Abstract
Protease-activated receptor-2 (PAR2) is abundantly expressed in the pulmonary compartment. House dust mite (HDM) is a common cause of allergic asthma and contains multiple PAR2 agonistic proteases. The aim of this study was to determine the role of PAR2 in HDM-induced allergic lung inflammation. For this, the extent of allergic lung inflammation was studied in wild type (Wt) and PAR2 knockout (KO) mice after repeated airway exposure to HDM. HDM exposure of Wt mice resulted in a profound influx of eosinophils in bronchoalveolar lavage fluid (BALF) and accumulation of eosinophils in lung tissue, which both were strongly reduced in PAR2 KO mice. PAR2 KO mice demonstrated attenuated lung pathology and protein leak in the bronchoalveolar space, accompanied by lower BALF levels of the anaphylatoxins C3a and C5a. This study reveals, for the first time, an important role for PAR2 in allergic lung inflammation induced by the clinically relevant allergens contained in HDM.
Introduction
Asthma is associated with chronic inflammation of the airways commonly triggered by allergens and characterized by infiltration of eosinophils in the lungs, mucus hypersecretion and airway remodeling. 1 House dust mite (HDM) allergens are an important cause for allergic lung inflammation in asthma patients. 2 The prevalence for HDM IgE Abs varies between 40 and 85% in different asthma populations.2,3 HDM allergens can induce lung inflammation by distinct protease-dependent and protease-independent mechanisms. 4 The allergenic capacity of HDM resides in the mites themselves and in their fecal pellets; 5 in this respect LPS is an important player that can be detected routinely in HDM extracts. 6
Protease-activated receptors (PARs) are 7-transmembrane G-protein coupled receptors that carry their own ligand hidden in the extracellular amino terminus. 7 Upon proteolytic cleavage by a receptor-specific protease the tethered ligand becomes exposed, triggering downstream activation. PARs are therefore considered as cellular sensors for extracellular proteases. 8 To date, four PARs have been described (1–4). PAR2 is abundantly expressed in the pulmonary compartment and previous research has implicated this receptor in airway responses to allergens.9,10 In the model of ovalbumin-induced lung inflammation PAR2 knockout (KO) mice developed less airway inflammation than wild type (Wt) mice,11–13 whereas mice with transgenic overexpression of PAR2 exhibited an exaggerated response. 11 In addition, PAR2 activation resulted in allergic sensitization to simultaneously inhaled allergens 13 and to enhanced allergen-induced airway inflammation in sensitized mice. 14 Of note, however, ovalbumin has little, if any, relevance for human asthma and more recent investigations have used allergens implicated in human disease.15–17 Moreover, whereas ovalbumin does not express endogenous protease activity, three important human allergen sources contain PAR2-activating proteases: cockroach, 15 the fungus species Alternaria alternata 18 and HDM. 19 Indeed, at least four HDM allergens from the common European HDM Dermatophagoides pteronyssinus (Der p) are known to function as PAR2 activating proteases: Der p 1, 19 Der p 3, Der p 6 and Der p 9. 20 The concept that allergens with protease activity can induce PAR2-dependent inflammation in the lungs was recently supported by studies using cockroach extract. 16
Considering the relevance of HDM allergens for human asthma and their documented capacity to activate PAR2,3,5,19,20 we here sought to study the role of PAR2 in HDM-induced allergic lung inflammation. For this we compared responses of PAR2 KO and WT mice in our recently established model of HDM-evoked asthma. 17
Materials and methods
Animals
Sex- and age-matched 8–9-wk-old C57Bl/6 mice were purchased from Charles River (Maastricht, the Netherlands) and used as Wt controls. PAR2 KO C57Bl/6 mice (Jackson Laboratories, Bar Harbor, ME, USA) were bred in the animal facility of the Academic Medical Center. Each experimental group consisted of eight mice. Animals were housed in standardized specific pathogen-free conditions and all experiments were approved by the Animal Care and Use Committee of the University of Amsterdam.
HDM-induced mouse asthma model
HDM allergen whole body extract (Greer Laboratories, Lenoir, NC, USA), derived from the common European HDM species D. pteronyssinus, was used to induce allergic lung inflammation as described. 17 Briefly, mice were inoculated intranasally on d 0, 1 and 2 with 25 µg HDM (sensitization phase) and on d 14, 15, 18 and 19 with 6.25 µg HDM (challenge phase). Controls received isotonic sterile saline intranasally on each occasion. Inoculum volume was 20 µl for every HDM and saline exposure and inoculation procedures were performed during isoflurane inhalation anesthesia. The experiment was ended at d 21 by euthanizing the mice and the subsequent collection and processing of samples: in one experiment bronchoalveolar lavage fluid (BALF) and citrated blood was collected; in a separate experiment one lung was obtained for pathology and one lung for homogenization using procedures as described in de Boer et al. 17 and in the online supplementary material.
Immunohistochemistry
After fixation in 10% formalin and paraffin-embedding we stained 4-µm-thick sections with hematoxylin-eosin (H&E) for gross pathology analysis and with periodic acid-Schif (PAS)-D for detection of mucus. A pathologist blinded for treatment group quantified H&E and PAS-D staining. Additionally, we digitally quantified eosinophils in lung tissue stained by an Ab against major basic protein (MBP; kindly provided by Dr Nancy Lee and Professor James Lee, Mayo Clinic Arizona, Scottsdale, AZ, USA). 17 Imaging analysis is further explained in the online supplementary material.
Assays
Immuno-assays are described in the online supplementary material.
Statistical analysis
Values are expressed as mean ± SE. Differences between groups were tested by Mann–Whitney U-test. GraphPad Prism version 5.0 (GraphPad Software, San Diego, CA, USA) was used for all analyses. Values of P < 0.05 were considered statistically significant.
Results
PAR2 KO mice have a decreased cell influx in BALF after HDM administration due to reduced eosinophil recruitment
Instillation of HDM elicited cell influx into BALF of both Wt (Figure 1A, P < 0.001 versus saline) and PAR2 KO mice (Figure 1A, P < 0.001 versus saline). The impact of HDM exposure on cell influx in BALF was significantly smaller in PAR2 KO mice compared with Wt mice (Figure 1A, P < 0.01). This difference was predominantly caused by a reduction of 80% in eosinophil numbers in PAR2 KO mice (Figure 1B, P < 0.01 versus Wt HDM). Additionally, differences between Wt and HDM-challenged PAR2 KO mice were observed in the absolute numbers of neutrophils (Figure 1C, P < 0.01) and lymphocytes (Figure 1D, P < 0.01). Although HDM caused a modest rise in the numbers of alveolar macrophages in BALF of both Wt and HDM-challenged PAR2 KO mice (Figure 1E, P < 0.001 versus saline), no significant differences were seen between the two mouse strains. Together, these data show PAR2 deficiency is associated with a reduced cell influx into BALF after HDM exposure, which is predominantly caused by a reduced eosinophil recruitment.
PAR2 KO mice demonstrate reduced cell influx in BALF after HDM exposure due to attenuated eosinophil recruitment. Total cell count (A), eosinophils (B), neutrophils (C), lymphocytes (D) and alveolar macrophages (E). Data are means ± SE (105 cells/ml) of eight mice per group. **P < 0.01, ***P < 0.001.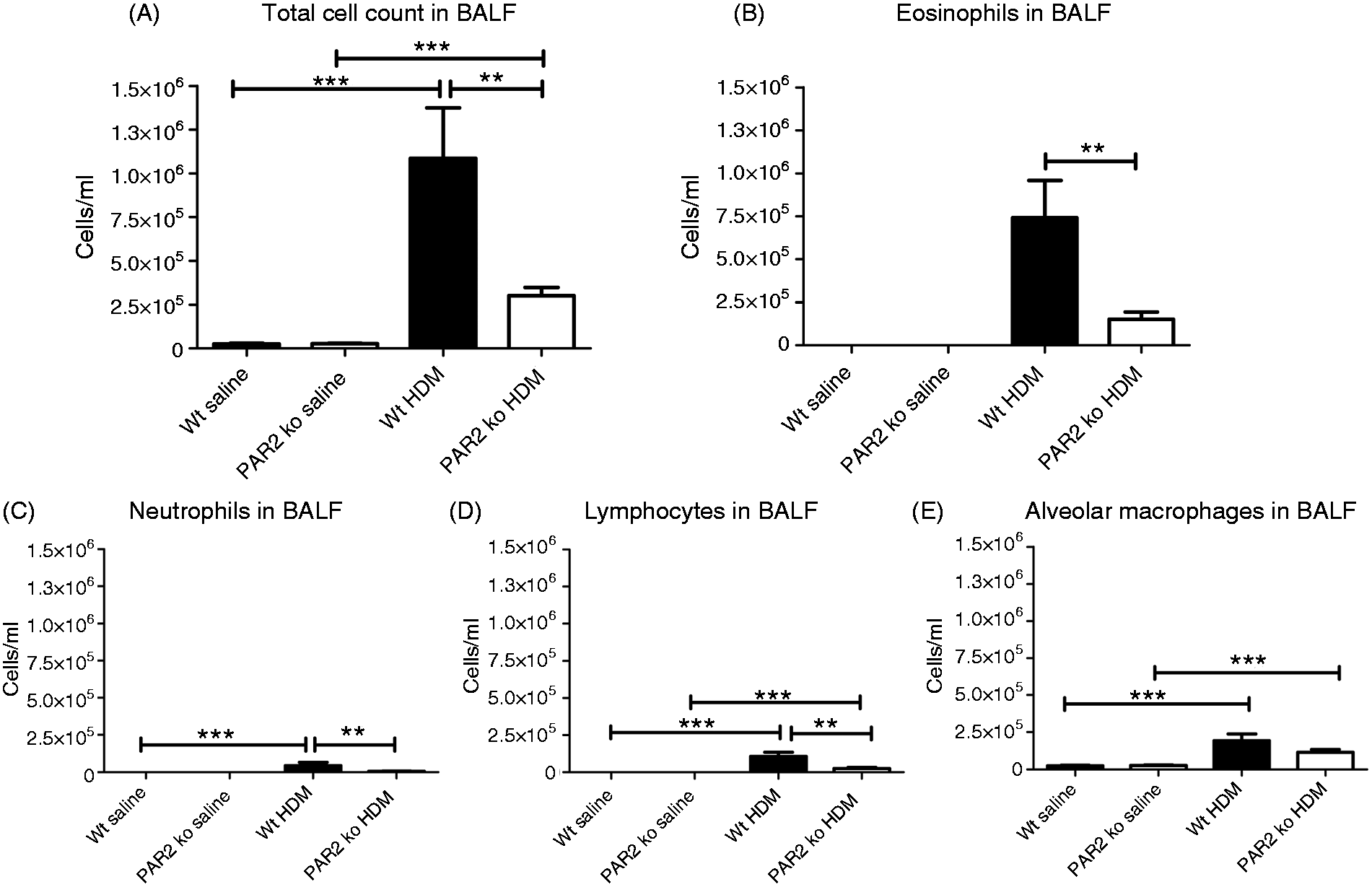
PAR2 KO mice show diminished eosinophil accumulation in lung tissue after HDM exposure
The impact of PAR2 on HDM-induced eosinophil influx was further investigated in lung tissue by immunostainings with an eosinophil-specific anti-MBP Ab (Figure 2). HDM caused a significantly increased accumulation of eosinophils in lung tissue in both Wt (Figure 2A, P < 0.001 versus saline) and PAR2 KO mice (Figure 2A, P < 0.05 versus saline). Eosinophil numbers in lung tissue of HDM-challenged PAR2 KO mice were significantly lower than in lungs of HDM-administered Wt mice (P < 0.05).
PAR2 KO mice demonstrate a reduced influx of eosinophils in lung tissue after HDM challenge. (A) Percentage of lung surface stained positive for eosinophils quantified by digitally imaging of MBP staining (see the online supplementary material). Data are means + SE of eight mice per group. *P < 0.05, ***P < 0.001. Representative MBP staining of lung tissue slides of Wt mice exposed to saline (B, F, 100× and 40× magnification, respectively), PAR2 KO mice exposed to saline (C, G, 100× and 40× magnification respectively), Wt mice exposed to HDM (D, H, 100× and 40× magnification respectively) and PAR2 KO mice exposed to HDM (E, I, 100× and 40× magnification, respectively).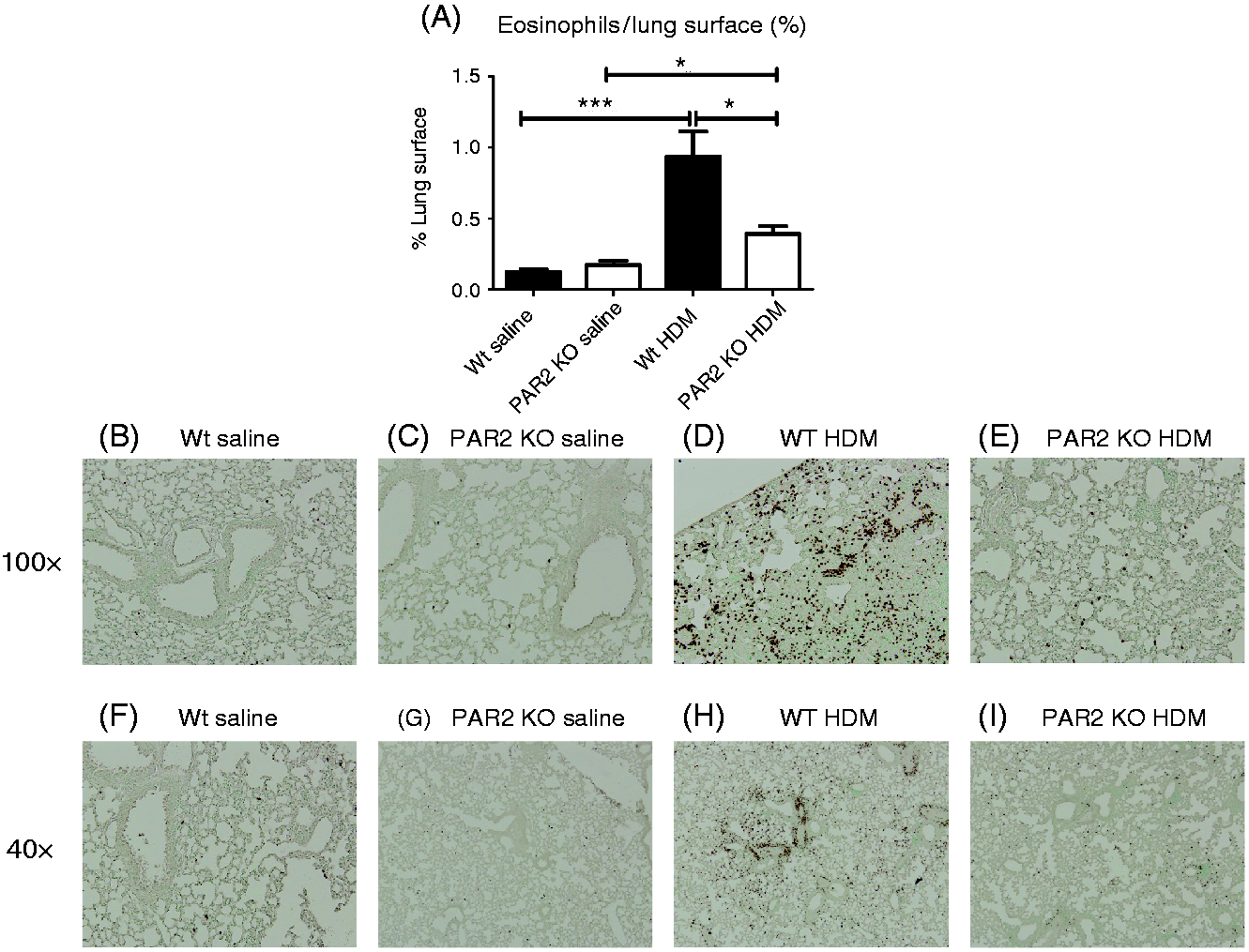
PAR2 KO mice demonstrate attenuated lung pathology after HDM exposure
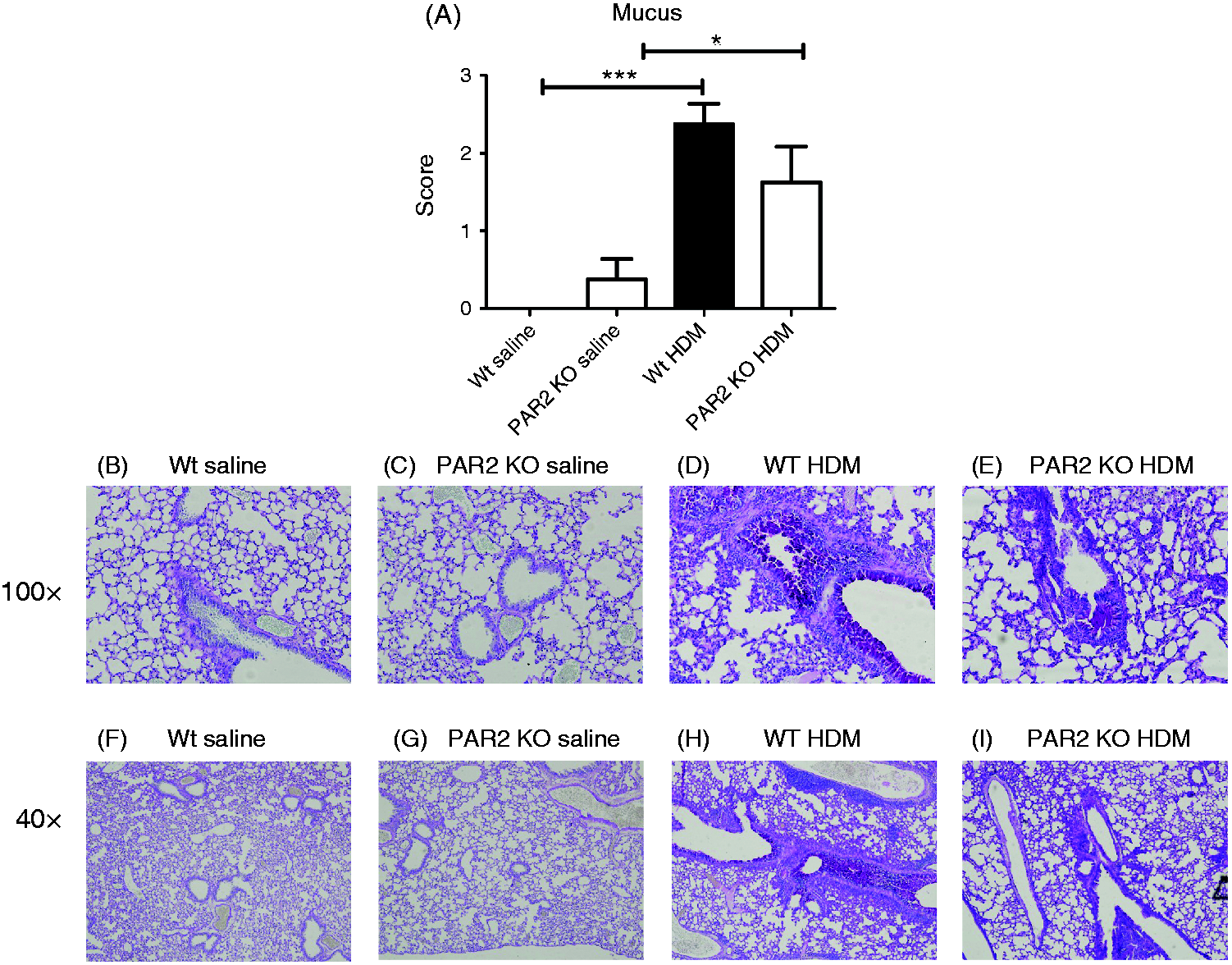
The impact of HDM exposure on lung pathology and the role of PAR2 herein were determined using the semi-quantitative scoring system described in the online supplementary material. Relative to saline controls, HDM elicited marked lung inflammation in both Wt and PAR2 KO mice (Figure 3A, P < 0.001 and P < 0.01 versus saline for Wt and PAR2 KO mice, respectively); the extent of HDM-induced lung inflammation was significantly lower in PAR2 KO mice (Figure 3A, P < 0.05 versus Wt mice). Remarkably, PAR2 KO, but not Wt, mice showed a modest inflammatory response in lung tissue upon repeated saline challenge (P < 0.05 Wt saline versus PAR2 KO saline). Together, these data show that PAR2 contributes to the general inflammatory response in the lungs after HDM challenge. In contrast, PAR2 deficiency did not impact on the extent of mucus formation in the bronchial tree after HDM exposure, as determined by PAS-D staining of lung tissue (Figure 4).
PAR2 KO mice have reduced lung pathology after HDM exposure. (A) Semi-quantitative pathology score (described in the online supplementary material). Data are means + SE of eight mice per group. *P < 0.05, **P < 0.01, ***P < 0.001. Representative H&E-stained lung tissue slides of Wt mice exposed to saline (B, F, 100× and 40× magnification, respectively), PAR2 KO mice exposed to saline (C, G, 100× and 40× magnification, respectively), Wt mice exposed to HDM (D, H, 100× and 40× magnification, respectively) and PAR2 KO mice exposed to HDM (E, I, 100× and 40× magnification, respectively). PAR2 KO mice show an unaltered mucus production in their airways upon HDM challenge. (A) Semi-quantitative mucus score (described in the online supplementary material). Data are means + SE of eight mice per group. *P < 0.05, ***P < 0.001. Representative PAS-D stained lung tissue slides of Wt mice exposed to saline (B, F, 100× and 40× magnification, respectively), PAR2 KO mice exposed to saline (C, G, 100× and 40× magnification, respectively), Wt mice exposed to HDM (D, H, 100× and 40×x magnification, respectively) and PAR2 KO mice exposed to HDM (E, I, 100×and 40× magnification respectively).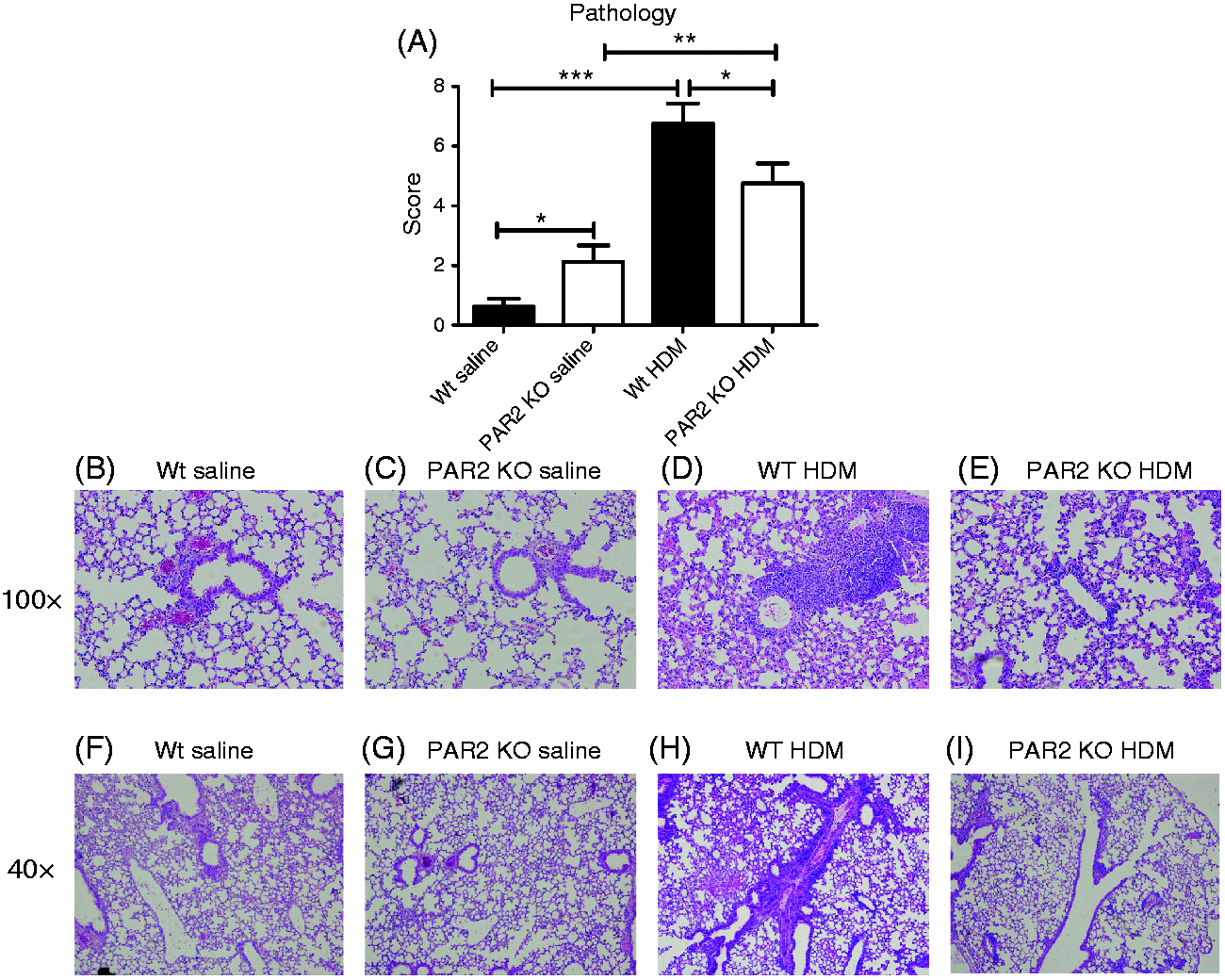
PAR2 KO mice show attenuated IgE and IgG1 responses after HDM challenge
Wt mice had elevated plasma levels of total IgE (Figure 5A, P < 0.01) and anti-HDM IgG1 (Figure 5B, P < 0.01) upon HDM exposure. Plasma IgE remained unchanged in PAR2 KO mice after HDM challenge (P < 0.001 Wt HDM versus PAR2 KO HDM), while plasma anti-HDM IgG1 showed a modest increase (P < 0.05 versus PAR2 KO saline).
PAR2 KO mice have attenuated IgE and IgG1 responses after HDM exposure. Plasma concentrations of IgE (A) and anti-HDM IgG1 (B). Data are means + SE of eight mice per group. *P < 0.05, **P < 0.01, ***P < 0.001. AU: arbitrary units.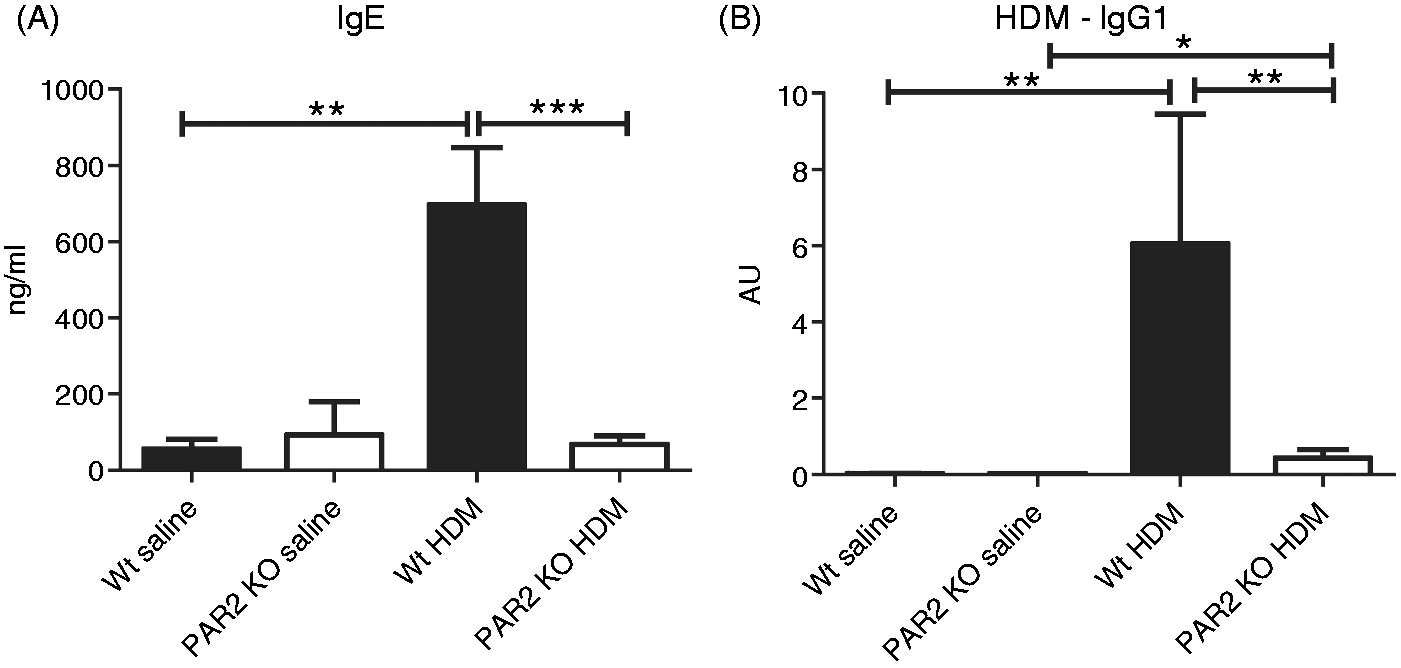
PAR2 KO mice have decreased protein leakage in BALF after HDM challenge
As an end-parameter of intrapulmonary leakage we measured BALF protein levels (Figure 6). Exposure of HDM in the airways caused increased BALF protein levels in Wt and PAR2 KO mice compared with saline (P < 0.001 and P < 0.05, respectively). Interestingly, the increase in BALF protein levels upon HDM challenge was less in PAR2 KO mice (P < 0.01 versus Wt HDM).
PAR2 KO mice have less protein leakage in BALF after HDM challenge. Mean (± SE) levels of protein in BALF (n = 8 per group). *P < 0.05, **P < 0.01, ***P < 0.001. Lower limit of detection (dotted line).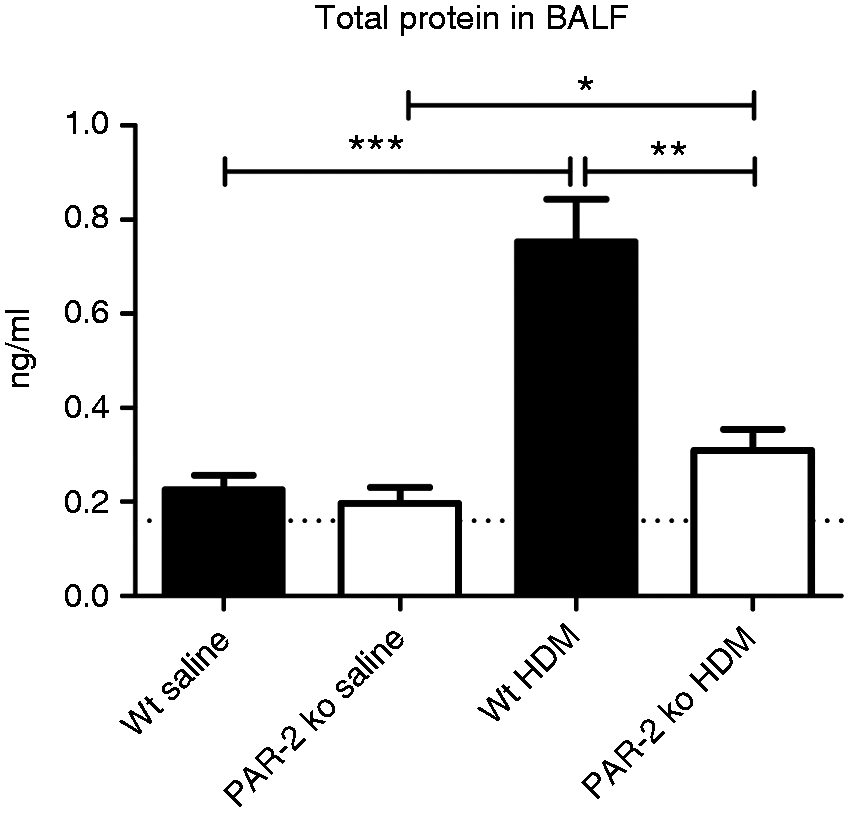
PAR2 KO mice have an attenuated release of complement anaphylatoxins C3a and C5a in BALF after HDM administration
We recently showed that HDM-induced allergic lung inflammation is associated with local activation of the complement system.
17
Therefore, we considered it of interest to determine the influence of PAR2 herein. Administration of HDM caused increased BALF levels of C3a (Figure 7A, P < 0.01 Wt saline versus Wt HDM and P < 0.05 PAR2 KO saline versus PAR2 KO HDM) and C5a (Figure 7B, P < 0.01 Wt saline versus Wt HDM and P < 0.05 PAR2 KO saline versus PAR2 KO HDM). Relative to Wt mice, PAR2 KO mice displayed reduced BALF C3a and C5a concentrations upon HDM exposure (Figure 7, both P < 0.05).
PAR2 KO mice have attenuated complement activation after HDM exposure. Mean (± SE) levels of C3a (A) and C5a (B) in BALF (n = 8 per group). C3a and C5a concentrations remained undetectable in saline control Wt and PAR2 KO mice. *P < 0.05, **P < 0.01.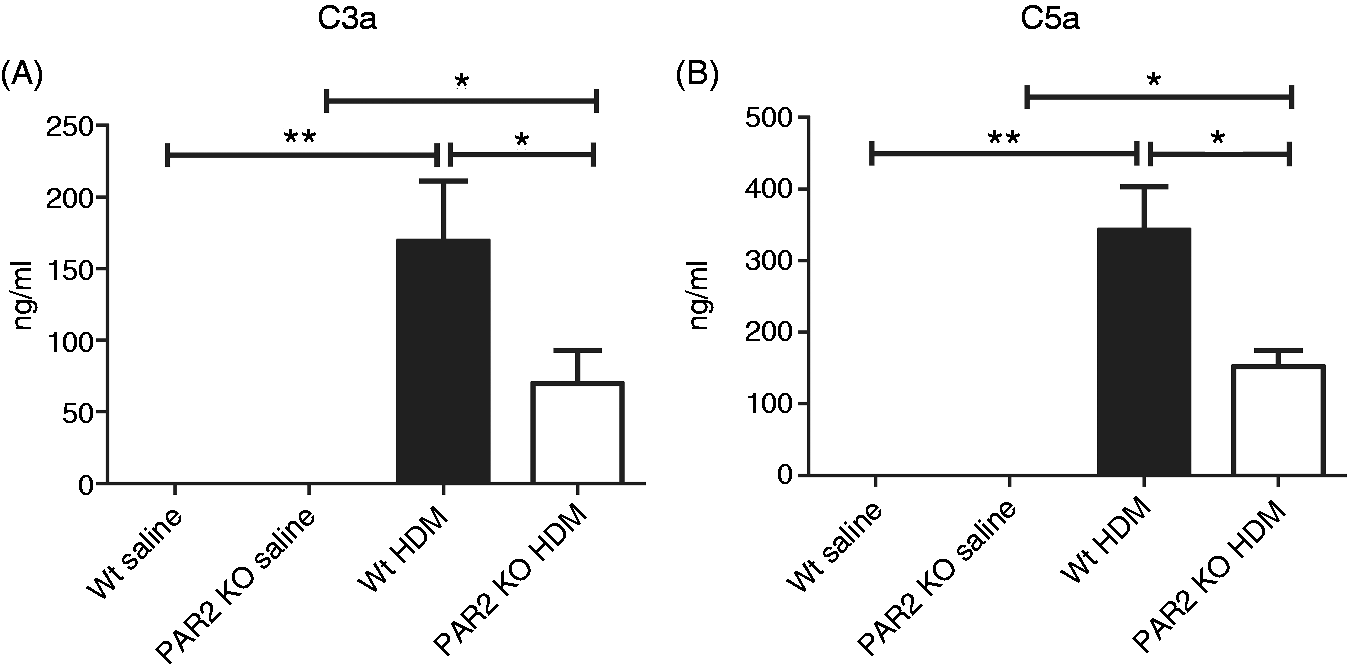
Discussion
Protease activity of allergens is suggested to contribute to allergic lung inflammation by exerting local pro-inflammatory effects. HDM is an important source of allergen for human disease and contains allergens with significant protease activity, including proteases capable of activating PAR2. Here we studied the role of PAR2 in a HDM-induced mouse asthma model using Wt and PAR2 KO mice. We showed that PAR2 KO mice demonstrate strongly attenuated allergic lung inflammation upon airway exposure to HDM, as indicated by a strongly decreased influx of eosinophils in BALF and lung tissue and attenuated lung pathology, accompanied by reduced plasma IgE and HDM-IgG1 responses. These data document a pivotal role for PAR2 in allergic lung inflammation induced by an allergen relevant for human asthma.
PAR2 is widely expressed in the pulmonary compartment of mice and men on resident cell populations (airway epithelium, smooth muscle cells, fibroblasts, endothelium, glands, alveolar macrophages 21 ), as well as cells that migrate to the lungs in allergic inflammation (eosinophils 22 and neutrophils 23 ). While PAR2 expression in alveolar macrophages was shown to be similar in asthma patients compared with healthy volunteers, 21 the expression of PAR2 in human nasal epithelial cells increased after Der p 1 stimulation. 24 Interestingly, PAR2 could be detected mainly intracellularly in human eosinophils and PAR2 agonist treatment with trypsin was suggested to alter eosinophil function by affecting shape change, release of cysteinyl leukotrienes and reactive oxygen species. 22 In our study, PAR2 KO mice showed reduced eosinophil influx in BALF and lung tissue upon HDM administration. Ovalbumin asthma models using PAR2 KO mice have shown similar reductions in eosinophil influx,11,12 further underlining the function of PAR2 in translating (allergen) pro-inflammatory signals from the environment into an inflammatory response. As ovalbumin does not contain PAR2 agonists itself, the protection of PAR2 KO mice in ovalbumin asthma models could be caused by decreased effects of PAR2 activating proteases of endogenous source. For the HDM model, it would be interesting for future experiments to investigate whether loss of PAR2 on airway epithelium decreases eosinophil influx and/or whether loss of PAR2 on eosinophils decreases eosinophil sensitivity to chemotaxis, for example by using cell-specific PAR2 KO mice (using the Cre-lox system) and/or PAR2 bone marrow chimeras.
PAR2 can have a broad variety of effects that may have relevance for asthma pathogenesis: it can activate epithelial cells, 19 smooth muscle cells and fibroblasts 25 to release pro-inflammatory cytokines, trigger pulmonary sensory neuron activation, 26 activate eosinophils 22 and induce fluid hyper secretion by airway submucosal glands. 27 Our current study strongly suggests a role for PAR2 in complement system activation as well. The levels of the anaphylatoxins C3a and C5a are elevated in the airways of asthma patients 24 h after allergen compared with volunteers, 28 and can contribute to bronchoconstriction, vascular permeability, chemotaxis and release of histamine from mast cells. Although C3a and C5a have been shown to be of importance for eosinophil chemotaxis, 29 it remains to be established whether the difference in C3a and C5a BALF levels contributed to the differences in pulmonary eosinophil influx between Wt mice and PAR2 KO mice.
Although PAR2 KO mice displayed less lung pathology after HDM exposure, PAR2 KO mice showed modest lung inflammation upon repeated saline challenge. As Wt mice did not demonstrate such a response, this finding pointed to a possible anti-inflammatory role of PAR2 in the relatively quiescent bronchoalveolar space. Indeed, PAR2 activation can mediate anti-inflammatory effects by triggering the production of cyclooxygenase and prostangladin E2 from airway epithelium, thereby inducing airway relaxation. A recent study reported that the pro-inflammatory effects of PAR2 in the airways are β-arrestin-2 dependent, whereas the protective anti-constrictor functions of PAR2 are not. 30 Possibly, these distinct PAR2-mediated pathways are differentially activated depending on the nature and extent of the inciting stimulus.
Mucus hypersecretion is an important feature of asthma. 1 Stimulation of human tracheal mucosa with PAR2-activating peptide resulted in mucus secretion by a Ca2+ dependent mechanism.31,32 In accordance, PAR2-activating peptide induced glandular secretion by nasal turbinates harvested from allergic rhinitis, an effect that was mimicked by exposure to HDM. 27 Yet, another study, using human bronchial epithelial cell lines, found only a weak effect of PAR2 stimulation on mucin secretion. 33 Our current results, showing unaltered PAS-D staining in PAR2 KO mice after HDM exposure, argue against an important role for PAR2 in mucus hypersecretion in the airways during HDM-induced allergic lung inflammation.
In conclusion, we have shown that PAR2-deficient mice are largely protected from allergic lung inflammation in a HDM extract asthma model. As HDM allergy is very prevalent in asthma patients and HDM effects are partly protease-dependent, this study further reveals PAR2 as an interesting target for anti-inflammatory treatment in allergic conditions.
Footnotes
Funding
J Daan de Boer is supported by the Netherlands Asthma Foundation (project 3.2.08.009). I Stroo is supported by the Landsteiner Foundation for Blood Transfusion Research.
Acknowledgements
We thank Joost Daalhuizen, Marieke ten Brink, Danielle Kruijswijk and Regina de Beer for excellent laboratory assistance. We are indebted to Dr Nancy Lee and Professor James Lee (Mayo Clinic Arizona, Scottsdale, AZ, USA) for generously providing monoclonal Ab against major basic protein. We thank Onno J de Boer for assistance with digital analysis of MBP immunostaining.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
