running suture for skin closure.
(a) Schematic of the laparotomy surgery in vivo rat model. Animals were anesthetized until after skin closure. Blood samples were taken at baseline and prior to sacrifice at 1 h and 6 h. Infusions (0.5 ml/kg/h) were with 0.9% NaCl only or 0.9% NaCl with ALM. (b) Schematic of a ROTEM Trace showing the key coagulation parameters measuring clot initiation, propagation and growth, and clot breakdown or lysis. CT is time to initiation of the clot from thrombin formation and start of fibrin polymerization. CFT is the time from initiation of the clot until a firmness of 20 mm is reached. Clot propagation and growth involves increasing stabilization of the clot until maximum firmness, followed by clot breakdown (see ‘Materials and methods’ for details).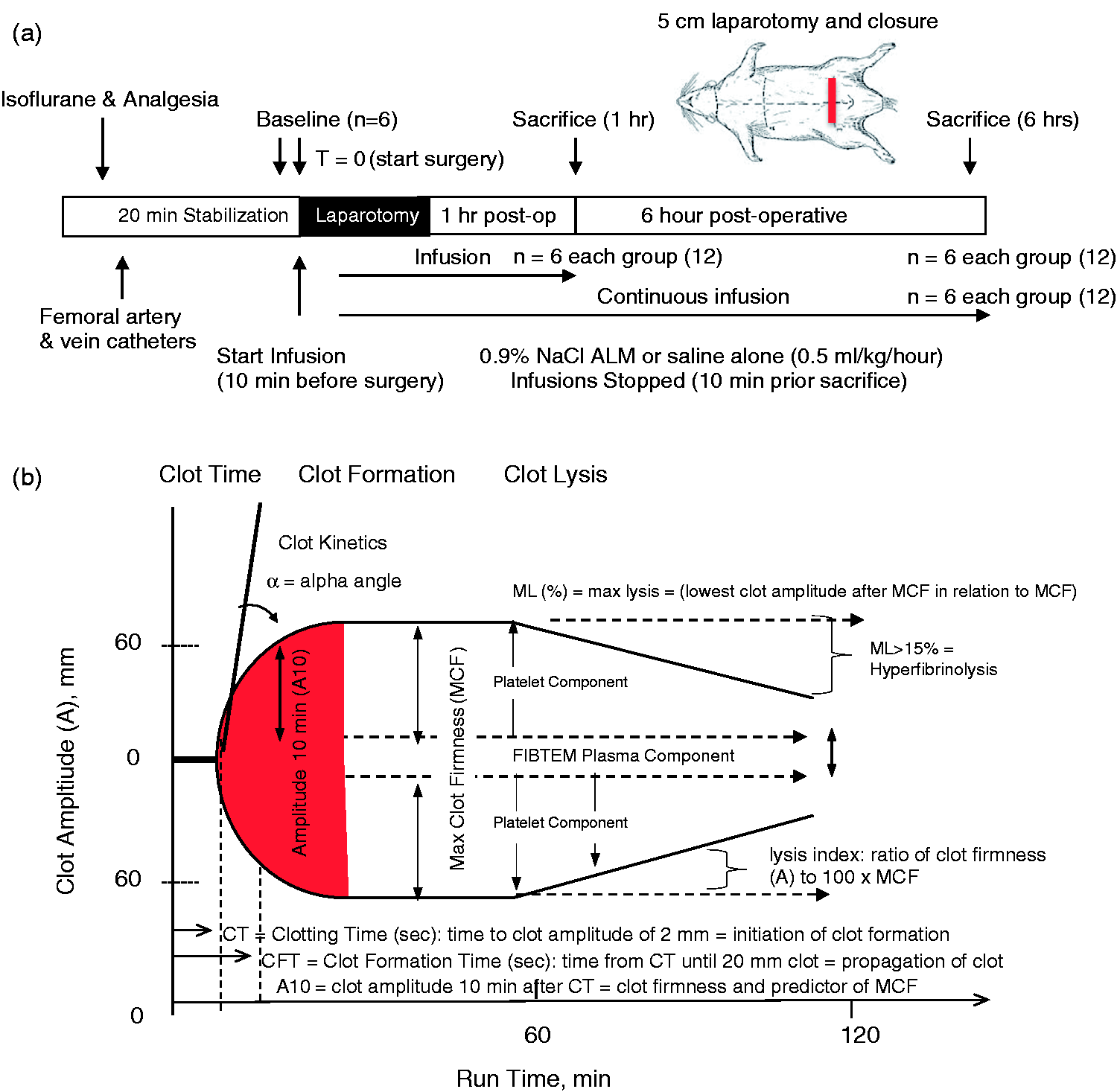
Experimental design
Animals were randomly assigned to one of seven groups (n = 6 per group) (see Figure 1): (1) baseline (n = 6); (2) 1 h infusion of 0.9% NaCl, sacrificed 1 h after laparotomy (Saline 1 h); (3) 1 h infusion of 0.9% NaCl, sacrificed 6 h after laparotomy (Saline 6 h); (4) 6 h infusion 0.9% NaCl infusion, sacrificed 6 h after laparotomy (Saline continuous); (5) 1 h infusion of 0.9% NaCl ALM treatment, sacrificed 1 h after laparotomy (1 h ALM); (6) 1 h infusion of 0.9% NaCl ALM treatment, sacrificed 6 h after laparotomy (ALM 6 h); or (7) 6 h infusion 0.9% NaCl ALM treatment, sacrificed 6 h after laparotomy (ALM continuous). ALM i.v. solution comprised 50 mg adenosine, 100 mg lidocaine and 50 mg MgSO4/10 ml 0.9% NaCl. Intravenous infusions (0.5 ml/kg/h) commenced 10 min prior to laparotomy (Figure 1).
Blood gases, electrolytes, lactate and complete blood count
Arterial blood gases, electrolytes (pH, pO2, pCO2, Cl, Na, K, Ca, sO2, base excess, HCO3) and lactate were measured at the end of monitoring using the Radiometer ABL800 blood gas analyzer (Radiometer Pacific, Mt Waverley, VIC, Australia). The VetScan HM5 Hematology system (REM Systems, Macquarie Park, New South Wales) was used for blood cell counts at baseline, 1 and 6 h.
Inflammatory analysis
Plasma concentrations of IL-6 and IL-10 were quantified with rat-specific ELISA kits (Novex® by Life Technologies, Mulgrave, VIC, Australia) using the Thermoscientific Multiskan FC. Kits were performed in accordance with manufacturer’s instructions, and samples were measured in duplicate as per literature standard with the average value of the duplicate used for each animal. Detections ranges, sensitivity, and intra- and inter-assay CVs for IL-6 were 23.5–1500 pg/ml, < 5 pg/ml, 5.8% and 8.8%, respectively; and for IL-10 were 15.6–1000 pg/ml, < 5 pg/ml, 7.5% and 9.3%, respectively. R2 values for the standard curves for IL-6 and IL-10 were 0.9947 and 0.9633, respectively.
Rotational thromboelastometry (ROTEM®)
ROTEM® (Tem International, Munich, Germany) was conducted according to manufacturer’s instructions. 15 Whole blood collected in 3.2% sodium citrate tubes was warmed to 37°C. Three assays were performed: EXTEM (extrinsically activated test using tissue factor), INTEM (intrinsically activated test using ellagic acid) and FIBTEM (fibrin-based EXTEM-activated test with 50 µg/ml cytochalasin D, to inhibit platelet contribution to clot formation). 15 All assays were run for 60 min. ROTEM parameters include clotting time (CT, sec); clot formation time (CFT, sec); alpha angle (α°); clot amplitude (A, mm); maximum clot firmness (MCF, mm); and maximum clot elasticity (MCE). Clot lysis parameters include maximum lysis (ML, %) and lysis index (LI, %). LI30, 45 and 60 represent the percentage of remaining clot firmness in relation to the MCF value at 30, 45 and 60 min after CT. Quality control measurements (ROTROL-N and ROTROL-P) were performed weekly. 15
Prothrombin time and activated partial thromboplastin time
Prothrombin time (PT) and activated partial thromboplastin time (aPTT) were measured on the KC4 delta coagulometer (TCoag, Wicklow, Ireland) as previously described. 14 Briefly, arterial blood collected in 3.2% sodium citrate vacutainers (BD, North Ryde, NSW, Australia) was centrifuged at 1500 g at room temperature, and plasma stored at −80°C until analysis. PT and aPTT were determined using STA-Neoplastine® Cl Plus (Lot: 250461) and TriniCLOT aPTT HS reagent (Lot: G203641) (Diagnostica Stago, Donacaster, VIC, Australia). Measurements were performed in triplicate, and assays were terminated after 5 min.
Statistical analysis
SPSS Statistical Package 22 was used for all statistical analysis (IBM, St Leonards, New South Wales). All values are expressed as mean ± SEM. Data normality was assessed numerically with Shapiro–Wilks, and Levene’s test was used to determine equality of variances. Parametric data were evaluated using ANOVA followed by LSD or Dunnett’s C post-hoc test. Two-way ANOVA comparison was used for within group comparisons. Non-parametric data was evaluated using Kruskal–Wallis. Data generated from ELISAs was analyzed using Prism 7 4-parameter-logistic curve fitting (GraphPad, La Jolla, USA). Statistical significance was defined as P < 0.05.
Results
Hemodynamics
There were no significant differences in hemodynamics between groups (data not shown). The mean arterial pressure at baseline was 107 ± 2 mmHg and increased by approximately 20% at 3 and 6 h in both groups. The pulse pressures and heart rates were not significantly different among the groups (data not shown).
Hematology profile
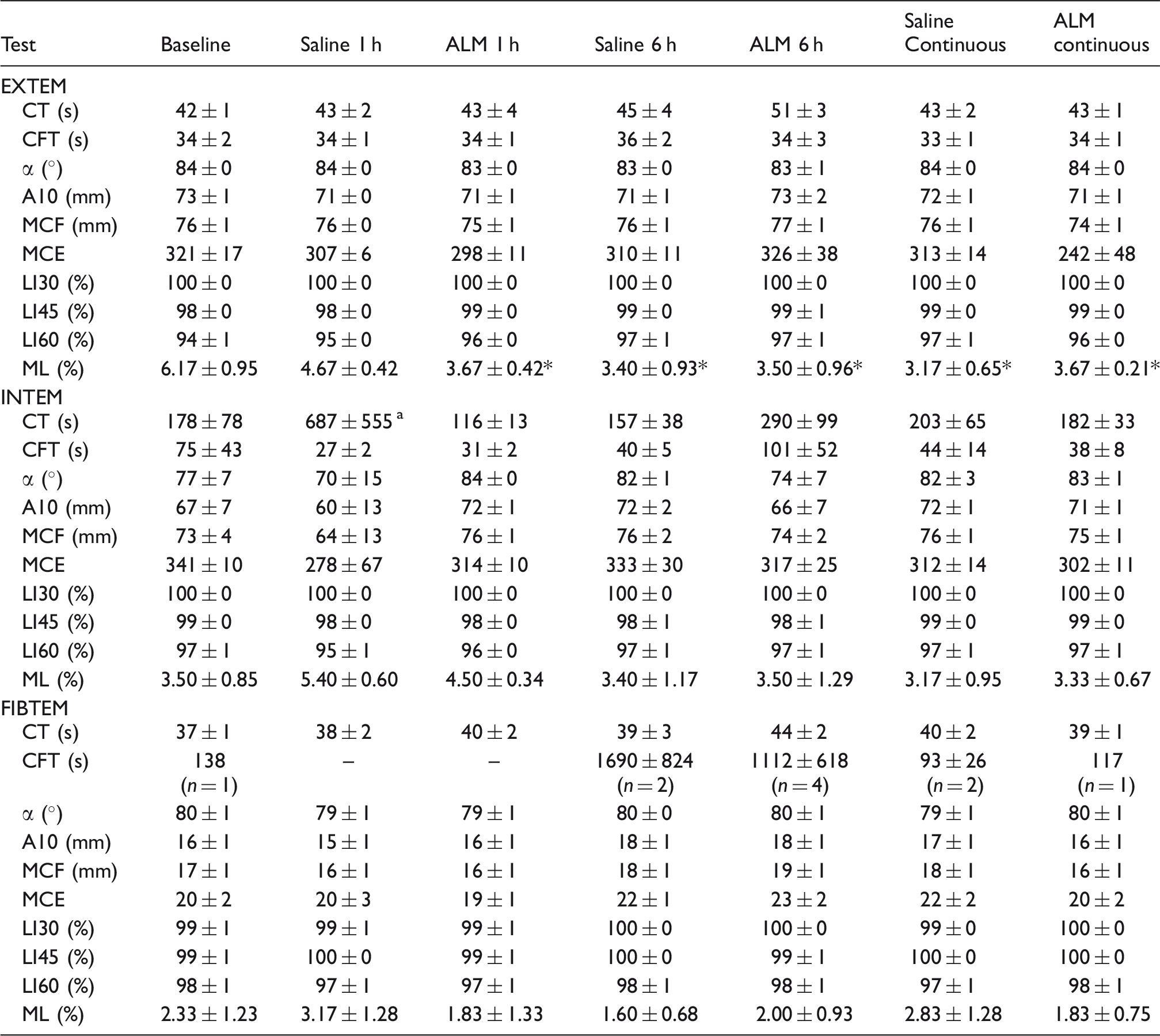
Complete blood counts at baseline, Saline 1 h, ALM 1 h, Saline 6 h, ALM 6 h, Saline continuous, ALM continuous.
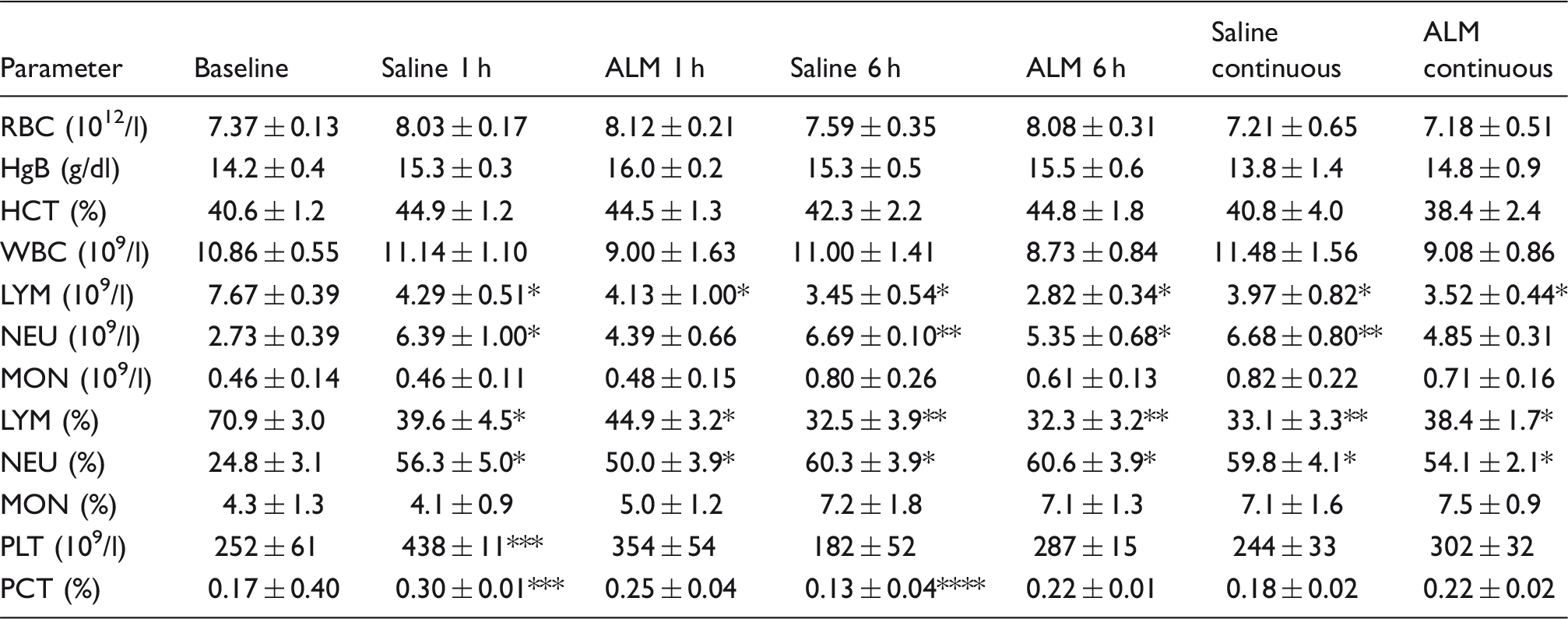
Data represent mean ± SEM. n = 6 for all values. Lymphocytes (LYM), monocytes (MON) and neutrophils (NEU) are presented as absolute values and percentage of total white blood cell (WBC) count. RBC: red blood cells; HgB: hemoglobin; HCT: hematocrit; PLT: platelets; PCT: platelet crit. *P < 0.05 compared with baseline; **P < 0.05 compared with baseline and ALM 1 h; ***P < 0.05 compared with Saline 6 h, ALM 6 h (after 1 h infusion), and saline continuous; ****P < 0.05 compared with ALM 6 h.
One-h infusion (1 h sacrifice)
Lymphocyte counts at baseline were 71% of total white cell counts and significantly decreased by 44–46% in both saline control and ALM groups after 1 h relative to baseline (Table 1). The identical lymphocyte decrease was accompanied by a significant increase in neutrophils in saline controls (2.3 times) and 1.6 times in ALM-treated animals compared with baseline. Monocytes did not change in both groups. After 1 h infusion, platelet counts in saline controls increased 1.74 times and 1.4 times in the ALM group (Table 1). The platelet hematocrit (%) also paralleled the changes in counts in both groups.
One-h infusion (6 h sacrifice)
In a separate group (see Figure 1a), lymphocyte numbers continued to decrease after 6 h compared with 1 h sacrifice. Lymphocytes fell by 55% in saline controls and 63% in ALM rats compared with baseline. Neutrophils also continued to increase after 6 h and were significantly higher in saline controls than ALM infused animals (at 1 h). This was accompanied by greater increases in monocytes in saline controls (1.7 times) than in the ALM group (1.3 times), but the differences were not significant. After 6 h, the platelet count in the saline group decreased by 28% relative to baseline, whereas ALM was 14% above baseline (Table 1).
Six-h continuous infusion (6 h sacrifice)
White cell differentials after 6 h continuous infusion were similar in both groups after one-h infusion and 6 h sacrifice. Lymphocytes remained low (33–38% of total white cell count) and neutrophils increased 2.4 times in saline controls and 1.8 times in ALM animals (∼25% less than saline) compared with baseline. Platelet counts in saline controls were identical to baseline after 6 h, and in the ALM group were 20% higher, with a significantly higher plateletcrit (0.22 ± 0.01 vs. 0.13 ± 0.04, P < 0.05) (Table 1).
Blood gases, electrolytes and lactate
Blood gases, electrolytes and lactate at baseline, Saline 1 h, ALM 1 h, Saline 6 h, ALM 6 h (all after 1 h infusion), Saline continuous (6 h) and ALM continuous (6 h).
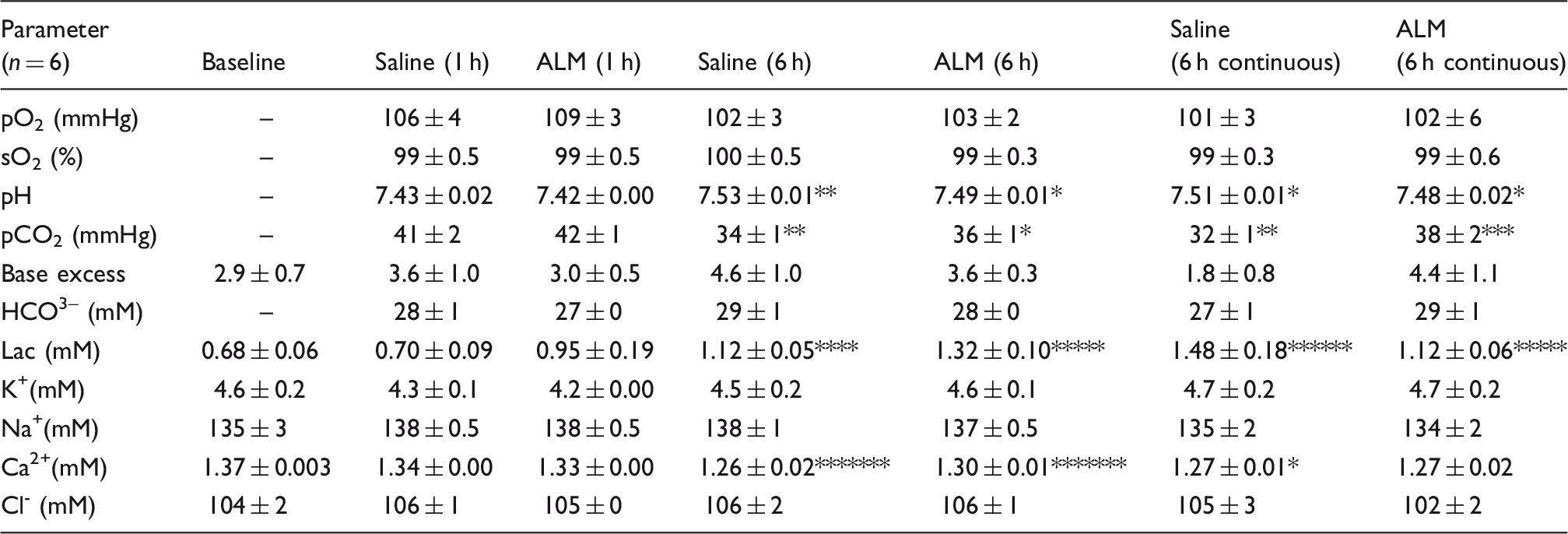
Data are mean ± SEM. Baseline oximetry could not be measured because animals were anesthetized with 5% isoflurane in 100% O2. Lac: lactate. *P < 0.05 compared with Saline 1 h and ALM 1 h; **P < 0.05 compared with Saline 1 h, ALM 1 h, ALM 6 h and ALM continuous; ***P < 0.05 compared with ALM 1 h; ****P < 0.05 compared with Baseline and Saline 1 h; *****P < 0.05 compared with Baseline, Saline 1 h and ALM 1 h; ******P < 0.05 compared with all groups except ALM 6 h; *******P < 0.05 compared with Saline 1 h.
In the 6 h constant infusion groups over 6 h, the saline group continued to have a higher pH (7.51 ± 0.01) and significantly lower pCO2 (32 ± 1 mmHg) and higher blood lactate (1.48 ± 0.18 mM) in direct contrast to the ALM group that had a more normal blood pH of 7.48 ± 0.02 and higher pCO2 (38 ± 2 mmHg) with lower lactate (1.12 ± 0.06 mM) (Table 2). This altered physiological state indicates a respiratory alkalosis in saline infusion groups at 6 h after 1 and 6 h infusion times (Table 2). There were no significant changes in plasma K+, Na+ or Cl– ions, but free Ca2+ decreased significantly after 6 h compared with control 1 h in the 1 h infusion groups. The same decrease in Ca2+ in the saline group was found after 6 h constant infusion, which suggests a mild hypocalcemic effect. Hypocalcemia appeared to be the result of the NaCl infusion, and not the addition of ALM because there were no differences in blood Ca2+ between groups (Table 2).
PT and aPTT clotting times
Baseline PT and aPTT values were 13 ± 0 and 53 ± 1 s, respectively (Figure 2). After 1 h infusion, the PT in the saline control and ALM group value were 16 ± 1 and 18 ± 2 s, respectively, indicating no activation of the extrinsic pathway. After 6 h, the PT was 16 ± 0 s in the saline controls and 15 ± 1 s in the ALM group (Figure 2). aPTT increased around 1.6 times after 1 h infusion for both groups (from 53 ± 1 s to 85–88 s), and the saline group returned close to baseline after 6 h and ALM group increased to 100 ± 43 s. These differences were not significant, indicating little or no activation of the intrinsic pathway. Similar findings were found after constant infusion for 6 h with no differences between treatment groups.
(a) PT and (b) aPTT times at baseline, 1 h and 6 h with only NaCl 0.9% or NaCl 0.9% with ALM. Tests were terminated after 5 min. Values represent mean ± SEM. n = 6 for all groups. *P < 0.05 compared with baseline.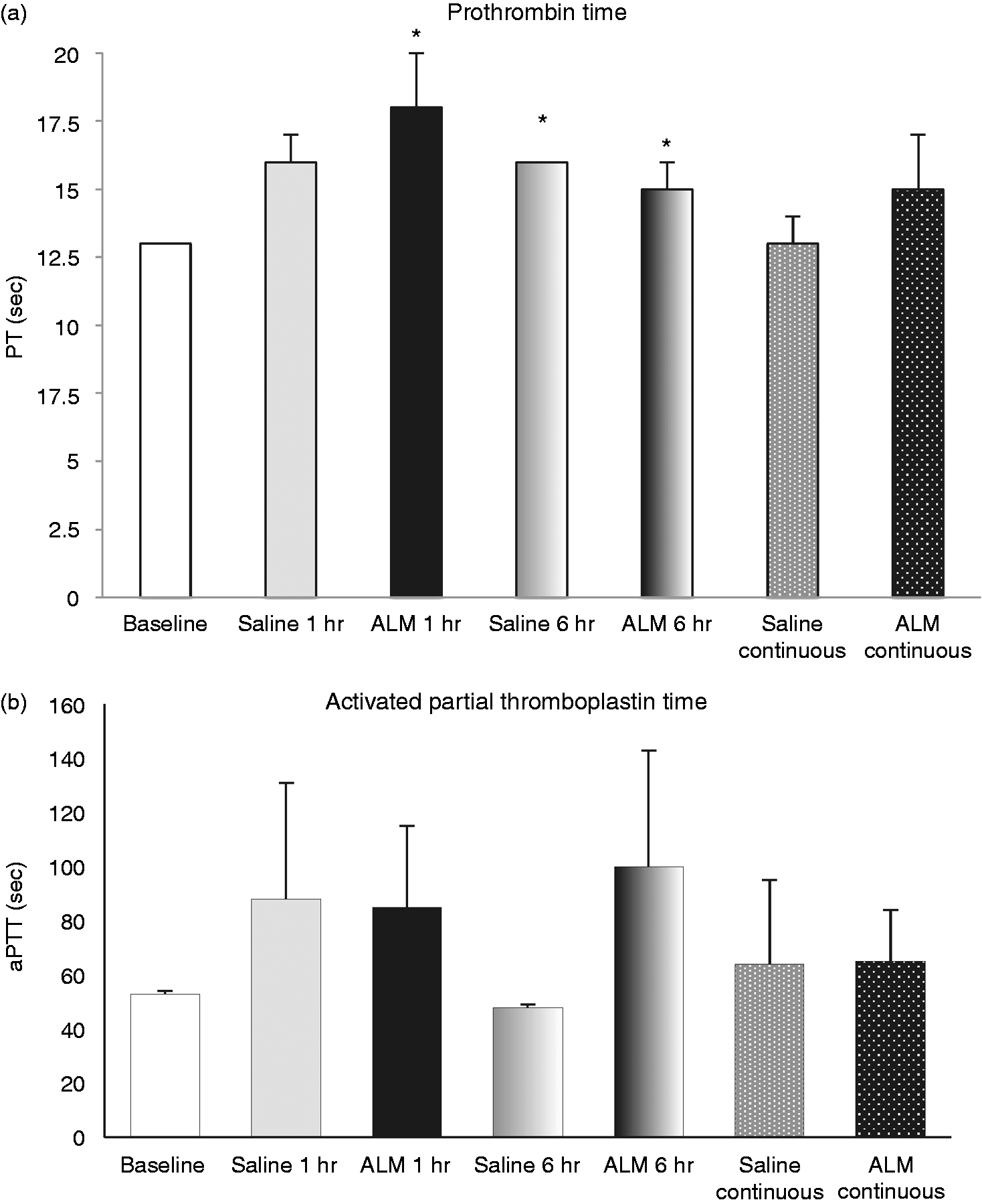
ROTEM parameters
Baseline EXTEM and INTEM CT were 42 ± 1 and 178 ± 78 s (Table 3). After 1 h infusion, the EXTEM CT in the saline control and ALM group value were both 43 s, indicating no activation of the extrinsic pathway. Similarly, INTEM changed little, indicating no activation of the intrinsic pathway. The same was found in EXTEM and INTEM CT after 6 h constant infusion, although INTEM CT was over 1.5 times higher in both groups at this time, but these increases were not significant owing to high variability (Table 3). FIBTEM CT was also unchanging, indicating that the time until thrombin generation in the absence of platelets is not different across treatment or time. Clot amplitudes (A10 and MCF) were not significantly different between the groups, and there was minimal fibrinolysis (Table 3). It was interesting that both groups significantly reduced EXTEM maximum lysis from baseline 6% to 3–4% after 1 and 6 h compared with baseline (Table 3). In our study, we found a weak linear relationship between EXTEM CT and PT having a correlation coefficient r = 0.145 (Figure 3a). A stronger correlation was found between INTEM CT and aPTT with r = 0.648 (Figure 3b). Similar correlations have been reported by other investigators in a number of animal models and patients, and the general consensus appears to be that both plasmatic PT and aPTT are not reliably interchangeable with ROTEM (or TEG) CT measurements.16,17
(a) Correlation between ROTEM EXTEM CT and PT where EXTEM CT = –0.2773 PT + 48.468, R2 = 0.02107 (r = 0.145); (b) correlation between ROTEM INTEM CT and aPTT where INTEM CT = 1.5581 aPTT + 77.879, R2 = 0.41996 (r = 0.648). ROTEM parameters for EXTEM, INTEM and FIBTEM tests. Data are mean ± SEM. α: alpha angle; A10: clot firmness after 10 min (mm). *P < 0.05 compared to baseline. an = 1 in Saline 1 h group did not form a clot > 20 mm on INTEM test.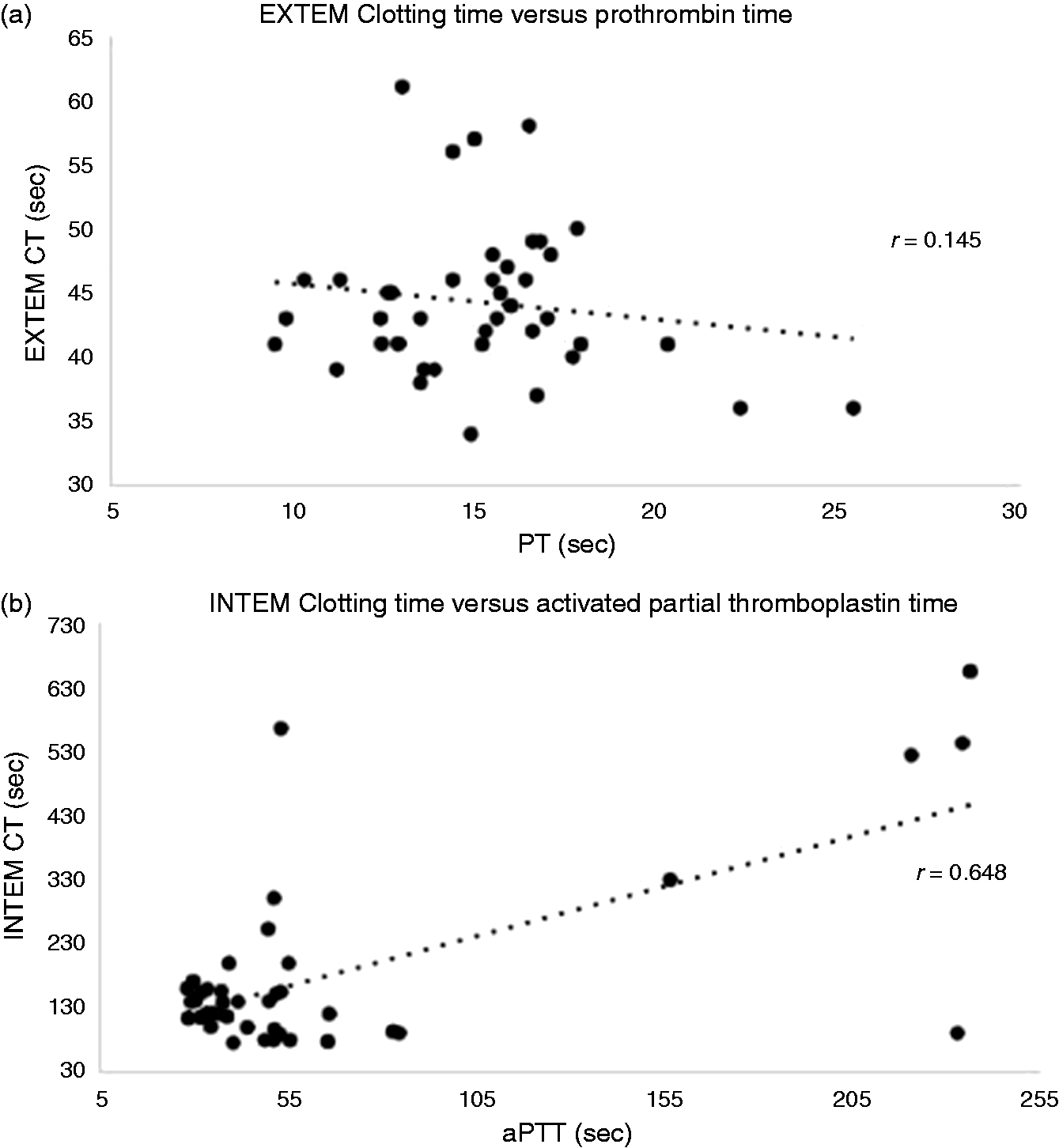
Inflammatory markers IL-6 and IL-10
Baseline IL-6 was below the detection limits of the assay and increased to 145 ± 76 and 128 ± 66 pg/ml after 1 h in saline and ALM groups, respectively (Figure 4a). After 6 h IL-6 increased fourfold over the 1-h value in the saline group and twofold in the ALM group. This difference between 6 h groups was not significant, but the saline group IL-6 (after 1 h infusion) was significantly higher than ALM continuous. IL-6 after continuous infusion was 426 ± 94 and 399 ± 60 pg/ml, respectively (Figure 4a). Anti-inflammatory cytokine IL-10 was also undetectable at baseline, and increased in both saline (154 ± 31 pg/ml) and ALM groups (261 ± 64 pg/ml) (Figure 4b). In contrast to the saline controls at 6 h (126 ± 77 pg/ml), IL-10 in the ALM group plummeted to 59 ± 9 pg/ml at 6 h after infusion stopped at 1 h. After 6 h continuous infusion, plasma IL-10 in the ALM group was 280 ± 102 pg/ml and saline group was 122 ± 29 pg/ml (Figure 4b).
(a) Plasma levels of IL-6 (pg/ml) and (b) IL-10 (pg/ml). Values represent mean ± SEM. n = 6 for all groups. *P < 0.05 compared with baseline; #P < 0.05 compared with baseline and ALM continuous; ¥P < 0.05 compared with baseline and ALM 1 h; ^P < 0.05 compared with ALM 1 h and ALM continuous. IL-6 in the baseline group was not detectable at the lowest sensitivity of the assay (<5 pg/ml) and was assigned a value of 0 pg/ml for statistical comparison.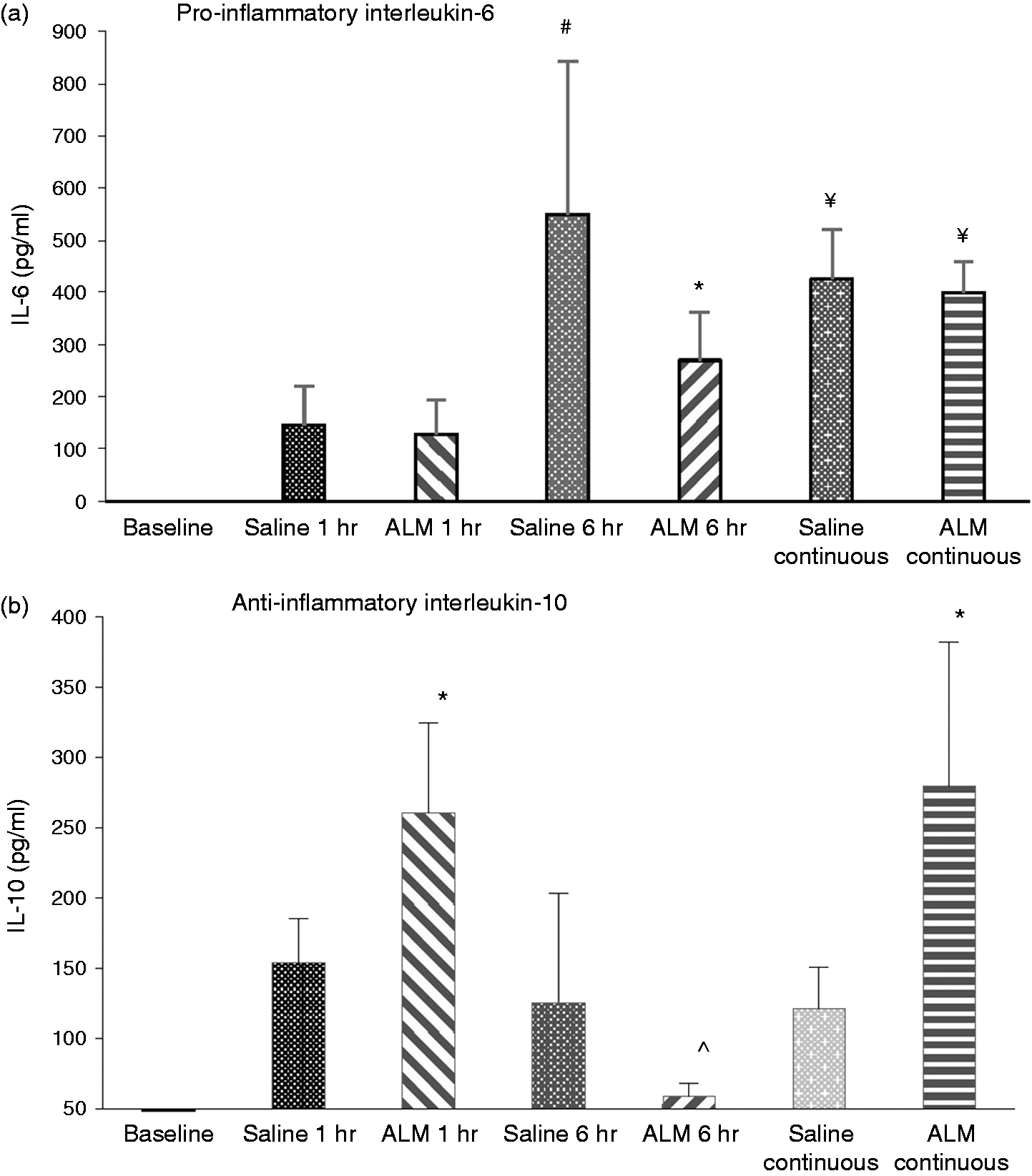
Discussion
New drug therapies are required to dampen a patient’s innate immune response to the trauma of surgery, which will possibly lead to improved outcomes.3,5,18 We report that a single transverse laparotomy and closure with no further manipulation was associated with the following:
A disproportionate increase in neutrophils in the saline vs. ALM groups was found after 1 h despite near identical losses of lymphocytes (∼45%). This loss of reciprocity appeared to be associated with a persistent 20% decrease in total white cell count in the ALM group at 1 and 6 h. ALM appeared to reduce neutrophil release from bone marrow. A disproportionate increase in platelet count was also found after 1 h, and the ALM group appeared to have reduced reactive thrombocytosis in response to the trauma of surgery, which was not present after 6 h. A heightened blood alkalosis was found in saline controls at 6 h after 1 h infusion, and after 6 h continuous infusion, compared with ALM groups. ALM infusion appeared to blunt the respiratory response after 6 h. Little or no activation of extrinsic or intrinsic coagulation pathways was apparent in either group at 1 and 6 h despite a significant reduction in fibrinolysis. Increased plasma pro-inflammatory IL-6 in saline and ALM groups (145 ± 76 and 128 ± 66 pg/ml) after 1 h, indicates a low-level inflammation in response to surgery. After 6 h (1 h infusion), IL-6 became more amplified in saline controls (550 ± 292 pg/ml) compared with the ALM group (270 ± 92 pg/ml). After 6 h continuous infusion, IL-6 was ∼400 pg/ml in both groups. ALM-infused animals after 1 h showed significantly 1.7 times higher anti-inflammatory cytokine IL-10 than in saline controls compared with baseline. In contrast to the saline group, this ALM effect was lost at 6 h when infusion stopped at 1 h. After 6 h continuous infusion, plasma IL-10 in the ALM group returned to higher values, whereas saline group remained low.
Lastly, the transverse laparotomy model may be useful to investigate underlying receptor recognition mechanisms of the innate immune system involved in the early activation of inflammation without activation of coagulopathy, but with significantly reduced fibrinolysis.
ALM infusion reduces neutrophil counts: implications for decreasing the emergency ‘stress granulopoiesis’ response to surgical trauma
Neutrophils play important functions in the innate immune system in acute response to tissue injury (or invading pathogens). 19 We report that saline infusion led to a significant 2.3-fold rise in neutrophil count after 1 h, whereas the ALM group increased only 1.6 times (31% higher in the saline group) (Table 1). This disproportionate neutrophil increase in saline vs. ALM rats may be due to differences in activation of the ‘emergency or stress granulopoiesis’ response to surgical trauma. 20 This curious result was also found in saline and ALM infusion groups after 6 h. Although we did not investigate the underlying mechanisms, two main possibilities exist: (1) ALM reduced the release of neutrophils from bone marrow, and/or (2) ALM led to enhanced neutrophil sequestration to remove inflammatory debris and promote healing at the sites of injury. Since 1–2% of mature neutrophils reside in the circulation, and the remainder mostly stored in bone marrow, 20 it is difficult to rationalize how ALM could lower blood neutrophil count from tissue sequestration without being quickly replenished from bone marrow over such short times. That ALM-induced neutrophil sequestration was unlikely, was also supported by lung myeloperoxidase levels, which were not detectable at 1 and 6 h (data not shown).
A more likely scenario is that ALM ‘suppressed’ neutrophil release from the post-mitotic pool in bone marrow. 20 This may have occurred from down-regulation of granulocyte colony stimulating factor, which is known to increase three bone marrow neutrophil pools: the stem cell pool, the mitotic pool and the post-mitotic pool release. 21 Other possible candidates include ALM inhibition of IL-6, IL-8, IL-17 and IL-23, chemokine receptor (e.g. CXCR4), leukotrines and other inflammatory mediators involved in neutrophil release and recruitment.19,21 A portion of the neutrophil decrease may have come from neutrophils having short half-lives of 1.5 to 8 h in the circulation; however, this is a topic of controversy. 19 The possibility of ALM-induced inhibition of neutrophil release may help explain the persistent 20% lower ‘steady-state’ total white cell count in the ALM group after 1 and 6 h compared with saline groups and baseline (Table 1).
ALM appeared to reduce reactive thrombocytosis after 1 h
Blood platelets normally play a major role in hemostasis and wound healing.15,22 Our study showed an elevated platelet count 1 h after infusion in both groups, with values of 1.74 times baseline in the saline group, and 20% higher counts than the ALM group (1.4 times baseline) (Table 1). Reasons for increased platelet counts are not known. Increased counts are common during acquired bone marrow disorders (e.g. malignancy); however, a more likely explanation is a stress-related mild reactive thrombocytosis. 23 Reactive thrombocytosis (> 450 × 109) is a normal physiologic response that can develop secondary to major surgery, tissue damage, inflammation or infection. 23 Increases in counts can occur in response to the hormone thrombopoietin that regulates bone marrow megakaryocyte differentiation and proliferation, and itself is influenced by many factors such as IL-6. 23 The condition is often underdiagnosed clinically because it is self-limiting and resolves within few days. In our study, platelet counts in both groups returned to near baseline values at 6 h after the infusions were stopped at 1 h (Table 1). Further, there was no elevation of platelets after 6 h constant infusion in both groups compared with baseline. Importantly, reactive thrombocytosis is separate from primary or autonomous thrombocytosis, which is associated with a significantly increased risk for thrombotic or hemorrhagic complications. 23
ALM infusion reduced respiratory alkalosis after 6 h: possible blunting of sympathoadrenal tone
Another interesting finding was blood alkalosis and lower pCO2 after 6 h following surgery (Table 2). A similar hyperventilation syndrome has been described in humans under stressful conditions, such as during or after anesthesia and/or surgery.24,25 Hyperventilation and lower pCO2 in humans is believed to reflect an enhancement of sympathoadrenal tone, 26 and if it persists, can lead to secondary hypocalcemia and hypokalemia that may cause cardiac complications, convulsive disorders and muscle spasm.24–26 In our study, respiratory alkalosis was not evident until 6 h (Table 2). After 6 h continuous infusion, respiratory alkalosis was most pronounced in saline controls (pH 7.51 ± 0.01) with a fall in pCO2 to 32 ± 1 mmHg, and this was associated with higher blood lactate (1.48 ± 0.18 mM). In contrast, pCO2 in ALM-treated animals maintained higher (38 ± 2 mmHg) and lower (1.12 ± 0.06 mM) lactates (Table 2). We also found that saline animals were noticeably more agitated than ALM groups despite receiving identical analgesia regimes, suggesting pain was not a primary cause. It appeared that the saline controls were more stressed than ALM animals, which may implicate ALM as a potential inhibitor of stress-related sympathoadrenal tone. However, without a no-treatment group, we do not know if NaCl infusion exacerbated the stress response or, alternatively, failed to blunt its progression. Further studies are required to include measurement of respiratory rates and extend the observation times beyond 6 h with HPA axis activation monitoring and behavioral tests quantifying post-operative stress. Unfortunately, we did not examine renal dysfunction as a possible underlying contributor to respiratory alkalosis.
Laparotomy led to systemic inflammation at 1 h and increased at 6 h
A local inflammatory response nearly always accompanies tissue injury. However, whether local activation develops into a systemic response depends upon the severity and duration of traumatic tissue injury.18,27,28 The early rise of pro-inflammatory cytokine IL-6 in plasma from activated immune cells is believed to be a measure of stress severity. 29 In 2006, Ishibashi et al. showed that IL-6 levels correlated with the length of surgical incision (1.0 cm vs. 3.0 cm) with the larger incision leading to doubling of plasma IL-6 values at 3 and 6 h. 30 Our study showed that plasma IL-6 increased to similar extents after 1 h in saline and ALM groups (145 ± 76 and 128 ± 66 pg/ml) (Figure 4). However, the small plasma increases reflect a low-level activation of inflammation in response to surgery in our model. After 6 h (1 h infusion), however, the inflammatory response appeared to be amplified as IL-6 increased in saline controls (550 ± 292 pg/ml) compared with the ALM group (270 ± 92 pg/ml). After 6 h continuous infusion, the inflammatory response was similar in both groups with IL-6 of ∼400 pg/ml (Figure 4).
The anti-inflammatory IL-10 is another clinically relevant cytokine that appears to act like an ‘immunological brake’ to prevent overexpression of inflammation, and therefore collateral tissue damage.27,28 Our study showed at 1 h the ALM group had IL-10 values 1.7 times saline control values, which was significantly higher than baseline (Figure 4). This difference may be significant to improve the body's ‘immune’ balance between pro-inflammatory and anti-inflammatory pathways, essential for optimal healing. This effect of ALM was lost at 6 h when the infusion was stopped at 1 h, whereas values in saline controls at 6 h remained low and close to their 1-h value (Figure 4). After 6 h continuous infusion, the ALM value of plasma IL-10 was maintained at higher values (280 ± 102 pg/ml), whereas the saline group remained low (122 ± 29 pg/ml). Further studies are required to investigate the immune cells in their different activated states such as macrophage M1/M2 (inflammatory/anti-inflammatory) phenotypes and neutrophil modulation in response to trauma of surgery.
Systemic inflammation with no coagulopathy
A curious result in the present study was that the activation of systemic inflammation was not accompanied by an acute coagulopathy. Plasmatic and whole blood viscoelastic ROTEM measurements for both infusion groups all showed no change in extrinsic and intrinsic pathway clotting times, clot elongation and clot amplitudes (Figure 2, Table 3). Inflammation and coagulopathy are closely interconnected via the state of activation of the endothelium. 31 Inflammation activates coagulation through three main mechanisms: (1) cytokine induction of tissue factor expression, (2) inflammation-induced regulation of the protein C/TAFI systems and (3) inhibition of fibrinolysis.31–34 In our surgical model, it appears that the severity of trauma was not sufficient enough to activate the first two mechanisms, but it did significantly reduce fibrinolysis in all groups at 1 and 6 h (Table 3). This uncoupling between inflammation and coagulation suggests that the endothelial thrombomodulin–thrombin complex, which can either activate protein C anticoagulant or TAFI coagulation pathways, remained poised in non-activated steady-state. 35 However, this balance does not explain the decreased fibrinolysis (Table 3), which may have occurred from lower plasmin levels from either decreased tissue-type plasminogen activator or increased of plasminogen activator inhibitor-1 (PAI-1), which slows fibrin degradation to soluble degradation products.
Possible clinical significance
New frontline drugs are urgently required to curb the overexpression of inflammation and the stress response during and following major surgery. This is clinically important because surgical stress is a global problem in terms of patient numbers, mortality, morbidity and costs.1,36,37 Our study may have relevance in woman undergoing planned or emergent cesarean sections, which is a single laparotomy but not without risks to mother or fetus. 38 In industrialized countries there is a 10% risk of post-operative infection, a 5% risk of excessive blood loss or clotting and a 1% risk of abnormal scar tissue formation.38,39 Another potential advantage is the ultra-small volumes of ALM infusion used that appear to protect the body at a number of levels following a single laparotomy. This may be important because aggressive administration of saline-containing infusions (>30 ml/kg/h) can lead to a number of complications in major surgery such as inflammatory stress, coagulopathy and metabolic acidosis. 40 In our study, we administered a total of 0.5 × 6 × 0.394 = 1.18 ml per 394 g rat over the 6 h period. Lastly, our work may be relevant to emergency patients or the elderly, who have reduced physiological reserve in response to surgical trauma,5,41 as well as the very young, who are sensitive to long periods of anesthesia. 42
Limitation and future studies
A potential limitation of the present study was that a single laparotomy was performed without any further manipulation, but this was deliberate to show that such a common procedure activates the immune and inflammatory systems without any further surgery. Another limitation was that we did not measure plasma levels of the major stress hormones of the HPA axis (e.g. epinephrine, cortisol) or the relevant cytokines and immune mediators involved in the regulation of granulopoiesis, activation of inflammation, fibrinolysis (tPA/PAI-1 ratio) and endothelial health. The inflammatory-based surgical stress response may also have been blunted by the analgesic non-steroidal anti-inflammatory drug Carprieve (Carprofen). 43 Higher ALM doses may have led to greater differences between groups. A future study would also benefit from an increase in sample number to improve statistical power. It would also be important to add a polytrauma to the present model to increase trauma severity such as gut ischemia, liver or splenic damage and hemorrhage, which would be expected to activate both inflammation and coagulation.
Conclusions
Following a single laparotomy without any further manipulation, ALM infusion appeared to reduce stress-related granulopoiesis, reactive thrombocytosis and respiratory alkalosis. After 1 h, both groups activated a mild systemic inflammatory status; however, after 1 h and continuous 6 h, ALM animals had higher IL-10 levels, indicating improved inflammatory balance. Surgery was not associated with activation of systemic coagulopathy. The surgical model may provide a useful investigative tool for understanding early inflammatory–coagulation relations with reduced fibrinolysis.
Footnotes
Acknowledgements
The author(s) would like to thank the College of Medicine and Dentistry, James Cook University. The authors would particularly like to thank the US Department of Defense for continued support of our work, and Maddison Griffin for technical assistance.
Declaration of Conflicting Interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Geoffrey Dobson is the sole inventor of the ALM concept for cardioplegia, organ preservation, surgery, infection, sepsis and trauma. LD and HLL have no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for the study came from internal funds from College of Medicine and Dentistry to GPD.
