Abstract
We previously observed that the human bitter taste receptor T2R38 is an important component of upper respiratory innate defense because it detects acyl homoserine lactone (AHL) quorum-sensing molecules secreted by Gram-negative bacteria. T2R38 activation in human sinonasal epithelial cells stimulates calcium and NO signals that increase mucociliary clearance, the major physical respiratory defense against inhaled pathogens. While mice do not have a clear T2R38 ortholog, they do have bitter taste receptors capable of responding to T2R38 agonists, suggesting that T2R-mediated innate immune mechanisms may be conserved in mice. We examined whether AHLs activate calcium and NO signaling in mouse nasal epithelial cells, and utilized pharmacology, as well as cells from knockout mice lacking important components of canonical taste signal transduction pathways, to determine if AHL-stimulated responses require taste signaling molecules. We found that AHLs stimulate calcium-dependent NO production that increases mucociliary clearance and thus likely serves an innate immune role against Gram-negative bacteria. These responses require PLCβ2 and TRPM5 taste signaling components, but not α-gustducin. These data suggest the mouse may be a useful model for further studies of T2R-mediated innate immunity.
Keywords
Introduction
Mucociliary clearance is the primary physical innate defense of both the upper and lower respiratory tract.1–3 Inhaled pathogens, toxins and irritants are trapped by the mucus that overlays the airway epithelium. Coordinated ciliary beating by airway epithelial cells drives the transport of this debris-laden mucus toward the oropharynx, where it is removed by expectoration or swallowing.4–6 In chronic rhinosinusitis (CRS), impairment of mucociliary clearance in the upper respiratory tract can result in recurrent bacterial infections that require prolonged medical therapy, which has a detrimental effect on patient quality of life4,5 and creates an annual aggregated healthcare cost of $6 billion. Because CRS is responsible for approximately one in five antibiotic prescriptions in adults, it has been hypothesized that antibiotic resistance may become a significant problem in CRS;7–9 therefore, new treatment modalities are needed. An attractive therapeutic strategy for CRS patients would be to target endogenous host defense pathways to stimulate mucociliary clearance and/or innate immune mechanisms, such as antimicrobial peptide secretion.
An emerging potential target pathway is that mediated by T2R bitter taste receptors.10–12 There are approximately 25 functional isoforms of human T2Rs, and many of these are also expressed in the ciliated epithelial cells of the human respiratory tract, 13 including the sinonasal cavity. 14 T2Rs in type II taste cells of the tongue protect against the ingestion of harmful compounds, including toxic bacterial and plant products, and at least one T2R in the human airway, described below, detects bacterial products and activates innate immune responses. 14 Ciliated epithelial cells have long been known to serve an important sentinel role in adaptive and innate sinonasal immunity,15,16 particularly through expression of TLRs that recognize PAMPs, such as LPS and flagellin.15,16 We previously demonstrated that the human bitter taste receptor T2R38 similarly contributes to the sentinel function of ciliated epithelial cells. T2R38 is expressed in the cilia of human upper respiratory epithelial cells and detects physiological concentrations of acyl-homoserine lactone (AHL) quorum-sensing molecules secreted by Gram-negative bacteria such as Pseudomonas aeruginosa. 14 Once activated, T2R38-mediated calcium signals stimulate NO synthase (NOS) and production of intracellular NO, which serves two innate defense roles. The first is stimulation of ciliary beating through protein kinase G activation to increase ciliary beat frequency and mucociliary clearance. The second role is direct bacterial killing upon diffusion of the NO into the airway surface liquid. NO and its reactive derivatives, including S-nitrosothiols and peroxynitrites, can damage bacterial DNA, inactivate enzymes containing reactive thiol groups or metal cofactors, and react with membrane lipids causing bacterial permeabilization.17,18
The AHL-induced antibacterial response of human sinonasal epithelial cells is correlated with genetic polymorphisms known to regulate T2R38 functionality. T2R38 is encoded by the TAS2R38 gene, which has two common polymorphisms in Caucasian populations. One polymorphism encodes a functional receptor variant containing a proline (P), alanine (A) and valine (V) at amino acid positions 49, 262 and 296, respectively, while the other polymorphism encodes a nonfunctional receptor variant with A, V and isoleucine (I) at these positions, respectively.19–22 Homozygous PAV/PAV individuals (∼ 20% frequency in Caucasian populations 19 ) are termed ‘supertasters’ for certain T2R38-specific agonists, such as phenylthiocarbamide (PTC) or 6-propyl-2-thiouracil (PROP), while AVI/AVI individuals (∼ 30% frequency in Caucasian populations) are ‘non-tasters’ for these bitter compounds. AVI/PAV heterozygotes have varying intermediate levels of taste for these compounds. Sinonasal epithelial cells derived from PAV/PAV supertaster individuals exhibited enhanced NO production, mucociliary clearance and bacterial killing compared with AVI/PAV and AVI/AVI cells. Furthermore, preliminary clinical data suggest that patients with enhanced T2R38 function (PAV/PAV supertasters) may be less susceptible to Gram-negative sinonasal infection than patients with low or intermediate levels of T2R38 function (AVI/AVI or AVI/PAV, respectively). 14 Additionally, T2R38 supertasters may be less likely to require surgical intervention for CRS. 23 The T2R38 pathway is thus a potential target to promote bacterial clearance and innate immunity in patients with upper respiratory infections, but because there is a large subset of patients that are sub-optimally responsive to T2R38 agonists, it is critical to further define the T2R38-mediated signaling pathway in airway epithelial cells.
The availability of biochemical and molecular biological tools in mice, including knockout (KO) animals, makes the mouse an attractive model organism for dissecting out airway T2R signaling. While mice express T2R bitter taste receptors that can respond to the T2R38 agonists PTC and PROP,24–27 they do not have a clear T2R38 ortholog and the mechanisms reported to underlie PTC and/or PROP avoidance in mice are complex and may not be solely based on taste.28–33 However, we sought to examine whether mice exhibit a sinonasal epithelial innate immune response to PTC and P. aeruginosa AHLs, and determine whether this response is dependent on taste signaling pathway. Our data suggest that important components of the sinonasal response to AHLs are conserved between mice and humans, revealing important mechanistic insights into this response. Also, this study suggests that the mouse is a useful model for future investigation of airway innate immune responses mediated by taste signaling molecules.
Materials and methods
Reagents and solutions
All reagents and solutions used were previously described.
14
Fluo-4, DAF-FM diacetate, thapsigargin, ionomycin, Texas Red dextran (10,000 MM), BAPTA-AM and fluorescent microspheres were from Invitrogen (Grand Island, NY, USA). N-Butyryl-
Mouse septal sinonasal air–liquid interface cultures
All mouse work was done with full approval of the University of Pennsylvania and Philadelphia VA Medical Center Institutional Animal Care and Use Committees (IACUCs). TRPM5 34 and α-gustducin 35 KO mice used were on an otherwise wild type (wt) C57BL/6 background. Air–liquid interface (ALI) cultures were set-up as previously described36,37 from excised nasal septae. Epithelial cells were isolated by collagenase and pronase digestion before culture on Costar 6.5 mm transwell permeable filter supports (Corning Life Sciences, Lowell, MA, USA) with apical side submerged. After 7 d, cells reached confluence and the medium was removed from the apical surface with feeding from the basolateral side. Differentiation and ciliogenesis were observed within 10–14 d after exposure to air, and cultures were used within 4–6 wks.
Bacterial culture
Bacterial culture was performed as previously described.
14
P. aeruginosa strains PAO1 (wt), PAO-JP2 (
Calcium and NO imaging
Calcium and NO imaging were performed as previously described14,36 using the calcium-sensitive fluorophore Fluo-4 and reactive nitrogen species indicator DAF-FM, respectively. The validation of DAF-FM use as a method for measuring NO produced by sinonasal ALI cultures was previously described.14,36 After loading of mouse sinonasal cultures with Fluo-4 AM (10 µM applied to apical side only for 2 h) or DAF-FM diacetate (10 µM applied to apical side only for 45 min), cultures were copiously washed and incubated in the dark at room temperature for 20 min to allow cells to de-esterify loaded dye and recover. Imaging was performed using the 488-nM laser line of an Olympus Fluoview confocal system with an Olympus IX-81 microscope and 10× (0.3 NA UPlanFLN objective; Olympus; Tokyo, Japan). Images were analyzed in Olympus Fluoview software and/or ImageJ as previously described.14,36 No offset or gamma alterations were used. Fluo-4 fluorescence changes were normalized after subtraction of the background fluorescence, which was estimated for each experiment by measuring unloaded ALIs at identical settings. Baseline Fluo-4 fluorescence (Fo) was determined by averaging the first 10 frames of each experiment. The magnitudes of DAF-FM fluorescence changes were used to approximate NO production, and thus care was taken to follow the loading protocol strictly to normalize DAF-FM fluorescence loading.
Imaging of mucociliary transport
Mucociliary transport velocity was measured as previously described 14 using 2-µm polystyrene fluorescent microspheres (0.0025% by mass in 30 µl) that were added to the apical surface of the cultures after copious washing with PBS to remove mucus clumps. Beads were imaged using an inverted Nikon TE2000E epifluorescence microscope (20×, 0.5 NA PlanFluor objective; Nikon, Tokyo, Japan) equipped with a 12-bit QImaging camera and computer running ImageJ and µManager. 41 To qualify for inclusion in the statistical analysis, a streak had to have a visible beginning and ending within the field of view.
Data analysis and statistics
All statistical analyses (Student’s t-test or ANOVA, as indicated) were performed in Excel and/or GraphPad Prism as indicated; P < 0.05 was considered statistically significant. For multiple comparisons, ANOVA with indicated post-test was performed.
Results
To examine sinonasal epithelial cell responses, we utilized ALI cultures derived from mouse nasal septum.36,37 ALI cultures are the state-of-the-art respiratory cell culture model, as they mimic the polarized respiratory epithelium with differentiated ciliated and goblet cells.42,43 We have found that the expression of the bitter taste receptor T2R38 is identically localized to respiratory cilia in human sinonasal ALI cultures and human sinonasal tissue explants, suggesting the receptor-mediated responses are physiologically relevant. Because taste receptors are linked to downstream calcium signaling, we loaded mouse septal ALI cultures with the calcium-sensitive fluorophore Fluo-4 to measure relative changes in cytoplasmic free calcium concentration as previously reported. 36
When mouse epithelial cells were stimulated by application of the bitter tastant PTC to the apical side of the ALI cultures, they exhibited sustained calcium responses with lower magnitude and slower kinetics relative to the purinergic receptor-induced responses stimulated by ATP (Figure 1a). PTC-induced responses were blocked by U73122 (Figure 1b), an inhibitor of PLCβ2, an important component of taste signaling10–12,44–46 at a concentration that did not inhibit the ATP-induced response (Figure 1c). These results appear to be very similar to the PTC-induced T2R38-mediated calcium signaling previously observed in human sinonasal ALI cultures and dissociated human sinonasal ciliated cells.
14
We thus hypothesized that mouse ALI cultures express bitter taste receptors that respond to PTC and potentially also to bacterial AHL molecules.
Mouse nasal septal epithelial cells respond to the bitter tastant PTC with an increase in intracellular calcium that is dependent upon the taste signaling component PLCβ2. (a) Average Fluo-4 trace (mean ± SEM) from mouse nasal septal ALIs (n = 5 cultures) during apical stimulation with 1 mM PTC and subsequent 100 µM ATP. Note the break in left y-axis because of the larger magnitude of the ATP response. (b) Average trace showing responses in the presence of the PLCβ2 inhibitor U73122 (5 µM; 10 min pre-incubation; apical side only; n = 5). (c) Bar graph of peak calcium responses (mean ± SEM from A–B) after 5 min stimulation with PTC (F/Fo = 1.24 ± 0.04 and 1.09 ± 0.03 in the absence and presence of U713122, respectively) and during ATP stimulation (F/Fo = 3.16 ± 0.1 and 2.92 ± 0.2, respectively). Symbols denote significance of indicated paired comparisons via one-way ANOVA with Bonferroni post-test; *P < 0.05. n.s.: not significant.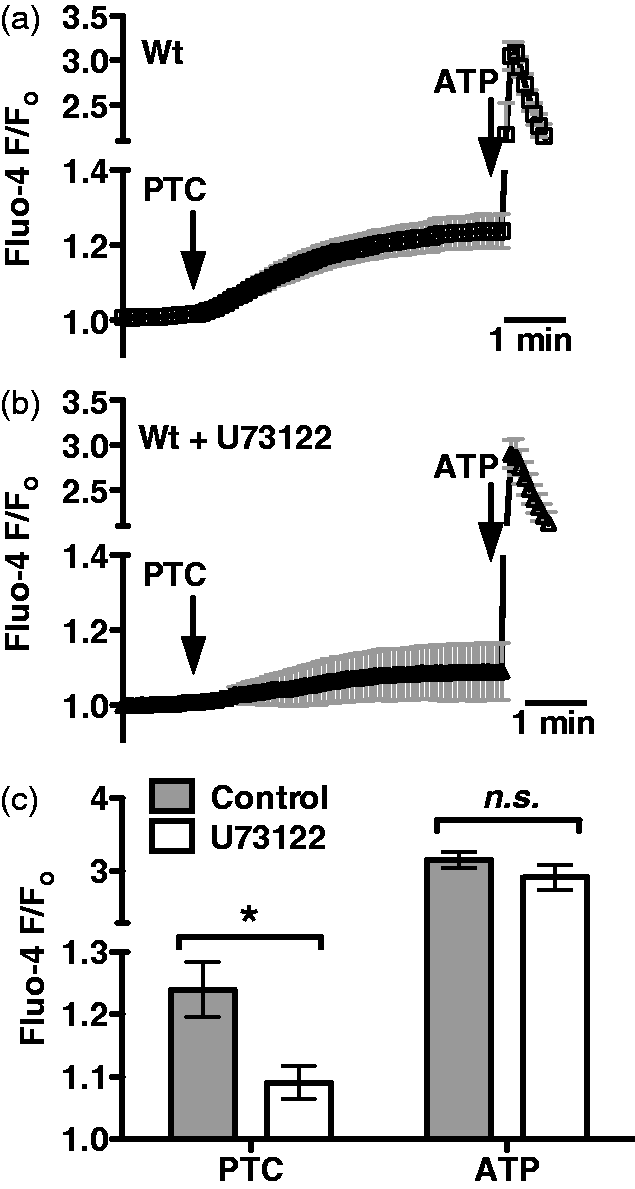
We next examined if mouse sinonasal epithelial cells used components of the classic taste signal transduction pathway to respond to the two most common AHL molecules used by airway pathogen P. aeruginosa, C4HSL and C12HSL. AHL-mediated quorum sensing is a mechanism for bacteria to regulate gene expression in response to the physical constraints of their environment and external signals, including those coming from other individual bacteria. The quorum sensing in Gram-negative bacteria can regulate many processes, including virulence, motility and swarming, and biofilm formation.38,47–49 Mouse ALI cultures stimulated with either C12HSL or C4HSL (100 µM each) exhibited calcium responses that appeared very similar to those elicited by PTC (Figure 2a,b). Interestingly, the magnitude and kinetics of this calcium response were altered significantly in TRPM5 KO mouse cultures (Figure 2c). In taste cells, TRPM5 is not itself calcium permeable, but is activated by initial receptor-dependent intracellular calcium release and serves to allow cation influx and support sustained signaling by depolarizing the cell membrane potential and activating voltage-gated channels.34,46,50–52 From these data, TRPM5 appears to be required for maintaining sustained calcium signaling in response to C4HSL in mouse sinonasal epithelial cells. Interestingly, C4HSL-induced calcium signaling was completely intact in ALI cultures derived from mice lacking the taste G protein subunit α-gustducin (Figure 2d).53–56 However, C4HSL-induced calcium signaling was inhibited by the PLCβ2 inhibitor U73122 (Figure 2e), but not by its inactive analogue U73343 (Figure 2f). These results are summarized in Figure 2g, and suggest that AHLs induce calcium signals in mouse sinonasal epithelial cells that go through two important proteins also used in taste signaling, PLCβ2 and TRPM5. The involvement of PLCβ2 and TRPM5, but not α-gustducin, was initially surprising, so this was confirmed by testing the responses to PTC in wt and α-gustducin KO cultures, which were not significantly different (Figure 2h–j). Wt and α-gustducin KO status of cultures was confirmed by PCR genotyping of animals before isolating and culturing cells, as well as afterwards by genotyping the same cultures used for the experiments shown in Figure 2.
P. aeruginosa AHLs stimulate mouse nasal calcium signaling that also requires components of taste signaling. (a, b) Average traces showing calcium responses in wt cultures stimulated with 100 µM C12HSL (A; n = 3 cultures) or C4HSL (B; n = 5 cultures). (c, d) Traces showing calcium responses to C4HSL in TRPM5 KO (TRPM5-/-; C, n = 7) and α-gustducin KO (α-gustducin-/-; D, n = 3) ALIs. (d, e) Traces showing C4HSL-induced calcium responses in wt cultures treated with the PLCβ2 inhibitor U73122 (D, n = 4) and the inactive U73343 (E, n = 4). (f) Bar graph showing calcium response after 5 min stimulation with C4HSL. Fluo-4 F/Fo values (mean ± SEM) after 5 min stimulation were 1.32 ± 0.07 (C12HSL; wt), 1.25 ± 0.05 (C4HSL; wt), 1.08 ± 0.02 (C4HSL; TRPM5-/-), 1.25 ± 0.07 (C4HSL; gustducin-/-), 1.07 ± 0.05 (C4HSL + U73122; wt) and 1.22 ± 0.05 (C4HSL ± U73343). (h–i) To confirm the lack of role for α-gustducin, cultures from wt (n = 5) and α-gustducin-/- (n = 5) mice were also stimulated with 1 mM PTC. (j) Bar graph showing calcium response after 5 min stimulation with PTC and peak calcium response after stimulation with ATP. Fluo-4 F/Fo values (mean ± SEM) after PTC were 1.26 ± 0.05 (wt) and 1.24 ± 0.04 (α-gustducin-/-). Fluo-4 F/Fo values (mean ± SEM) after ATP were 2.45 ± 0.25 (wt) and 2.68 ± 0.49 (α-gustducin-/-). Symbols denote significance determined by one-way ANOVA with Dunnett’s (g) or Bonferroni (j) post-tests; *P < 0.05. n.s.: not significant.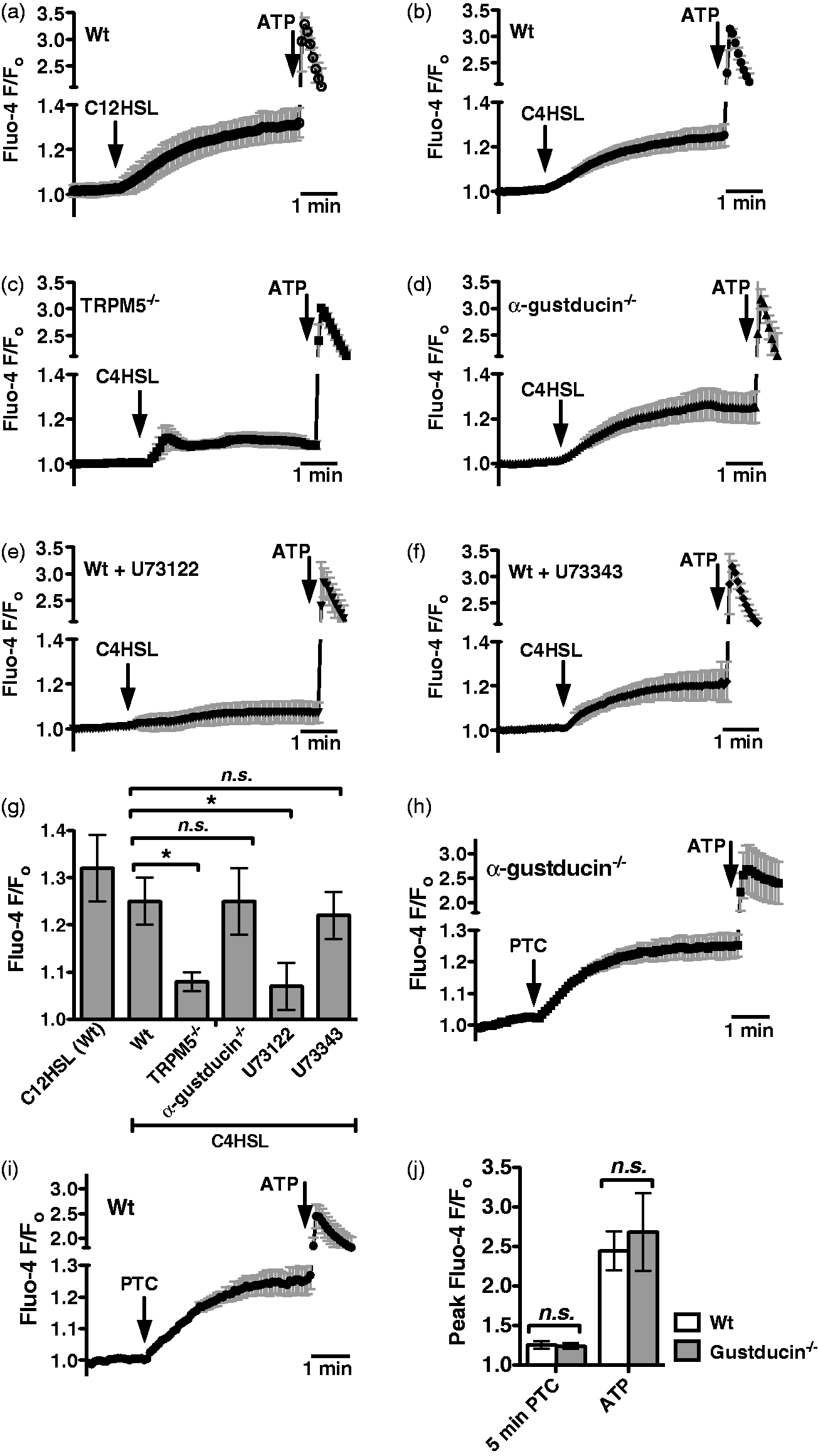
ALI cultures are made up of numerous cell types, including ciliated epithelial cells, and goblet cells.37, 57–59 We previously showed T2R38 expressed in human sinonasal ciliated epithelial cells detects AHLs and activates calcium and NO responses directly in the ciliated cells.
14
However, other studies in mice have shown that AHLs and bitter tastants stimulate calcium responses from dissociated non-ciliated epithelial cells in the nose, termed solitary chemosensory cells (SCCs), that do require α-gustducin in addition to PLCβ2 and TRPM5.27,60,61 We tested whether the responses observed in our cultures originated from ciliated epithelial cells by taking dissociated ciliated epithelial cells from mouse airway (visualized under simultaneous differential interference contrast and confocal fluorescence microscopy), loading them with Fluo-4, and stimulating them with PTC (Figure 3a) or C4HSL (Figure 3b). Ciliated epithelial cells did, indeed, respond to both agonists with similar calcium signals, suggesting that the responses we observed in ALI cultures do, indeed, originate from the ciliated epithelial cells themselves.
Dissociated mouse septal ciliated epithelial cells exhibit calcium responses to PTC and C4HSL. (a, b) Mouse septal ciliated epithelial cells were dissociated as previously described and stimulated with 1 mM PTC (a) or 150 µM C4HSL (b). Three responses from each condition are shown, representative of 5–7 experiments each.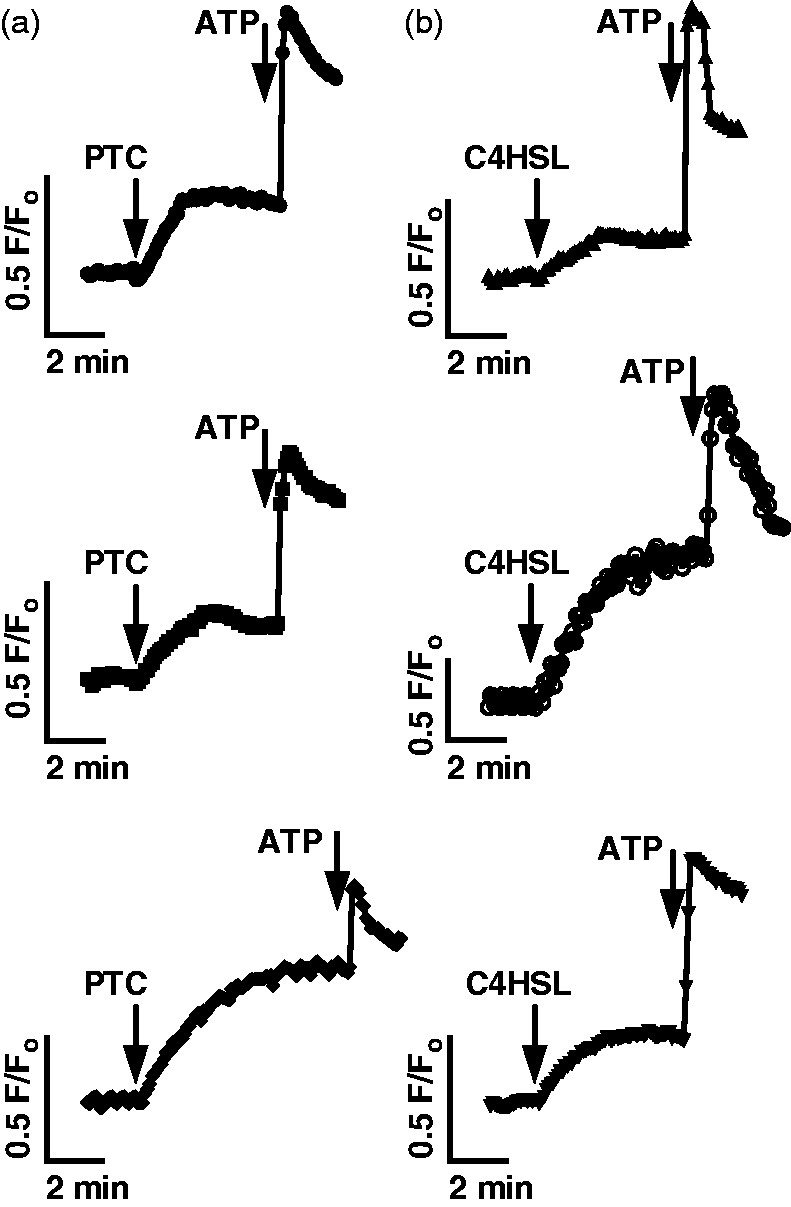
We next examined whether AHL-induced mouse sinonasal calcium signaling results in production of NO, an important signaling component that can regulate two important components of airway innate immunity by increasing ciliary beating and mucociliary clearance,14,62 as well as by directly killing bacteria.14,17,18 NOS isoforms can be activated in response to low-level calcium signaling and calmodulin activation in airway epithelial cells.14,63 Mouse sinonasal ALI cultures were loaded with the fluorophore DAF-FM, which exhibits an increase in fluorescence when it reacts with NO and derived nitrogen radicals.
14
A DAF-FM fluorescence increase in mouse nasal septal ALIs was previously shown to reflect NO production.
36
C4HSL (100 µM) stimulated a robust increase in DAF-FM fluorescence (Figure 4a). The non-specific NO donor SNAP was added at the end of all experiments as a control for dye loading. C4HSL-induced fluorescence increase was inhibited when calcium signals were eliminated by loading cultures with the calcium chelator BAPTA-AM (10 µM for 15 min) and using extracellular media that contained the calcium chelator EGTA (0.5 mM; Figure 4b), despite the fact that SNAP was still able to induce robust fluorescence increases. This result suggests that DAF-FM fluorescence increases in response to C4HSL reflect calcium-stimulated NOS activity, likely activation of endothelial NOS.
63
Like the calcium signals observed in Figure 2, the C4HSL-induced DAF-FM fluorescence increases were inhibited by the PLCβ2 inhibitor U73122 (Figure 4c), but not its inactive analogue U73343 (Figure 4d). The DAF-FM fluorescence increases were also inhibited in cultures derived from TRPM5 KO mice (Figure 4e), as well as wt cultures in the presence of triphenylphosphine oxide (TPPO; 80 µM Figure 4f), a TRPM5 inhibitor.
64
However, the DAF-FM fluorescence increases did not depend on α-gustducin, as they were completely intact in cultures derived from α-gustducin KO mice (Figure 4g). These data are summarized in Figure 4h, and suggest that C4HSL activates NO production in mouse sinonasal epithelial cells and that this response also requires two important components of taste signaling, PLCβ2 and TRPM5.
C4HSL stimulates mouse septal epithelial cell NO production that requires taste signaling components. (a) Average trace (n = 7 cultures) showing DAF-FM fluorescence (increase reflects reactive nitrogen species production) in wt cultures during stimulation with 100 µM C4HSL, as well as the non-specific NO donor SNAP (10 µM). (b–d) Average traces of C4HSL-stimulated fluorescence increases in wt cultures under 0-calcium conditions (BAPTA/EGTA; n = 7, B), in the presence of U73122 (n = 5, C) and U73343 (n = 6, D). (e, f) Average traces of C4HSL-induced fluorescence increases in TRPM5-/- cultures wt cultures in the presence of the TRPM5 inhibitor TPPO (n = 5 each). (g) Traces of DAF-FM fluorescence increase in α-gustducin-/- cultures (n = 7). (H) Bar graph of fluorescence changes after stimulation with C4HSL as shown in A–F. DAF-FM fluorescence increases were 426 ± 47 (wt; control), 24 ± 18 (wt with 0-Ca2+), 65 ± 32 (wt + U73122), 330 ± 38 (wt + U73343), 90 ± 60 (TRPM5-/-), 108 ± 50 (wt + TPPO) and 308 ± 54 (α-gustducin-/-). Symbols denote significance compared with control determined via one-way ANOVA with Dunnett’s post-test; **P < 0.01; n.s.: not significant.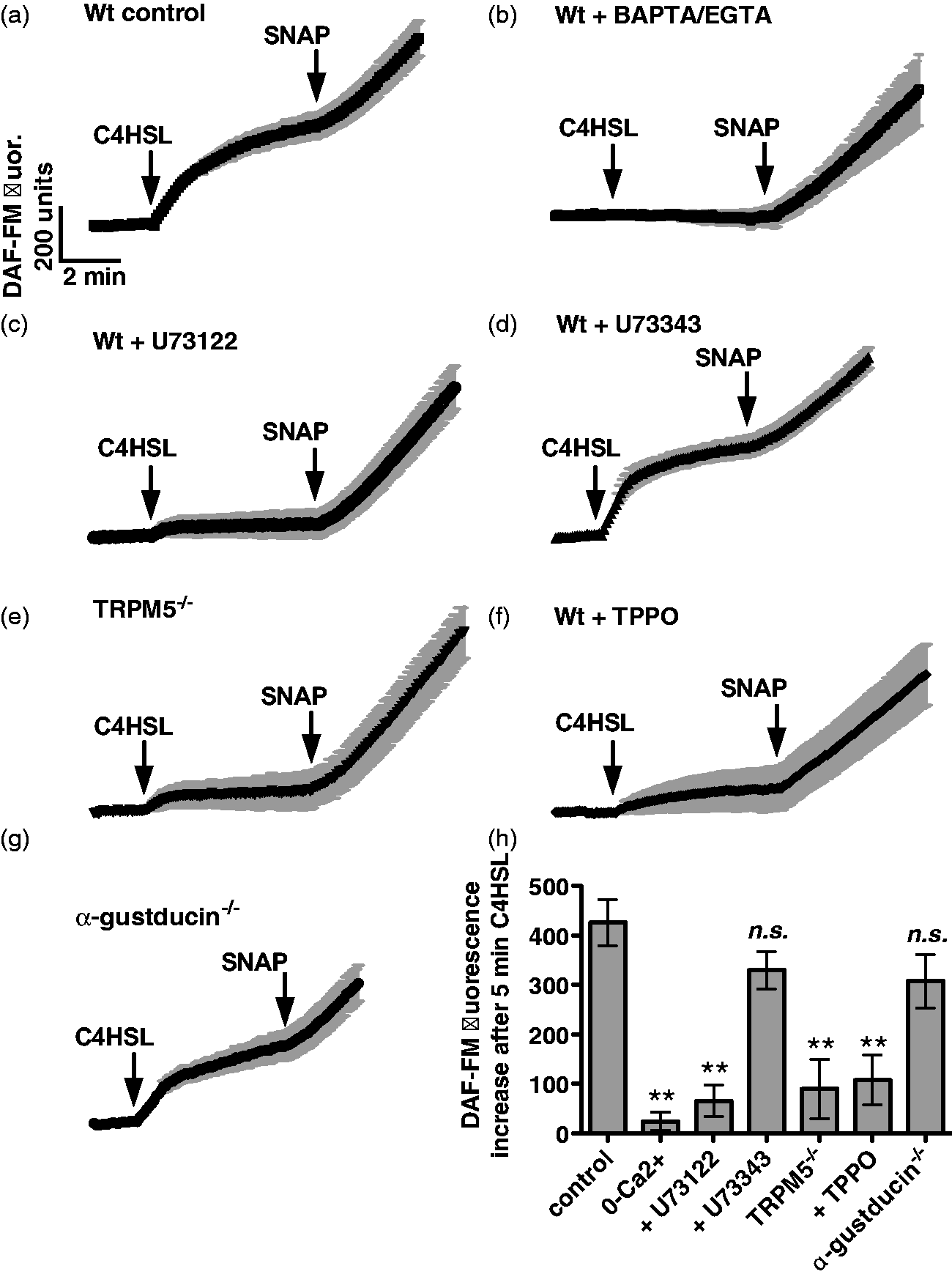
To examine whether AHLs secreted by intact P. aeruginosa bacteria can likewise activate mouse sinonasal epithelial cell NO, we took DAF-FM-loaded ALIs and stimulated them with dilute concentrations of P. aeruginosa- CM from a 3-d planktonic growth culture as previously described.
14
CM from a wt P. aeruginosa (strain PAO1) induced robust DAF-FM fluorescence increases (Figure 5a), while medium from a strain that cannot produce AHLs (PAO-JP2) did not (Figure 5b). DAF-FM fluorescence increases in response to PAO1 CM were also blocked by U73122 (Figure 5c), suggesting they require the PLCβ2 component of taste signaling. These data are summarized in Figure 5d.
CM from wt, but not AHL-deficient, P. aeruginosa stimulates taste-dependent NO production. (a–c) Traces of DAF-FM fluorescence from wt mouse nasal septal ALI cultures stimulated with dilute CM from wt P. aeruginosa (PAO1; A; n = 6 cultures), AHL-deficient P. aeruginosa (PAO-JP2; B; n = 7 cultures), and wt P. aeruginosa in the presence of U73122 (C; n = 6 cultures). (d) Bar graph showing DAF-FM fluorescence increases (mean ± SEM) after 3 min stimulation with 6.25% CM and 12.5% CM. DAF-FM fluorescence increases were [6.25% CM] 103 ± 7 (PAO1), 33 ± 10 (PAO-JP2), 42 ± 16 (PAO1 + U73122) and [12.5% CM] 138 ± 7 (PAO1), 27 ± 20 (PAO-JP2) and 26 ± 20 (PAO1 + U73122). Asterisks indicate significance determined by one-way ANOVA with Bonferroni post-test; *P < 0.05.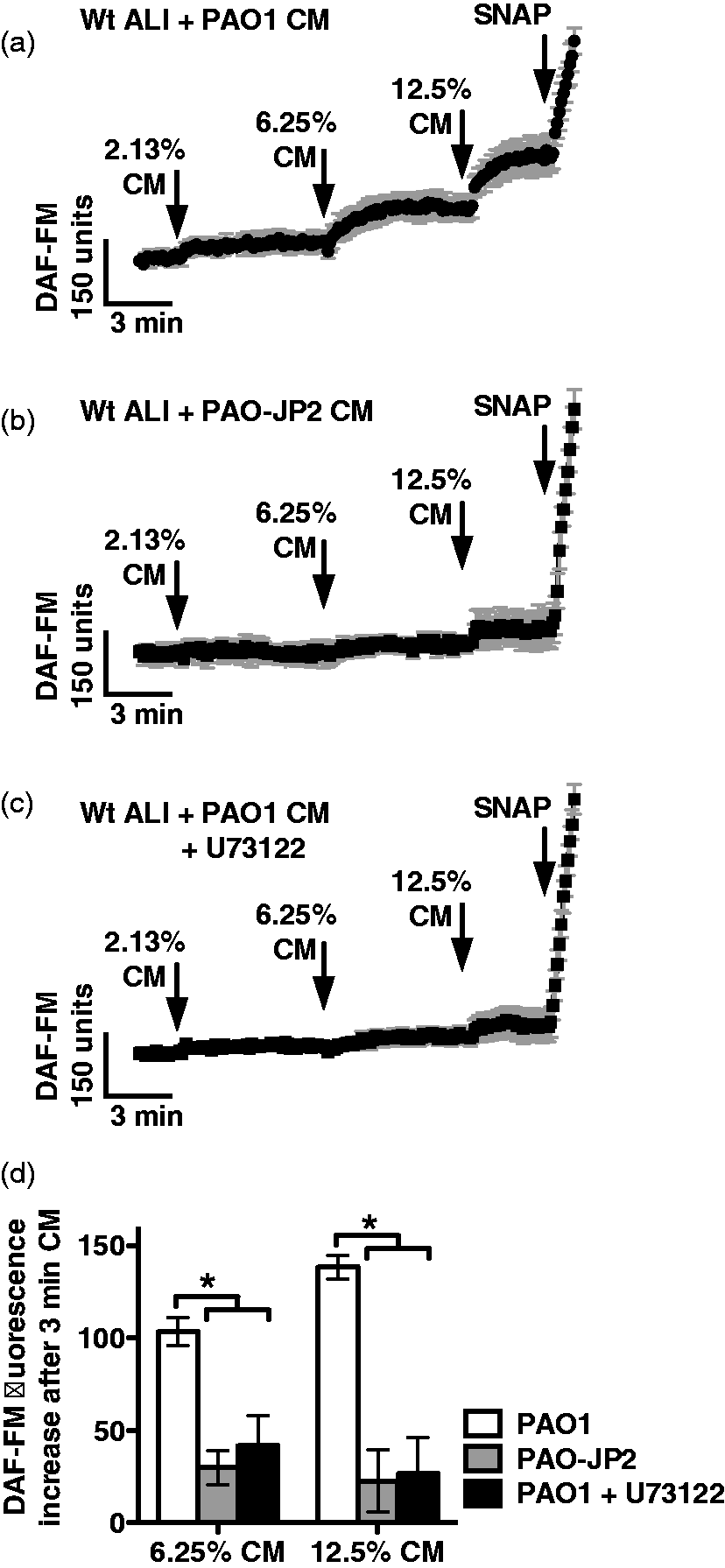
We next examined if AHL-activated taste signaling and NO production stimulates mucociliary clearance to confirm that this reflects an innate immune response. A particle-tracking assay was used to measure relative changes in mucociliary transport, as previously described.14,65 Fluorescence microspheres were overlayed on the ALI culture and images during 2-s camera exposures, resulting in images of ‘streaks’ denoting the distance traveled during the 2-s period. Relative streak length approximates the relative increase in mucociliary transport rate. Stimulation of wt cultures with 10-µM C4HSL induced a ∼ twofold increase in mucociliary transport rate (Figure 6A) that was blocked by U73122 or the NOS inhibitor L-NAME (20 µM for 15 min pre-incubation; Figure 6b) or the NO scavenger carboxy-PTIO (10 µM; Figure 6b). This suggests that C4HSL increases mucociliary clearance that requires both PLCβ2/taste signaling and NOS activity and NO production. Likewise, we found that dilute (12.5%) PAO1 CM induced an increase in mucociliary clearance that required PLCβ2/taste signaling, while 12.5% LB alone had no effect (Figure 6b). CM from the AHL-deficient strain PAO-JP2 had no effect (Figure 6b), while CM from a flagellin-deficient flgK mutant strain (Sad36) activated responses similar to PAO1 CM (Figure 6b), supporting the specific role for secreted AHLs in initiating this response.
C4HSL and wt P. aeruginosa-CM stimulate an increase in mucociliary transport. (a) Representative images of one field of view showing streaks representing particle transport by a wt mouse septal ALI culture before stimulation (left) and after 3 min stimulation with 100 µM C4HSL (right). (b) Graph showing mean ± SEM (n = ALI 3–5 cultures each) of normalized changes in particle transport after addition of PBS alone (reflecting the mechanical force of pipetting alone, 1.08 ± 0.12), C4HSL (2.25 ± 0.29), C4HSL + U73122 (1.21 ± 0.2), C4HSL + L-NAME (1.11 ± 0.15), C4HSL + cPTIO (1.21 ± 0.08), 12.5 % LB alone (1.15 ± 0.10), 12.5% PAO1 CM (2.21 ± 0.25), PAO1 CM + U73122 (1.21 ± 0.20), PAO-JP2 CM (1.25 ± 0.12) and Sad36 CM (1.98 ± 0.05). Symbols denote significance versus appropriate control condition (PBS or LB alone) determined via one-way ANOVA with Dunnett’s post-test; *P < 0.05, **P < 0.01.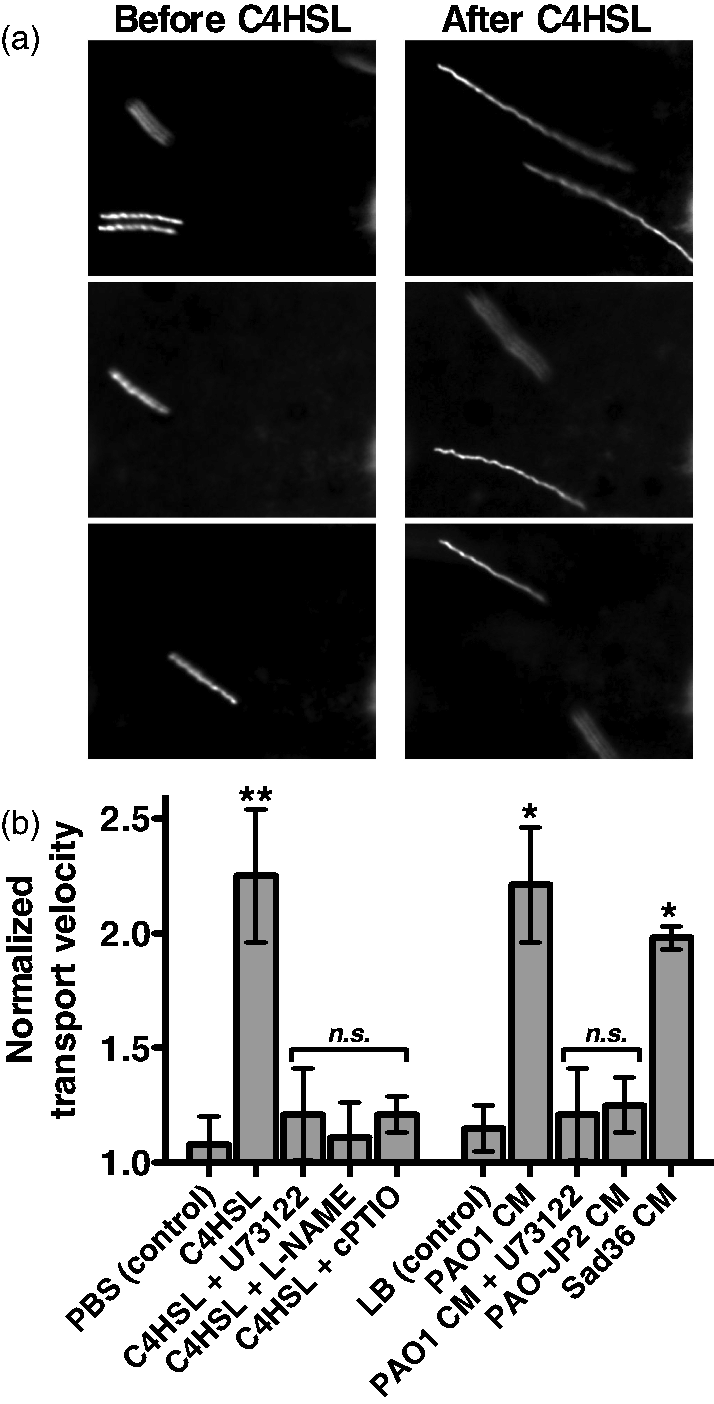
Discussion
T2R bitter receptors are emerging as important players in the innate immunity of the upper14, 23 and lower 13 airways, with preclinical 14 and clinical14,23 data suggesting the human receptor T2R38 plays a key sentinel role in upper airway Gram-negative bacteria infection and CRS by detecting AHL quorum-sensing molecules secreted by Gram-negative bacteria. The role of taste receptors in innate immunity is particularly intriguing because taste receptors have a uniquely high density of naturally-occurring genetic variants; 66 variation in individual T2R function may partly underlie differences in susceptibility to CRS and partly explain previous studies that have suggested CRS has one or more unknown genetic components.67–69 Because of the potential importance of this signaling pathway to human innate immunity, we examined if molecules involved in taste signaling are also required for detection of AHLs in mouse nasal septal epithelial cells, both to define mechanisms of this pathway and to determine if the mouse is a useful model in which to study it.
The data shown above demonstrate that the bitter tastant PTC and P. aeruginosa quorum-sensing AHLs stimulate calcium signals with similar magnitude and kinetics. The AHL-dependent calcium response drives production of NO that stimulates an increase in mucociliary clearance, the ‘first-line’ of defense in sinonasal innate immunity. This response requires two important components that are also involved in taste signaling, namely PLCβ2 and TRPM5, identically as previously observed in human sinonasal epithelial cells. 14 Notably, neither the PTC-induced nor C4HSL-induced calcium and NO responses observed here require the G protein subunit α-gustducin, a central component of the canonical signaling pathway in taste receptor cells of the tongue.44,46 Gustducin-independent taste-receptor signaling has been previously observed,35,54,55,70–73 but to our knowledge, the finding that a T2R-receptor-linked signaling pathway is completely intact in the absence of α-gustducin would be highly novel. While our results are strongly suggestive of a T2R receptor-mediated response, they do not demonstrate a requirement for T2R receptors because specific KO mice do not yet exist. Further work must be directed at determining which T2R receptors may be required for this response in order to confirm whether or not this pathway is truly an α-gustducin-independent T2R-dependent chemosensory pathway. The Gα-protein(s) involved in the sinonasal mucosal response to AHLs also remain to be determined. It is also notable that, because no specific pharmacological gustducin inhibitor exists, this elucidation of this molecular insight into AHL airway signaling required use of the mouse model and α-gustducin KO.
The responses observed here originate from ciliated epithelial cells. As mentioned above, other researchers have observed AHL-dependent calcium responses originating from discreet non-ciliated cells termed solitary chemosensory cells (SCCs) that synapse with trigeminal neurons and regulate breath-holding in response to bitter chemicals.27,60,61 While the responses reported here were independent of α-gustducin, the responses of SCCs to AHLs have been reported to require α-gustducin.27,60,61 This likely reflects a difference in the signaling pathways of these two very different cell types. While the role of reported SCC responses in the mouse airway appears to be to prevent further inhalation of toxic substances,27,60,61 our data suggest that the ciliated epithelial cells themselves can also detect AHLs and mount rapid local innate immune responses mediated by calcium-dependent NO production. While it is yet to be determined whether the SCC responses27,60,61 are mimicked in the human nasal epithelium, the data here suggest that the ciliated epithelial cell NO response is partially conserved between species.
In addition to its role in controlling airway innate immunity by increasing mucociliary clearance rates, NO is thought to be an important direct mediator of innate immunity. NO is thought to be able to rapidly diffuse into bacteria, and NO and its reactive derivatives (e.g. S-nitrosothiols and peroxynitrites) can damage bacterial DNA, react with proteins containing sulfhydryl or thiol groups, inactive enzymes with metal cofactors, and damage membrane lipids.17,18 It has been suggested that the majority of airway NO originates in the nose74–76 with high levels of NOS expression in the cilia and microvilli of the paranasal sinus epithelium.77,78 Polymorphisms in the NOS1 gene have been correlated with severity of CRS, 79 but levels of exhaled NO have been both positively and negatively correlated with CRS.80–84 The evidence is not yet consistent enough nor are there reliable enough measurement techniques to make exhaled NO a useful measure for or predictor of sinonasal infection. 85 Nonetheless, high concentrations of NO can be toxic to the common nasal pathogens P. aeruginosa 14 and Staphylococcus aureus, 86 making NO an attractive therapeutic molecule for treatment of sinonasal disease. Thus, there are significant potential clinical implications for the ability to harness the T2R-mediated NO production pathway for treatment of respiratory infections. The exact mouse T2R receptor(s) that may be involved in AHL-induced NO production are yet unknown owing to the lack of a clear T2R38 ortholog. However, the data outlined in this study demonstrate that important components of this pathway are conserved between human and mouse, revealing important molecular insights into this pathway and suggesting that the mouse is a very useful and important model with which to study the mechanisms and downstream effects of sinonasal AHL-detection and potentially T2R signaling in vitro and, potentially, in vivo.
Footnotes
Funding
This work was supported by a grant from the Flight Attendants Medical Research Institute (082478) and a charitable donation from the RLG Foundation, Inc., both to N.A.C., as well as a National Institutes of Health grant (DC03055) to R.F.M
Conflict of interest
The authors do not have any conflicts of interest to declare.
Acknowledgements
We thank BH Igelwski and JM Schwingel for P. aeruginosa strain PAO-JP2 and G. O’Toole for strain Sad36.
