Abstract
The monocyte activation test (MAT) is a promising replacement of the currently used rabbit pyrogen test to detect the presence of pyrogens in injectable drugs. In the MAT, drugs are incubated with a source of human monocytes and production of pyrogenic cytokines used as readout. The best results are obtained with human mononuclear cells (MNC). However, donor variation requires testing on four different donors, and for most laboratories access to fresh MNCs is a problem. The current study shows how to overcome these problems using frozen pooled MNCs. The MAT is performed by thawing pooled MNC and co-culture overnight with a test substance, LPS or non-endotoxin pyrogens, with IL-6 production as the readout. The study demonstrates that fresh and frozen pooled MNC have comparable sensitivity. The reproducibility of the MAT performed with different batches of frozen pooled MNC was excellent. Different non-endotoxin pyrogens induce IL-6, confirming the ability of the MAT to detect a variety of pyrogens. In conclusion, the MAT using frozen pooled MNC is a highly sensitive, specific and reproducible pyrogen test, able to detect and quantify endotoxin and non-endotoxin pyrogenic contaminations in parenteral pharmaceuticals.
Introduction
Pharmaceutical products intended for parenteral use must be free of pyrogens, which may induce in recipients a systemic inflammation becoming apparent as fever and in severe cases hypotension upon administration of the drug. Examples of substances that can induce systemic inflammation are endotoxins from the Gram-negative bacterial cell envelope and non-endotoxin pyrogens from the Gram-positive bacterial cell envelope, in addition to viruses and fungi. Two methods to test drugs for pyrogenicity are widely used: the rabbit pyrogen test (RPT) and the bacterial endotoxin test (BET), making use of Limulus amoebocyte lysate. Both tests have their shortcomings. The RPT is not always able to detect human pyrogens. Some parenteral products, despite the fact that they passed the RPT, have caused pyrogenic responses in patients.1,2 Furthermore, the method is not quantitative and it only gives a pass/fail result. The BET is specifically intended for endotoxin pyrogens and cannot detect non-endotoxin pyrogens. In addition, test substances containing high protein levels and/or β-
In recent years an alternative pyrogen test, the monocyte activation test (MAT), has been developed to detect and quantify endotoxin and non-endotoxin pyrogenic contaminations. It is based on the growing insight of how fever is induced. 5 Exogenous pyrogens act via induction in the recipient of endogenous pyrogens such as IL-1β, IL-6 and TNF-α. In human blood, the monocyte is the main IL-1β-, IL-6 - and TNF-α-producing cell, which responds to pyrogen triggering.6–11 Based on these findings, a number of test systems have been developed. In essence, the test sample is incubated with human monocytes and the production of cytokines is used as readout. The source of monocytes is either PBMCs, whole blood or cells of a monocytic cell line (for a review, see Poole et al. 11 and Hoffmann et al. 12 ). Advantages of the MAT are its sensitivity for all types of pyrogens and its quantitative nature. Monocytic cell lines have the disadvantage that cell culture is tedious and that the cell lines are not stable. 11 PBMCs and whole blood show donor variation. In addition, obtaining fresh human blood can be a problem. One way to overcome the problems inherent to obtaining fresh human blood is to use frozen cells. Indeed, such a product using frozen whole blood is commercially available. 13 Recently, Koryakina et al. described a test using cryopreserved PBMCs with excellent results. 14 An ideal situation would be if a large number of identical ampoules could be cryopreserved. That would allow quality testing of the PBMCs before use, thereby greatly enhancing the reproducibility of the MAT. Therefore, we have investigated whether we could use PBMCs isolated from different human volunteers, pool them and cryopreserve large numbers of ampoules for use in the MAT. Our results show that this procedure allows freezing of at least 300 identical ampoules, each sufficient to test one 96-well culture plate in the MAT, with excellent results. We validated the test according to the requirements of the European Pharmacopeia and applied it to test human albumin produced for clinical applications. 15
Materials and methods
Preparation of frozen PBMC ampoules
PBMCs from the buffy coat of four healthy donors were isolated through separation over a Percoll gradient. From each buffy coat, an equal number of cells was pooled and prepared in Iscove’s Modified Dulbecco’s Medium (IMDM) (Lonza, Verviers, Belgium) with 5% FCS and 5% DMSO (Merck, Darmstadt, Germany). This is aliquoted in 10 million cells per CRYO ampoule (2 ml; Greiner bio-one, Frickenhausen, Germany). Ampoules were frozen and stored in liquid nitrogen. Approximately 300 ampoules can be prepared from four buffy coats. In order to perform MAT, ampoules containing frozen PBMC were placed in a water bath of 37℃ to thaw and were thereafter directly washed once with IMDM, using a laboratory centrifuge (8 min, 528 g, 5℃). Finally PBMC were taken up in culture medium (vide infra) at a concentration of 400,000 PBMC/ml, and 100 μl was added to 100 μl of the sample to be tested in 96-well flat-bottom microtiter plates (Nunc, Roskilde, Denmark).
Culture medium
Culture medium consisted of IMDM, penicillin 100 U/ml/streptomycin 10 µg/ml (Gibco, Merelbeke, Belgium), 2-mercaptoethanol 50 μM (Sigma-Aldrich, Steinheim, Germany), heat-inactivated FCS 5% (Bodinco, Alkmaar, the Netherlands) and human apo-transferrin 20 µg/ml (Sigma-Aldrich).
Endotoxin standard
EDQM LPS Escherichia coli (E0150000 EDQM, Council of Europe, Strasbourg, France) calibrated against the International Endotoxin Unit (EU) was used as a standard curve for endotoxin pyrogens. Stock solution (200,000 ng/ml) was prepared in IMDM, aliquoted (50 μl) and stored at –80℃. The endotoxin stock solution was diluted further in the culture medium to the required endotoxin concentrations, and prepared such that the final concentrations of LPS in the 96-well flat-bottom microtiter plates range from 0.4 to 100 pg/ml (0.004–1 EU/ml).
Albumin
Different batches of human albumin from Sanquin (Albuman 200 g/l; Sanquin, Amsterdam, the Netherlands) were tested in the MAT with a dilution ranging from 2 to 1458×.
Performing MAT
Samples were diluted in the culture medium and 100 μl was added to the microtiter plate in quadruplicate. The plate containing the test concentrations of the product and the LPS standard curve, cultured together with PBMC, was incubated in a humidified incubator at 37℃ in the presence of 5% CO2 for 20 h.
Detection of non-endotoxin pyrogens
Different endotoxin-free agonists such as lipoteichoic acid (LTA) (tlr-pstla; Invivogen, San Diego, CA, USA), Staphylococcus aureus (SAC) (PANSORBIN cells, standardized, heat-killed, formalin-fixed S. aureus cells; Millipore, Darmstadt, Germany), R848 (tlr-r848-5; Invivogen), Poly I;C (21072010; Invivogen), Pam3Cys (tlr-pms; Invivogen) and Flagellin BS (tlr-bsfla; Invivogen) were tested in MAT. In order to confirm that these non-endotoxin pyrogens were not contaminated with endotoxin, 40 µg/ml polymyxin B (PMB) (Sigma Aldrich) was used.
Cytokine measurement
IL-6 was measured in two dilutions using a commercially available ELISA kit (PeliKine Compact human ELISA kit; Sanquin). The lower limit of quantification of this validated ELISA kit is found to be equal to 1.4 pg/ml IL-6. IL-1β, IL-8 and TNF-α were also measured in similar commercially available ELISA kits (Pelikine Compact; Sanquin).
Product-specific validation test
To exclude interference between the test and the product being tested, the MAT has to be validated for that product according to the pharmacopeia guidelines. Therefore, endotoxin is spiked into an uncontaminated batch of the product and tested in the MAT. The product is considered free from interfering factors if the mean recovery of the added endotoxin is in the range of 50–200%. 15
We reported previously that reagents used in the MAT test, such as FCS and culture medium, can be contaminated with pyrogens. 16 Therefore, it should be emphasized that in the entire process of this test, all materials and reagents were sterile and pyrogen-free.
Results
Effect of pooling and freezing
PBMCs were isolated from the buffy coat of four individual donors and tested as such in MAT. Simultaneously, we mixed equal amounts of cells of each donor and tested the pool either directly or after freezing and thawing. As presented in Figure 1, a mixture of cells derived from four individual donors responds to LPS comparable to the average of the response of the individual donors. Frozen and thawed cells are as good as fresh cells (two-way ANOVA, multiple comparisons; not significant).
Comparison of fresh and frozen PBMC pool. Isolated PBMC from the buffy coat of four individual donors pooled equally, along with the frozen pooled PBMC after thawing, were diluted in the culture medium to a final concentration of 400,000 PBMC/ml. PBMCs were added to the LPS standard, concentration ranging from 0.02 to 4 EU/ml. As presented in this figure, frozen pooled PBMC showed comparable reactivity to fresh individual and pooled MNC upon endotoxin LPS (two-way ANOVA, multiple comparisons; not significant). Results are shown as mean ± SD of duplicate measurements for the MNC pools. Data of the average activation of individual donors are shown as mean ± SD of four individual donors measured in duplicate.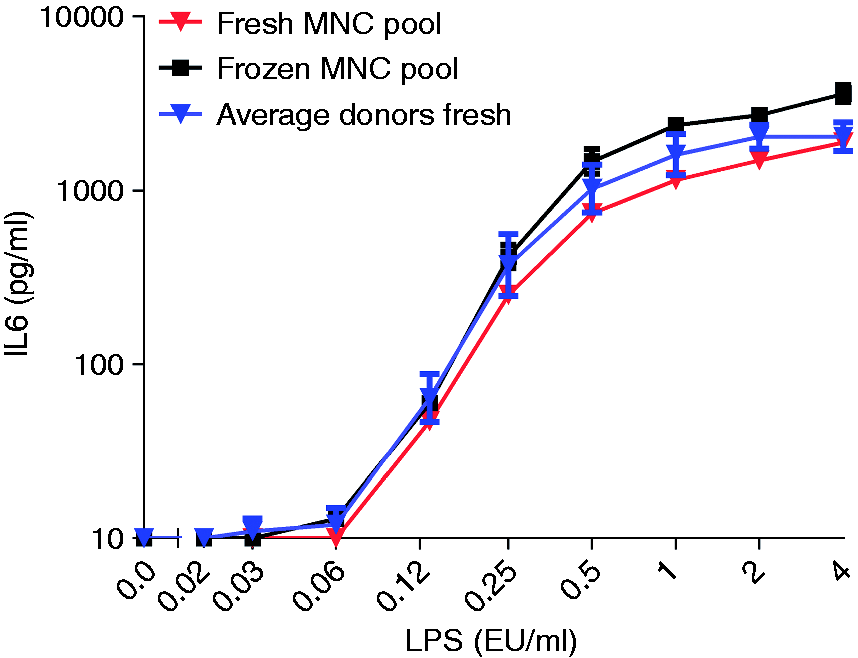
Reproducibility of the MAT performed with frozen pooled PBMC
To investigate the reproducibility of MAT, we tested different batches of frozen pooled PBMC. In total, we used 20 different donors and made five different pools of PBMC. In addition, we compared the response of the fresh individual cells to that of the pool. In all five cases the pools responded similarly to the average of the individual donors (data not shown). Moreover, the different pools are comparable in their reactivity towards LPS as shown in Figure 2 for three pools generated out of four different donors for each pool (two-way ANOVA, multiple comparisons; not significant). Figure 2 also illustrates the sensitivity obtained by measuring IL-6 production compared with IL-1β, IL-8 and TNF-α.
Reproducibility of the MAT performed with frozen pooled PBMC. Three independent frozen pooled PBMC batches were diluted in the culture medium to the final concentration of 400,000 PBMC/ml, and added to the LPS standard concentration, ranging from 0.001 to 1 EU/ml. As presented in this figure, three independently frozen pools of PBMC showed comparable reactivity upon the endotoxin LPS (two-way ANOVA, multiple comparisons; not significant). Results are shown as mean ± SD of quadruplicate measurements. Some SDs are so small that error bars are hardly visible. This figure also illustrates the sensitivity obtained by measuring IL-6 production compared with IL-1β, IL-8 and TNF-α.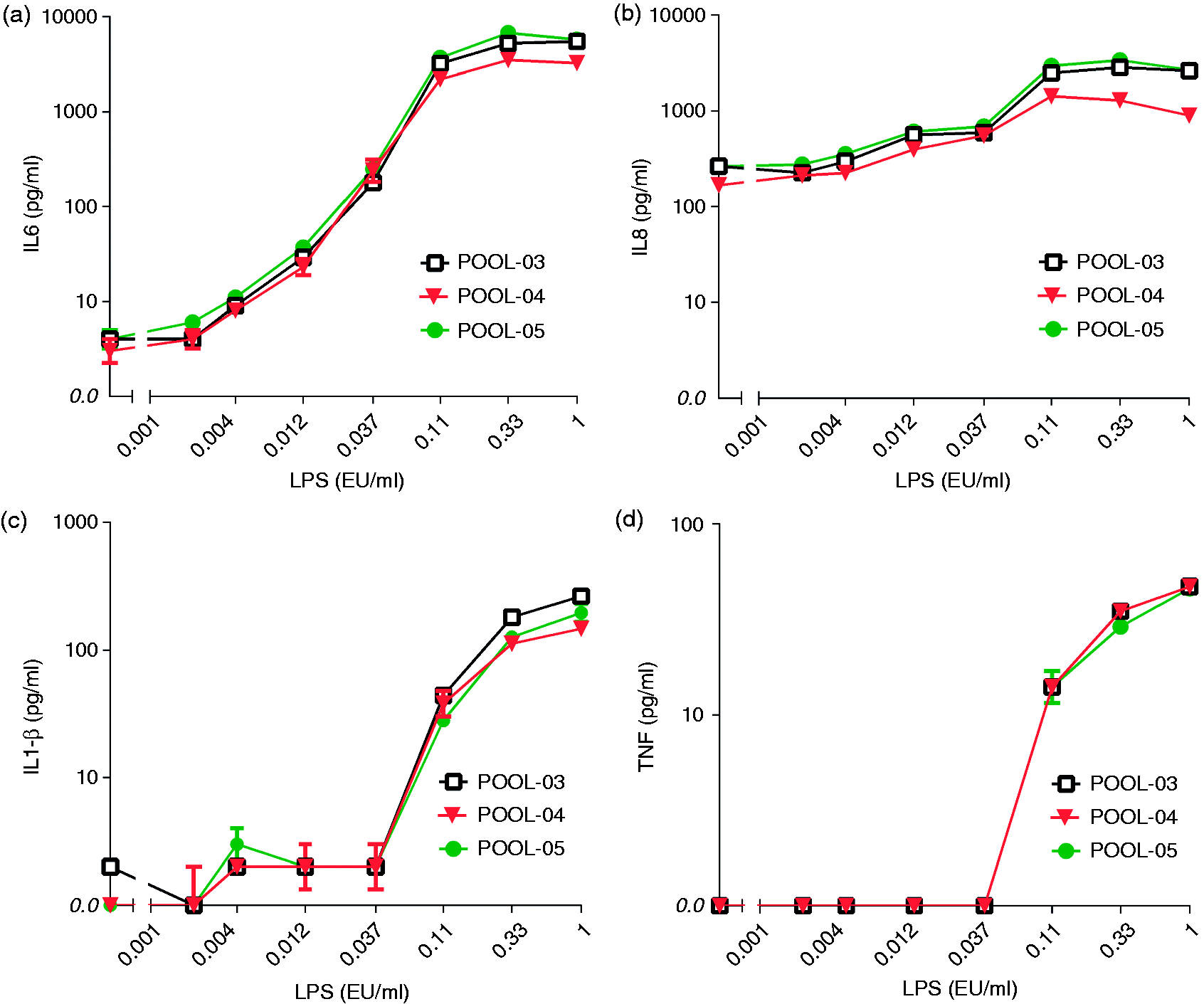
Sensitivity of the MAT performed with frozen pooled MNC
MAT sensitivity.
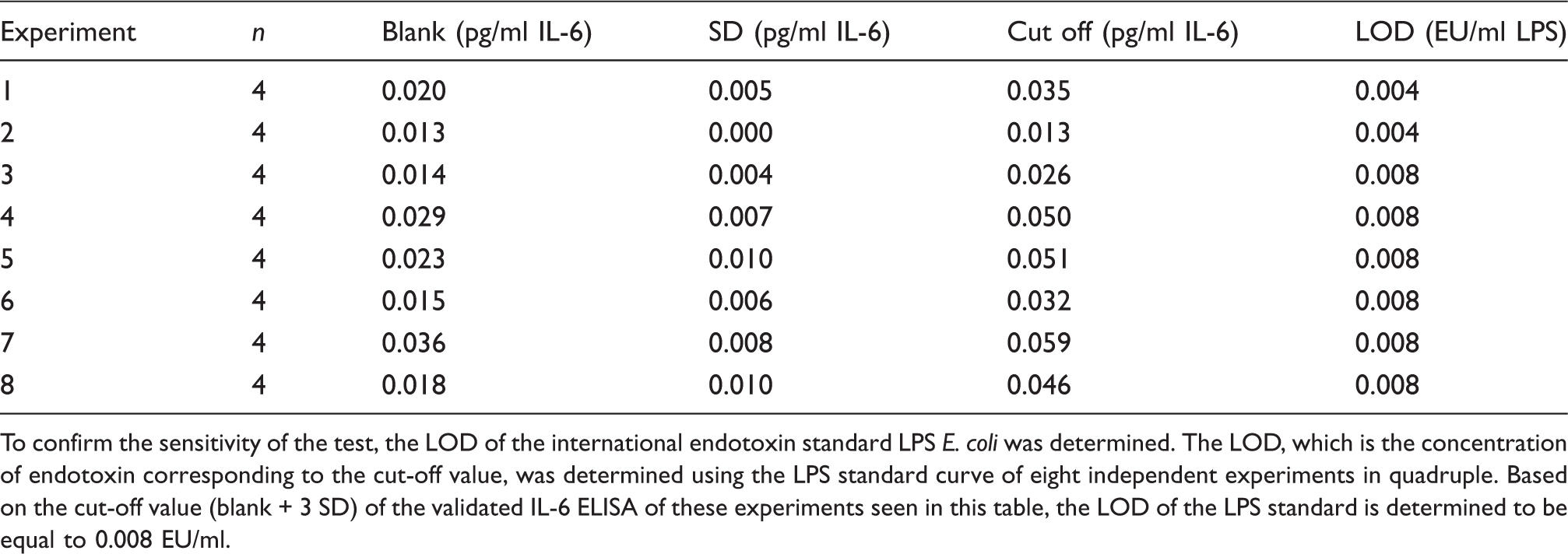
To confirm the sensitivity of the test, the LOD of the international endotoxin standard LPS E. coli was determined. The LOD, which is the concentration of endotoxin corresponding to the cut-off value, was determined using the LPS standard curve of eight independent experiments in quadruple. Based on the cut-off value (blank + 3 SD) of the validated IL-6 ELISA of these experiments seen in this table, the LOD of the LPS standard is determined to be equal to 0.008 EU/ml.
Ability of the MAT to detect non-endotoxin pyrogens
To investigate the ability of the MAT to detect a variety of pyrogens, the MAT is carried out using different agonists such as LPS, LTA, SAC, R848, Poly I;C, Pam3Cys and flagellin, which can activate monocytes via TLR4, TLR2, TLR2, TLR7/8, TLR3, TLR1/2 and TLR5, respectively. Again, four individual donors were compared with the frozen pool of these donors. As shown in Figure 3, variation in donor responses towards all tested non-endotoxins were comparable to the variations in the LPS response. This variability is corrected using the pooled PBMC as the reactivity of pooled PBMC is comparable to the average reactivity of individual donors (multiple t-test with alpha = 0.05; not significant). All experiments were carried out in the presence and absence of 40 µg/ml PMB. PMB completely inhibited the LPS response and had no effect on the other stimuli (data not shown), indicating that all TLR agonists were endotoxin-free and the observed cytokine response was only due to stimulation of the respective TLR.
Ability of the MAT to detect a variety of pyrogens and comparison of the activation of pooled PBMC of different donors upon endotoxin and non-endotoxin ligands. Isolated PBMC of four donors, individually or equally pooled, were diluted to the final concentration of 400,000 PBMC/ml and added to the (a) LPS standard concentration, ranging from 0.001 to 1 EU/ml, (b) SAC ranging from 10,000 × to 7,290,000×, (c) LTA ranging from 1.4 to 1000 ng/ml, (d) flagellin ranging from 0.13 to 2000 ng/ml, (e) Poly I;C ranging from 0.005 to 80 µg/ml, (f) R848 ranging from 0.003 to 40 µg/ml and (g) PAM-3 CYS ranging from 0.05 to 40 ng/ml. As can be seen, the agonists of different TLRs were able to stimulate the monocytes and induce IL-6. Additionally, the activation of pooled PBMCs upon both endotoxin LPS and non-endotoxin ligands, SAC, LTA, flagellin, Poly I;C, R848 and PAM-3 CYS, is comparable to the average activation of individual donors (multiple t-test with alpha = 0.05; not significant). Data of the average activation of individual donors is shown as mean ± SD of four individual donors measured in duplicate. The down error bars are missing from some points because the bottom of the error bar would go to a negative Y value, and negative numbers cannot be shown on a logarithmic axis.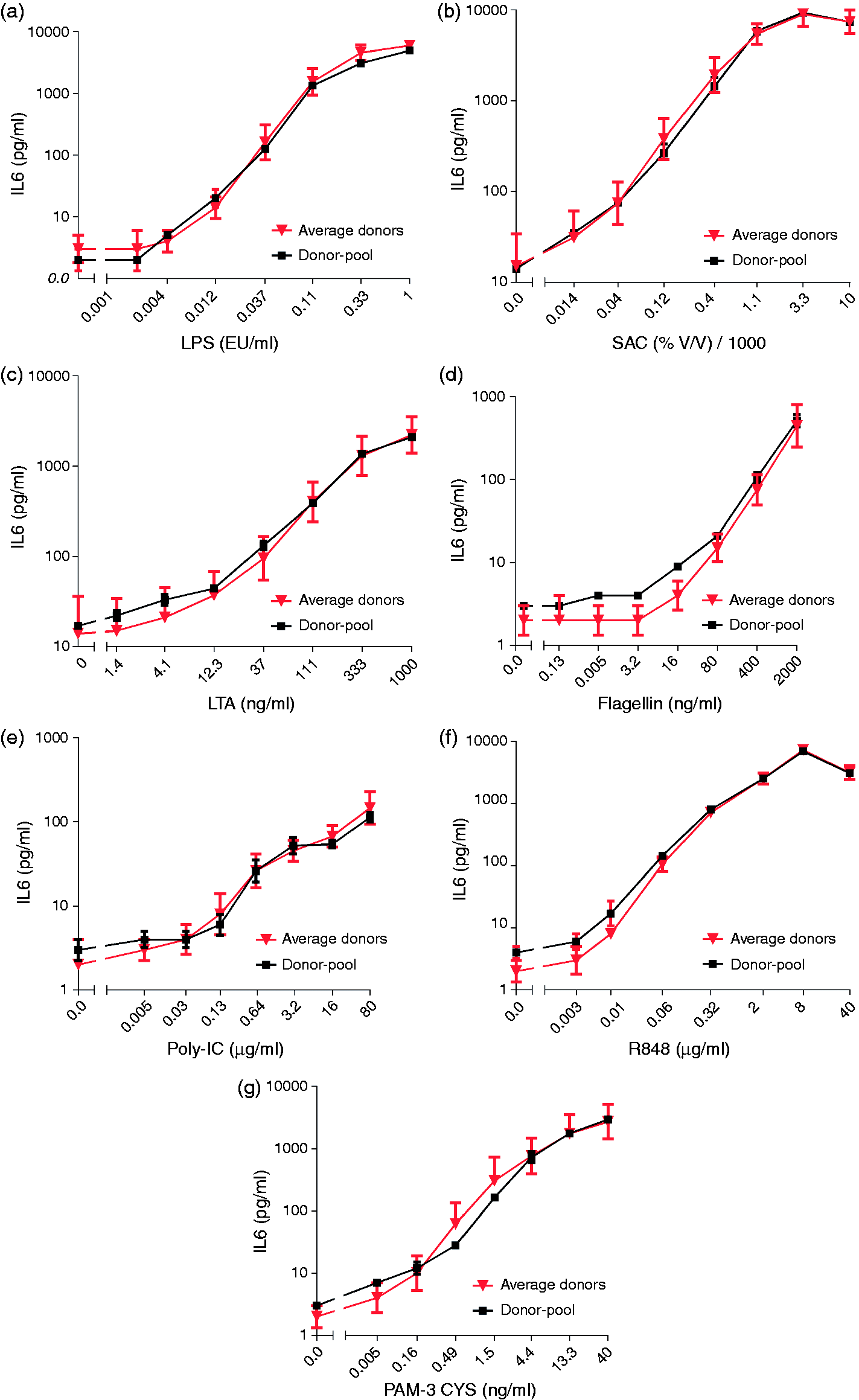
Non-endotoxin standardization
SAC stability.
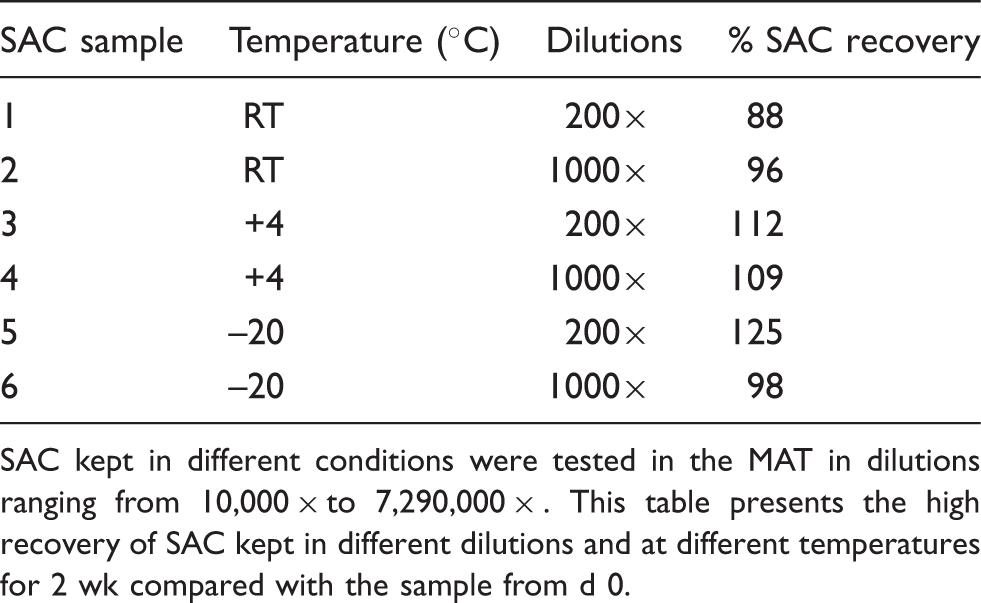
SAC kept in different conditions were tested in the MAT in dilutions ranging from 10,000 × to 7,290,000 × . This table presents the high recovery of SAC kept in different dilutions and at different temperatures for 2 wk compared with the sample from d 0.
Albumin product-specific validation for endotoxin and non-endotoxin pyrogens
Albumin product-specific validation of the MAT.
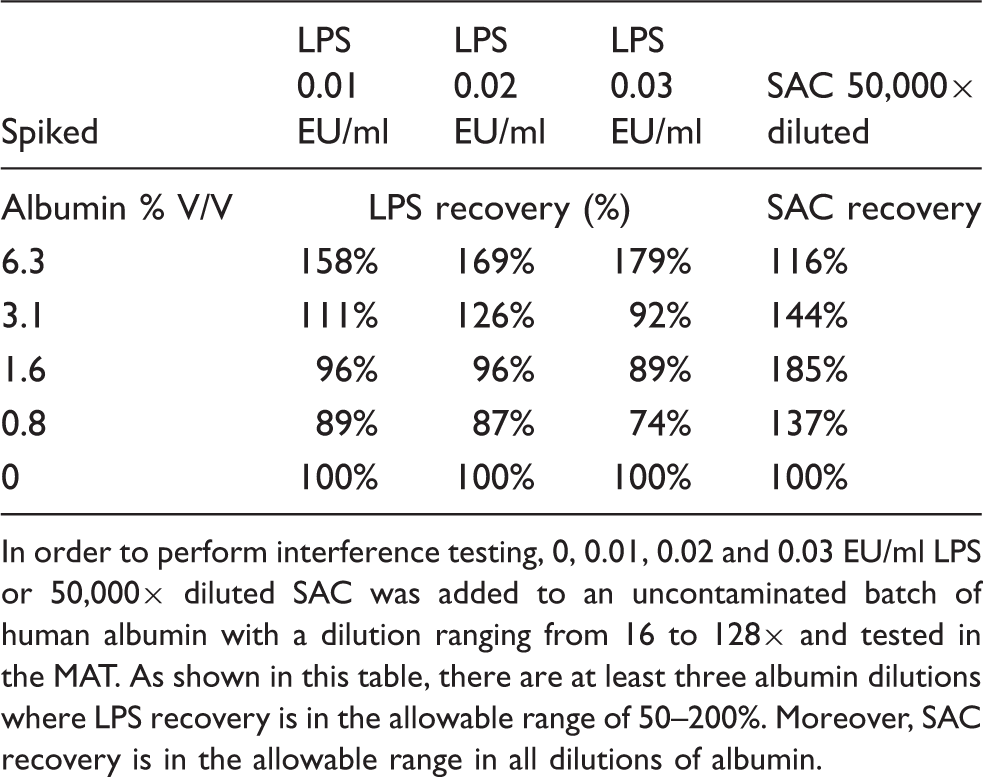
In order to perform interference testing, 0, 0.01, 0.02 and 0.03 EU/ml LPS or 50,000× diluted SAC was added to an uncontaminated batch of human albumin with a dilution ranging from 16 to 128× and tested in the MAT. As shown in this table, there are at least three albumin dilutions where LPS recovery is in the allowable range of 50–200%. Moreover, SAC recovery is in the allowable range in all dilutions of albumin.
Testing the pyrogenicity of different batches of human albumin.
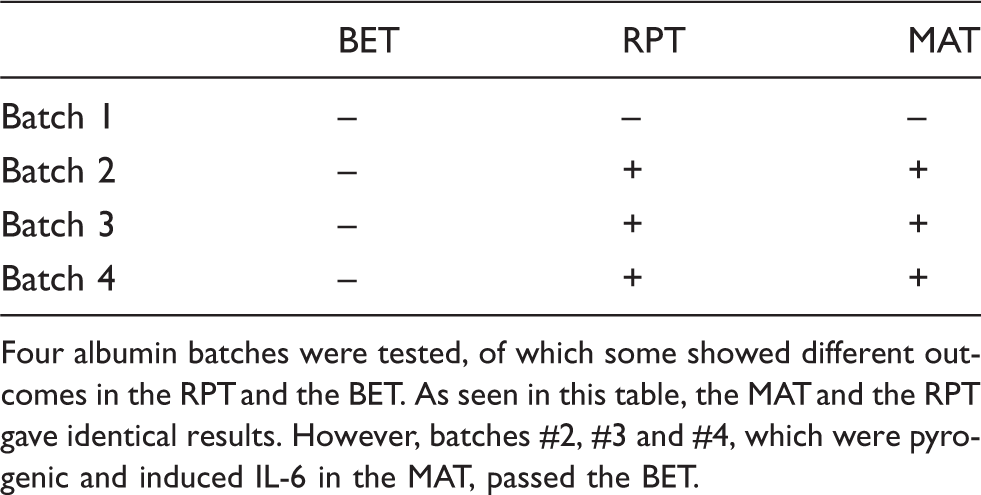
Four albumin batches were tested, of which some showed different outcomes in the RPT and the BET. As seen in this table, the MAT and the RPT gave identical results. However, batches #2, #3 and #4, which were pyrogenic and induced IL-6 in the MAT, passed the BET.
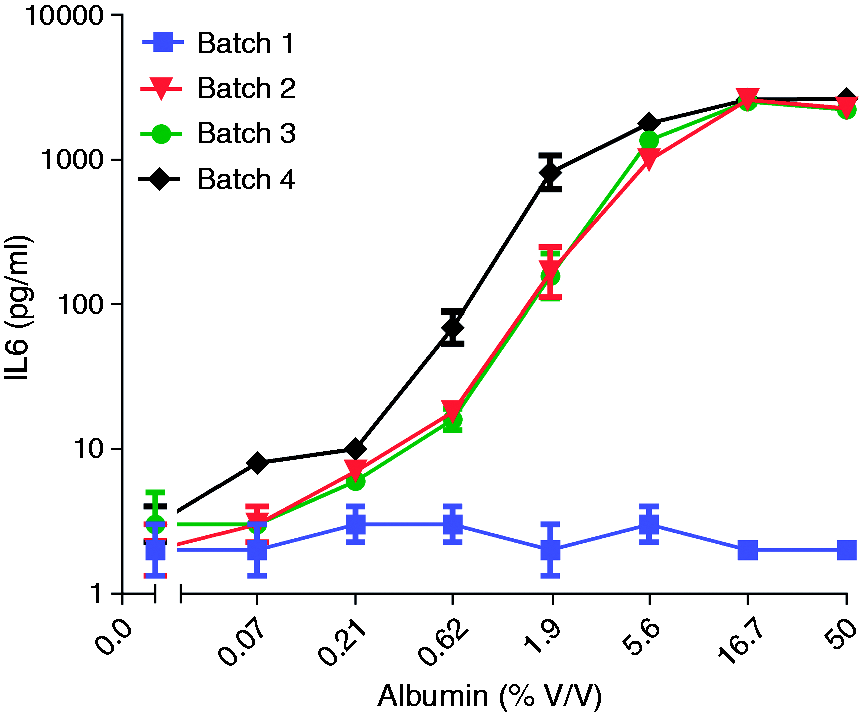
Albumin batches tested in the MAT. Different batches of human albumin were tested with a dilution ranging from 2 to 1458×. This figure presents the results for a number of these batches, among them several that were not passed by the MAT. Results are shown as mean ± SD of duplicate measurements.
Discussion
In search of a replacement for the RPT to test and quantify endotoxin and non-endotoxin pyrogenic contaminations in pharmaceutical products for parenteral use, the BET and the MAT have been developed. The BET is far more simple than the MAT but unfortunately it cannot detect non-endotoxin pyrogens. The reluctance to introduce the MAT is based on difficulties in obtaining fresh human blood and the variation in donor responses. Hence, for each test four different donors are required. In this paper we show that these problems can be overcome by preparing large pools of human PBMCs and storing the cells frozen. We have now tested batches that have been stored for 4 yr in liquid nitrogen and have observed no change in function and sensitivity. To explore the possibilities of shipment we also kept the cells at –80 ℃ in dry ice; storage for up to 2 wk at this temperature did not affect the behavior of the cells in the MAT.
The use of frozen PBMCs for the MAT was reported recently by Koryakina et al. 14 They used PBMCs isolated from leukocyte filters from single donors with a limited recovery of cells. The isolation of PBMCs from the buffy coats of four donors and the use of them as pools allows preparation of batches containing approximately 300 identical ampoules. Each ampoule suffices to test one 96-well microtiter plate. From the outset of these experiments we were afraid that the mixing of untyped cells would lead to a mixed lymphocyte reaction and possibly a high background stimulation of cytokine production. However, usually it takes 6 d in round-bottom wells (to increase cell–cell contact) to develop cytotoxicity. The 20 h incubation in the flat-bottom wells of the MAT is too short to lead to (background) cytokine production. Indeed, in the five pools that we have prepared until now we have seen no problems of background IL-6 production or of interactions between donor cells; the pools behave as the average of the four donors tested separately. Another advantage of the preparation of large batches of ampoules is that it allows for quality control. If, in the future, we run into a donor causing problems, it will become evident when the new batch is tested. However, to date, all tested donors responded well to all tested TLR stimuli.
We have used IL-6 production as readout but other cytokines were tested as well (Figure 2). In line with the observations of Poole et al. we observed IL-6 to be more sensitive than IL-1β and TNF-α. 11 However, it should be mentioned that in our experience TNF-α production peaks earlier than the other cytokines. Earlier sampling might lead to higher levels of TNF-α. IL-8 production is also very sensitive but has a much higher background than IL-6.
One major advantage of the MAT is that it responds to non-endotoxin pyrogens. All TLR ligands tested so far gave a high IL-6 response in the MAT. At the same time this leads to the necessity to develop standards for these pyrogens. Our experiments show that SAC is an interesting candidate for such a standard. Monocytes are very sensitive to SAC and the formulation is stable for at least 2 wk at room temperature. However, one should realize that SAC is only one of the many possible non-endotoxin pyrogens and that other stimuli using other TLRs should be tested. To include controls for every individual TLR would be problematic because the controls would consume most of the experimental space available. In that sense, the approach presented in this paper could be advantageous. Response to the various TLR agonists could be part of the quality control of a batch of frozen PBMC and a condition to use the ampoules in MAT.
Our experiment on the four batches of human albumin shows the usefulness of the MAT. Recovery of LPS is fine from a dilution of 1:32 on and because the MAT is so sensitive the detection limit for LPS at this albumin concentration is 0.26 EU/ml albumin (32 × 0.008), whereas the contamination limit as set by the European Pharmacopeia is 1.4 EU/ml. Moreover, recovery of the non-endotoxin (SAC) in the same albumin dilutions was also within the allowable range. This underlines the potency of the MAT to detect non-endotoxin pyrogens in pharmaceutical products such as albumin.
From our production plant we selected four batches of albumin that behaved anomalously in the RPT and the BET. It is clear that the RPT and the MAT correlate well, whereas the BET deviates from the two, which is in line with the fact that the BET does not detect non-endotoxin pyrogens.
We conclude that the MAT, using frozen pooled PBMCs and IL-6 detection, is a highly sensitive, specific and reproducible pyrogen test that is able to detect and quantify endotoxin and non-endotoxin pyrogenic contaminations in parenteral pharmaceuticals, and replace the RPT.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
