Abstract
During Helicobacter pylori (Hp) infections, innate immune cells may be positively or negatively modulated by Hp compounds or by Hp-induced cytokines. We have shown previously that the natural cytotoxic activity of PBMC was lower in Hp-infected [Hp(+)] than Hp-uninfected individuals [Hp(−)]. Here, we asked whether the Hp-modulated cytotoxic amplitude is associated with changes in the number of NK cells, their activation or intracellular cytokine expression. Flow cytometry immunophenotyping of PBMC was performed with regard to the surface receptors CD3, CD56 and CD25, and intracellular cytokine expression of IL-2, IFN-γ and IL-10 after in vitro stimulation with Hp glycine acid extract (GE), Hp LPS or standard Escherichia coli LPS. Hp GE-driven enhancement of lymphocyte cytotoxic activity was associated with the expansion of CD3−CD56+CD25+ NK cells and the up-regulation of IFN-γ and/or IL-2 synthesis, up to the higher level in Hp(−) than in Hp(+), while Hp LPS-mediated decrease in lymphocyte cytotoxicity was accompanied by the lack of CD3−CD56+CD25+ NK propagation, the inhibition of pro-inflammatory cytokine expression and intense expansion of IL-10–producing NK cells. Thus, the cytotoxic and cytokine activities of NK cells were dependent on the type of antigenic challenge and the Hp status, that is, NK cells could be modulated positively by Hp GE Ags and negatively by Hp LPS.
Introduction
NK cells are the components of the innate immune system demonstrating direct cytolytic activity towards target cells possessing inappropriate MHC class I Ags or against cells with surface exposure of foreign molecules.1,2 In contrast to classic T-lymphocytes, NK cells lack a CD3 molecule but express, with variable intensity, CD56 and CD16 receptors. 1 According to recent findings via natural cytotoxicity receptors such as Nkp46 or Nkp44, NK cells may recognize bacterial, viral and parasite Ags and thus contribute to the elimination of infected cells.3,4 The receptors for the Fc fragment of immunoglobulins enable NK cells to kill Ab-coated cells via an Ab-dependent cell cytotoxicity mechanism (ADCC). 5 Recent studies have revealed that NK cells also mediate adaptive-like responses and could play an important role in confining and defeating infections and cancer cells before their spread and induction of pathologic manifestation.2,5 When stimulated, NK cells rapidly express cytotoxic activity. In addition, prior to activation, NK cells secrete cytokines such as IL-1, IL-10, IL-12, IL-15 and IL-18. The most prominent cytokines produced by NK cells include TNF-α and IFN-γ. Thus, secondary to triggering of innate immune cells by direct recognition, NK cells can relay and amplify responses by cytokine release, which influences their activity or the activity of other cells in an autocrine or paracrine manner, respectively. 1
NK cells are important players during microbial infections caused by pathogens that induce extensive cell damage, apoptosis or cancerogenic changes. Helicobacter pylori (Hp), a major cause of chronic gastric inflammation, involved in the development of gastric cancer, is a representative of this group of microbes.6,7 Predominance, heterogeneity and distribution of NK cells at different sites within the gastric mucosa reflect a potential, functional role of these cells during Hp infection. 8 CD56+ NK cells consist of two major subpopulations: CD56bright cells, which express higher numbers of CD56, and CD56dim cells characterized by lower CD56 expression. It has been suggested that CD56bright cells possess regulatory rather than cytotoxic properties, demonstrated by the intensive production of IL-10, IFN-γ and TNF-α, whereas CD56dim NK cells possess stronger cytotoxic activity and co-express CD16 receptor involved in the ADCC mechanism. In particular, CD56bright cells constitutively express the IL-2 receptor (IL-2 R, CD25) with enhanced affinity, which makes them more prone to proliferation in response to IL-2 and/or IL-15.9,10 It has also been shown that CD8−CD16−CD56bright NK cells of the gastric mucosa are adapted to respond to microbial infections with cytokine production, and in this way influence the effectiveness of the innate defense. 8
In the course of Hp infections in vivo, the activity of NK cells and other immune cells may be modulated, positively or negatively, by the cellular and soluble compounds of these bacteria or by Hp-driven cytokines produced by the immune cells. For instance, an intense secretion of IFN-γ by NK cells in response to direct interaction with Hp has been shown. 11 In addition, the ability of several Hp Ags to diminish proliferation of peripheral blood T-lymphocytes has been observed in several studies.12–16 Special attention has been paid to the Hp LPS as an immunomodulator with a dual activity. It may either promote chronic infections and the development of gastric pathologies, or diminish the activity of immune cells such as macrophages, NK cells and lymphocytes, thus promoting the Hp persistence. This strategy helps Hp to survive in the colonized gastric mucosa of the stomach and resist eradication for years.16–18
In a previous study, we found that the natural cytotoxic activity of peripheral blood lymphocytes from Hp-infected donors was lower than that observed in Hp-uninfected individuals. It was also postulated that the mechanism of lymphocyte cytotoxicity may depend on both the type of Ag used for stimulation, namely Hp LPS, Hp glycine acid cellular extract (GE) or standard Escherichia coli LPS, and the network of cytokines secreted by immune cells under stimulation. It was demonstrated that Hp GE Ags and E. coli LPS enhanced, whereas Hp LPS down-regulated, lymphocyte cytotoxicity towards target cells. The E. coli LPS-driven cytotoxic activity was accompanied by IFN-γ and IL-2 production, whereas Hp GE-dependent cytotoxicity could have been a result of IL-10 and IFN-γ signaling. The inhibition of lymphocyte cytotoxicity in response to Hp LPS was associated with low amounts of IL-10 and the lack of pro-inflammatory cytokines (IFN-γ and IL-2) in the culture milieu. 18
In this study we asked whether the cytotoxic activity amplitude observed by us in response to Hp GE or Hp LPS and E. coli LPS is consistent with variations in NK cell numbers, their activation measured by IL-2 R-CD25 expression, as well as intracellular accumulation of pro-inflammatory (IL-2, IFN-γ) and anti-inflammatory (IL-10) cytokines, and we examined if those responses depend on the Hp status.
Materials and methods
Twenty healthy volunteers (9 men and 11 women with a mean age of 37 yrs, range 22–61 yrs) were included in the study. Hp status was estimated on the basis of a capsule-based urea breath test [13C]UBT 19 and an in-house ELISA using Hp GE Ags from the reference Hp strain CCUG 17874 (Culture Collection University of Gothenborg, Sweden) for detection of anti-Hp serum immunoglobulin (IgG), as previously described. 20 The research was conducted according to the principles expressed in the Declaration of Helsinki and approved by the local ethics committee. Peripheral blood for cellular and serological study was obtained with written informed consent from all volunteers.
Stimulation of cells and the cytotoxicity assay
PBMC from Hp(+) and Hp(−) individuals were isolated by Histopaque gradient centrifugation (Sigma, St Louis, MO, USA), washed twice, adjusted to a density of 2.5 × 106/ml and distributed into 24-well culture plates (1 ml/well). The Hp LPS from the reference strain of Hp CCUG 17874 or the standard E. coli LPS (serotype O55:B5; Sigma) were added to the selected wells at a final concentration of 25 ng/ml. The Hp LPS was extracted and purified according to a previously described procedure. 21 Briefly, the Hp LPS was prepared by hot phenol–water extraction after pretreatment of the bacterial biomass with pronase. Then, the LPS crude preparation was purified by RNase, DNase and proteinase K treatment, and by ultracentrifugation. The Hp GE Ags were used in a final protein concentration of 5 µg/ml. The GE preparation contained < 0.001 EU/ml of LPS, as shown by the chromogenic Limulus amebocyte lysate test (Lonza, Braine-Alleud, Belgium), whereas the purified Hp LPS was used at a concentration of 0.7 EU/ml. After 24 h incubation (37℃, 5% CO2) non-adherent lymphocytes were collected, washed twice, adjusted to a density of 2 × 107 cells/ml. The viability of leukocytes from each donor was evaluated before and after stimulation, and it was in the range of 98–100%. The HeLa cell line (European Cell Culture Collection, Salisbury, UK), was used as the target in the cytotoxic assay, as described previously. 18 Effector cells, untreated or pre-incubated with Hp GE, Hp LPS or E. coli LPS, were added to the wells containing settled target cells at a ratio 100:1 (adjusted experimentally) and incubated (4 h at 37℃, 5% CO2). Then, the lymphocytes were removed and the remaining cells were washed out. Washing effectiveness was controlled under an inverted microscope. The wells containing HeLa cells alone (without lymphocytes) were used as controls for target cell viability. The cytotoxic activity of the lymphocytes was estimated on the basis of the viable target cells ability to reduce 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) salt using the TACS MTT Cell Proliferation Assay (R&D Systems, Minneapolis, MN, USA), as recommended by the manufacturer. The intensity of MTT reduction was estimated spectrophotometrically at a wavelength of 570 nm.
Granzyme B quantification as a soluble indicator of lymphocyte cytotoxicity
The granzyme B concentration was evaluated in supernatants from mixed target/effector cell cultures (ELISA Granzyme B kit; Diaclone SAS, Besancon, France), as recommended by the manufacturer. There was a positive correlation between the number of viable target cells (MTT reduction assay) and the concentration of granzyme B (ELISA) after the cytotoxic assay.
Cell surface and intracellular staining of NK cells by flow cytometry
The effector lymphocytes obtained from the PBMC fractions as described in the previous section, untreated (incubated in the culture medium) or stimulated with Hp GE, Hp LPS or E. coli LPS, were washed twice and adjusted to a density of 1 × 107 cells/ml. For FACS analysis, prior to staining cells were incubated with 4% autologous serum in FACS buffer (PBS/1% FCS, 30 mins at 4℃). Surface staining of CD markers was performed on ice and in the dark for 30 mins, in the presence of the experimentally-adjusted amount of mAb conjugated with fluorochromes, that is, FITC, phycoerythrin (PE) and peridinin chlorophyll protein (PerCP). The following labeled mAbs were used: anti-CD3-PerCP, anti-CD56-FITC, or anti-CD25-PE (eBioscience, San Diego, CA, USA). For intracellular staining, the cells, after incubation with 4% autologous serum (PBS/1% FCS, 30 mins at 4℃), were washed twice with the FACS buffer and fixed in the dark for 20 mins at room temperature (RT) (21–23℃) by adding 200 µl of 2% (vol/vol) paraformaldehyde in PBS. Cells were then washed twice and permabilized with 0.5% saponin for 30 mins at RT. Again, the cells were washed and stained intracellularly with directly conjugated Abs towards human cytokines in PBS containing 1% BSA and 0.5% saponin. For identifying cytokine-positive lymphocytes staining was performed with anti-IFN-γ-PE, anti-IL-10-PE or anti-IL-2-allophycocyanine (APC) mAb. After staining, all samples were washed twice in PBS and the pellets were suspended in 300 µl PBS. To ensure the proper identification of positive versus negative Ag expression and to exclude non-specific binding, additional internal negative controls were used, that is, unstained cells for the detection of autofluorescence and an isotype control corresponding to the IgG clone used in the analysis (IgG1, IgG2a or IgG2b).
Flow cytometry data acquisition and analysis
The acquisition and immunophenotypic analysis were performed within 1 h on a LSR2 Flow Cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) and the FlowJo software was used for cell analysis. Approximately 80,000–100,000 events were acquired, and lymphocyte gating was set by a forward (FSC) and a side light scatter (SSC). NK cell subpopulations were expressed as a percentage of the total number of gated lymphocytes. For the NK subpopulations analysis, a sequential gating strategy was employed to detect CD3−CD56+-positive cells. Briefly, a gate was set on viable lymphocytes (FSC versus SSC), and the cells were further analyzed versus CD3 on a forward scatter. CD3-negative population was then gated and further analyzed for the simultaneous expression of the CD56 receptor. To evaluate the percentage of CD25-positive cells among NK cell subpopulations, the viable lymphocytes were gated, and the CD3-negative cells were analyzed for the simultaneous expression of CD56 and CD25 molecular markers. In order to calculate the percentage of total cytokine producing lymphocytes, the gate was set on viable lymphocytes (a FSC versus a SSC), and the cells were analyzed on a forward scatter versus IFN-γ-PE, IL-10-PE or IL-2-APC. For the NK cell data analysis, the CD3− cells were analyzed for the co-expression of the CD56 NK cell marker and the intracellular cytokine production: IFN-γ-PE, IL-10-PE and IL-2-APC.
Statistical analysis
For both examined groups, Hp(−) and Hp(+) individuals, the NK cell percentages in total gated lymphocytes were expressed as the mean values ± SD. The differences between groups were tested using Mann–Whitney’s U-test for impaired data preceded by the evaluation of normality and homogeneity of variances. For statistical analysis Statistica 5.5 PL software was used. Results were considered statistically significant when P ≤ 0.05.
Results
The elevation of the lymphocyte cytotoxic activity and the expansion of NK cells and CD25+ NK cells in response to Hp GE or E. coli LPS but not to Hp LPS
We have demonstrated that Hp Ags may modulate positively or negatively the MHC-unrestricted cytotoxic activity of peripheral blood lymphocytes towards tumor epithelial HeLa cells. The cell line was selected for this study to ensure that the cytotoxic effect is a result of the NK cell and not cytotoxic T-lymphocytes (CTL) activity. As CTL use the specific T cell receptor for the recognition of Ags in the context of self-MHC molecules their involvement in the cytotoxicity towards HeLa cells is rather marginal. The untreated PBMCs from Hp(−) and Hp(+) donors contained similar percentages of CD3−CD56+ NK cells (Figure 1A, A’). In the milieu of Hp GE the CD3−CD56+ NK cells expanded significantly in both Hp(+) and Hp(−) PBMC cultures (P = 0.001 and P = 0.009, respectively). Similarly, in response to E. coli LPS, but not to Hp LPS, the number of CD3−CD56+ NK cells from Hp(+) and Hp(−) strongly increased (P = 0.003 and P = 0.03, respectively) (Figure 1A, A’). Further analysis regarding the expression of a CD25 receptor on nonstimulated PBMC showed a higher percentage of CD25+ NK cells in Hp(+) than in Hp(−) donors (P = 0.04) (Figure 1B, B’). In the stimulation assay, the PBMC from Hp(+), as well as Hp(−), donors responded to Hp GE with an intense expansion of CD3−CD56+CD25+ NK cells (P = 0.001 and P = 0.03, respectively). However, the Hp GE-driven propagation of CD3−CD56+CD25+ NK cells was greater than that observed in Hp(+) individuals (P = 0.001Δ) (Figure 1B, B'). The observed expansion of CD3−CD56+CD25+ NK cells in response to Hp GE corresponded to the elevated levels of lymphocyte cytotoxicity, measured by the soluble granzyme B concentration in the PBMC culture supernatants [P = 0.0009 for Hp(+) and P = 0.02 for Hp(−) donors] (Figure 1C, C’). Similarly to the natural cytotoxicity, the Hp GE-driven cytotoxic activity of the lymphocytes from Hp(−) donors was higher than that observed in Hp(+) individuals (P = 0.04 ∅) (Figure 1C, C’).
The comparison of CD3−CD56+ NK cells (A) and CD3−CD56+CD25+ NK cells (B) expansion in response to the Hp GE, Hp LPS or standard E. coli LPS in relation to the cytotoxic activity of previously stimulated (as above) lymphocytes measured by the granzyme B concentration (C) in the mixed cultures of HeLa target cells and lymphocytes from Hp(−) or Hp(+) individuals. The pie charts show the percentages (gray) of CD3−CD56+ and CD3−CD56+CD25+ in the total PBMCs after culture. The statistically significant differences between the Hp(−) and Hp(+) subjects in regard to CD3-CD56+CD25+ propagation are presented: *P = 0.04; ΔP = 0.001; and the cytotoxic activity ØP = 0.04.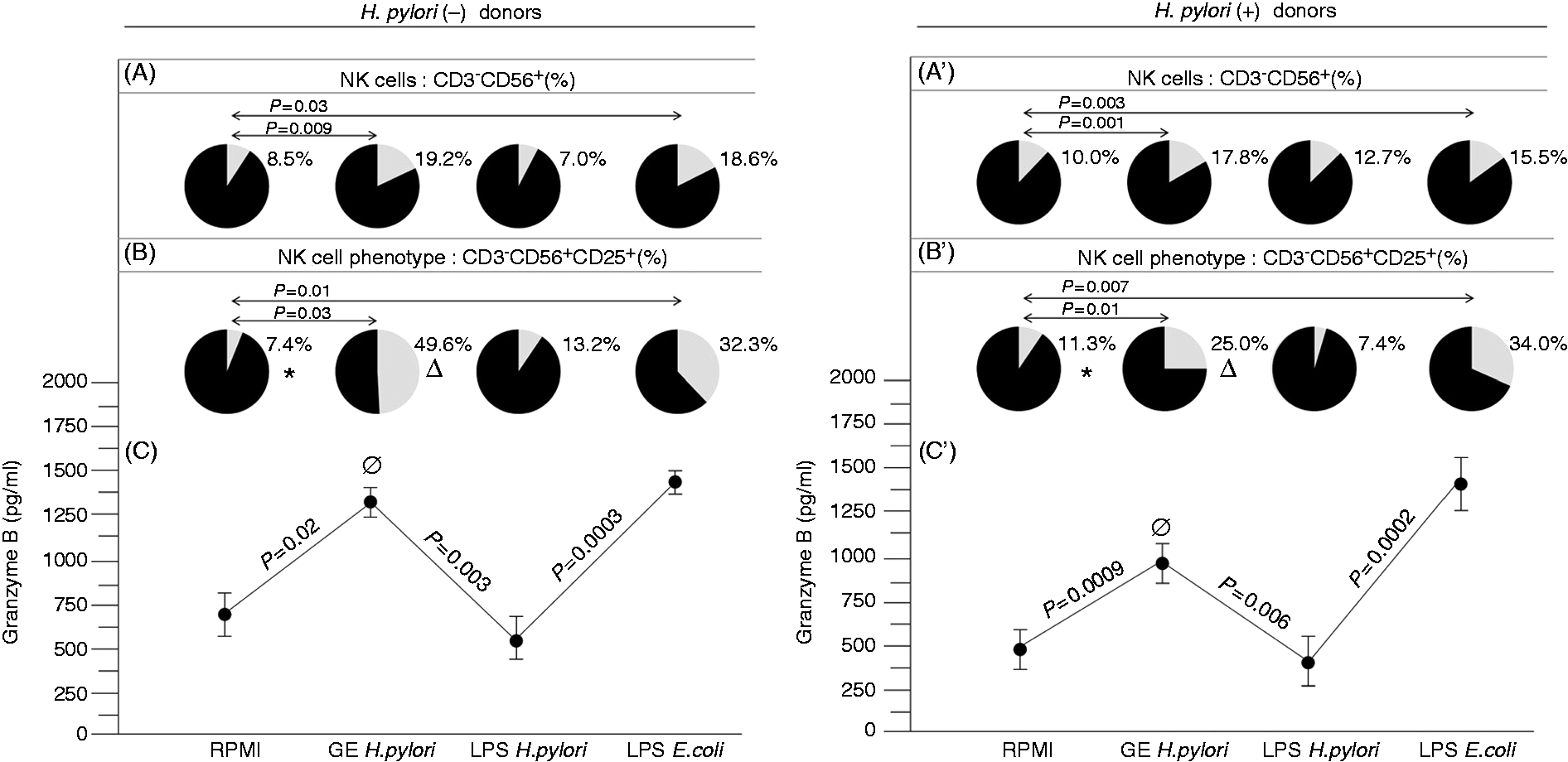
In the PBMC cultures supplemented with E. coli LPS we observed an increase in the number of CD3−CD56+CD25+ NK cells in both Hp(+) and Hp(−) study groups compared with the untreated cultures (i.e. P = 0.007 and P = 0.01, respectively) (Figure 1B, B’). This E. coli LPS-driven expansion correlated with an increased concentration of granzyme B in the culture supernatants after cytotoxic assay conducted with the PBMC from Hp(+) (P = 0.0002), as well as from Hp(−) donors (P = 0.0003) (Figure 1C, C’). In contrast, Hp LPS activity did not lead to the expansion of either Hp(+) or Hp(−) CD3−CD56+CD25+ NK cells as compared with untreated PBMCs. The lack of CD3−CD56+CD25+ NK cell propagation in the milieu of Hp LPS was associated with a significant decrease in the granzyme B concentration compared with the mixed cultures containing untreated lymphocytes of Hp(−) and Hp(+) donors (P = 0.003 and P = 0.006, respectively) (Figure 1C, C’).
Patterns of intracellular cytokine expression in the total lymphocyte fraction, and CD3−CD56+ NK cells depend on the type of Hp Ag used for stimulation, as well as the Hp status
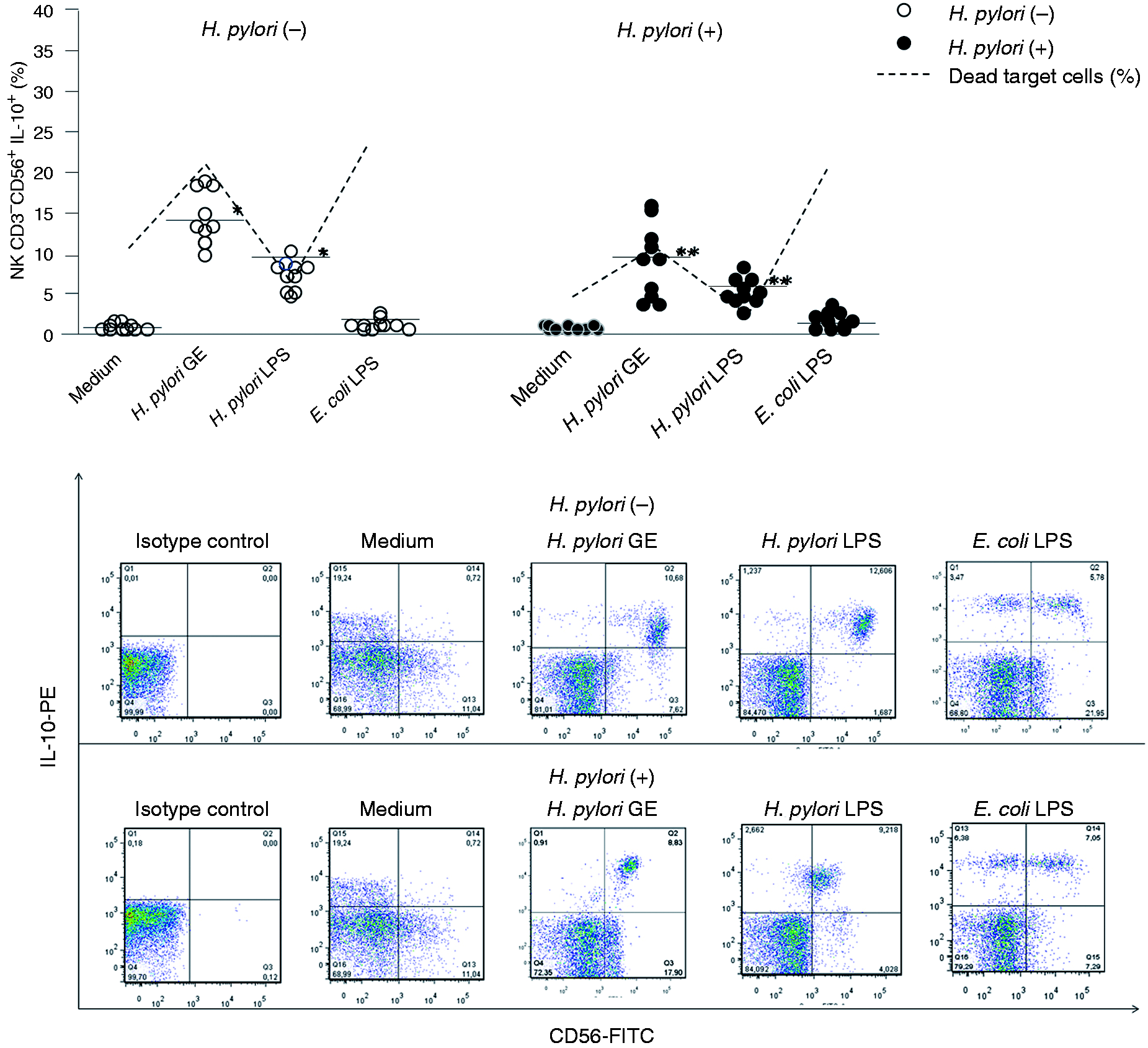
Hp GE-driven enhancement of lymphocyte cytotoxicity towards HeLa cells, expressed as a percentage of dead target cells, was associated with the expansion of the total lymphocyte fraction (Figure 2), as well as NK cell population (Figure 3), with the intracellular expression of IFN-γ in both the Hp(−) and Hp(+) study group. We observed that Hp GE induced a significant increase in both IFN-γ-positive lymphocytes [Hp(−), P = 0.009; Hp(+), P = 0.03] and IFN-γ+ NK cells [Hp(−), P = 0.004; Hp(+), P = 0.04]. However, the increase in IFN-γ+ NK cells of Hp(−) donors was at a higher level (P = 0.03 #) (Figure 3). Supplementation of cell cultures with Hp GE resulted in an intense expansion of total lymphocytes positive for IL-10 in both PBMCs from Hp(−) and Hp(+) donors (P = 0.01 and P = 0.03, respectively) (Figure 4) in comparison to untreated cultures. This phenomenon was also observed in NK cells from both Hp(−) and Hp(+) PBMCs (P = 0.008 and P = 0.04, respectively) (Figure 5).
The percentages of IFN-γ+ lymphocytes in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars show the statistically significant differences in comparison to unstimulated cultures of PBMC from Hp(−)* and Hp(+)** individuals. The representative dot plots show changes in the percentages of IFN-γ positive lymphocytes. The percentages of IFN-γ+-NK cells in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars show the statistically significant differences in comparison to unstimulated PBMC cultures from Hp(−)* and Hp(+)** individuals. The statistically significant differences between the Hp(−) and Hp(+) patients are also marked (#P = 0.03) The representative dot plots show changes in the percentages of IFN-γ positive lymphocytes. The percentages of IL-10+ lymphocytes in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars show the statistically significant differences in comparison to unstimulated PBMC cultures from Hp(−)* and Hp(+)** individuals. The representative dot plots show changes in the percentages of IL-10+-positive lymphocytes. The percentages of IL-10+-NK cells in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars show the statistically significant differences in comparison to unstimulated PBMC cultures from Hp(−)* and Hp(+)** individuals. The representative dot plots show changes in the percentages of IL-10+-positive lymphocytes.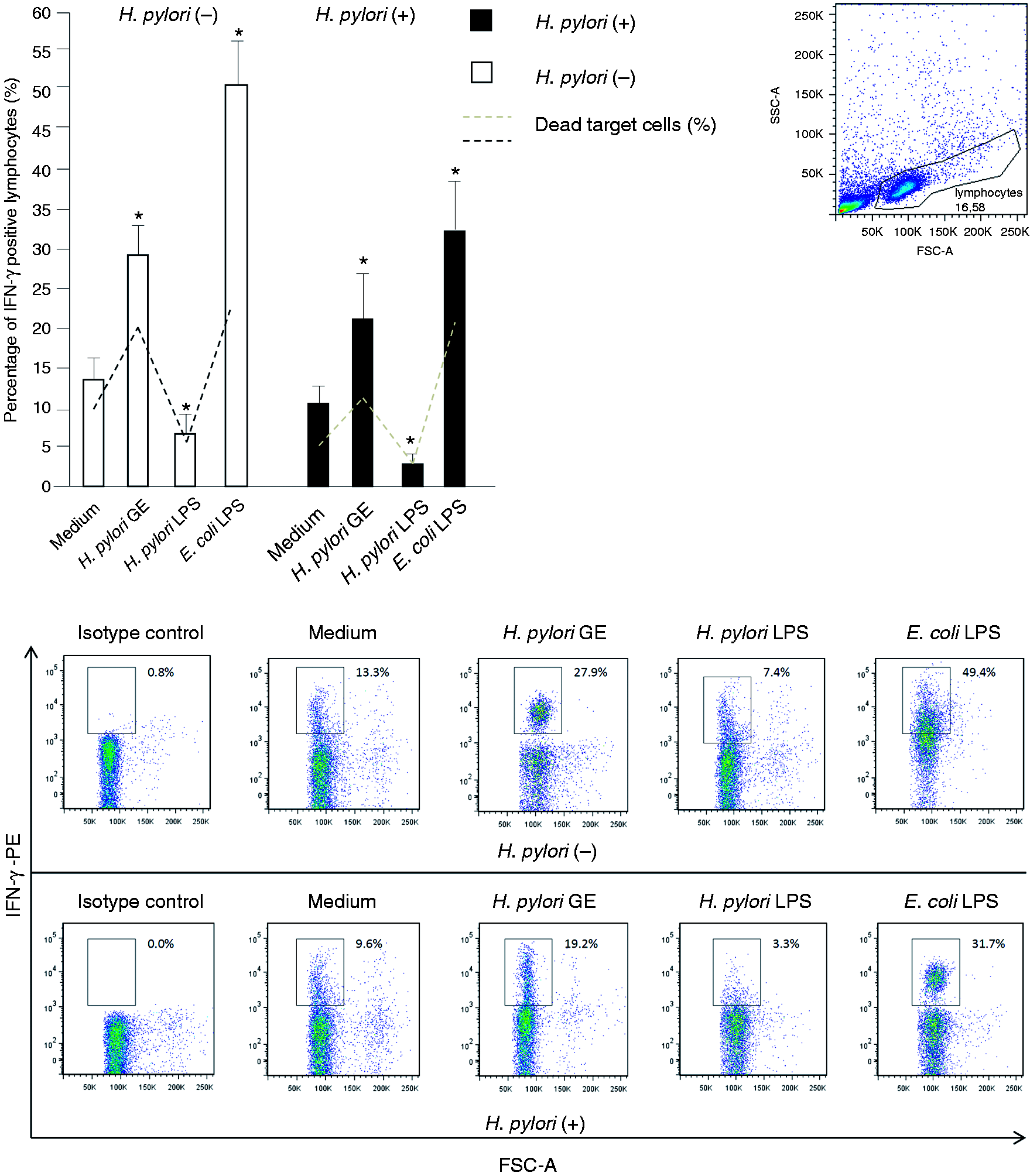
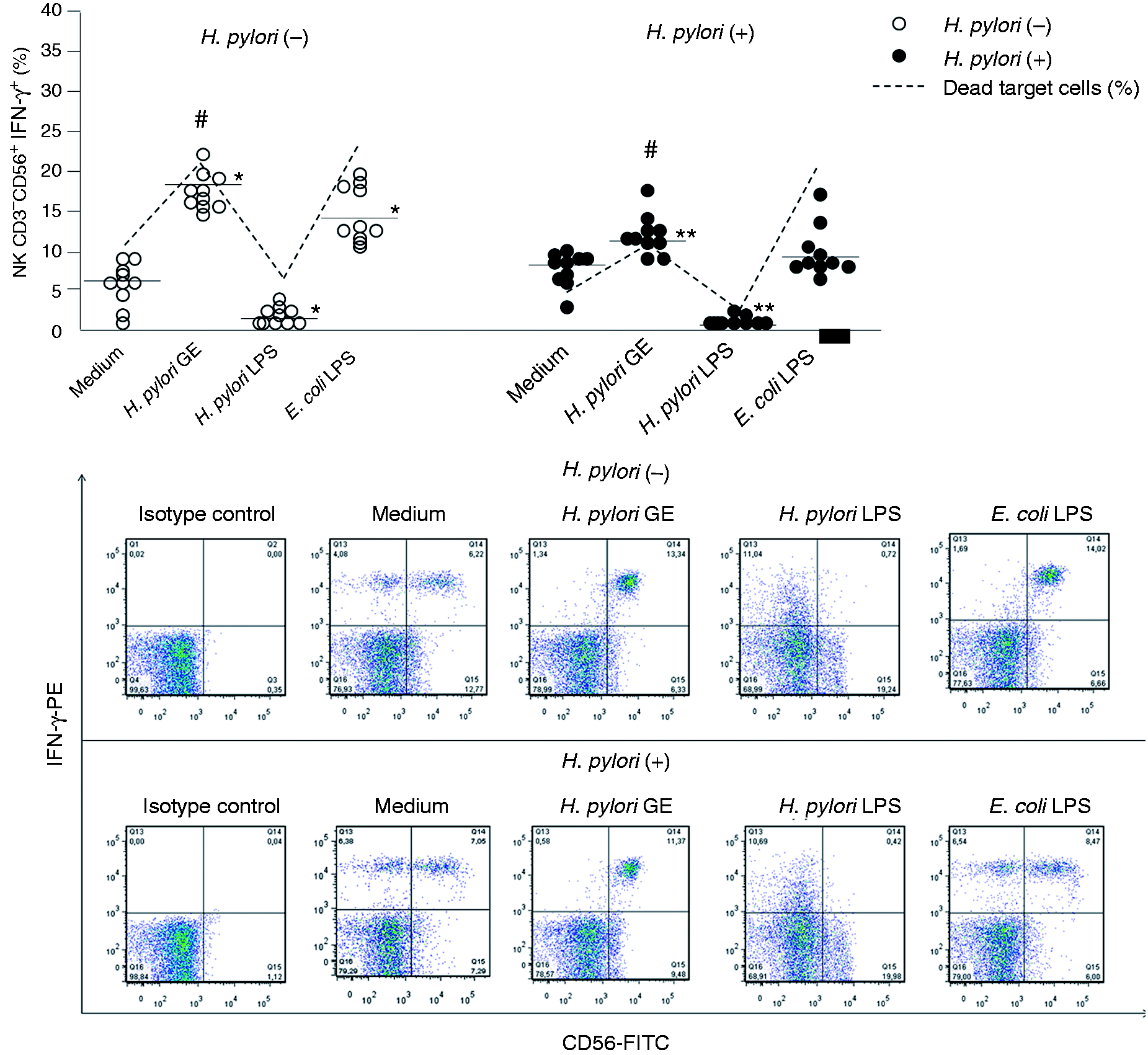
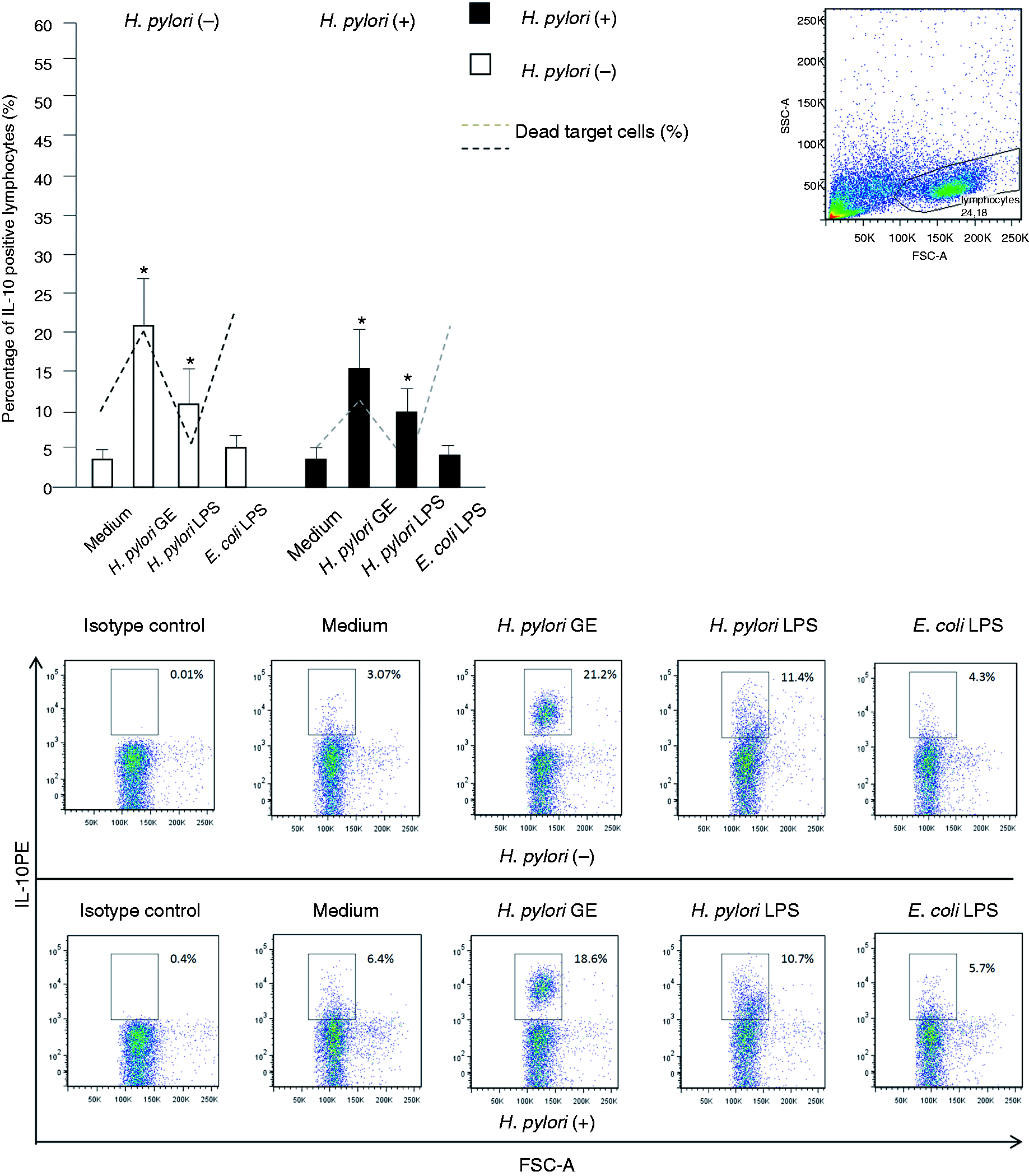
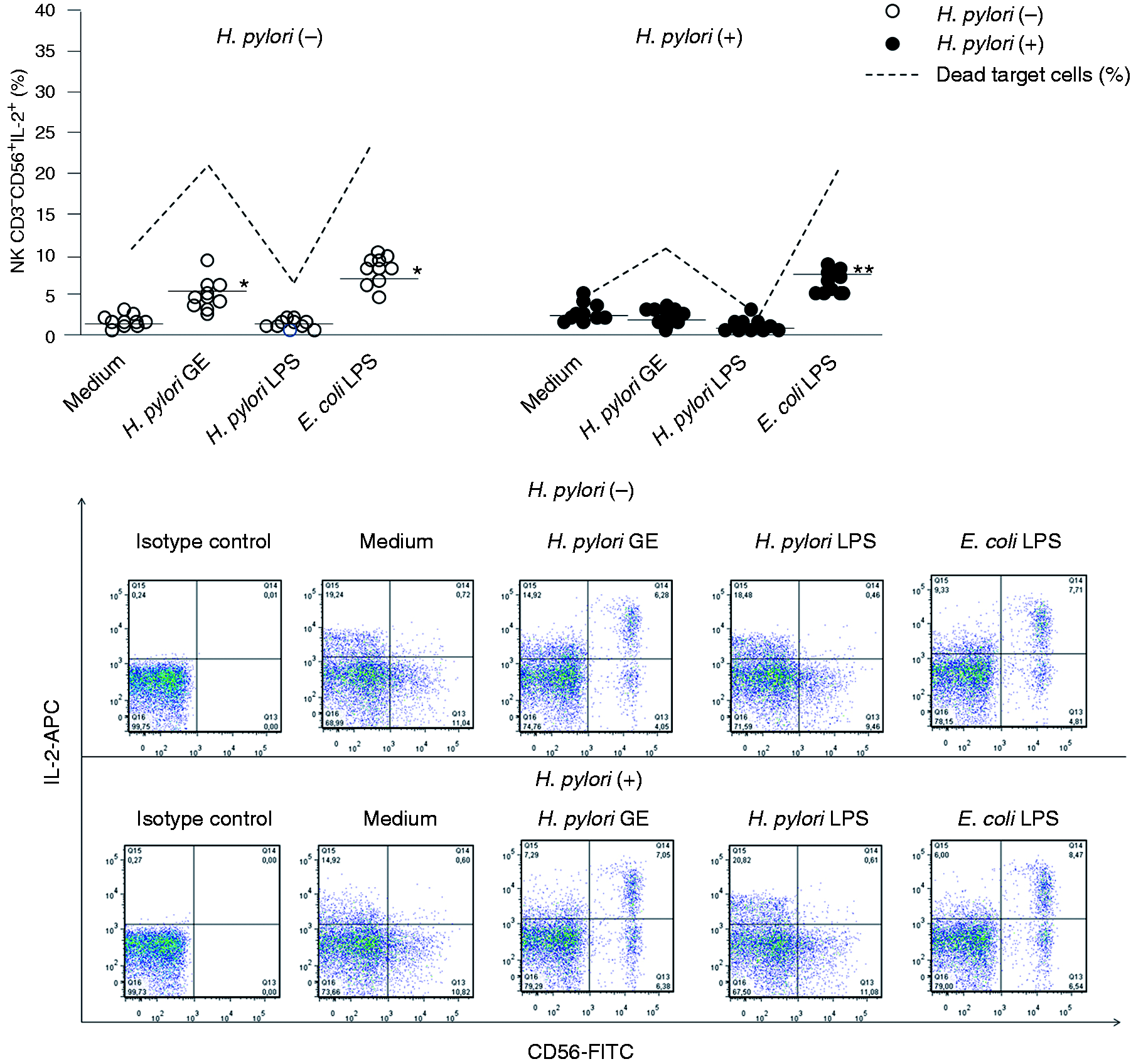
In the Hp(−) PBMC cultures exclusively, the Hp GE-driven increase in the cytotoxic potential of lymphocytes was associated with a significant increase in IL-2+ NK cells (see Figure 7). The lymphocyte cytotoxicity was also significantly enhanced in response to E. coli, but not to Hp LPS (Figure 1C, C’). Stronger lymphocyte cytotoxicity in the milieu of E. coli LPS was related to the expansion of the total lymphocyte population with the intracellular expression of IFN-γ in the Hp(+) and Hp(−) group (P = 0.003 and P = 0.001, respectively) (Figure 2), and exclusively in Hp(−) individuals, with the propagation of IFN-γ+ NK cells (Figure 3). The E. coli LPS stimulation resulted in the expansion of IL-2+ lymphocytes, in both Hp(−) and Hp(+) cell cultures (P = 0.007 and P = 0.004, respectively) (Figure 6), and in the propagation of CD3−CD56+ NK cells positive for IL-2 in Hp(−) (P = 0.04 and in Hp(+), P = 0.03 cell cultures) (Figure 7). However, the control E. coli LPS did not induce an increase in the total number of IL-10+ lymphocytes (Figure 4) or IL-10+ NK cells (Figure 5). By comparison, the Hp LPS-mediated lack of elevation in lymphocyte cytotoxic activity was accompanied by a significant expansion of lymphocytes (Figure 4), including NK cells (Figure 5), with intracellular IL-10 expression, in both study groups. In contrast, the magnitude of the total lymphocyte population (Figure 2) and NK cells (Figure 3) positive for intracellular IFN-γ was diminished in these cell cultures. The treatment of PBMC with Hp LPS did not induce the expansion of either lymphocytes (Figure 6) or NK cells (Figure 7) with the cytosolic localization of IL-2 in comparison to the untreated cultures.
The percentages of IL-2+ lymphocytes in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars shows the statistically significant differences in comparison to unstimulated PBMC cultures from Hp(−)* and Hp(+)** individuals. The representative dot plots show changes in the percentages of IL-2+-positive lymphocytes. The percentages of IL-2+ NK cells in response to the Hp GE, Hp LPS or standard E. coli LPS versus the cytotoxic activity of lymphocytes treated with Hp GE, Hp LPS or standard E. coli LPS. The marked bars shows the statistically significant differences in comparison to unstimulated PBMC cultures from Hp(−)* and Hp(+)** individuals. The representative dot plots show changes in the percentages of IL-2+ positive lymphocytes.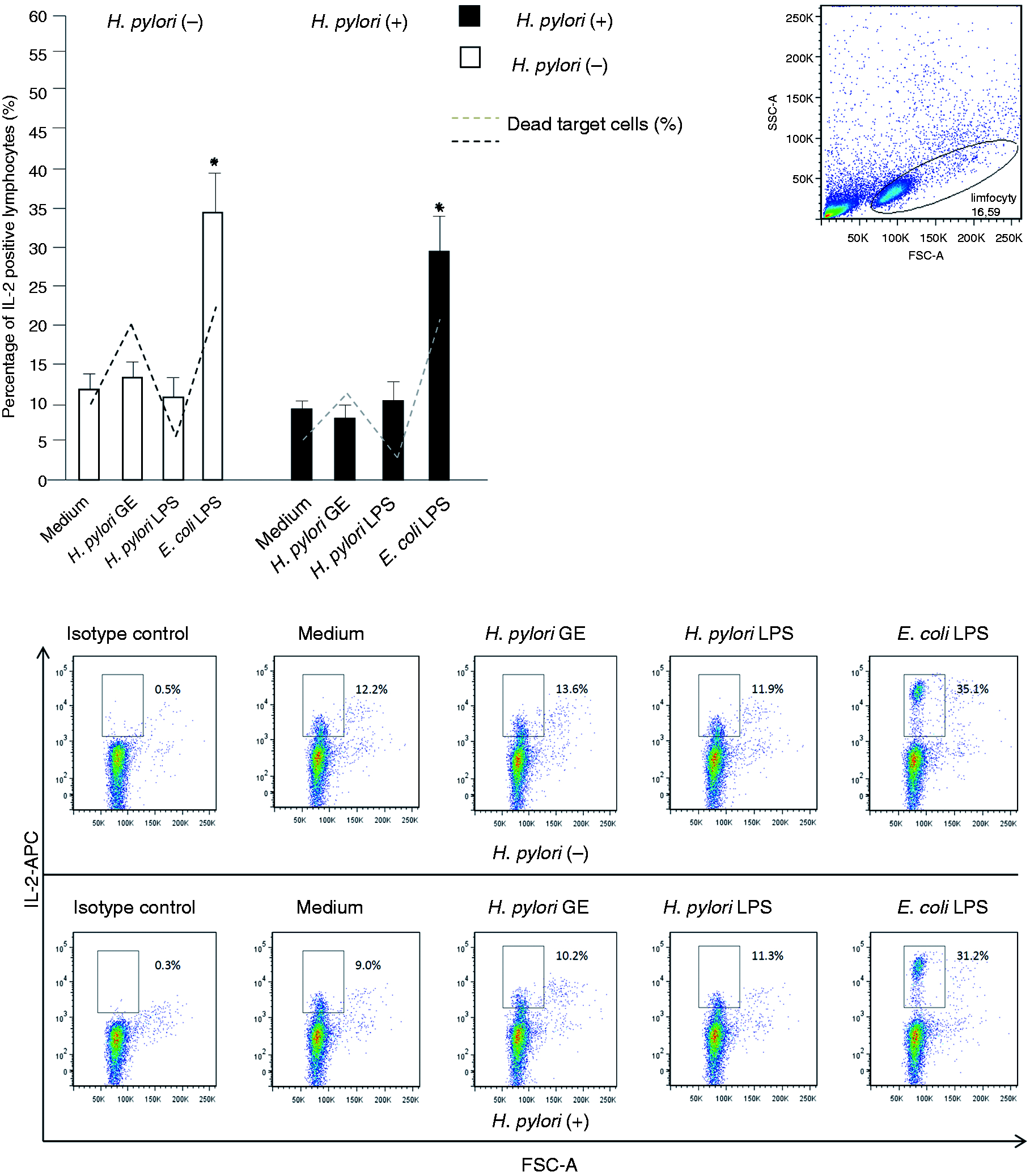
Discussion
Hp express a diverse array of virulence factors that may promote the evasion of the immune cells, especially owing to the ability to negatively modulate both innate and adaptive immune responses.12–17,22 The efficiency of non-specific immune mechanisms may limit the development of Hp infection to the first stage and prevent the development of bacteria-driven pathologies. NK cells, with their cytotoxic potential, play a central role among the non-specific cellular mechanisms of immune components. They reduce a risk of malignant transformation and kill cells with cytoplasmic or surface expression of viral and bacterial pathogens. NK cells also influence the responsiveness of other immunocompetent cells by providing them with cytokine signals.1,2,5 In this study, we hypothesized that NK cell cytotoxic and secretory functions could be modulated positively or negatively by Hp Ags in the inflammatory milieu. If so, NK cells might support either the elimination or the persistence of these bacteria and actively influence the course of infection. Numerous clinical and basic research studies have been performed in order to define what makes Hp a pathogen and why so many individuals remain asymptomatic despite infection. It is possible that Hp-infected individuals, without dyspeptic symptoms, develop a very limited acute inflammatory response to Hp infection and, owing to this, bacteria are not eliminated. In this study, we focused on the cytotoxic potential of lymphocytes from Hp-infected or uninfected donors, and asked whether Hp Ags, that is, GE or LPS, can modulate the lymphocyte cytotoxic activity, the propagation of NK cells and the synthesis of selected cytokines by enhancing or diminishing their functions.1,9,10
We have shown previously that lymphocytes from Hp-uninfected individuals were characterized by stronger natural cytotoxic activity than that observed in Hp-infected patients. This conclusion was based on the lower percentage of dead target HeLa cells in the mixed effector/target cell cultures containing lymphocytes from Hp-uninfected than lymphocytes from Hp-infected donors. We also found that the Ags of Hp GE and standard E. coli LPS positively modulated the cytotoxic activity of lymphocytes from both study groups, whereas Hp LPS affected it negatively. 18 This shows that Ags used in our studies posses universal properties that are independent of Hp status. However, the amplitude of the observed effects could have been related to previous in vivo exposure of the immune cells to Hp Ags or different susceptibility of the cells from Hp(+) and Hp(−) donors. Thus, the reason for the lower natural cytotoxicity of Hp(+) than Hp(−) lymphocytes may be due to the differences in the number of lymphocytes engaged in the cytotoxicity process. The cytotoxic cells may also vary in regard to their maturity, the degree of activation and regulatory potential. Therefore, we compared the percentages of total NK cells from Hp(+) and Hp(−) donors, and their propagation in the in vitro cultures containing Hp GE, Hp LPS or standard E. coli LPS. The immunophenotypic analysis showed no differences between Hp(+) and Hp(−) donors in the total number of NK cells. However, the NK cells of both study groups expanded significantly in response to Hp GE and E. coli LPS, but not in the presence of Hp LPS. The increased number of CD3−CD56+ NK cells responding to Hp GE and E. coli LPS was in line with the enhancement of NK cell cytotoxicity in the milieu of these compounds, thus confirming the responsiveness of the cells with the NK cell phenotype.
In trying to find the explanation for differences in natural cytotoxic activity between Hp(+) and Hp(−) NK cells, and the reason for increased cytotoxic activity of NK cells in response to Hp GE and E. coli LPS, we analyzed the number of NK cells that expressed IL-2 R (CD25)—a marker of cell activation. We observed a higher percentage of CD25+ NK cells in the peripheral blood of Hp(+) than in the blood of Hp(−) individuals. It is possible that CD25+ NK cells from Hp(+) individuals expanded in vivo in response to Hp Ags. 11 However, in in vitro cultures the natural cytotoxic activity of Hp(+) lymphocytes was lower than that observed in Hp(−) individuals. Our previous study showed the domination of the CD56bright NK phenotype in the peripheral blood of Hp(+).23 Interestingly, CD56bright NK cells constitutively express the CD25 receptor. This study revealed that the mean fluorescence index for CD56 expression on NK cells from Hp(+) individuals was greater (892 ± 32) than that on NK cells from Hp(−) (313 ± 32) patients, which suggested that the dominating population of CD3−CD56+ NK cells from the Hp(+) group expressed CD56bright phenotype. The cytototoxic activity of CD56bright cells is rather low compared with the activity of CD56dim NK cells. It has been suggested that CD56bright NK cells can represent a population not mature enough to act as cytotoxic cells, but ready to express regulatory functions mediated by cytokine secretion. If so, it cannot be ruled out that the differences in cytotoxic activity of NK cells from Hp(+) versus Hp(−) donors, observed by us, may be due to differences in their maturity. However, NK cells from Hp(+) donors can be strongly influenced in vivo by the regulatory IL-10, which has been found to be elevated in the sera of Hp(+) but not Hp(−) individuals 23 and in the gastric biopsies of Hp(+) individuals. 24 Thus, it is possible that CD56bright NK cells dominating in Hp(+) donors were better producers of IL-10 by themselves. O’Keeffe et al. 8 have suggested that, in the course of Hp infection, the CD56bright NK cells may deeply influence the effectiveness of the innate immune defense by delivering cytokines to the site of inflammation and releasing them into the circulation. However, NK cells by themselves could also act as targets for cytokines. For instance, IL-10 may down-regulate the NK cell cytotoxicity in an autocrine manner or act in a paracrine way, by inducing NK cells or other immune cells to produce various cytokines. 25
The activation of the immune cells related to the enhanced expression of the IL-2 receptor could be of great importance for the elevation of lymphocyte cytotoxic activity. In this study, we showed that CD25+ NK cells from both Hp(+) and Hp(−) donors expanded significantly in the milieu of Hp GE compared with the control unstimulated lymphocytes, although the expansion of NK cells from Hp(−) donors was more significant. The propagation of CD25+ NK cells in response to Hp GE was related to an increased lymphocyte cytotoxic activity, which was consistent with the principle of ‘higher cytotoxic activity of Hp(−) than Hp(+) lymphocytes’, suggesting that the cells from Hp(−) donors express stronger cytotoxic potential, probably owing to the regulatory function of IL-2. Our results indicate that the cytotoxic activity of lymphocytes from Hp(+) donors was lower than that observed in Hp(−) participants. This could be owing to the domination of CD56bright lymphocytes with low cytotoxic properties. However, it was shown that Hp(+) donors possess a higher percentage of CD25+ NK cells than Hp(−) patients. However, in the milieu of Hp GE the CD25+ NK cells from Hp(−) patients propagated to the higher numbers than those from Hp(+) individuals. These results do not contradict each other. As all NK cells possess CD25 receptors it cannot be excluded that in Hp(−) donors cytotoxic NK cells of CD25+CD56dim phenotype are more expansive. This NK phenotype expresses a rather cytotoxic than a regulatory function. In comparison, in Hp(+) donors the CD25+CD56bright cells, regulatory rather than cytotoxic, propagated to a higher level. Moreover, the cytotoxic activity of lymphocytes of Hp(+) donors could be modified in vivo by Hp Agsor cytokines. If so, different populations of NK cells from Hp(+) and Hp(−) donors could be propagated in in vitro cell cultures.
There was no expansion of CD25+ NK cells, either from Hp(+) or Hp(−) donors, in response to Hp LPS, which was in line with the lack of cytotoxicity enhancement in the same milieu. However, a significant propagation of CD25+ NK cells appeared after stimulation of lymphocytes with E. coli LPS, and it was consistent with the enhancement of lymphocyte cytotoxicity in response to this LPS. These results indicate that the NK cells’ cytotoxic potential in response to Hp GE and E. coli LPS, but not Hp LPS, depends on their activation level related to CD25 expression.
Considering the role of the cytokine network in the development of the cytotoxic function of NK cells, we estimated the total lymphocyte fraction and, specifically, the population of NK cells with an intracellular expression of IFN-γ, IL-2 and IL-10 responding to Hp GE, Hp LPS and E. coli LPS. It was shown that cytotoxic activity of lymphocytes, in response to Hp GE Ags, was associated with the expansion of IFN-γ producing NK cells, which remained in balance with, or dominated, the IL-10+ NK cells. The inhibition of cytotoxic activity in the milieu of Hp LPS was accompanied by a percentage of IFN-γ+ NK cells and the intense propagation of NK cells producing IL-10, negatively regulating their cytotoxic activity. In comparison, the cellular cytotoxicity of NK cells in the milieu of E. coli LPS was a result of the NK cells producing IFN-γ and IL-2, but not the IL-10, which generated a ‘positive signal’, not balanced with IL-10-driven ‘negative regulation’. Our results indicate that the cytotoxic potential of NK cells in response to Hp GE depended on the ability of the cells of both Hp(−) and Hp(+) donors to produce IFN-γ, whereas diminished cytotoxic responsiveness of NK cells in the milieu of Hp LPS was probably a consequence of the domination of IL-10-producing NK cells. Previously, it was shown that contact of lymphocytes with Hp augmented NK cell activity followed by IFN-γ production.11,26 It is interesting that, exclusively in Hp(−) PBMC cultures, the Hp GE-driven increase in the cytotoxic potential was associated with a significant expansion of IL-2+ NK cells. These results may point to a regulatory function of NK cells by releasing IL-2, which is decreased in Hp(+) patients and may regulate the cytotoxicity of NK cells. Previously, it was shown that IL-2 synergizes with IL-12 to induce lymphokine-activated cytotoxicity and perforin, as well as granzyme gene expression in fresh human NK cells. 27 In this study we also showed that Hp LPS and E. coli LPS possesses adverse potentials to modulate NK cells. Recently, it has been suggested that dynamic regulation of signaling by mannose and fucose-expressing pathogens might lead to the intense suppression of pro-inflammatory responses. 28 It has been also shown that Hp LPS, through fucose-containing glycans, interacts with the C-type lectin receptor on dendritic cells (DCs).29,30 Gringhuis et al. 28 have proved that LewisX (LeX) determinants of Hp LPS, with antigenic specificity related to fucose moieties, enhanced IL-10 production but down-regulated IL-12 and IL-6 expression in DCs, and that this modulation occurred via the TLR4 signaling. In this study, the Hp LPS of the LeXY type, with fucose-containing antigenic determinants, stimulated the propagation of IL-10+ NK cells with diminished cytotoxic activity. In addition, other unique structural characteristics of Hp LPS such as the presence of long (>14 atoms of carbon) fatty acids, but few short (4–5 atoms of carbon) fatty acids, and lower charge of lipid A, could affect the Hp LPS binding to pathogen recognition receptors, such as TLR4, which, according to recent findings, may affect or even inhibit LPS-mediated cell signaling pathways.31,32
The results obtained in this study showed that NK cells could be engaged in the immune response during the course of Hp infection. Interestingly, NK cells with rather low cytotoxic activity dominated in the blood of Hp(+) donors. We also showed that response of NK cells to Hp probably depended on the type of antigenic challenge. It could be modulated positively by Hp GE (through IFN-γ and IL-2) or negatively by Hp LPS (through IL-10). It is possible that in Hp-infected individuals the Ags with negative immunomodulating potential such as LPS,13,16–18 cytotoxine associated gene A antigen (CagA) 12 or vacuolating toxin A (Vac A), 14 can suppress the elimination of Hp from the gastric mucosa and facilitate the development of chronic infections.
Footnotes
Funding
This work was supported by
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
