Abstract
How very high exposure levels to endotoxin in a farming environment provide protection against respiratory allergic symptoms and low-to-moderate levels of endotoxin in urban homes promote allergic response is unclear. Dose-specific bacterial endotoxin or LPS-induced tolerance mechanisms can affect lung inflammations, coupled with the Th2 immune responses. Here, we explored the effects of intranasal exposure of LPS at two different doses (based on occupational exposures during handling of agricultural wastes) in OVA-sensitized allergic wild type (WT) and TLR4-KO mice, particularly, with respect to Th2 cytokines and Tregs level. Low-dose LPS (100 ng) exposure prohibited airway tolerance and failed to generate T-cell-dependent protection against lung inflammations in allergic mice. Furthermore, low Tregs at the inflammatory site and induced Th2 cytokines, as well as IL-6 and IL-25, suggested that low-dose LPS might be associated with the suppression of tolerance mechanisms. In contrast, high-dose LPS (20 µg) favored the suppression of Th2 cytokines, IL-6 and IL-25, but failed to induce Th1 cytokines (e.g. IFN-γ). Our results suggest that low-dose LPS can enhance airway allergic inflammation through failing of antigen-dependent immune regulatory homeostasis. The exposure levels of LPS can determine the generation of inflammatory responses in airway allergy.
Introduction
Bacterial endotoxin or LPS is one of the most common environmental microbial immunomodulators and a common air pollutant present everywhere. However, LPS exposure levels vary from place to place and environment to environment. LPS exposure can be associated with the induction of airway allergy and aggravation of asthma symptoms,1,2 as well the induction of protective effects against airway allergy and allergic asthma.3,4 In contrast to urban children, the risk of atopic asthma was reported to be less frequent in children growing in farms and rural areas and the environmental LPS exposure was considered as a key player behind this apparent fact.5,6 According to the revised hygiene hypothesis, the generation of robust anti-inflammatory network, as a result of persistent challenge of allergens and microbial immunomodulators, and involvement of regulatory T cells (Tregs), are jointly associated with the suppression of allergic and inflammatory diseases. The mechanisms behind this hypothesis, however, are not completely investigated yet, and most of the previous mechanistic studies explored lower doses of LPS. The prime objective of the present study was to test how different higher doses of LPS (representing occupational exposures during handling of agricultural wastes or other farm works) influence immune regulatory network in airway allergy involving regulatory Tregs.
TLRs recognize a wide range of microbial products and generate innate immune responses. 7 Ligand-induced activation of TLRs triggers effector response by providing a direct bridge between the innate and adaptive immunity.7,8 The mechanisms associated with the development of immune tolerance through allergen inhalation have gained significant interests recently.9,10 To understand the mechanisms associated with LPS tolerance, most of the recent studies were mainly focused on cell surface molecules, 11 signaling proteins, 12 pro- and anti-inflammatory cytokines, 13 and other cellular mediators. 14
Tregs regulate key molecules related to the inhaled Ag-induced immune response, either suppressing resultant Th2 immune response or by maintaining a tolerant Th2 state. 15 The early engagement of Tregs during inflammations helps to establish successful suppressive immune response against foreign invaders, including allergens and microorganisms. 7 This process includes rapid generation of Tregs followed by migration of the induced Treg cells (iTregs) to the inflammatory sites (e.g. the lung) coupled with the production of associated cytokines, such as IL-10 and TGF-β. Whether the activation of Treg cells occur either through direct recognition of microbial products or through the host inflammatory response at the site of inflammation, is still unclear.
Both Th1 and Th2 inflammatory responses have been reported to be influenced by LPS exposures. 16 Apart from Th1 and Th2 cells, another subset of Th cells expressing IL-17 were also reported to regulate allergic airway inflammations. 17 Several studies demonstrated that IL-25 (also known as IL-17E) is structurally related to IL-17 and may act as an essential mediator of Th2 response.18,19 The main source of IL-25 production is airway epithelial cells, subsets of T lymphocytes (Th2), and also eosinophils and basophils. 20 How IL-25 is involved in generating Th2 immunity in airway allergic inflammations, particularly when an inhalation exposure to LPS occurs, is still an enigma yet to be solved. Furthermore, the mechanistic study of LPS, as well as air pollution-induced allergy and asthma, is unclear and require further explorations.
The present study examined the airway associated regulatory and functional immune responses against two different doses of LPS in wild type (WT) and TLR4 knockout (TLR4-KO) mice during the development of OVA-induced airway allergic inflammations. Furthermore, we determined the homeostasis of immune regulatory cells such as Tregs, in LPS-induced inflammatory responses in allergic and control mice of WT and TLR4-KO groups. We have analyzed the histological changes in lungs of allergic mice exposed intranasally to two different LPS doses, with consequent allergen sensitizations and challenges, and also estimated the levels of eosinophils, neutrophils, and several Th2 and Th1 cytokines.
Materials and methods
Mice
C57BL/6 female mice of 8–10 wk age were used in all experiments. Experimental mice were housed in individually ventilated cages in the pathogen-free animal care facility at the Department of Environment Health, University of Cincinnati, USA, following the appropriate Institutional Animal Care and Use Committee (IACUC) guidelines. Animal experiments were performed using the experimental protocol approved by the IACUC, University of Cincinnati.
Abs and reagents
The following Abs and immunological reagents were used: rat anti-mouse CD4 (GK1.5, Isotype Rat IgG2b) was purchased from BioLegend (San Diego, CA, USA); CD8 (53-6.3; Isotype Rat IgG2a), CD25 (PC61.5; Isotype RatIgG1), foxp3(FJK-16s; Isotype Rat IgG2a), CD11c (N418; Isotype Armenian Hamster IgG), CD205 (205yekta; Isotype Rat IgG2a), Ly-6 G/Gr-1 (RB6-8C5; Isotype Rat IgG2b) and IL-10 (JES5-16E3; Isotype Rat IgG2b) were purchased from eBiosciences (San Diego, CA, USA); and CD125 (T21; IsotypeRat IgG1) and CD16/CD32 (2.4G2; Isotype Rat IgG2b) were purchased from BD Pharmingen (San Jose, CA, USA). For most of the cytokines (IL-4, IL-5, IL-6, IL-10, IL -13, IL-25 and IFN-γ) assays, we used mouse ELISA Ready-SET-Go analysis kit from eBiosciences. For IL-12p40, we used BD optEIA from BD Biosciences (San Jose, CA, USA). For the estimation of IgGE, IgG2a and IgG1 from serum, we purchased ELISA kit from Immunology Consultants Laboratory (Portland, OR, USA).
Study of allergen-induced airway inflammation
The allergen sensitization and challenge assessment consisted of a 26-d protocol involving both intraperitonial (i.p) and intranasal (i.n.) immunization (Figure 1). Mice were sensitized i.p. with 20 µg OVA (Sigma, St. Louis, MO, USA) along with 2% Alhydrogel (InvivoGen, San Diego, CA, USA) in 1:1(v/v) of 100 μl/dose, and boosted with the same Ag with same concentration after 14 d. On d 1, 7, 14 and 21, mice were exposed i.n. with the low-dose (100 ng) and high-dose (20 µg) ultrapure LPS from Escherichia coli (InvivoGen), both dissolved in 20 µl PBS. On d 24, 25 and 26, mice were challenged i.n. with 100 µg OVA. Mice were scarified within 24 h of the final OVA challenge with i.p. sodium pentabarbitol injection.
Experimental protocol of the study. Mice were sensitized by i.p. injection with 20 µg OVA on d 1 and d 14. Low-dose (100 ng) and high-dose (20 µg) ultrapure LPS from E. coli were delivered on the d 1, 7, 14 and 21, through intranasal route. On d 24, 25 and 26, mice were challenged with 100 µg OVA through intranasal route. After 24 h of last challenge mice were assessed.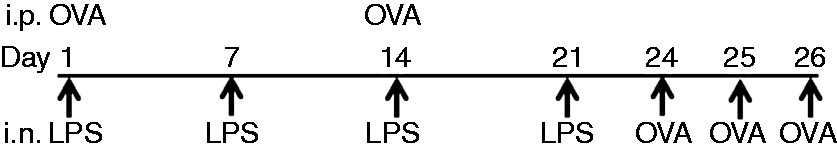
Lung histology
For the analysis of airway inflammation and pathological changes in the lungs, formalin-fixed paraffin embedded lung sections (5 -µm thick) were stained with hematoxylin and eosin.
Isolation of lung inflammatory cells
Lung tissues were isolated within 24 h after the final allergen challenge from the scarified mice. Lungs were manually minced into fragments (1–2 mm), and incubated at 37℃ for 1 h in 1 mg/ml collagenase (Sigma) and 20 mg/ml DNaseI (Sigma) in Hanks Balanced Salt Solution. After collagenase digestion, tissue suspension was passed through a syringe at every 5–10-min interval. To isolate single cells, a 23% and 70% bilayer, Percoll (Sigma) gradient-based cell separation method was followed and cells at the interface were finally collected for the further analysis.
Flow cytometric analysis
Single-lung-cell suspensions from lungs (∼106 cells/ml) were blocked with anti-mouse CD16/CD32 (BD Pharmingen) before cell-surface staining. Cells were stained with mAbs for CD4, CD8, CD25, CD11c, CD125, Gr-1, IL-10 and foxp3, as per the specific type of analysis. For Treg analysis, cells were stained with CD4, CD25 and foxp3 Abs. For intracellular staining of foxp3 and IL-10, cells were fixed and permeabilized with IC fixation and permeabilization buffer (eBioscience), according to the manufacturer’s instructions. For flow cytometric analysis of intracellular IL-10, purified cells (∼106 cells/ml) were incubated with 2 μl/ml cell stimulation cocktail plus protein transport inhibitors (eBioscience) for 5 h. The anti-CD125 and anti-Gr-1 Abs were used for the analysis of eosinophils and neutrophils, respectively.
Real-time PCR analysis
RNA was prepared from frozen lung tissues and the IL-4, IL-5,IL-13 and IL-10 mRNA transcript were quantified by qRT-PCR using following primers: IL-4: forward 5’-GGTCTCAACCCCCAGCTAGT-3’; reverse 5’- GCCGATGATCTCTCTCAAGTGAT-3’; IL-5: forward 5’- CTCTGTTGACAAGCAATGAGACG-3’; reverse 5’- TCTTCAGTATGTCTAGCCCCTG-3’; IL-10: forward 5’-CCCTGGGTGAGAAGCTGAAG-3’; reverse 5’-CACTGCCTTGCTCTTATTTTCACA-3’; IL-13: forward 5’-CCTGGCTCTTGCTTGCCTT-3’; reverse 5’-GGTCTTGTGTGATGTTGCTCA-3’; β-actin: forward 5’-AGAGGGAAATCGTGCGTGAC-3’; reverse 5’-CAATAGTGATGACCTGGCCGT-3’. Data were normalized using β-actin and fold change was calculated over PBS.
Cytokine assays
One part of the lung tissue from the scarified mice was snap-frozen and stored at −80℃ until further analysis. Lung tissue homogenates were prepared with 1 ml tissue protein extraction reagent (T-PER; Thermo Scientific, Waltham, MA, USA) containing working concentration of Halt Protease Inhibitor Cocktail (Thermo Scientific). The lung tissues were homogenized at 4℃. Lung homogenates were then centrifuged at 5000 g for 10 min at 4℃. Total protein concentration in the supernatants of lung homogenates were measured by BCA kit (Sigma). For the measurement of ELISA-based cytokines from lung tissue homogenates, we used 20 µg total proteins in each sample. In brief, 20 µg protein from the lung tissue homogenates was added in each cytokine-specific capture Ab-coated plate. The cytokine levels were detected using biotin–avidin–HRP conjugated detection Ab. All procedures were appropriately followed as per the instructions provided by suppliers.
ELISA for the measurement of immunoglobulin isotypes
Blood serum was collected from scarified mice within 24 h of the last OVA challenge. Total IgG1, IgG2a and IgE in the serum were measured by using anti-IgG1-, IgG2a- and IgE-coated 96-well plates. The procedures and instructions given in the supplier’s (Immunology Consultants Laboratory, Portland, OR, USA) protocol were followed properly.
Statistical analyses
Data were analyzed using SigmaPlot (version: 12.0; Systat Software, Inc., San Jose, CA, USA) and statistically significant differences were determined by using one-way ANOVA followed by Bonferroni multiple comparison test. Kruskal–Wallis tests were conducted if the data did not have a normal distribution. The significant differences were calculated between PBS vs. OVA, OVA vs. LPS and different doses of LPS, in both WT and TLR4-KO mice. All the data were represented as means ± SD and a P-value of < 0.05 was considered as statistically significant.
Results
Low-dose exposure of LPS was more effective for the generation of airway allergic responses
Mice sensitized with low-dose LPS showed significantly higher infiltrations of cells in the lungs of WT, as well as TLR4-KO mice (Figure 2a). Further, we measured serum IgE, IgG1 and IgG2a in all mice groups, and results showed significant up-regulation of IgE, IgG1 and IgG2a in both WT and TLR4-KO mice (Figure 2b–d). As shown in the figures, both high and low doses of endotoxin elevated serum IgE in WT and TLR4-KO mice, however, this effect was more robust in TLR4-KO mice. Particularly, the low dose was more effective in elevating the serum IgE levels in the TLR-KO mice than the high dose, but the same variation was not observed in case of WT mice.
Hematoxylin and eosin staining of lung section showing cell infiltration and pathology upon low- (100 ng) and high- (20 µg) dose LPS intranasal exposure and OVA challenge in WT and TLR4-KO mice (a). Bar graph showing serum IgE, IgG1 and IgG2a levels in these mice (b–d: WT and TLR4-KO mice). Mice were sensitized by i.p. administration of OVA (20 µg) along with Alum (1:1 v/v) on d 1 and 14, followed by intranasal exposure of PBS, 100 ng LPS and 20 µg LPS, on d 1, 7, 14 and 21; then challenged i.n. with 100 μg OVA on d 24, 25 and 26. Data are represented as mean ± SD per mouse (n = 8); magnification 200×. *P < 0.05 and **P < 0.005 with respect to PBS treated control and between WT and TLR4-KO mice groups.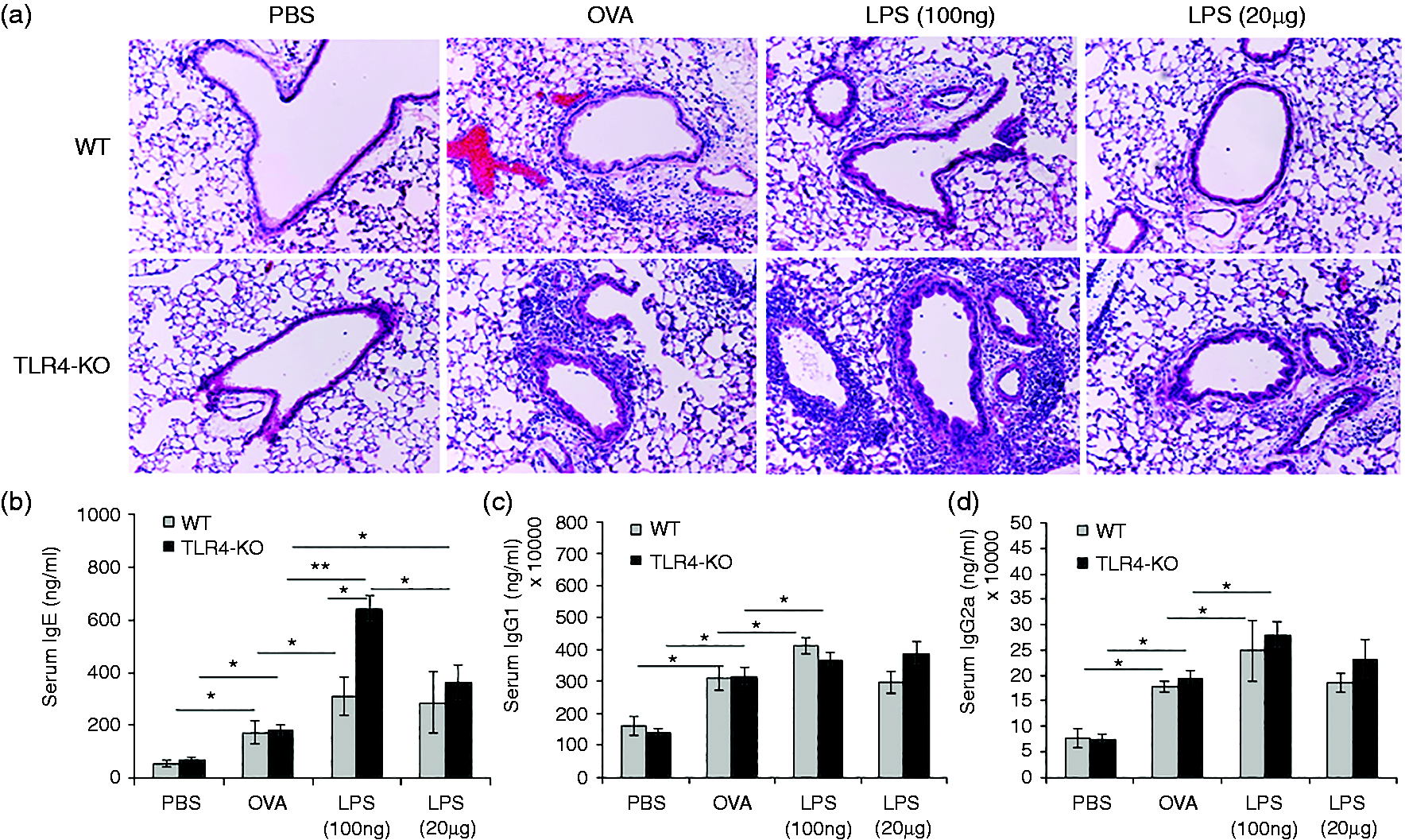
Further, we analyzed total cell counts (Figure 3a), eosinophils (CD125+) (Figure 3b) and neutrophils (CD11c−Gr-1+) (Figure 3c) in the lung tissue homogenates of both mice groups treated with high and low doses of LPS. Results showed that elevation level of total no of cells in lung (Figure 3a), eosinophil and neutrophils (Figure 3b, c) were more robust at low dose of LPS exposure. In contrast, mice exposed to high dose of LPS demonstrated milder airway inflammatory response (Figure 3a–c).
Bar graphs showing total and differential cell counts in collagenase-digested lung single-cell suspension by haemocytometer (a). Flow cytometer-based counting was performed for eosinophils and neutrophils in lung single-cell suspension. Eosinophil count (CD125+) was presented in WT mice and TLR4-KO mice (b). Neutrophil count (Gr-1+ CD11c−) was presented in WT mice and TLR4-KO mice (c). Data are represented as mean ± SD of respective types of cells per mouse lung (n = 8). *P < 0.05 with respect to PBS-treated control and in between WT and TLR4-KO mice groups.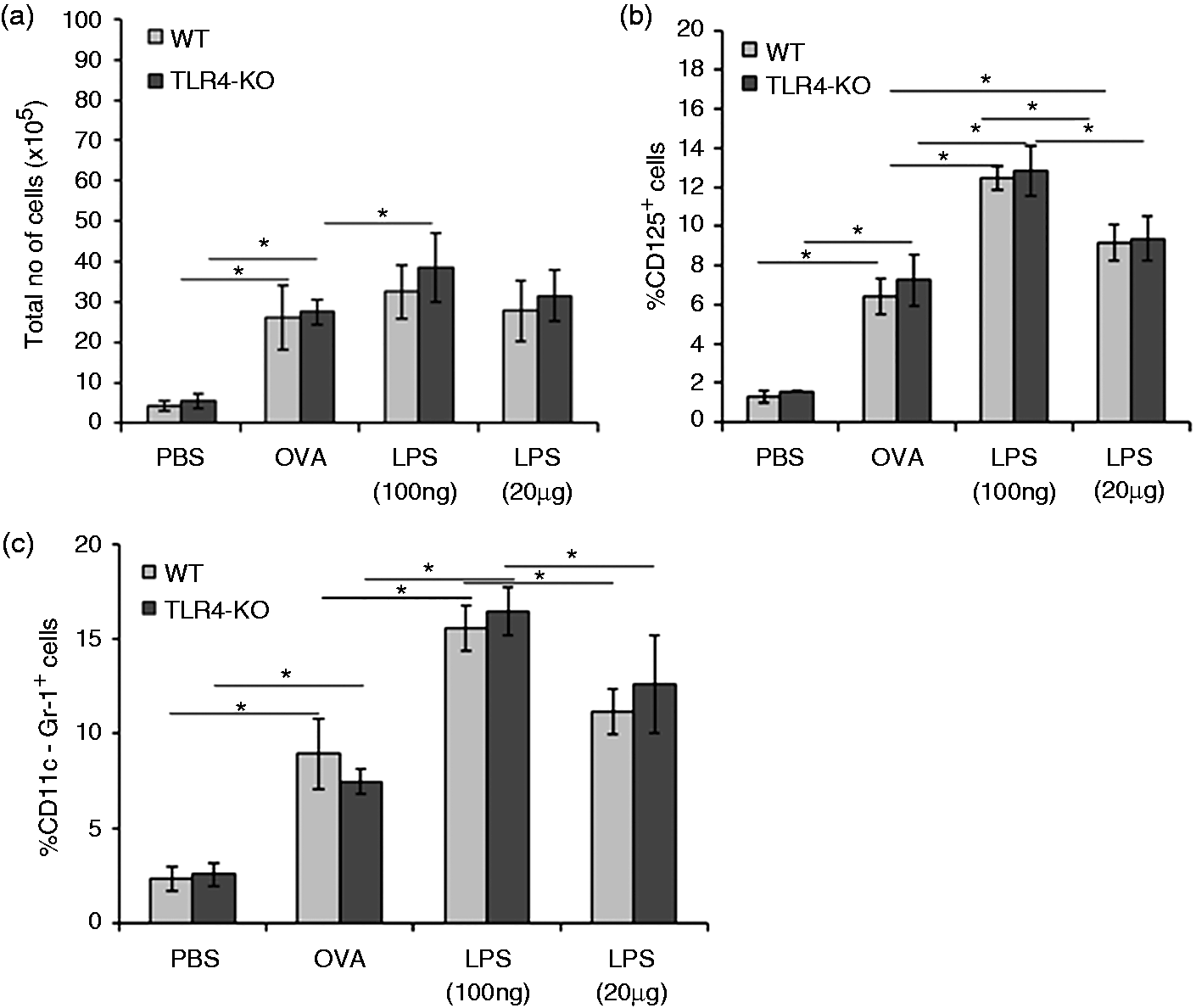
High-dose LPS exposure was associated with the elevation of Tregs
We analyzed the LPS-induced CD4+CD25+foxp3+ T cells and observed that low-dose LPS exposure did not induce CD4+CD25+foxp3+ Tregs in the lung of WT mice (Figure 4a), whereas the same cell population was found to be increased in TLR4-KO mice (Figure 4a). Additionally, we observed increased levels of intracellular IL-10-producing Tregs in WT (Figure 4b) and TLR4-KO (Figure 4b) mice exposed to high dose of LPS.
Low dose of LPS suppresses induction of CD4+CD25+foxp3+ Treg cells. Single-cell suspension from collagenase-digested lungs was analyzed by using flow cytometer. Bar graph showing percent populations of CD25+foxp3+ cells gated on total CD4+ cells (a); and IL-10-positive Treg cells gated on CD4+CD25+foxp3+ T cells (b). Data are represented as mean ± SD per mouse (n = 8). *P < 0.05) with respect to PBS-treated control and between WT and TLR4-KO mice groups.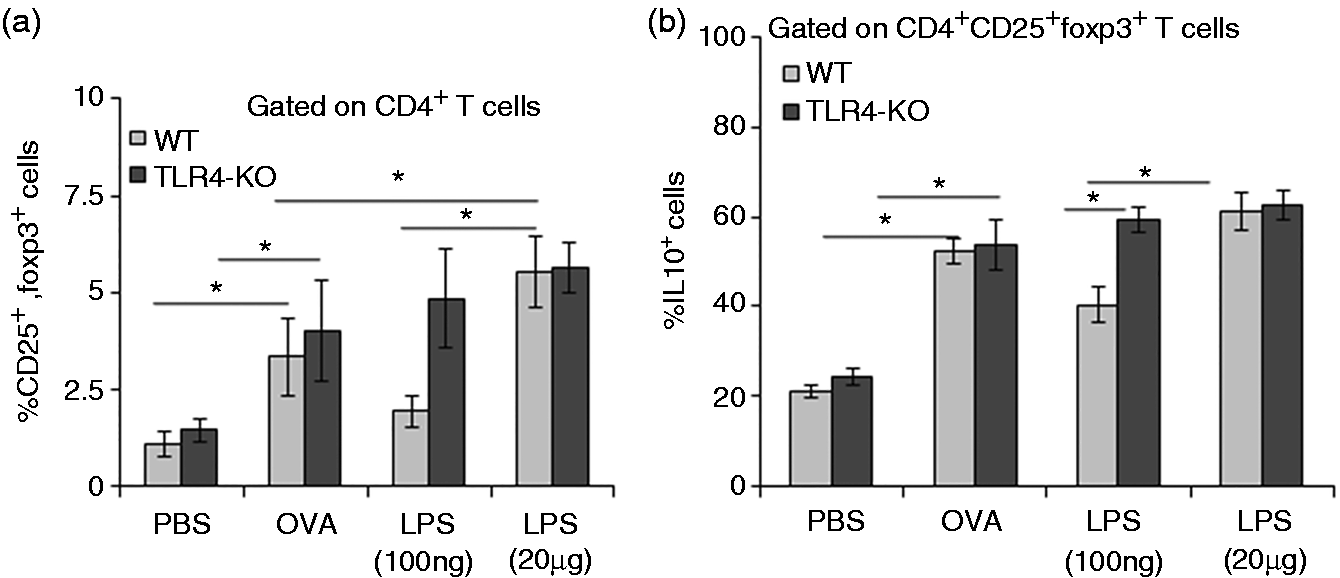
Increased Th2 cytokine production in low-dose LPS exposed mice
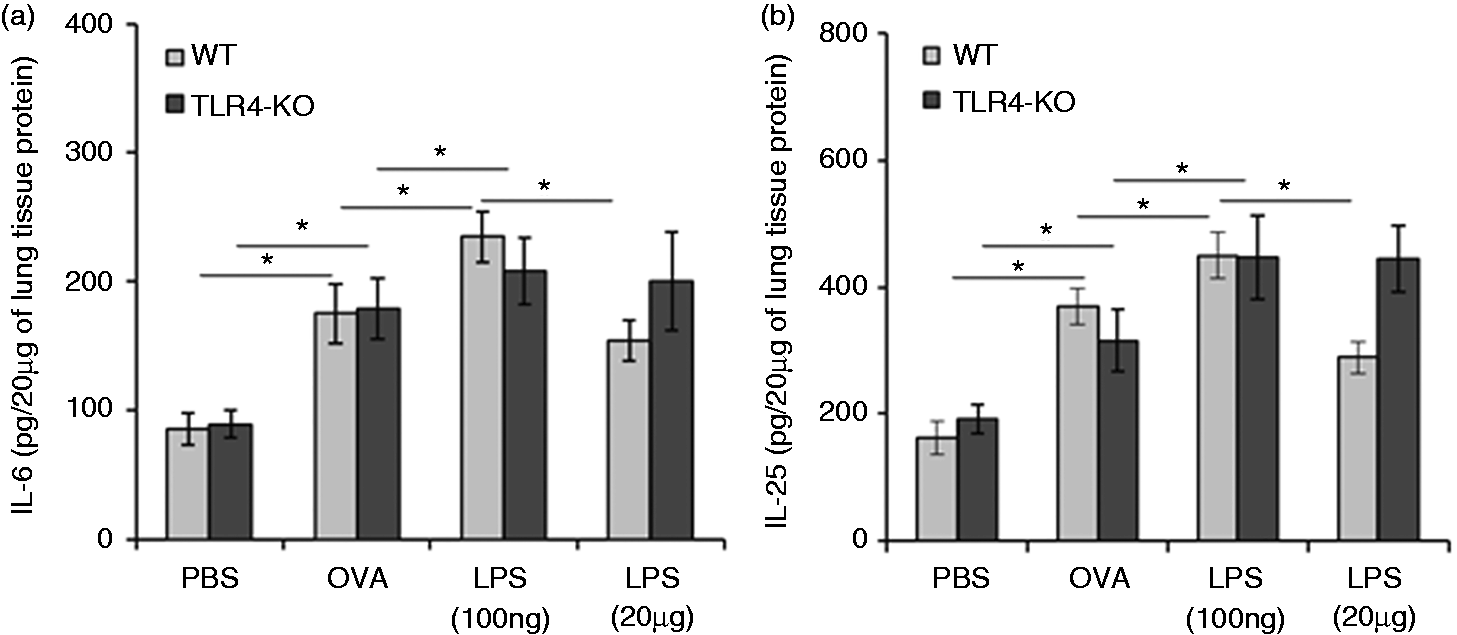
The above-described results suggested that increase in allergic response occurs more robustly at low-dose LPS exposure than at highdose exposure. To provide additional support to this finding, we further analyzed various cytokines using real-time PCR-based analysis in frozen lung and ELISA-based analysis in lung tissue homogenates, of all test mice groups. We observed that low-dose LPS exposure resulted in higher concentration of IL-4, IL-5, IL-10 and IL-13 in lungs of WT and TLR4-KO mice (Figure 5a–d; see Supplementary Material, Figure S1A–D). In addition to these cytokines, production of IL-6 and IL-25 were found higher upon exposure of low dose of LPS in WT mice, as well as TLR4-KO mice (Figure 6a, b). We also examined the levels of IFN-γ and IL-12p40 in lung homogenates of all test mice groups. The level of IFN-γ was significantly low only in WT mice at low-dose LPS exposure and this difference was not observed in TLR4-KO mice (see Supplementary Material, Figure S2). We did not find any significant changes in IL-12p40 levels during different doses of LPS exposures in both groups of mice.
Effects of LPS doses on in vivo Th2 cytokine responses in the lung. Low dose of LPS enhances Th2 cytokines. Total lung tissue proteins were isolated from PBS, OVA or different doses of LPS-exposed mice, by using tissue protein extraction reagent (T-PER) buffer containing complete protease inhibitors cocktail. Twenty µg lung tissue proteins were used for estimation of different cytokines by ELISA. Bar graphs show levels of IL-4, IL-5, IL-10 and IL-13 cytokines in WT mice and TLR4-KO mice (a–d). Data are represented as mean ± SD concentrations of cytokines (pg/20 µg of lung tissue proteins). *P < 0.05 and **P < 0.005 with respect to PBS-treated control and between WT and TLR4-KO mice groups. Effects of LPS doses on in vivo cytokine responses (other than Th2). Low dose of LPS enhances IL-6 and IL-25 production. Total lung tissue proteins were isolated from PBS, OVA or different doses of LPS exposed mice by using tissue protein extraction reagent (T-PER) containing complete protease inhibitors cocktail. Twenty µg lung tissue proteins were used for estimation of different cytokines by ELISA. Bar graphs are showing cytokines levels of IL-6, and IL-25 in WT mice and TLR4-KO mice (a, b). Data are represented as mean ± SD concentrations of cytokines (pg/20 µg of lung tissue proteins). *P < 0.05 with respect to PBS-treated control and in between WT and TLR4-KO mice groups.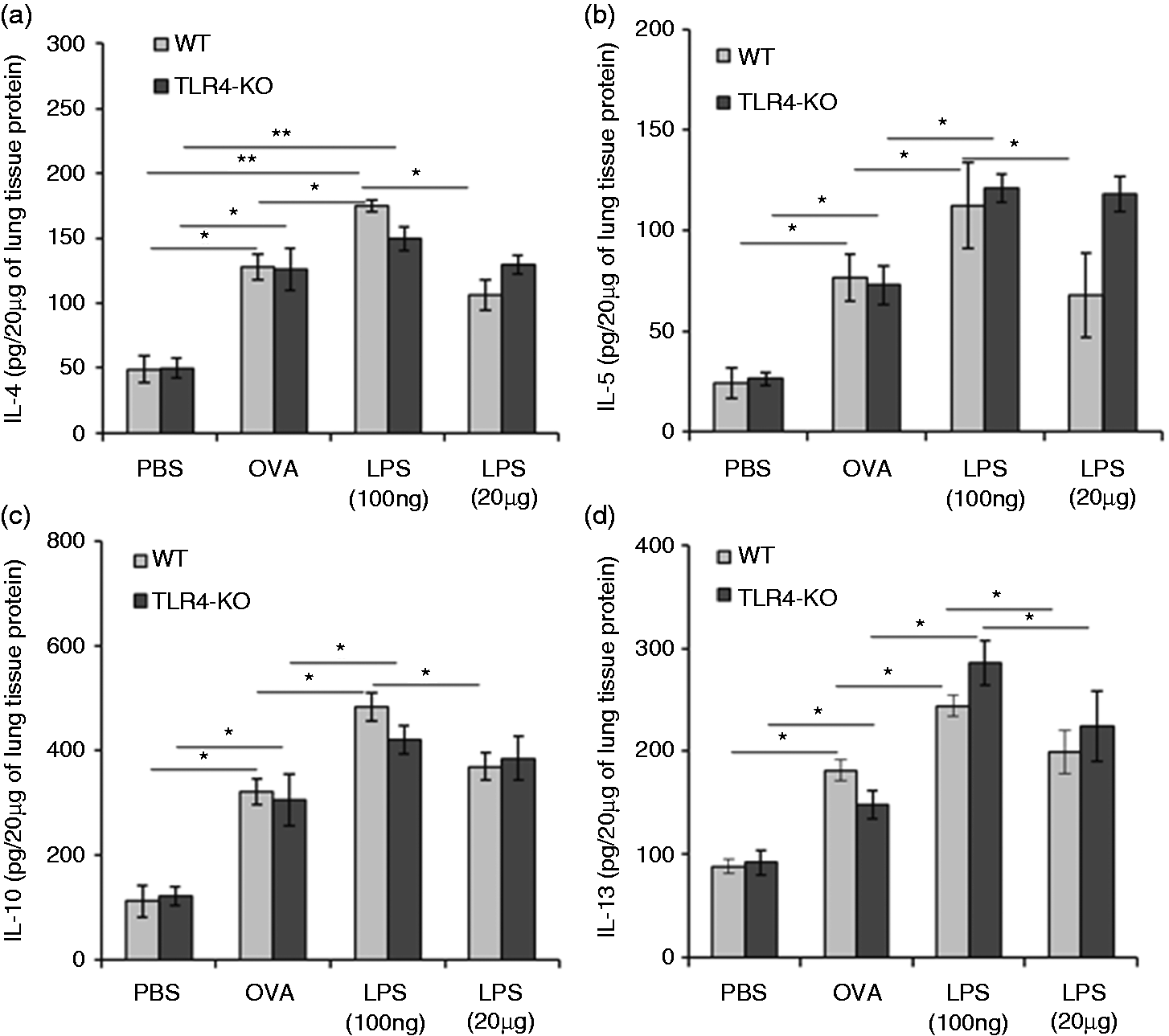
Discussion
The present study illustrated the effect of different doses of LPS on allergic responses in the lung, particularly the modulation of immune regulatory network and changes in associated regulatory cytokines. We have explored how different doses of LPS can be associated with the maintenance and/or breakdown of immunological tolerance. We have observed that the low dose of LPS exposure enhances the allergic airway inflammations in OVA-sensitized mice, which, in turn, was found to be associated with higher production levels of eosinophils, serum IgE, IgG1 and IgG2a. Although this enhanced response was common in WT, as well as TLR4-KO, mice, the TLR4-KO mice group was found to show more robust responses in comparison to the WT mice, particularly in terms of the induction of IgE in the presence of both low and high dose of LPS.
Akdis suggested that Tregs can mediate tolerance against allergic immune responses. 15 Both naturally occurring Treg cells (nTregs), as well as Ag-specific adoptive/iTregs have been reported to suppress immune responses; however, iTreg cells might be more competent in certain cases.21–23 Additionally, selective expression of TLRs occurs on Tregs upon LPS stimulation, as well as through microbial product-induced inflammatory molecules, associated with activation of Tregs. 24 Indeed, we have observed that high dose of LPS exposure maintains airway tolerance by induction of Treg cells and subsequent inhibition of Th2 type response and lung inflammations. Here, we noticed that low dose of LPS exposure was unable to expand CD4+CD25+foxp3+ Treg cells in WT OVA-sensitized mice; however, we did not observe any differential effects in TLR4-KO mice at either low or high dose of LPS exposures. Enhanced airway inflammation with no change in iTregs as observed in TLR4-KO mice, indicates the pivotal role of TLR4 signaling through Treg-mediated suppression of allergic inflammation. In the absence of TLR4 signaling, LPS-induced immune response might be attributed to TLR4-independent but MyD88-dependent signaling pathways. 25 The above findings were also supported by previous researchers who found that adoptive transfer of Tregs suppressed the development of lung inflammations, and reported increased induction of Tregs during lung inflammations to countermeasure it. 26 IL-10 producing Tregs were reported to be more competent for blocking lung inflammations. 26 Our study suggested that low-dose LPS exposure inhibited the recruitment of IL-10-positive Tregs in the lung as compared with high-dose LPS exposure.
Further, we found higher levels of Th2 cytokine production during the low-dose LPS exposure. This observation suggested that low dose of LPS may suppress the coordination of Ag-specific immune regulation network by inappropriate generation of LPS Ag specific immune responses. Although effects of different doses of endotoxin in airway allergy demonstrated that TLR4 signaling regulates inflammatory responses, 27 the data presented here also indicated that TLR4 signaling regulates allergic inflammation especially at low dose of endotoxin exposure in the lung.
Although conventionally IL-6 has been considered as an inflammatory marker rather than as a regulatory cytokine, the role of IL-6 in promoting effector T-cell subsets indicated that IL-6 may play a functional role in allergic immune responses. Previous studies demonstrated that IL-6 can suppress the development and functional activity of Tregs in the lung and promotes Th2 immune responses in allergic airway disease.12,28,29 In the present study, we found a significant difference of IL-6 levels between the low- and high-dose LPS-treated mice groups. Additionally, we observed an inverse correlation between IL-6 production and levels of Treg cells under allergic conditions. Therefore, we can assume that IL-6 plays an important role in dose-specific LPS-mediated regulation of allergic immune response through Tregs.
Th2 cytokines such as IL-4, IL-5 and IL-13 16 were previously reported to be well correlated with airway allergic responses. The prospect of IL-25 has been also reported in airway allergy, 18 but how IL-25 transmits signals to the cells and what types of cells are targeted by this cytokine has been explored very little. 18 Keeping this background in mind we wanted to explore the role of LPS exposures in the release of IL-25 in the lung immune environment. We found that high-dose LPS significantly down-regulates the production IL-25 in OVA-sensitized WT mice, but in case of OVA-sensitized TLR4-KO mice IL-25 production was higher during both low- and high-dose LPS exposures.
Further, down-regulation of IFN-γ suggested that low dose of LPS might be associated with the breakdown of airway allergic tolerance, whereas high dose of LPS might inhibit airway allergic response by generating Ag-specific immune responses.
Thus, the findings presented in this article show that different doses of LPS influence ongoing allergen-mediated inflammatory as well as regulatory immune responses by either opposite ways or at significantly different levels, which is a unique finding. The high dose of LPS inhibits allergic inflammation through requirement of Tregs cells at inflammatory, whereas low dose of LPS fails to maintain Ag-specific immune regulatory homeostasis and resulted in decreased levels of Tregs in the lung of WT mice. These findings further supported the fact that high-dose LPS helps to maintain allergic immune tolerance through elevation of Tregs. Tregs probably respond directly to LPS and it may control the inflammatory response either in a positive or in a negative manner. Moreover, association of a high dose of LPS was found with inhibition of Th2 response and generation of associated Th1 cytokines. The TLR4-dependent reduction of allergic inflammation seen in WT mice is not associated with the production of Th1 cytokine at high dose of LPS exposure. However, high-dose LPS significantly decreased the production of IL-25 and IL-6 in WT mice. Thus, we can say that a high dose of LPS may inhibit the allergic airway inflammations through the inhibition of Th2 polarity in an iTreg-dependent manner. Taken together, the present study shows that exposure to different doses of LPS leads to alteration in balance between Tregs and effector T-cell immune response, thus influencing the susceptibility to allergic inflammation.
Footnotes
Acknowledgements
We are grateful to Professor Christopher L. Karp of Cincinnati Children's Hospital Medical Center for kindly providing us the WT and TLR4-KO mice. Thanks are due to Drs Jayati Chakrabarti and Eric Kettleson for their assistance in mice handling and maintenance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grant number R21ES017316 from the National Institute of Environmental Health Sciences and Grant No. OHLHH0199-09 from the Healthy Homes Technical Studies Program of the U.S. Department of Housing and Urban Development (HUD). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Environmental Health Sciences or the National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
