Abstract
This study aimed to investigate the levels of matrix metalloproteinase-8 (MMP-8) and tissue inhibitors of MMP-1 (TIMP-1) in saliva and serum samples of women with polycystic ovary syndrome (PCOS; n = 80) and matched systemically healthy controls (n = 45), with varying degrees of gingival inflammation. Salivary levels of MMP-8 and the MMP-8/TIMP-1 ratio were significantly elevated in women with PCOS, who also exhibited more gingivitis than systemically healthy women. No major changes were observed in salivary TIMP-1 levels with regard to PCOS. Serum levels of MMP-8 and the MMP-8/TIMP-1 ratio were significantly higher in women with PCOS, irrespective of the presence of gingivitis, while there were no differences in TIMP-1 levels. A positive correlation was indicated between probing depth, bleeding on probing, plaque index and salivary or serum MMP-8 levels or MMP-8/TIMP-1 ratio in the case of PCOS, while a negative such correlation was revealed for TIMP-1 in systemically healthy women. Increased levels of MMP-8 in saliva and serum seem to be more pronounced in women with PCOS, and potentiated in the presence of gingival inflammation. Alterations in MMP/TIMP system triggered by local and systemic inflammation may be implicated in the pathogenesis of PCOS, or the deterioration of its clinical presentation.
Introduction
Polycystic ovary syndrome (PCOS), or Stein–Leventhal syndrome, is one of the most common endocrine disorders, affecting about 6.5–8.0% of women. It is heterogeneous with reproductive and metabolic dysfunction.1,2 Patients with PCOS are at high risk for insulin-dependent diabetes, 3 visceral obesity 4 and cardiovascular disease. 5 Changes in the ovarian extracellular environment such as follicular development, ovulation and subsequent formation and regression of the corpus luteum, as well as follicular atresia, are regulated, in part, by the actions of a specific family of proteolytic enzymes known as the matrix metalloproteinases (MMPs). 6 MMPs and their tissue inhibitors [tissue inhibitors of metalloproteinases (TIMPs)] control the site and remodeling of the ovarian tissue, and are associated with a variety of physiological and pathological processes. 7 PCOS is characterized by abnormalities of ovarian tissue degradation and alterations of MMP expression. 8 It was previously demonstrated that serum MMP-9 concentrations and MMP-9/TIMP-1 ratios were significantly higher in women with PCOS than those in systemically healthy women. 7 An altered balance between serum levels of MMPs and TIMPs in women with PCOS has indeed recently been reported. 9 MMPs and TIMPs are also produced locally in periodontium, and altered levels of MMPs are associated with inflammation related to gingivitis and periodontitis.10,11 MMP-8 is a proteolytic enzyme secreted mainly by neutrophils that is present in inflamed gingiva, gingival crevicular fluid (GCF), saliva and serum. MMP-8 can be activated both by host and bacterial enzymes. 12 In several studies it was reported that GCF MMP-8 levels were found to differentiate between periodontal health and the conditions of gingivitis and periodontitis,13,14 and play an important role in periodontal tissue destruction. 13 Furthermore, serum MMP-8 and TIMP-1 levels may especially reflect systemic chronic inflammatory and immune responses.15–17 During the onset and progression of periodontal disease, at the cellular level, increased secretion of cytokines and inflammatory markers such as IL-6, IL-1 and TNF-α results in the release of MMPs when gingival health progresses to disease. 18 It was recently demonstrated that PCOS is particularly associated with the severity of gingival inflammation, especially on the pro-inflammatory cytokines such as IL-6, IL-17, TNF-α.19–21 Moreover, higher levels of suspected periodontal pathogens, such as Porphyromonas gingivalis and Fusobacterium nucleatum, and higher Ab responses to those, were observed in women with PCOS, particularly on the substrate of increased gingival inflammation. 22
Despite intense research, the pathophysiology of PCOS is not fully understood. It has been suggested in recent studies that the MMP–TIMP system may be implicated in the pathogenesis of PCOS through regulating the degradation and turnover of ovarian extracellular matrix. 23 As saliva is a suitable biological analyte to study in a global manner the oral health state, including gingival inflammation,24,25 and as PCOS is a systemic condition, it was deemed that saliva was appropriate for the design of this case–control study. Therefore, the aim of the study was to investigate the salivary and serum levels of MMP-8 and TIMP-1 in females with PCOS and systemically healthy controls, in the presence or absence of gingivitis.
Materials and methods
Study population and clinical examination
The Ethics Committee of Ege University approved the study protocol (13–5.1/13); all patients received information on the study and signed the informed consent before enrolment. The study was conducted in full accordance with ethical principles, including the World Medical Association’s Declaration of Helsinki, as revised in 2008, and conforms to STROBE guidelines for observational studies. 26 In four study groups a total of 125 patients (45 women with PCOS and healthy periodontium, 35 with PCOS and gingivitis, 25 systemically and periodontally healthy women, and 20 systemically healthy women with gingivitis) were recruited from the outpatient clinic of the Department of Endocrinology, School of Medicine, Aydın Government Hospital, Turkey, between October 2012 and April 2013. For each individual participant, comprehensive medical and dental histories were completed. A diagnosis of PCOS was assigned according to the 2003 criteria of Rotterdam with the presence of at least two of the following: polycystic ovaries, oligomenorrhea and/or anovulation, hyperandrogenism (clinical and/or biochemical). 27 Systemically healthy women were included as a control group with history of regular menstrual cycle, no clinical or biochemical signs of hyperandrogenism, and after verifying absence of PCOS by ultrasound examination. Patients were excluded if any of the following criteria were present: hyperandrogenism (tested by levels of 17-α-hydroxyprogesterone), diabetes mellitus, hyperprolactemia, congenital adrenal hyperplasia, thyroid disorders, Cushing’s syndrome, hypertension, hepatic or renal dysfunction, body mass index > 30 kg/m2, cardiovascular diseases. None of the patients were using medications such as oral contraceptive agents, steroid hormones, insulin-sensitizing drugs and antibiotics or anti-inflammatory drugs that could affect their periodontal status during in the 6 mo before inclusion in the study. Enrolled patients were referred to the clinic for clinical periodontal measurements, including probing depth (PD), plaque index (PI), 28 and the presence of bleeding on probing (BOP). BOP was assumed positive if it occurred within 15 s of application of the probe. Clinical periodontal measurements were recorded at six sites on each tooth, except for third molars, using a Williams periodontal probe (Hu-Friedy, Chicago, IL, USA). All clinical measurements were performed by a single calibrated examiner (AA).
Patient selection was based on consensus report of the 1999 International Workshop for the Classification of Periodontal Diseases and Conditions. 29 Diagnosis of gingivitis was assigned when BOP scores > 50% of all sites, PD < 3 mm at 90% of the measured sites and no more than one site had a PD > 4 mm or clinical attachment level (CAL) ≤ 1 mm, and no clinical and/or radiographic sign of periodontitis was evident.
Collection and processing of saliva and serum
All salivary samples were collected in the morning, between 8:00 am and 9:00 am. The individuals were first asked to rinse their mouth with distilled water, before expectorating whole saliva into sterile 50-ml tubes for 5 min. The collected saliva-containing tubes were then placed on ice, supplemented with EDTA-free Protease Inhibitor Cocktail (Roche Applied Science, Basel, Switzerland) and then centrifuged at 10,000 g for 15 min at 4℃. The resulting supernatants were then immediately aliquoted and frozen at –80℃. For serum collection, a total of 5 ml of venous blood was collected in BD Vacutainer blood collection tubes with silicone-coated interiors (BD Diagnostics, Franklin Lakes, NJ, USA) by a standard venipuncture method. The collected serum samples were left at room temperature to allow for blood clotting and then centrifuged at 1500 g for 15 min at 4℃ in order to remove the fibrin clot and other cellular elements. The serum samples were then immediately aliquoted and frozen at –80℃.
MMP-8 analyses by immunofluorometric assay
Salivary and serum levels of MMP-8 were analysed by a time-resolved immunofluorometric assay using the methods previously described by Hanemaaijer et al. 30 and Tuomainen et al. 17 MMP-8-specific Abs 8708 (Medix Biochemica Oy Ab, Kauniainen, Finland) were used as the catching and 8706 (Medix Biochemica Oy Ab, Kauniainen, Finland) as the tracer Ab, which was labeled using europium-chelate. All samples were assayed singly. Then, samples were diluted in assay buffer consisting of 20 mM Tris-HCl (pH 7.5), 0.5 M NaCl, 5 mM CaCl2, 50 µM ZnCl2, 0.5% BSA, 0.05% sodium azide and 20 mg/l diethylenetriaminepentaacetic acid, and incubated for 1 h, followed by tracer Ab incubation for 1 h. Five min after the addition of enhancement solution fluorescence was measured using a fluoremeter (1234 Delfia Research Fluoremeter, Wallac, Turku, Finland). The specificity of the mAbs against MMP-8 was the same as that of polyclonal MMP-8 Abs.30,31
TIMP-1 analyses by ELISA
ELISA (Duoset ELISA Development Systems; R&D Systems, Minneapolis, MN, USA) was used to evaluate salivary and serum levels of TIMP-1. 32 All samples were assayed in duplicate. The ELISA kit was capable of detecting the active, pro, complexed and fragmented forms of the studied TIMP-1, as mentioned by the manufacturer. MMP-8/TIMP-1 ratios were calculated as described previously. 17
Statistical analysis
A statistical software program (GraphPad Prism version 6.00c for Mac OS X; GraphPad Software, La Jolla CA, USA) was used for statistical analyses. The distribution of the variables was validated by D’Agostino-Pearson omnibus normality test. Comparisons between all groups for non-normal distributed variables (MMP-8, TIMP-1 levels and MMP-8/TIMP-1 ratios in serum and saliva) were performed by the Kruskal–Wallis test, and Dunn’s test was used in order to correct for multiple comparisons. For normally distributed variables (age, PI, BOP and PD) a one-way ANOVA test with Holm–Sidak’s multiple comparison test (family-wise significance and confidence level 0.05) was used. Correlations between BOP, PI scores, PD and biochemical data were analyzed by Spearman's correlation test. All tests were performed at α = 0.05.
Results
Clinical findings
The clinical and demographic data of this patient population have been reported previously. 22 Briefly, there were no statistically significant differences in age between groups, whereas the clinical measurements of PI, BOP and PD were significantly higher in the gingivitis groups, than the periodontally healthy groups, either systemically healthy or with PCOS. Clinical periodontal parameters were similar in the gingivitis groups with or without PCOS.
Salivary and serum levels of MMP-8 and TIMP-1
The salivary and serum MMP-8, TIMP-1 levels and MMP-8/TIMP-1 ratios were investigated. In women with PCOS, salivary MMP-8 levels were significantly elevated in gingivitis compared with periodontal health. In the systemically healthy group, no differences were found between periodontal health and gingivitis (Figure 1A). In systemically healthy women, salivary TIMP-1 levels were significantly higher in periodontal health compared with gingivitis. Nevertheless, no differences in TIMP-1 levels were found between the two PCOS groups (Figure 1B). Interestingly, the salivary MMP-8/TIMP-1 ratio was significantly higher in women with PCOS and gingivitis compared with periodontally healthy women, with or without PCOS (Figure 1C).
MMP-8 levels in serum and saliva. MMP-8 levels in serum and saliva from PCOS with periodontal health (Ph) (n = 45); PCOS with gingivitis (Pg) (n = 35); systemically healthy with periodontal health (Hh) (n = 24); systemically healthy with gingivitis (Hg) (n = 20). The individual values represent (A) MMP-8 levels in saliva; (B) TIMP-1 levels in saliva; (C) MMP-8/TIMP-1 levels in saliva; (D) MMP-8 levels in serum; (E) TIMP-1 levels in serum and (F) MMP-8/TIMP-1 levels in serum in both groups. The horizontal lines and numbers in the box plots represent the median values for each patient group and the whiskers represent the 5th–95th percentiles. * P < 0.05, ** P = 0.01, *** P < 0.001, **** P < 0.0001.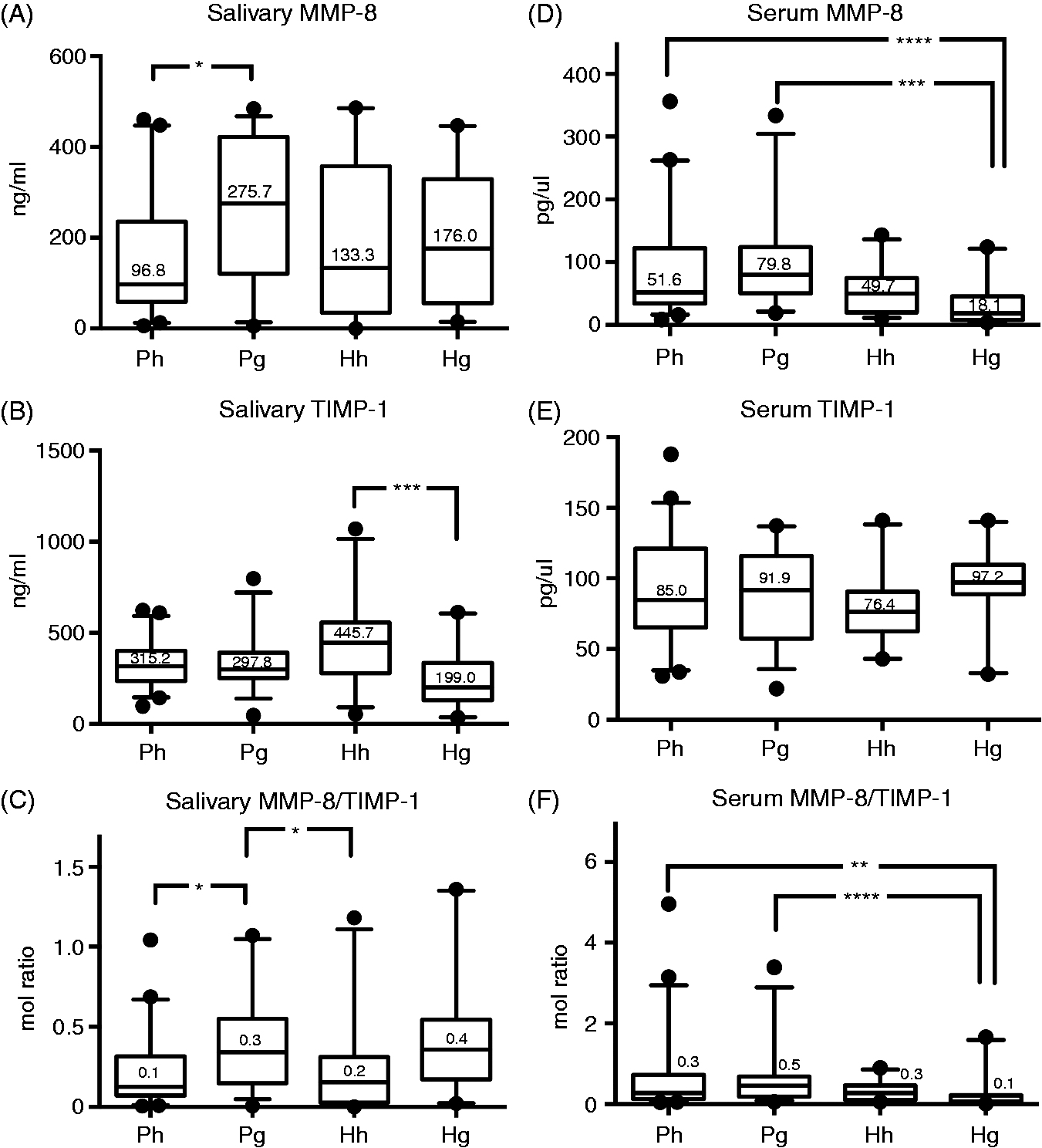
The corresponding serum levels of MMP-8 and TIMP-1 were then investigated. MMP-8 levels were significantly higher in women with PCOS and gingivitis compared with systemically healthy women with gingivitis. Significantly elevated serum MMP-8 levels were also found in periodontally healthy women with PCOS compared with systemically healthy women with gingivitis (Figure 1D). Serum TIMP-1 levels were not significantly different among the study groups (Figure 1E). Nevertheless, the serum MMP-8/TIMP-1 ratio was significantly higher in women with PCOS and gingivitis compared with systemically healthy women with gingivitis. It was also significantly higher in periodontally healthy women with PCOS compared with systemically healthy women with gingivitis (Figure 1F), following a trend similar that to MMP-8.
Correlations
Correlation analysis between clinical periodontal parameters and both serum and salivary MMP-8, TIMP-1 levels and MMP-8/TIMP-1 ratios. Significant correlations are shown in bold (p < 0.05).
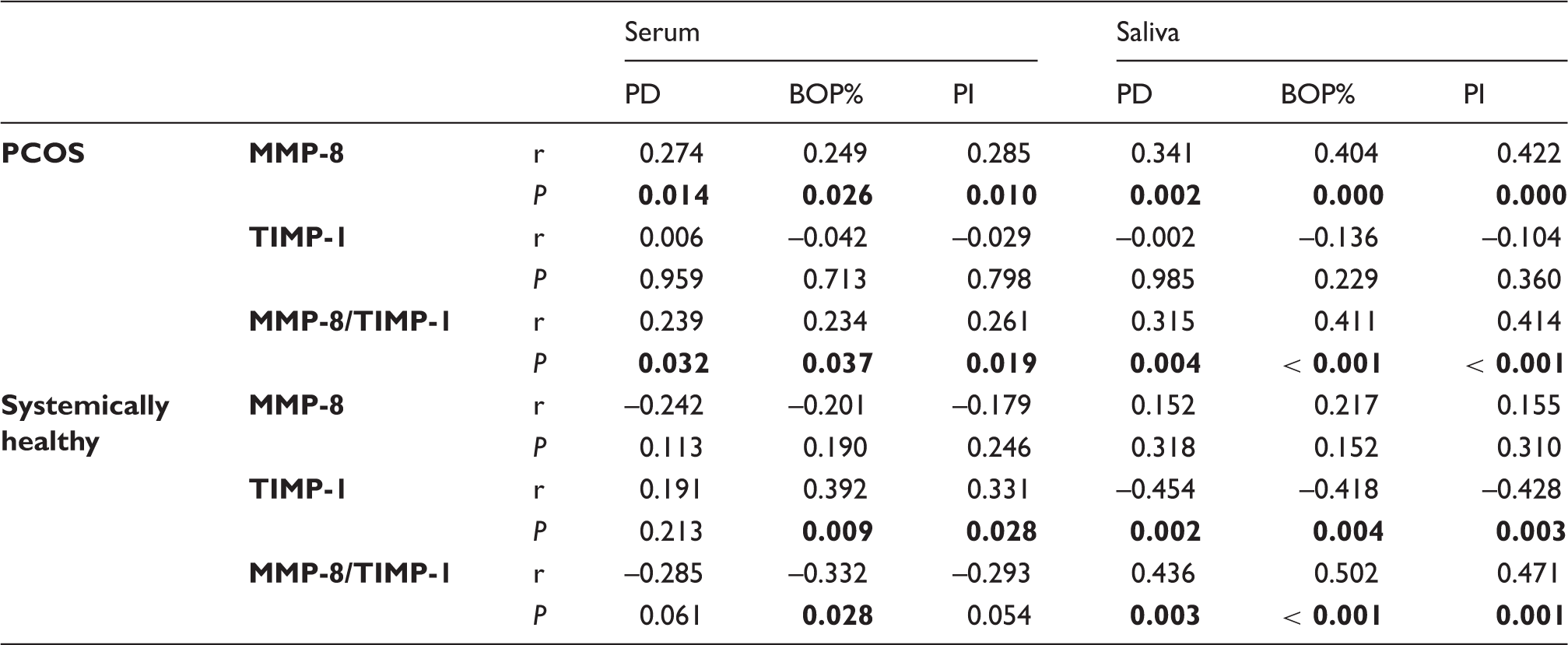
Regarding serum, there was a positive correlation between MMP-8 levels or the MMP-8/TIMP-1 ratio and all clinical periodontal parameters in PCOS. However, these correlations were not seen in systemically healthy women, who exhibited instead a positive correlation between TIMP-1 levels in serum and BOP% or PI.
Discussion
MMPs have been implicated in various physiological and pathological processes, including inflammatory response, 16 cardiovascular diseases 33 and ovarian dysfunction, as recently identified. In the present study, salivary and serum MMP-8, TIMP-1 levels and MMP-8/TIMP-1 ratio were analyzed in patients with PCOS and systemically healthy controls. The two groups were very similar in terms of demographic and clinical periodontal parameters, apart from higher plaque and bleeding on probing scores in the gingivitis groups. We demonstrated that women with PCOS had elevated serum and saliva MMP-8 concentrations, particularly in the presence of gingival inflammation (i.e. gingivitis). While TIMP-1 levels in serum remained unaffected, in saliva these proved to be lower in systemically healthy women with gingivitis. Interestingly, the serum MMP-8/TIMP-1 ratio was elevated in women with PCOS, irrespective of the presence of gingivitis. Although MMP-8 and TIMP-1 concentrations in plasma samples have been evaluated in women with PCOS, 9 their salivary levels or their association with gingival inflammation, have not been previously demonstrated. The lack of differences in clinical periodontal parameters between the groups (PCOS or systemically healthy) may imply a relationship between the elevated levels of MMP-8 and PCOS.
Gingivitis is a reversible condition associated with inflammation caused by the microbial biofilm accumulating on the tooth surfaces. If left untreated, gingivitis may progress to periodontitis in some individuals. 34 Determination of the progression of the inflammatory lesion could be explained by differential host responses attributed to an individual’s susceptibility. 35 Host-derived inflammatory mediators, such as cytokines, and proteolytic enzymes, including MMPs, are triggered and activated as a response to the bacterial infection.16,36 PCOS is characterized by chronic low-grade inflammation, 37 and a prolonged low-grade inflammatory state can be caused by chronic infections such as gingivitis, which is a common condition seen in the general population worldwide. It was reported that gingivitis is frequently observed in patients with PCOS.19–21 It is postulated that such peripheral and ovarian inflammation might be the precursor for insulin resistance, hypothalamic–pituitary–ovarian dysfunction and anovulation. 38 At least half of women with PCOS are obese, 39 or insulin resistant and hyperinsulinemic, independent of obesity. 40 Hence, obesity and diabetes could influence inflammatory status, which, in turn, may affect MMP-8 and TIMP-1 levels. Hence, it should be noted that non-obese and non-diabetic patients were included in the present study.
Regarding the association of PCOS with gingival inflammation, it was recently demonstrated that they appear to act synergistically on the production of pro-inflammatory cytokines IL-6, IL-17 and TNF-α.20,21 Moreover, a reciprocal link between PCOS and oral health was reported by elevated salivary levels of the periodontal pathogens P. gingivalis, F. nucleatum and Tannerella forsythia in women with PCOS, particularly in the presence of gingivitis. 22 In addition, certain microorganisms, such as Aggregatibacter actinomycetemcomitans are highly associated with the release and activation of MMP-8. 41 In the present study, a positive association between clinical periodontal parameters and the salivary or serum levels of MMP-8 was revealed, particularly in the case of PCOS but not in systemically healthy individuals. This positive correlation could denote an additive effect of PCOS on salivary MMP-8 levels, especially in the presence of gingival inflammation and since both systemically healthy groups showed similar levels of this biomarker. In contrast, systemically healthy women exhibit a negative correlation between TIMP-1 and clinical periodontal parameters, which is not observed in PCOS. Thus, considering the findings of the present study together with the previous data,20–22 it is suggested that PCOS has a confounding effect on gingival inflammation with regard to the MMP–TIMP system in serum and saliva.
It was previously demonstrated that TIMPs are protective against periodontal disease by controlling MMP activity, and are found in higher levels in saliva of periodontally healthy individuals. 42 The higher TIMP-1 levels in systemically healthy women with healthy periodontium, compared with women with gingivitis, confirm these previous findings. Still, the similar salivary TIMP-1 levels in both PCOS groups may reveal a direct or indirect effect of this underlying systemic condition on periodontal tissues. Although the mechanism still remains obscure, decreased salivary TIMP-1 levels in PCOS might imply decreased protection against MMP-8 activity and increased susceptibility to gingival inflammation. However, serum TIMP-1 levels were not significantly different among groups in the present study, which is in line with a previous report. 9 As molecules present in saliva could be either produced locally or derive from serum, 43 the discrepancy in TIMP-1 levels between serum and saliva observed here could be attributed to local (oral) rather than systemic production. Based on the present findings it is apparent that in the absence of gingival inflammation TIMP-1 levels are not consumed and can keep up the anti-MMP-8 protective shield, whereas during gingivitis salivary TIMP-1 is already being consumed and levels in saliva are reduced. As gingivitis is a less severe or initial form of periodontal pathology, it may not be surprising that the changes detected in saliva do not match with corresponding changes in serum. Nevertheless, one should not exclude the possibility that such changes may eventually occur in serum, once periodontal disease progresses.
The MMP-8/TIMP-1 ratio could be an indicator of the proteolytic activity within a biological medium. It was recently demonstrated, as a salivary biomarker, that this ratio correlated significantly with clinical periodontal variables, such as PD and BOP. 36 Our findings also show significant positive correlations between PD, BOP, PI and salivary MMP-8/TIMP-1 ratio. These could be owing to the degree of gingival inflammation, as postulated by a previous study. 36 It has also been demonstrated that women with PCOS had elevated serum concentrations of MMP-2, MMP-9 and MMP-9/TIMP-1 ratio compared to healthy women with regular ovulation. 7 Lewandowski et al. 8 reported similar results with significantly higher serum concentrations of MMP-2, MMP-9 and TIMP-1 levels in women with PCOS compared with healthy controls.
Elevated serum and saliva levels of the MMP-8/TIMP-1 ratio can be indicators of a critical equilibrium between MMPs and TIMPs that determines extracellular matrix degradation, and alterations in this ratio may lead to disease. Fundamental aspects of ovarian function (e.g. secretion of hormones and the production of oocytes) require dynamic, extensive, cyclic tissue remodeling. Therefore, deregulation of their balance may contribute to ovarian pathologies characterized by improper follicular development and atresia, such as PCOS.
It can be concluded from the present study that increased levels of MMP-8 and MMP-8/TIMP-1 ratio in saliva and serum seem to be more pronounced in women with PCOS, and potentiated by gingival inflammation. While the examined population is of a relatively young age and free of periodontitis, the elevated concentrations of MMP-8 pose a risk for future disease progression. Although the present findings highlight an important interplay between PCOS and gingival inflammation, owing to the case–control nature of the study it is difficult to conclude on the exact underlying mechanism for the involvement of the MMP-8-TIMP-1 system. Longitudinal prospective studies would be more suitable to probe this link, and reveal if there is a causal or occasional relationship.
Footnotes
Funding
This study was supported by a Visiting Research Fellowship (Aliye Akcalı) of the Continental European Division of the International Association of Dental Research, and the Institute of Oral Biology, Center of Dental Medicine, University of Zürich.
Acknowledgements
Timo Sorsa is an inventor of oral diagnostic US patents (5652247, 57336341, 5866432, 6143476).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
