Abstract
As innate immunity factors in breast milk (BM) modulate infants’ immune responses, we investigated whether soluble CD14 (sCD14) and defensin levels in BM are associated with the emergence of allergy in childhood. The randomly selected group of 260 mother–child pairs belonged to a randomized, double-blind placebo-controlled trial where 1223 mothers with fetuses at high risk for allergy received for the 4 last wk of pregnancy a mixture of probiotics, or placebo; after birth, the child received the treatment for 6 mo. Children were followed for the emergence of sensitization and allergic symptoms for 5 yr. IgE-mediated allergic disorder was diagnosed in 80 children by the age of 5 yr. Levels of sCD14, human neutrophil peptide (HNP) 1–3 and β-defensin 2 (HBD2) in colostrum and in BM 3 mo post-partum were measured with ELISA. BM sCD14 levels decreased from 0 to 3 mo. HNP1–3 and HBD2 were detected in colostrum, but not in BM 3 mo post-partum. High sCD14 levels in BM 3 mo post-partum were associated with children developing an IgE-mediated allergic disorder by the age of 5 yr. BM HNP1–3, HBD2 or sCD14 levels were not associated with probiotics treatment. Our results suggest that sCD14 in BM influences the emergence of allergy in children with atopic heredity.
Introduction
Breastfeeding affects an infant’s immune system in numerous ways. 1 To date, research on whether breastfeeding affects the development of allergy is, however, inconclusive. 2 Which of the numerous immunomodulatory factors in breast milk (BM), if any, affect the risk of developing allergy, remains unclear. Candidates for the induction of tolerance include IgA, TGF-β and Ag–IgG complexes.1,3–7 As innate immunity plays a key role in shaping the immune responses in infancy and early childhood, 8 we investigated innate immunity factors contained in BM in relation to the development of allergy in early childhood.
Soluble CD14 (sCD14) is one of many innate immunity factors present in BM. CD14 is part of a complex that recognizes LPS, which is a main component of Gram-negative bacterial cell envelope. 9 Most CD14 resides on monocytes and other immune cells, but it can also be found in soluble form in human serum and secretions. Soluble CD14 induces B cell growth and differentiation, and immunoglobulin production. 10 An interaction between polymorphisms in CD14 gene and endotoxin exposure modulates the development of allergy. 9 A few studies have indicated an association of sCD14 levels in BM and the development of allergy in the child. Jones et al. observed that lower sCD14 levels in BM 3 mo postpartum were associated with eczema in infants at 6 mo of age. 11 We previously showed that sensitization and allergic symptoms by the age of 4 yr were associated with low sCD14 levels in mother’s colostrum. 3 Rothenbacher et al. reported a positive association of sCD14 levels in BM 6 wk postpartum and decreased risk of asthma by the age of 2 yr. 12
Defensins are another group of innate immunity factors found in BM. These antimicrobial peptides target a wide range of pathogens from Gram-negative and Gram-positive bacteria to fungi and viruses. 13 Based on their structure, human defensins are categorized into α-defensins and β-defensins. α-Defensins are expressed in neutrophils (human neutrophil peptides; HNP) and in intestinal Paneth cells (human Paneth cell defensins; HD) in a mostly constitutive manner. 13 A variety of epithelial cells express human β-defensins (HBDs). HBD1 expression is mostly constitutive, whereas HBD2–5 expression is induced by pathogens and cytokines. 13 In addition to their direct antimicrobial action, defensins may act as chemoattractants, 14 modulate T helper cell function and activate APCs.15–18 Abnormal expression of defensins has been linked to inflammatory bowel diseases, psoriasis and infection susceptibility in atopic dermatitis. 19 We recently showed that high fecal HBD2 levels in infancy were associated with an increased risk for sensitization by the age of 5 yr, and that in contrast high fecal HNP1–3 levels were associated with a decreased risk for atopic dermatitis. 20 We also found that probiotics treatment compared with placebo was associated with slower decrease in fecal HBD2 levels. 20 Indeed, certain probiotics appear to induce HBD2 expression in the gut.21–23
We investigated whether sCD14, HBD2 and HNP1–3 levels in colostrum and in BM 3 mo postpartum were associated with the emergence of eczema, sensitization and allergy between the ages of 6 mo and 5 yr in a population of healthy, full-term infants with atopic heredity. We also studied whether probiotic treatment affected the levels of these innate immunity factors in BM.
Materials and methods
Cohort
The current study is based on a randomized double-blind allergy prevention trial on pro- and prebiotics. We have previously described the protocol in detail and reported the main clinical findings.24,25 The ethics committee of the Hospital for Children and Adolescents of Helsinki University gave its approval for the study protocol. Mothers signed a written informed consent.
We recruited 1223 pregnant women who carried high-risk fetuses (one or both parents had doctor-diagnosed allergic disease), and randomized them to treatment and placebo groups at 35 wk of gestation. The study was double blinded. The treatment for mothers was one capsule containing a mixture of probioitics Lactobacillus rhamnosus GG (ATCC 53103) 5 × 109 CFUs, L. rhamnosus LC705 (DSM 7061) 5 × 109 CFUs, Bifidobacterium breve Bb99 (DSM 13692) 2 × 108 CFUs and Propionibacterium freudenreichii ssp. shermanii JS (DSM 7076) 2 × 109 CFUs, twice daily for approximately 4 wk before delivery. Infants followed the protocol from birth to the age of 6 mo. They received one opened capsule containing the same probiotics plus 0.8 g galacto-oligosaccharides in syrup once daily. Placebo capsules contained microcrystalline cellulose only, and placebo syrup contained no galacto-oligosaccharides. The supplier (Valio, Helsinki, Finland) delivered the products to the mothers in sequentially numbered packages. We asked parents not to feed other probiotic preparations during the intervention. Exclusion criteria were pre-maturity (< 37 wk of gestation), major malformations or being a B-twin.
We examined the children at the ages of 3 and 6 mo, as well as at 2 and 5 yr, and additionally if parents reported that their child showed allergic symptoms. Primary outcome measures were the cumulative incidence of any allergic disease and any IgE-mediated (atopic) allergic disease until the age of 2 or 5 yr. Eczema, food allergy and asthma with and without IgE sensitization, allergic rhinitis and IgE sensitization alone were secondary outcome measures. We based eczema diagnosis on the UK Working Party’s criteria. 26 When eczema was associated with sensitization, the diagnosis of IgE-associated atopic eczema was established. We diagnosed food allergy with an open food challenge, 27 except in the case of doctor-diagnosed anaphylaxis. Criteria for asthma diagnosis were two or more doctor-diagnosed wheezy episodes coinciding with persistent cough or exercise induced symptoms between episodes, or with bronchial hyper-responsiveness in oscillometry. Allergic rhinitis was diagnosed based on two or more recurrent symptoms (nasal discharge, blockage, sneeze/itch) during contact with a specific Ag that the patient was sensitized to. 28 Children underwent skin prick tests (SPTs) on a panel of food and inhalant allergens applied with a standard technique. 29 We measured allergen-specific IgE against cow’s milk, egg white, birch, timothy, dog and cat, and, at 5 yr, also to wheat, fish, peanut, mugwort and Dermatophagoides pteronyssinus in sera with an immunoassay (ImmunoCAP® system; Pharmacia Diagnostics, Uppsala, Sweden). We defined sensitization as one or more positive SPT results and/or serum Ag-specific IgE > 0.7 kU/l to the allergens tested. In case allergic symptoms coincided with sensitization, the child was considered to have IgE-mediated allergy.
We collected BM samples at the maternity hospital (colostrum samples, 0–3 d post-partum) and 3 mo later from mothers participating in the study. The mother manually expressed 5–20 ml BM immediately before breastfeeding her child during the daytime. For the current study, we had randomly selected samples available from 260 mothers: 210 colostral samples and 227 BM 3 mo post-partum. We used SPSS (IBM, Armonk, NY, USA) software for random selection of half of the available colostral samples from mothers of the intention-to-treat children who had attended the 2-yr follow-up visit, and selected all BM pairs available (364 colostrum and 321 BM samples). Among these samples, for the present study, we used those samples for which we had the results of the 5-yr follow-up available. BM samples were immediately frozen (−20℃), brought frozen to the laboratory and stored at −70℃. After thawing, we centrifuged the samples at 10,000 g for 30 min, discarded the cellular debris and fat layer, and used the clear middle layer for analyses. We performed ELISA according to manufacturers’ instructions for measuring HNP1–3 (Hycult Biotechnology, Uden, the Netherlands; detection limit 0.050 ng/ml) and HBD2 (Immundiagnostik, Bensheim, Germany; detection limit 0.077 ng/ml) and sCD14 (Hycult Biotechnology; detection limit 1.56 ng/ml, lowest standard at 3.1 ng/ml). The intra-assay coefficients of variation (CV) were 6.5% and for HNP1–3, 4.3% for HBD2 and 5.9% for CD14. Interassay CV for CD14 was 8.0% (12 assays), for HBD2 it was 7.3% (seven assays) and for HNP1–3 it was 19.1 % (three assays). The number of assays to determine interassay CV% is low for defensins because the levels were below the detection limits in most BM samples 3 mo post-partum.
Statistical analyses
As measurement data were skewed, we performed logarithmic transformations. Values under detection level were computed as lowest detection level divided by two. We compared sCD14, HBD2 and HNP1–3 levels, and continuous clinical variables between groups with independent two-sample t-test. Dichotomic clinical variables were compared with the χ2 test. We performed binary logistic regression analysis with IgE-mediated allergy at the age of 0–5 yr as the dependent variable, and mother’s atopy, the mode of delivery, the number of siblings, presence of pets in the household, treatment with probiotic or placebo and one of the measured BM components at a time as covariates. We analyzed the effect of time on sCD14 levels with the general linear model of repeated measures. We considered a P-value ≤ 0.05 to be significant. We analyzed the data with SPSS version 15.0 (IBM).
Results
Characteristics of the study population and the whole original clinical study group divided into children with IgE-mediated allergic disorder at the age of 0–5 yr and those without. IgE-mediated allergic disorder is defined as allergic symptoms combined with a positive SPT and/or specific IgE levels > 0.7 kU/ml for at least one allergen on a broad panel. None of the variables differs significantly between the two groups (P > 0.1, χ2 test or t-test).
Levels of sCD14 in BM 3 mo post-partum were higher in mothers whose children developed an IgE-mediated allergic disorder by the age of 5 yr (Figure 1; Table 2). BM 3 mo post-partum from mothers with children who developed eczema and sensitization by the age of 5 yr had higher levels of sCD14 (n = 50, mean 6.1, 95% CI 5.3–6.9) than samples from mother with non-sensitized children without eczema (n = 167, mean 4.6, 95% CI 4.1–5.3 (t-test P = 0.03).
Levels of sCD14 in colostrum and in BM samples 3 mo post-partum in children who developed an IgE-mediated allergic disorder at the age of 0–5 yr (shaded bars, n = 70 for colostral samples and n = 68 for BM samples 3 mo post-partum) and in children who did not (open bars, n = 132 and n = 149). IgE-mediated allergic disorder is defined as allergic symptoms combined with a positive SPT and/or specific IgE levels > 0.7 kU/ml for at least one allergen on a broad panel. Bar height denotes geometric mean and lines 95% CI. P-Value is based on t-test (on logarithmic transformations). Levels of HBD2, HNP1–3 and sCD14 in colostrum samples, and sCD14 in BM samples at 3 mo post-partum in children with or without an IgE-mediated allergic disorder at the age of 0–5 yr. IgE-mediated allergic disorder is defined as allergic symptoms combined with a positive SPT and/or specific IgE levels > 0.7 kU/ml for at least one allergen on a broad panel. Values change significantly with time (P < 0.05, general linear model of repeated measures). bValues differ significantly: P < 0.01, t-test on logarithmic transformations.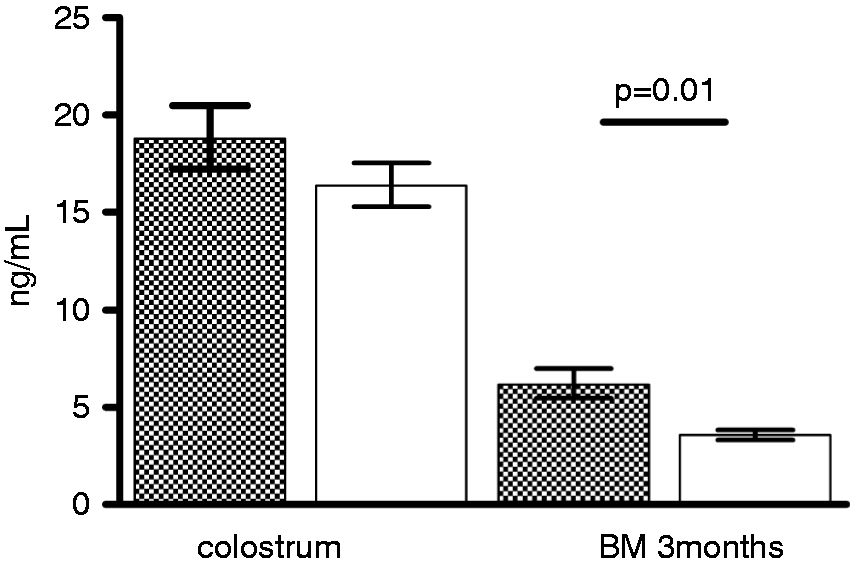
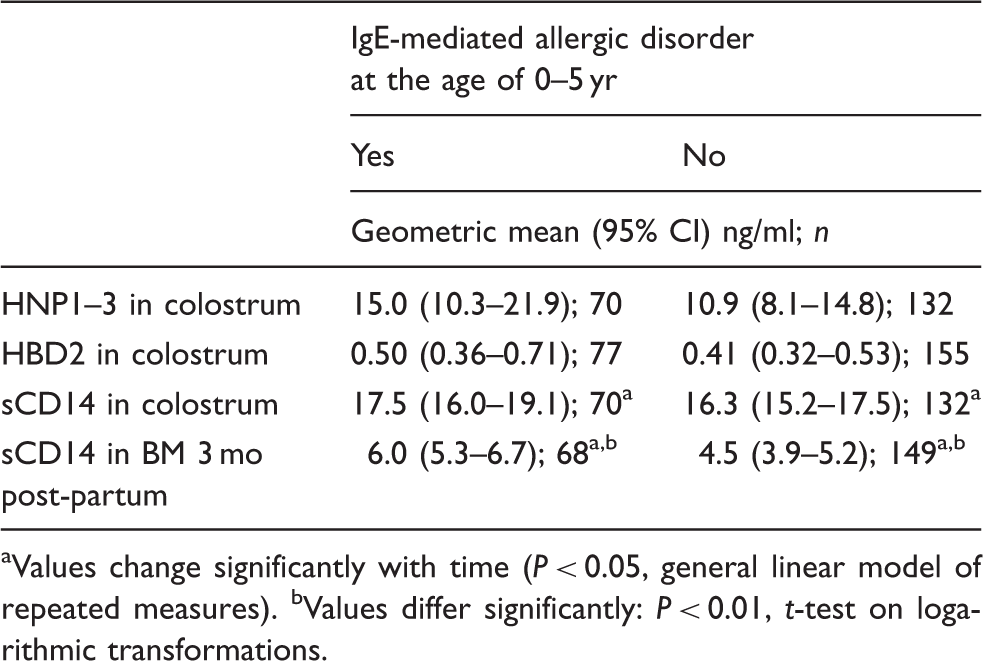
HNP1–3 and HBD2 levels in BM were not associated with children’s later allergy (Table 2).
sCD14 was detectable in all BM samples. HNP1–3 was over detection limit in 99% of colostral samples and HBD2 in 75%. Defensins were mostly undetectable in BM samples 3 mo post-partum.
Levels of sCD14 in BM decreased from 0 to 3 mo post-partum (Figure 1; Table 2). According to analysis with a general linear model of repeated measures, the temporal difference in sCD14 levels from 0 to 3 mo was significant (P < 0.0001), whereas the temporal change did not differ between mothers whose children developed IgE-mediated allergy or did not (Figure 1; Table 2). HBD2 and HNP1–3 levels also decreased over time as they were detectable in most colostral samples, but not in BM samples 3 mo post-partum.
We tested the association of sCD14, HBD2 and HNP1–3 levels in colostrum, as well as of sCD14, in BM 3 mo post-partum with several other clinical variables: eczema at the age of 0–6 mo or by the age of 5 yr, sensitization or allergic symptoms without sensitization by the age of 5 yr. We observed no statistically significant associations with these variables (data not shown). Binary logistic regression analysis with variables listed in methods revealed no significant cofactors (data not shown).
Treatment with pro- and prebiotics or placebo was not associated with sCD14, HBD2 and HNP1–3 levels in colostrum, or with sCD14 levels in BM 3 mo post-partum (data not shown).
Discussion
High levels of sCD14 in BM 3 mo post-partum were associated with increased risk of IgE-mediated allergy in the child by the age of 5 yr, while colostral sCD14, HBD2 and HNP1–3 levels were not associated with allergy development.
The 260 mother–child pairs included in this study were part of a large (n = 1223) randomized, double-blind, placebo-controlled trial that investigated the effect of preventive pro- and prebiotics treatment on the emergence of sensitization and allergy in a high-risk population.24,25 We followed the children until the age of 5 yr. The clinical outcomes until the ages of 6 mo and 2 and 5 yr were based on prospective questionnaires and children’s visits at our outpatient clinic where the same doctor (AKK) thoroughly assessed the clinical status, sensitization and allergic disorders, which were diagnosed based on strict criteria. We asked the participating mothers for BM samples in the maternity hospital (colostrum) and 3 mo post-partum. Although the number of samples available for the current study could ideally have been larger given the scale of the clinical trial, we are confident that the study population of 260 mother–child pairs is large and representative enough for the results to be reliable. As the protocol of the allergy prevention trial stipulated, most of the mothers were atopic and all children had at least one parent with atopy. As the effect of pro- and prebiotics treatment,30,31 as well as of breastfeeding, 32 may depend on the hereditary status of the mother and/or the child, our results may not unequivocally apply to the general population.
Our result that high sCD14 levels in BM are associated with increased emergence of allergy appear to contradict previous studies. Snijders et al. found no association between sCD14 and the development of atopy by the age of 2 yr. 33 The observation remained unchanged, even when the CD14 genotype was taken into account. 34 Other studies have, however, reported an association between low sCD14 levels in BM and the development of atopy or asthma.3,11,12 Several factors may explain some of the discrepancy. Previous studies have had shorter follow-ups than our current study, which lasted until the age of 5 yr. In the current study we did not find any association of atopy and allergy at 6 mo and 2 yr with BM sCD14 levels. The time of BM collection also varies between studies, from colostrum to 6 mo post-partum. We studied both colostral and BM samples at 3 mo, and observed no association with colostral sCD14 levels and child’s allergy. Nevertheless, contradicting results insinuate that BM sCD14 has a relatively mild effect, which can be superseded by other factors influencing the development of allergy. Studies that have investigated the association between cord blood levels of sCD14 and the development of allergy, have found no association,35,36 whereas low levels of circulating sCD14 later in infancy have been associated with atopy. 36 Both BM and serum levels of sCD14 appear to be higher in atopic mothers than non-atopic mothers.33,35 As our study population was selected to have atopic heredity unlike populations in the other aforementioned studies investigating BM, we propose that atopic heredity affects the association between BM sCD14 and later allergy in the child. Genetic factors, along with environmental cues, may drive the B cell activation induced by BM sCD14 towards the production of IgE class Abs. 37
Our result that probiotics treatment 4 wk before delivery was not associated with sCD14 levels in BM is in line with a previous study that measured sCD14 levels in colostrums and in BM 3 and 6 mo post-partum. 38 In our study, mothers did not continue the treatment after delivery, whereas in the study by Prescott et al., 38 mothers used probiotics 2–5 wk before delivery and as long as they were breastfeeding. These two studies investigated different strains of probiotics. Thus, at least two lines of evidence imply that probiotics do not induce sCD14 secretion to BM. A study on the early intestinal colonization and circulating sCD14 has furthermore shown that only Staphylococcus aureus colonization was associated with high serum sCD14 levels in infants. 39
Immunomodulatory factors in BM have been linked to the emergence of allergy in the child.1,3–6 To our knowledge, however, defensins have not previously been investigated in this context. Our previous study on fecal defensin levels in infancy was based on a subpopulation drawn from the same trial as in the present study. 20 We investigated fecal samples at the age of 3 and 6 mo, and only at the latter time point did we see associations between defensin levels and later allergy. Given these and our present results, it is plausible that fecal defensin levels reflect maturation of the immune system and of the gut flora. 40 Our results suggest that defensins in BM do not have a significant role in modulating the immune system early in a way that would affect the later emergence of allergy.
We propose that the association of sCD14 levels in BM and later allergy may depend on atopic heredity, as our results in a high-risk population differed from earlier studies. According to the results from our current and previous studies, intestinal defensin secretion in infancy, but not defensin levels in BM, is associated with the development of allergy in childhood.
Footnotes
Funding
The study was supported by the Helsinki University Central Hospital Research Funds, the Foundation for Pediatric Research and Valio Ltd, Helsinki, Finland. Part-time monthly salaries that MK, and monthly salaries that KK received during 2003 and 2007 from the Clinical Research Institute Helsinki University Central Hospital Ltd, were funded by Valio Ltd.
Conflicts of interest
The authors do not have any potential conflicts of interest to declare.
Acknowledgements
We are grateful to Professor Tari Haahtela from the Skin and Allergy Hospital, University of Helsinki, for his valuable contribution. We warmly thank Elsa Valtonen for her excellent technical assistance.
