Abstract
Alongside the intestinal border, dendritic cells (DCs) sample large amounts of endogenous and potentially pathogenic antigens followed by initiation of protective immune responses or induction of tolerance. Breakdown of oral tolerance towards commensal bacteria is suggested to be crucial for the development of both human and canine inflammatory bowel disease (IBD). The aim of this study was to investigate canine intestinal DCs in the steady state and in dogs with IBD using multicolour immunofluorescence. In the healthy gut, DC-like cells expressed MHC II, CD1a8.2 and CD11c, and, in lower amounts, CD11b, within lamina propria, Peyer’s patches (PPs) and mesenteric lymph nodes (MLNs), whereas those expressing CD80 and CD86 were only present in PPs and MLNs. Occasionally, DC-like cells were in contact with the intestinal lumen through transepithelial projections. In canine IBD, CD1a8.2+, CD11b+ and CD11c+ DC-like cells were decreased within the stomach, duodenum and colon, whereas the colonic mucosa revealed elevation of CD86+ DC-like cells. The complex network of DC-like cells in the gut indicates their important role in canine mucosal immunity, including active sampling of luminal antigens. Furthermore, their shift in diseased dogs suggests a pathogenetic significance for canine IBD.
Introduction
Dendritic cells (DCs), first described by Ralph Steinman in 1973, 1 rank among professional APCs, which incorporate pathogenic antigens in the body periphery (immature DCs) and migrate to secondary lymphoid organs where antigen presentation to naïve T cells (mature DCs) and induction of immunity takes place. 2 Additionally, specialised DCs can mediate immunological tolerance and therefore play an important role in immune regulation. 3 Alongside the intestinal border, DCs take up large amounts of both pathogenic and beneficial antigens and can induce expansion and gut homing of regulatory T cells (Tregs), which is essential for the establishment of oral tolerance and maintenance of intestinal homeostasis.4,5
Canine idiopathic inflammatory bowel disease (IBD) is characterised by chronic or recurrent gastrointestinal symptoms of at least 3 wks duration, including diarrhoea, vomiting and weight loss without obvious underlying cause, and histological evidence of mucosal inflammation.6,7 Histopathologically, IBD can be distinguished into different forms based on the dominant cellular infiltrates and the anatomical sites where inflammation takes place (i.e. stomach, small intestine or colon). 8 Regarding this, lymphoplasmacytic enteritis and eosinophilic gastroenteritis (EGE) represent the most common forms of canine IBD characterised by either lymphoplasmacytic or eosinophilic inflammation. 9
In humans, IBD comprises Crohn’s Disease and Ulcerative Colitis, which share several similarities with their canine counterparts, such as clinical symptoms and therapeutic strategies. 10 Additionally, genetic susceptibilities, disturbances of intestinal microbiota and mutation of innate immune receptors leading to a breakdown of oral tolerance are suggested in both humans and dogs, and therefore canine IBD represents a potential model to study human Crohn's Disease and Ulcerative Colitis.11–13
Founded in their huge heterogeneity and plasticity, various DC subsets with different phenotypes and functions are described, 14 implying the lack of a single marker for this cell type. In dogs, the phenotype of DCs has been characterised in detail in vivo in the context of related neoplastic disorders, 15 and has been further analysed in vitro using monocyte- and bone marrow-derived DCs.16,17 Therefore, canine DCs are known to express several markers, including MHC II, CD1 and CD11, as well as co-stimulatory molecules, such as CD40, CD80 and CD86. According to their important role in immunity, canine DCs have been investigated in several diseases, including infections, 18 arthropathies19,20 and skin disorders. 21 Furthermore, recent studies regarding canine cancer have implied their therapeutic potential in veterinary medicine.22,23 However, the few studies of canine IBD used markers that are not exclusively expressed by DCs, such as MHC II and CD11c.24,25 Comprehensive data about intestinal DCs either in the steady state or in gastrointestinal diseases are not available.
Therefore, the aim of this study was to investigate the distribution and phenotypes of DCs in the canine gut in healthy dogs and dogs suffering from IBD.
Materials and methods
Animals
Dogs without gastrointestinal diseases.
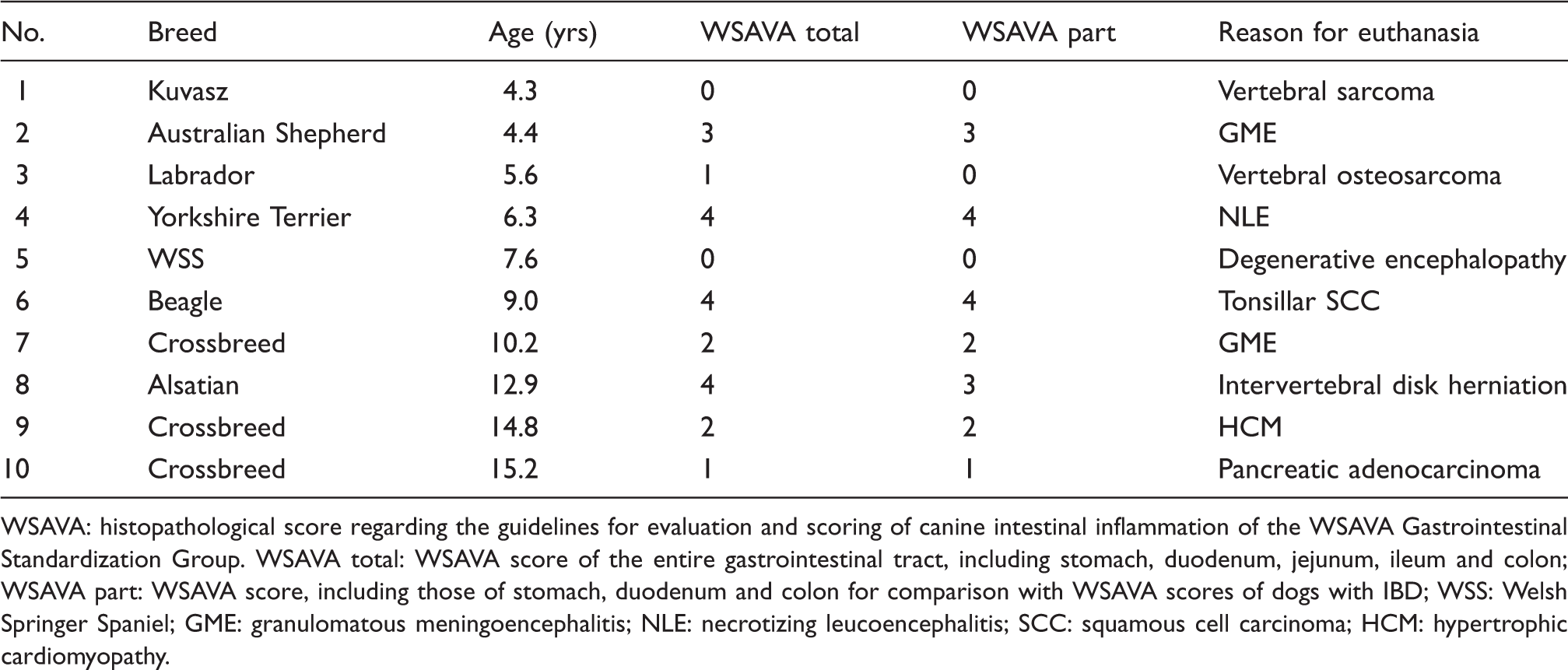
WSAVA: histopathological score regarding the guidelines for evaluation and scoring of canine intestinal inflammation of the WSAVA Gastrointestinal Standardization Group. WSAVA total: WSAVA score of the entire gastrointestinal tract, including stomach, duodenum, jejunum, ileum and colon; WSAVA part: WSAVA score, including those of stomach, duodenum and colon for comparison with WSAVA scores of dogs with IBD; WSS: Welsh Springer Spaniel; GME: granulomatous meningoencephalitis; NLE: necrotizing leucoencephalitis; SCC: squamous cell carcinoma; HCM: hypertrophic cardiomyopathy.
Dogs with IBD.
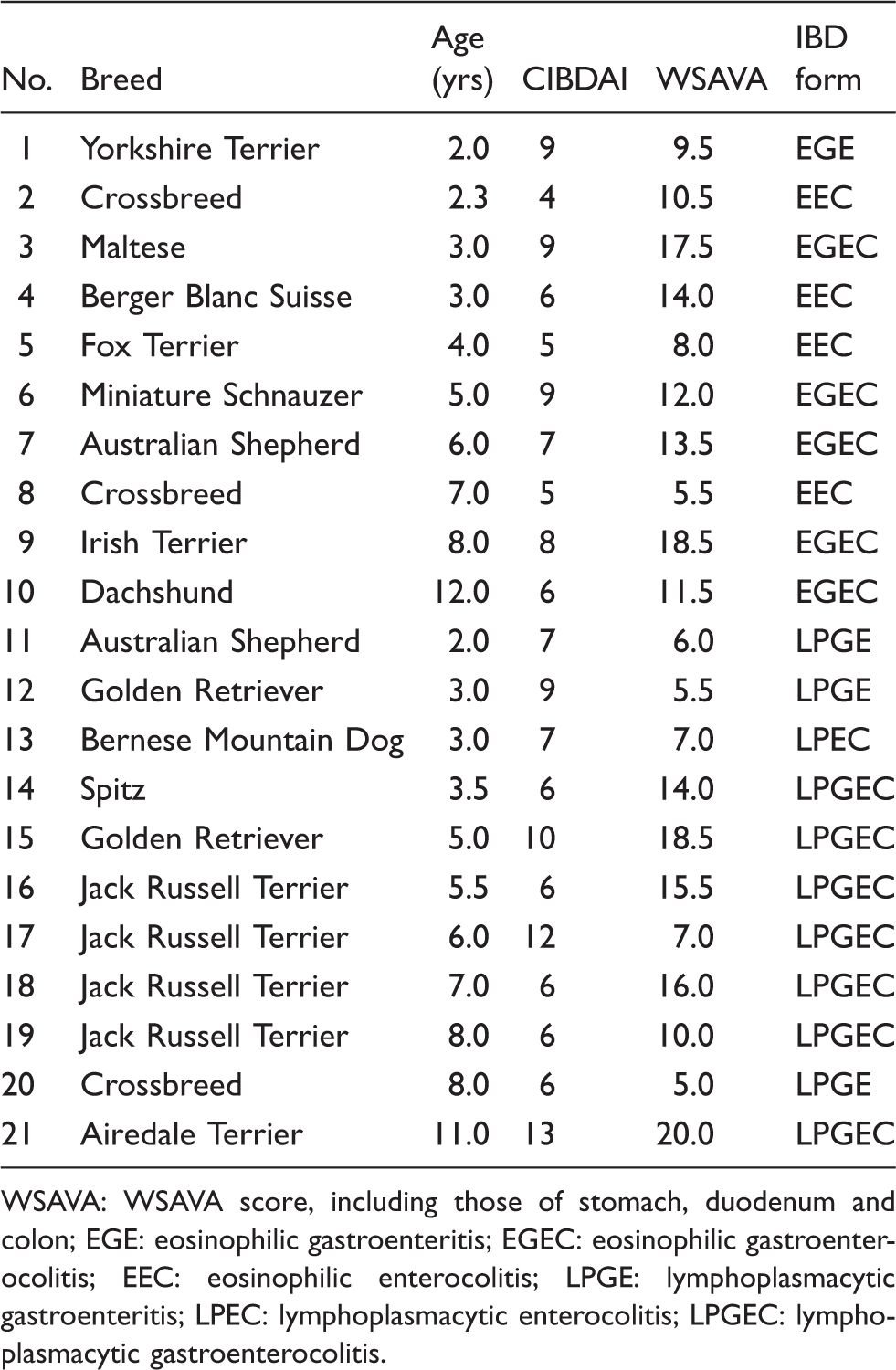
WSAVA: WSAVA score, including those of stomach, duodenum and colon; EGE: eosinophilic gastroenteritis; EGEC: eosinophilic gastroenterocolitis; EEC: eosinophilic enterocolitis; LPGE: lymphoplasmacytic gastroenteritis; LPEC: lymphoplasmacytic enterocolitis; LPGEC: lymphoplasmacytic gastroenterocolitis.
Histopathological examination
For histopathological examination, 2–4-µm thick sections of paraffin-embedded samples were prepared and stained with haematoxylin and eosin (H&E) according to standard histological procedures. Slices were independently evaluated by three pathologists (JJ, US, MHT) using light microscopes (Carl Zeiss, Oberkochen, Germany), according to histopathological guidelines for the evaluation and scoring of canine intestinal inflammation published by the World Small Animal Veterinary Association (WSAVA) Gastrointestinal Standardization Group.
27
In this study, all dogs used as healthy controls had no or only minimal sings of autolysis, ensuring that they were comparable to endoscopic biopsy specimens (Figure 1).
Intestinal pathology of dogs suffering from IBD. Diseased dogs revealed varying degrees of mucosal inflammation in addition to villus stunting, increase in intraepithelial lymphocytes and lacteal dilation (A, B) and histopathological scores (D) were significantly increased in of both eosinophilic (IBD-E) and lymphoplasmacytic (IBD-L) canine IBD compared to controls (C). Scatter plots are shown with mean and SD. Scale bars: A = 200 µm; B, C = 100 µm.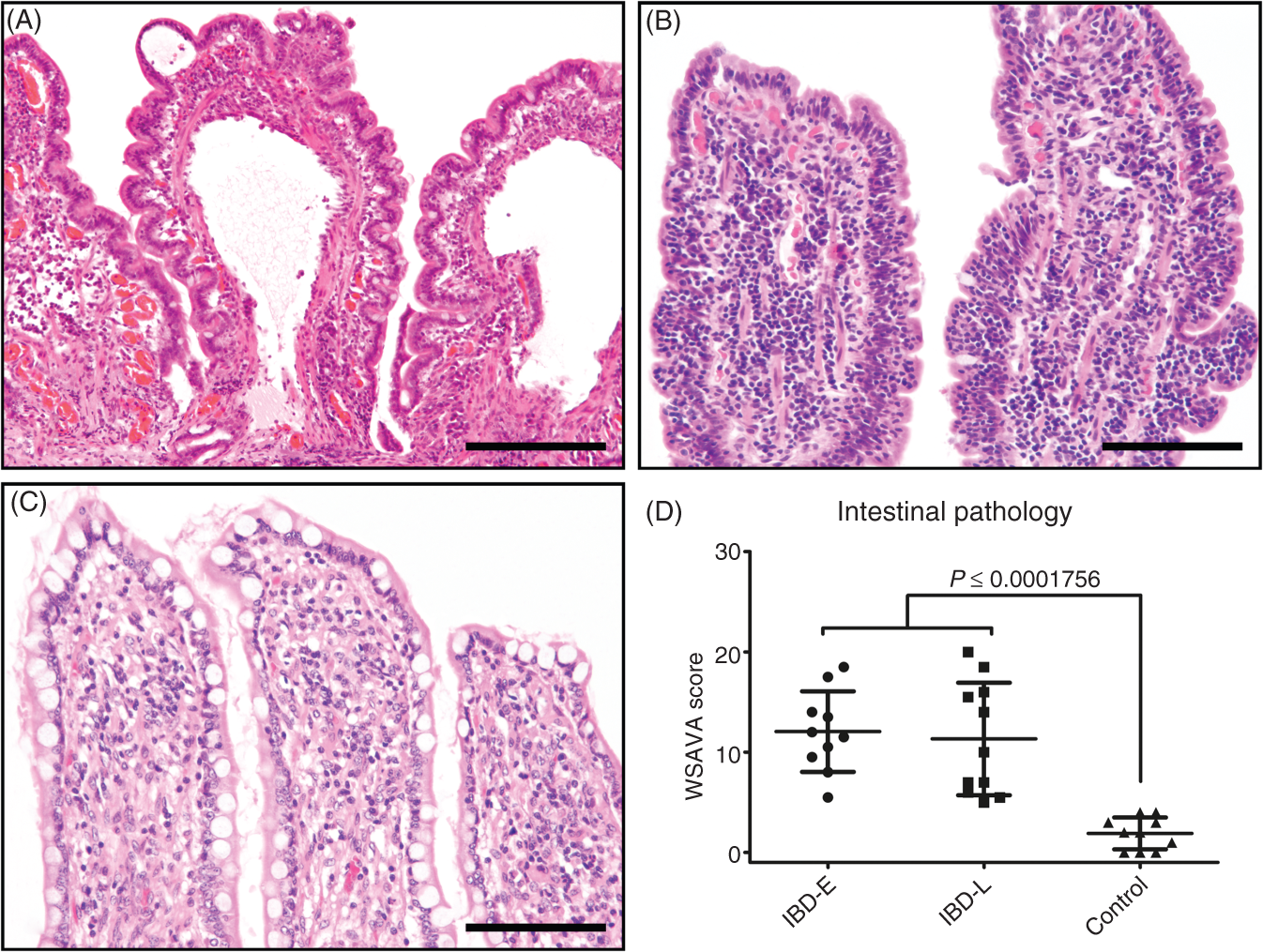
Multicolour immunofluorescence
For immunofluorescence staining, 4–5-µm thick serial sections were prepared with a cryotome (Leica CM1950, Leica Microsystems, Wetzlar, Germany), mounted on SuperFrost® Plus slides (Thermo Scientific, Waltham, MA, USA), dried, fixed in cooled acetone (Carl Roth) and stored at −80℃ until staining and analysis. The presence of histology similar to respective paraffin-embedded sections was ensured by evaluation of H&E stained cryosections. For immunofluorescence staining procedures, slides were defrosted, rinsed twice in PBS (pH 7.2, 0.15 M) and incubated with normal goat serum for 30 min. Subsequently, sections were double-incubated for 2 h at room temperature (RT; approximately 20–22℃) with rat anti-canine MHC II monoclonal Abs (clone YKIX334.2; diluted 1:1000 in PBS containing 1% of BSA; Serotec, Oxford, UK) in combination with a panel of different murine monoclonal Abs against canine CD1a6 (clone Fe1.5F4), CD1a8.1 (clone CA9.AG5), CD1a8.2 (clone CA13.9H11 further cross-reacting with CD1a8.1 [P. Moore, personal communication]), CD11b (clone CA16.3E10), CD11c (clone CA11.6A1), CD80 (clone CA24.5D4) and CD86 (clone CA24.3E4; all diluted 1:50 in PBS/BSA; all kindly received from P. Moore). Sections were then simultaneously incubated with goat anti-rat Cy™ 3-conjugated (minimal cross-reactions to mouse, human, bovine, horse and rabbit serum proteins) and goat anti-mouse Alexa Fluor® 488-conjugated (minimal cross-reactions to rat, human, bovine, horse and rabbit serum proteins) secondary Abs (Jackson ImmunoResearch, Newmarket, UK) diluted 1:200 in PBS with BSA for 1 h at RT. Thereafter, nuclei were counterstained using 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI; diluted at 1 µg/ml in PBS; Serotec) for 3 min at RT before covering slides with fluorescence mounting medium (Dako, Hamburg, Germany). Between each step, sections were rinsed twice in PBS.
Sections of normal feline (for clone Fe1.5F4) and canine (for remaining Abs) lymph nodes were used as staining positive controls. For negative controls, primary Abs were replaced by rat serum (Sigma-Aldrich, Munich, Germany) in combination with mouse IgG2a (for clone CA13.9H11; Dako), mouse IgG2b (for clone CA24.5D4; Serotec) or mouse IgG1 isotype control Abs (for all remaining clones; Millipore, Schwalbach, Germany) in equal concentrations.
Examination of sections
Double-stained slides were visualised and digitalized on an AxioVert 200 M microscope (Carl Zeiss) using AxioVision software (Carl Zeiss). Maximal light exposure time was calibrated using negative control sections. Accordingly, at least four areas of the lamina propria (LP) of double-stained slides were photographed at 200× magnification within the following compartments: gastric fundus; small intestine—villi, crypts; large intestine—crypts. Digital images were visualised on a monitor followed by manual examination of double-stained markers as follows. Simultaneous expression of two molecules within a single cell—called co-expression—was assessed by comparing the individual channels, such as the red channel for MHC II and the green channel for CD11c. This was further checked in merged images that comprised the red, green and blue (DAPI-stained nuclei) channel concurrently. Thereby, an overlay of red and green pixels, which may occur for closely adjacent epitopes, resulted in a yellow colour, indicating co-localisation of the proteins. Despite this, we limited the decisive criterion for cell quantification to co-expression, as determination of true co-localisation may have been difficult. 28 Therefore, mononuclear medium-to-large-sized cells (DAPI + nuclei) with clear cytoplasmic projections and co-expression of MHC II and simultaneously applied markers were suggested to be canine DCs and counted manually. However, we cannot totally exclude a slight overlap with macrophages, as both cell types share some similarities concerning their expression of applied surface molecules in dogs in vitro. 29 Thus, we use the term ‘DC-like cells’ below. Within villi, the area of the LP was measured and number of cells was computed per area. Results were expressed as double-positive cells per 150,000 µm2.
Statistical analysis
Statistical analyses were performed using R v. 2.15.1 (The R Foundation for Statistical Computing, Vienna, Austria). Initially, each group was assessed for normal distribution using the Shapiro-Wilk test. Wilcoxon rank-sum test was applied to evaluate differences between WSAVA scores of dogs with gastrointestinal diseases and controls. Additionally, differences in numbers of LP DCs between small intestinal localisations (duodenum and jejunum) and compartments (villi and crypts) were assessed using the Wilcoxon signed-rank test with (small intestinal localisations) or without (small intestinal compartments) correction of P-Values using the Holm method. Moreover, differences between distinct CD molecules were calculated by Wilcoxon signed-rank test with (CD1a8.2, CD11b, CD11c, CD80 and CD86) or without (CD1a8.1 and CD1a8.2) Holm’s correction of P-values. Numbers of DCs in dogs with IBD were compared with controls by applying the Wilcoxon rank-sum test. Furthermore, Spearman’s rank correlation coefficient was used to examine possible correlations between numbers of DCs and CIBDAI or WSAVA scores in dogs suffering from chronic idiopathic gastrointestinal disorders. P-Values ≤ 0.05 were defined as statistically significant.
Results
Clinical examination and histopathology
Histopathological examination of the intestines of dogs without clinical and pathomorphological signs of gastrointestinal disorders occasionally revealed only mild variation of normal histology, such as a slight increase in LP lymphocytes and plasma cells, minimal crypt distension or mild mucosal fibrosis (Table 1, Figure 1).
Clinical data of dogs suffering from IBD, including the CIBDAI, are summarised in Supplementary Table 1. Detailed diagnostic workup did not give any hint to possible underlying causes, such as infections, food allergy or neoplasia.
Endoscopic biopsies of dogs suffering from chronic idiopathic gastrointestinal disorders, however, displayed mucosal inflammation of a varying degree, in addition to further alterations, including epithelial injury, villous stunting and fusion, increased intraepithelial lymphocytes and loss of crypt architecture (Figure 1); WSAVA scores, given in Table 2, were significantly increased (P < 0.001) compared with healthy dogs. Inflammation was either dominated by lymphocytes and plasma cells (n = 11) or eosinophils (n = 10). Therefore, canine IBD was diagnosed—in accordance with its current definition 8 —by exclusion of possible underlying causes and the presence of mucosal inflammation.
Intestinal MHC II expression
Within the normal canine gut, various cells, including lymphocytes and those of macrophage and DC morphology, revealed positive cytoplasmic and membranous labelling for MHC II. Additionally, small intestinal enterocytes revealed immunoreactivity for MHC II with increased intensity of the individual cells from duodenum to ileum and from villi to crypts (semi-quantitative evaluation without statistical validation). Similarly, MHC II was expressed by colonic epithelial cells with increased intensity of the individual cells from the apical to the basal crypt epithelium.
Canine DC-like cells in the steady state
Numbers of DC-like cells in the LP of the canine control intestines are summarised in Table 3, given as medians per group. As different CD1A genes are described in dogs,
30
we used Abs against different CD1a proteins to assess their expression and distribution within the canine gut (Figure 2). CD1a8.2 was expressed by DC-like cells within the LP of the stomach, small intestinal villi and crypts, and large intestine of all control dogs. Furthermore, CD1a8.2 was regularly expressed in organised structures of the canine gut-associated lymphoid tissue (GALT), including lymphoid follicles, Peyer’s patches (PPs) with predominance in the subepithelial domes (SEDs), colonic lymphoglandular complexes and MLNs. In contrast, only 40% of control animals revealed CD1a8.1+ cells in the LP and MLNs. Additionally, CD1a8.1+ DC-like cells were present in significant lower numbers than those expressing CD1a8.2 in the LP (P ≤ 0.0079). Interestingly, CD1a8.1+ DC-like cells were present within SEDs beneath the follicle-associated epithelium (FAE) in 70% of dogs, including those with and without CD1a8.1 expression in the LP. However, CD1a6 was totally absent from the normal canine gut.
Expression of different CD1a proteins within the canine healthy gut. CD1a8.2 (green) was co-expressed with MHC II (red) in both structured (D–F) and diffuse parts (J–L) of the canine GALT in all animals (M, N), whereas CD1a8.1 (green) was only detected in the LP (G–I) and MLN of 4 dogs (N). Interestingly, CD1a8.1 was expressed in PPs (A–C) of 70% of the animals (N). In contrast, CD1a6 was absent from the canine GALT (M, N). Nuclei were counterstained with DAPI (blue). Scatter plots are shown with median. Scale bars = 50 µm. DC-like cells in dogs without gastrointestinal diseases. Medians of DC-like cells within the LP (double-positive for MHC II) are given as cells per 150,000 µm2. uo_villus: duodenal villus; duo_crypt: duodenal crypt; jej_villus: jejunal villus; jej_crypt: jejunal crypt; ile_villus: ileal villus; ile_crypt: ileal crypt.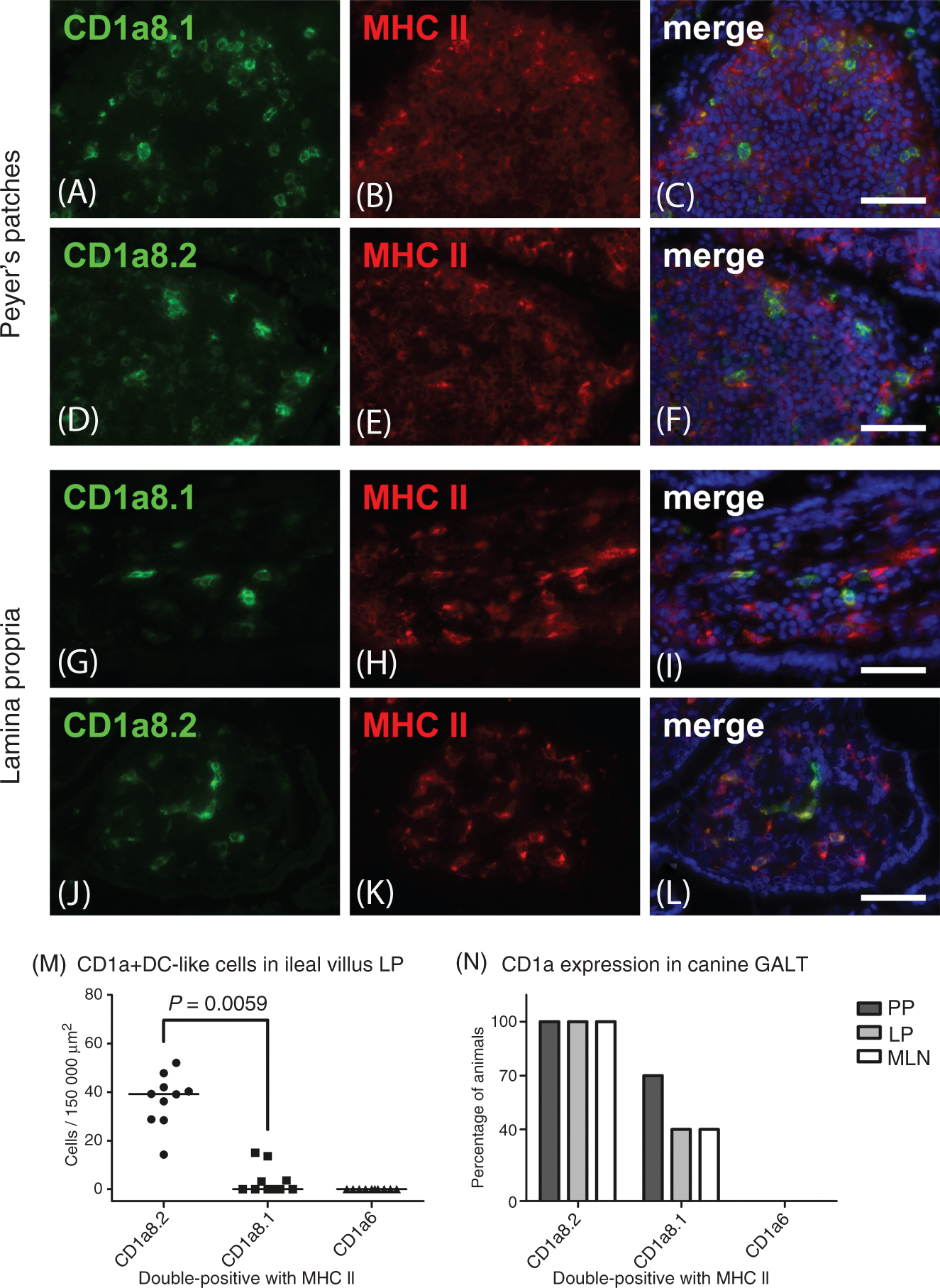
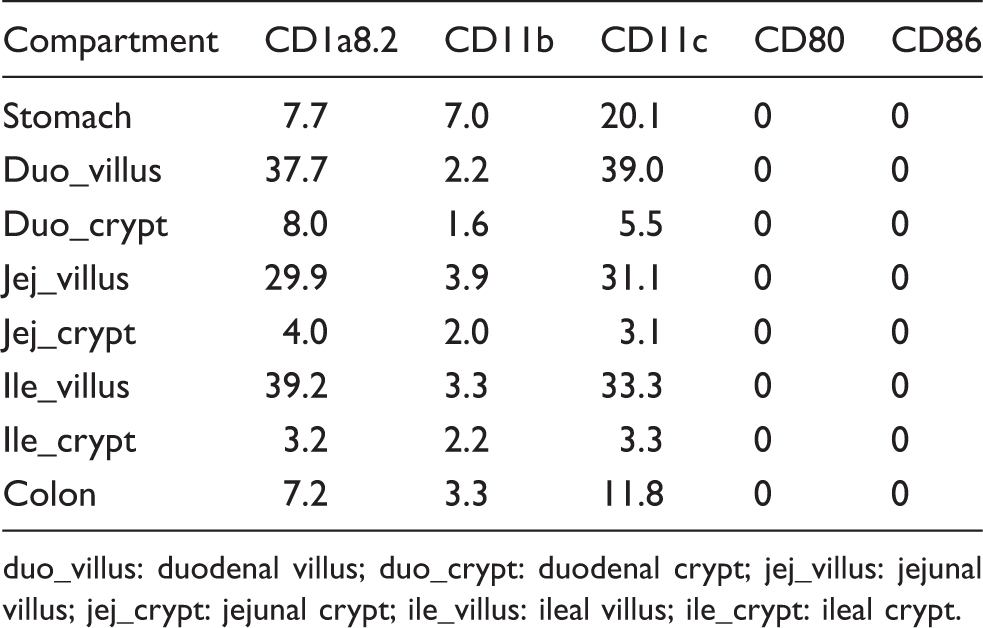
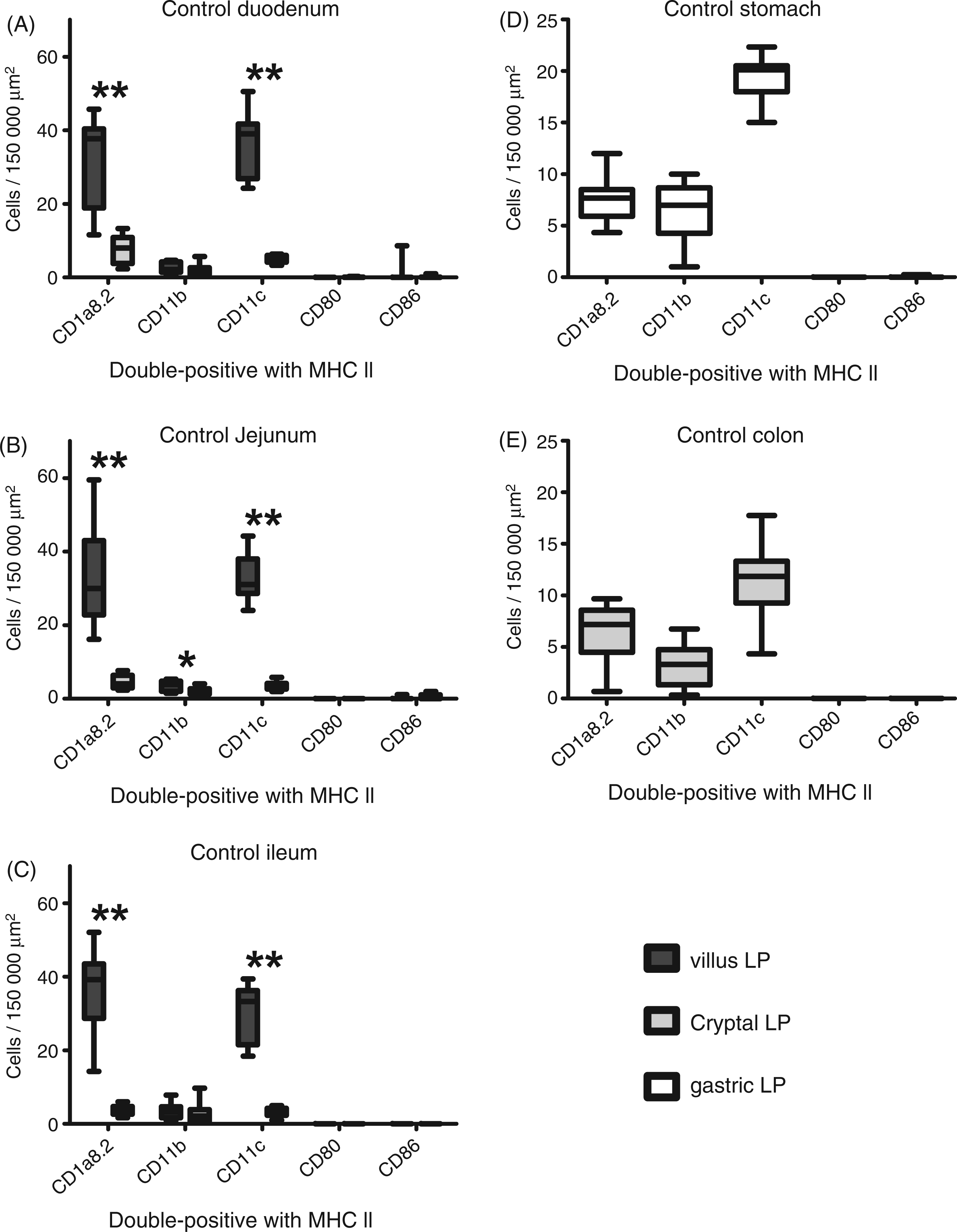
Despite differences concerning the expression of distinct CD1a proteins, the majority of canine LP DC-like cells in small intestinal villi expressed CD1a8.2, CD11c and MHC II, while significantly lower numbers were positive for CD11b (P = 0.0059; Figures 3 and 4). Concerning small intestinal crypts, equal differences were only present in the duodenum and jejunum (P ≤ 0.012). CD11c was further expressed by DC-like cells within the gastric and colonic LP, while the quantities of CD1a8.2+ DC-like cells were significantly lower than CD11c (P ≤ 0.0435), but higher (colon, P = 0.0488) or equal (stomach) to those expressing CD11b (Figure 4). In contrast, CD80+ and CD86 + DC-like cells were not expressed within the gut LP of control dogs.
Phenotypes of DC-like cells in dogs without gastrointestinal disorders. Phenotypically, canine intestinal DC-like cells revealed expression of CD1a8.2 (green), CD11c (green) and in lower amounts CD11b (green) within the healthy LP (A, D, G). In PPs, SEDs revealed high numbers of CD11c+ (H) and lower quantities of CD1a8.2+ (B) DC-like cells, while only few CD11b+ cells (E) were present. In MLNs, phenotype of DC-like cells were similar to PPs and they were mainly located in T cell areas (C, F, I). In contrast, CD80 and CD86 were expressed in T cell areas of both PPs and MLNs, while they were absent in the LP. The yellow colour indicates co-localisation with MHC II (red). Nuclei were counterstained with DAPI (blue). Scale bars = 50 µm. Canine gastrointestinal LP DC-like cells in the steady state. Within the steady state, canine DC-like cells in the small intestinal LP mainly expressed CD1a8.2, CD11c and MHC II, while lower amounts of CD11b+ cells were present (A–C). Within the gastric and colonic LP, numbers of CD1a8.2+ DC-like cells were lower than those expressing CD11c (D, E). Concerning their vertical distribution in the small intestine, CD1a8.2 and CD11c, as well as CD11b (only within the jejunum), were increased in villi compared with crypts (A–C). In contrast, CD80 and CD86 were absent from DC-like cells of the gastrointestinal LP. Boxplots always extend from minimal to maximal value and include median, as well as the lower and upper quartile (box). *P < 0.05; **P < 0.01 by Wilcoxon signed-rank test.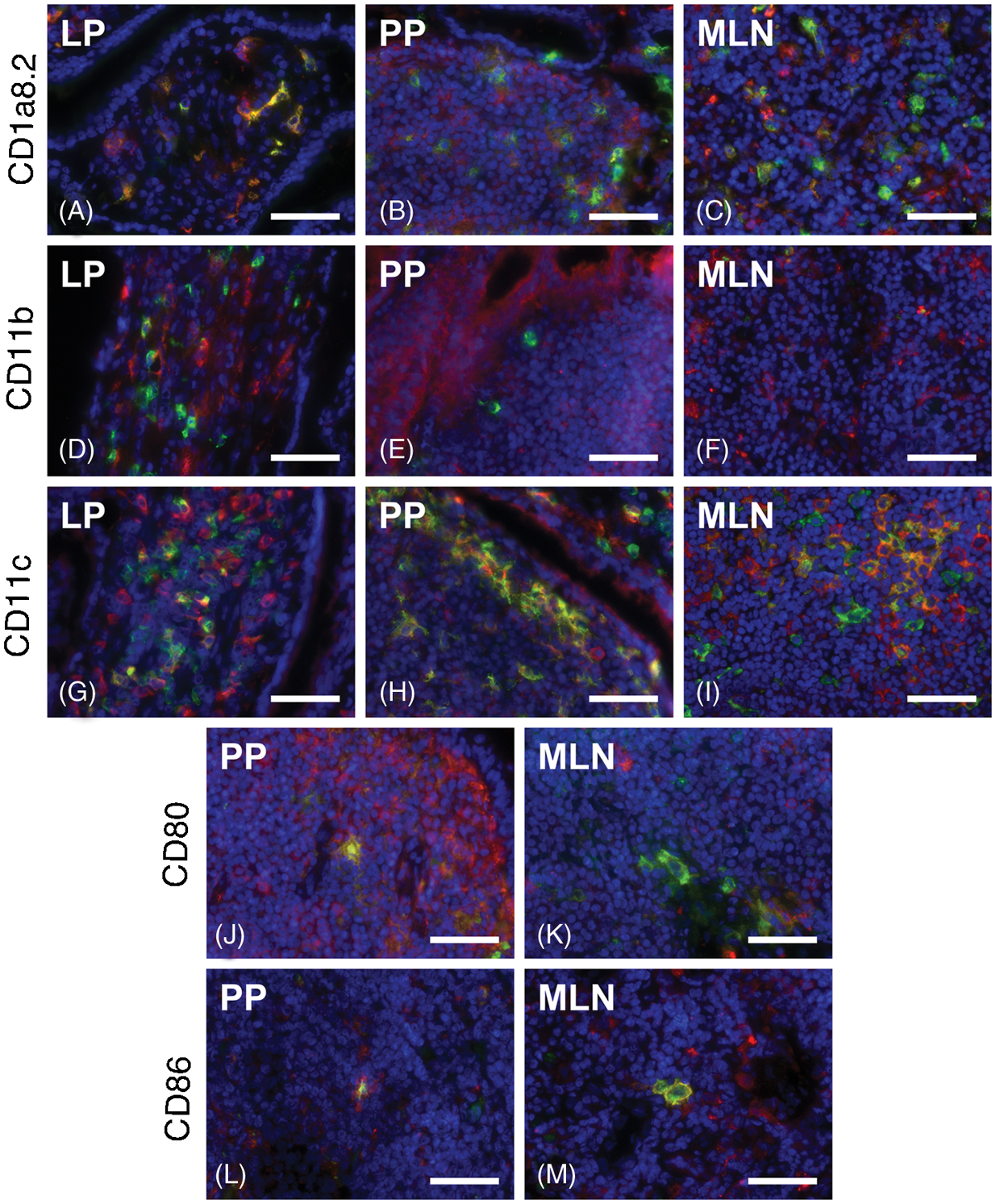
Within the organised GALT, high numbers of DC-like cells were positive for CD11c, while lesser quantities expressed CD1a8.2 and only few DC-like cells were labelled for CD11b (Figure 3). Within PPs, CD11c was highly expressed in SEDs and interfollicular regions (IFRs), whereas a lesser number was present within perifollicular areas and germinal centres. In contrast, we found moderate quantities of CD1a8.2+ and a low number of CD11b+ DC-like cells within PP; these were mainly restricted to SEDs. Within MLNs, DC-like cells were mainly located in perifollicular and paracortical areas, although those expressing CD11c were also present within germinal centres. In contrast to the LP, low numbers of CD80+ and CD86+ DC-like cells were visible within SEDs and IFRs of PPs and in slightly higher numbers in perifollicular and paracortical areas of MLNs. The aforementioned DC-like cells showed impressive cytoplasmic processes, being in close proximity to adjacent lymphocytes (Figure 3J–M).
Furthermore, the number of small intestinal LP DC-like cells was assessed concerning differences in their vertical (villi to crypts) and horizontal (duodenum to ileum) distribution. Within the villus LP numbers of both CD1a8.2+ and CD11c+ DC-like cells were significantly increased (P = 0.001953) compared with crypts (Figure 4). However, similar differences between villus and crypt CD11b+ DC-like cells were only present in the jejunum (P = 0.02734). Regarding their horizontal distribution, numbers of both CD1a8.2+ and CD11c+ crypt DC-like cells significantly decreased from duodenum to ileum and lower amounts of CD11c+ DC-like cells were present in villus LP of the ileum compared with the duodenum (P < 0.05, Figure 5). According to the Spearman correlation, there was no relation between the numbers of DC-like cells and WSAVA scores in control dogs.
Horizontal distribution of DC-like cells in healthy dogs. Concerning their horizontal distribution, canine intestinal DC-like cells significantly decreased from duodenal to ileal LP within villi (CD11c) and crypts (CD1a8.2 and CD11c) in the steady state. Boxplots always extend from minimal to maximal value and include median, as well as the lower and upper quartile (box). *P < 0.05; **P < 0.01 by Wilcoxon signed-rank test and Holm’s correction of P-values.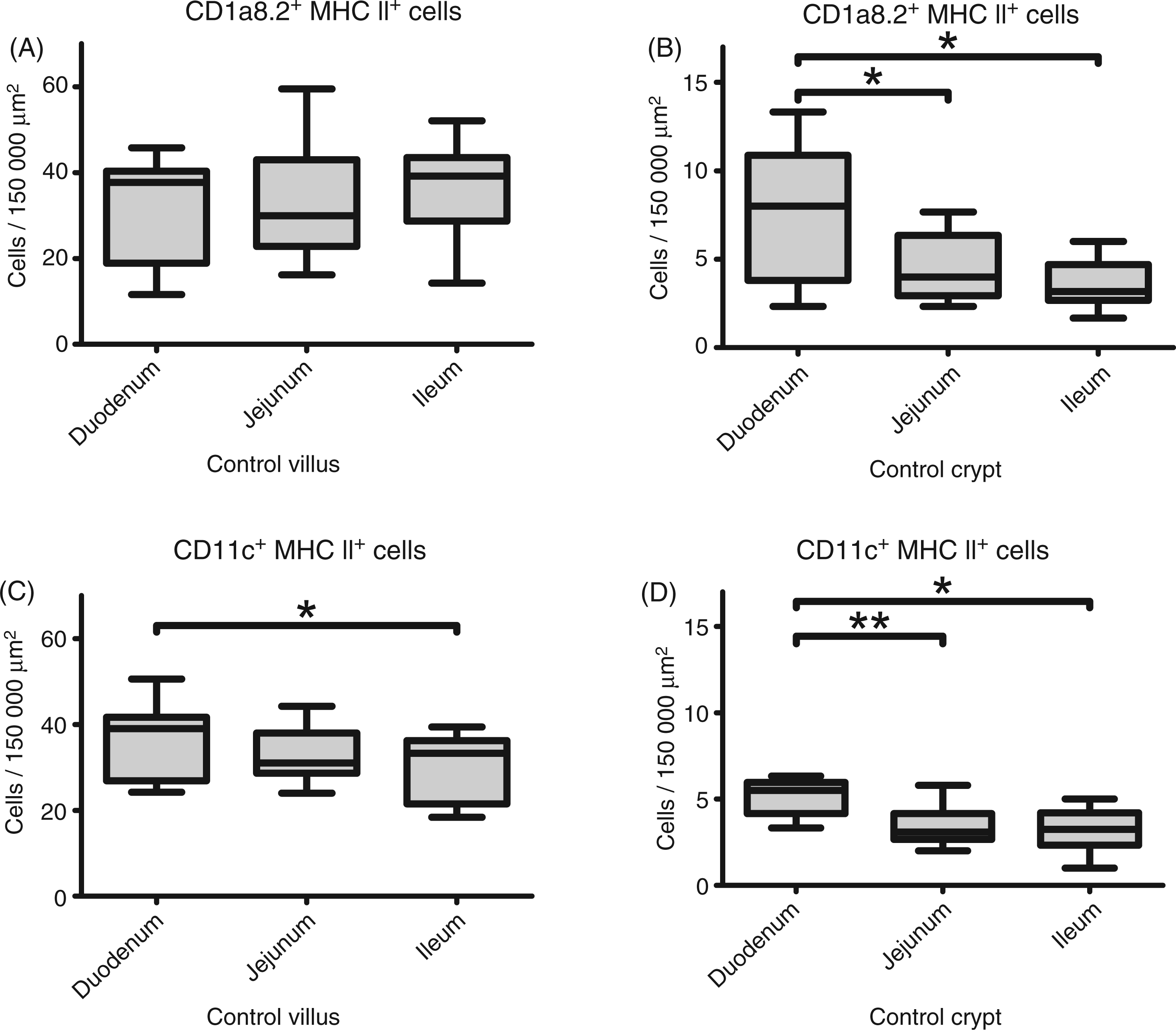
Intestinal transepithelial projections
Occasionally, intestinal DC-like cells located within the LP beneath enterocytes showed cytoplasmic extensions projecting through the epithelium into the gut lumen (Figure 6). Those transepithelial processes were mainly present within colonic and small intestinal crypts, but also within the FAE of PPs, and expressed CD1a8.2, CD11c and MHC II.
Transepithelial projections. CD1a8.2+ and CD11c+ (green) intestinal DC-like cells sending transepithelial projections between enterocytes into the gut lumen were present within colonic (A–D) and small intestinal LP of crypts (E), as well as beneath the FAE (F) of dogs without gastrointestinal diseases. The yellow colour indicates co-localisation with MHC II (red). Nuclei were counterstained with DAPI (blue). Scale bars = 20 µm.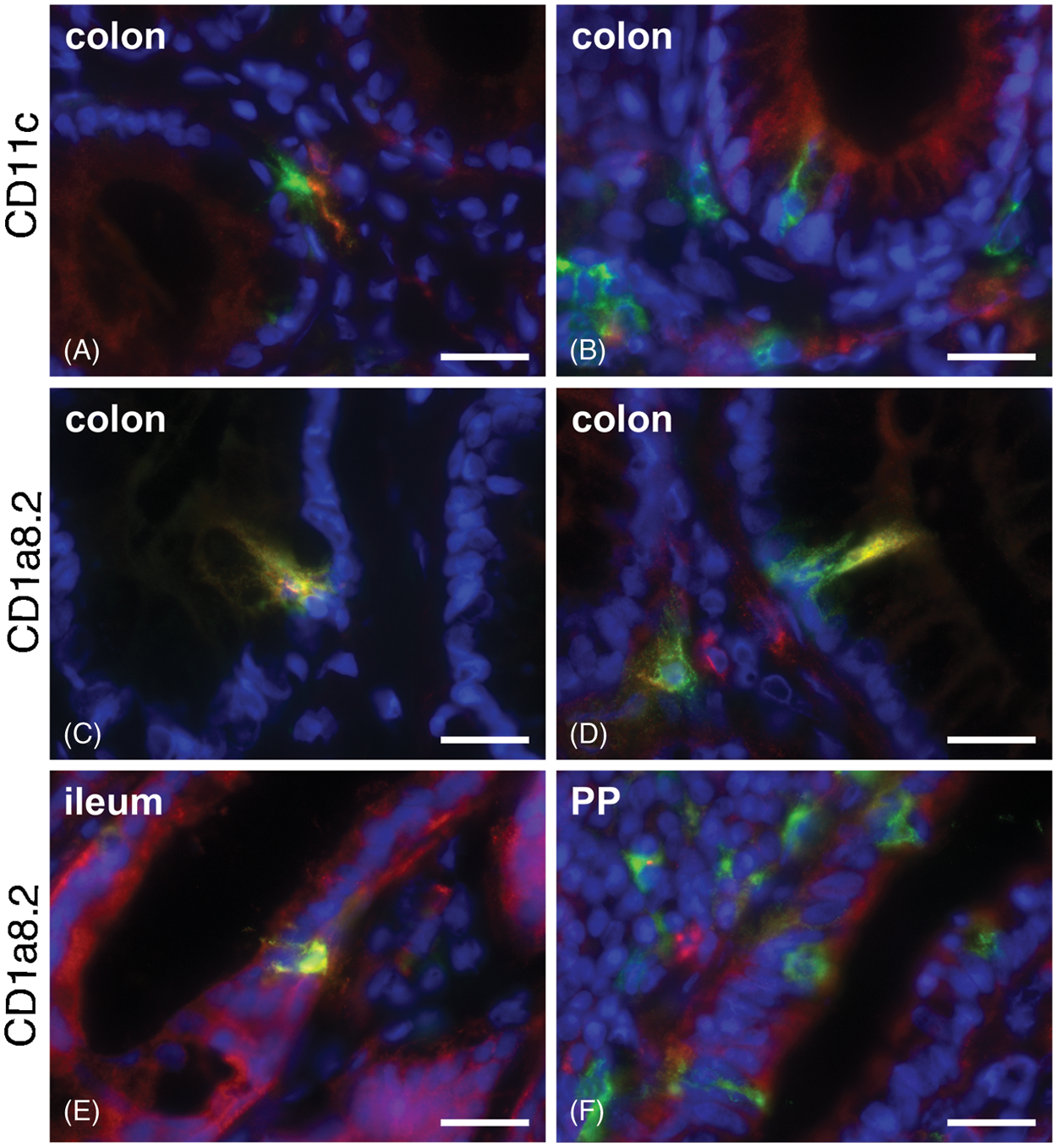
DC-like cells in canine IBD
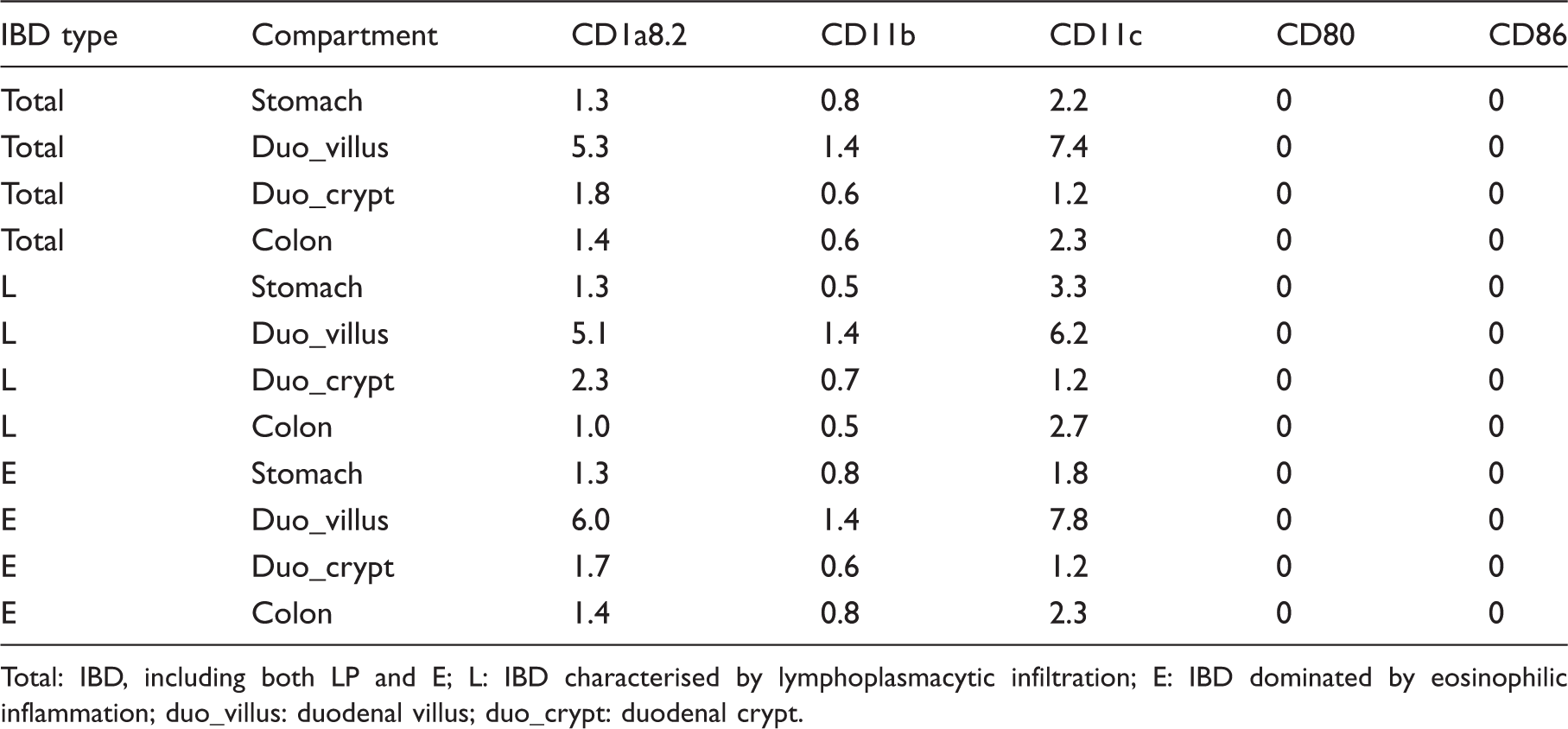
The number of LP DC-like cells in dogs with IBD is summarised in Table 4, given as median per group. In general, intestinal MHC II expression was increased in diseased dogs (semi-quantitative evaluation). However, numbers of CD1a8.2+ (P ≤ 1.993e-04), CD11b+ (P ≤ 0.03482) and CD11c+ DC-like cells (P ≤ 2.432e-05) were significantly decreased within the gastric, duodenal (villi and crypts) and colonic LP of dogs with IBD compared with controls (Figure 7).
DC-like cells in dogs suffering from IBD. In diseased dogs, numbers of CD1a8.2+, CD11b+ and CD11c+ DC-like cells were significantly decreased within the LP compared to controls (A–D). In contrast, amounts of CD86+ DC-like cells were slightly increased in the colonic LP (D). Boxplots always extend from minimal to maximal value and include median, as well as the lower and upper quartile (box). *P < 0.05; ***P < 0.001 by Wilcoxon rank sum test. DC-like cells in canine IBD. Medians of DC-like cells within the LP (double-positive for MHC II) are given as cells per 150,000 µm2. Total: IBD, including both LP and E; L: IBD characterised by lymphoplasmacytic infiltration; E: IBD dominated by eosinophilic inflammation; duo_villus: duodenal villus; duo_crypt: duodenal crypt.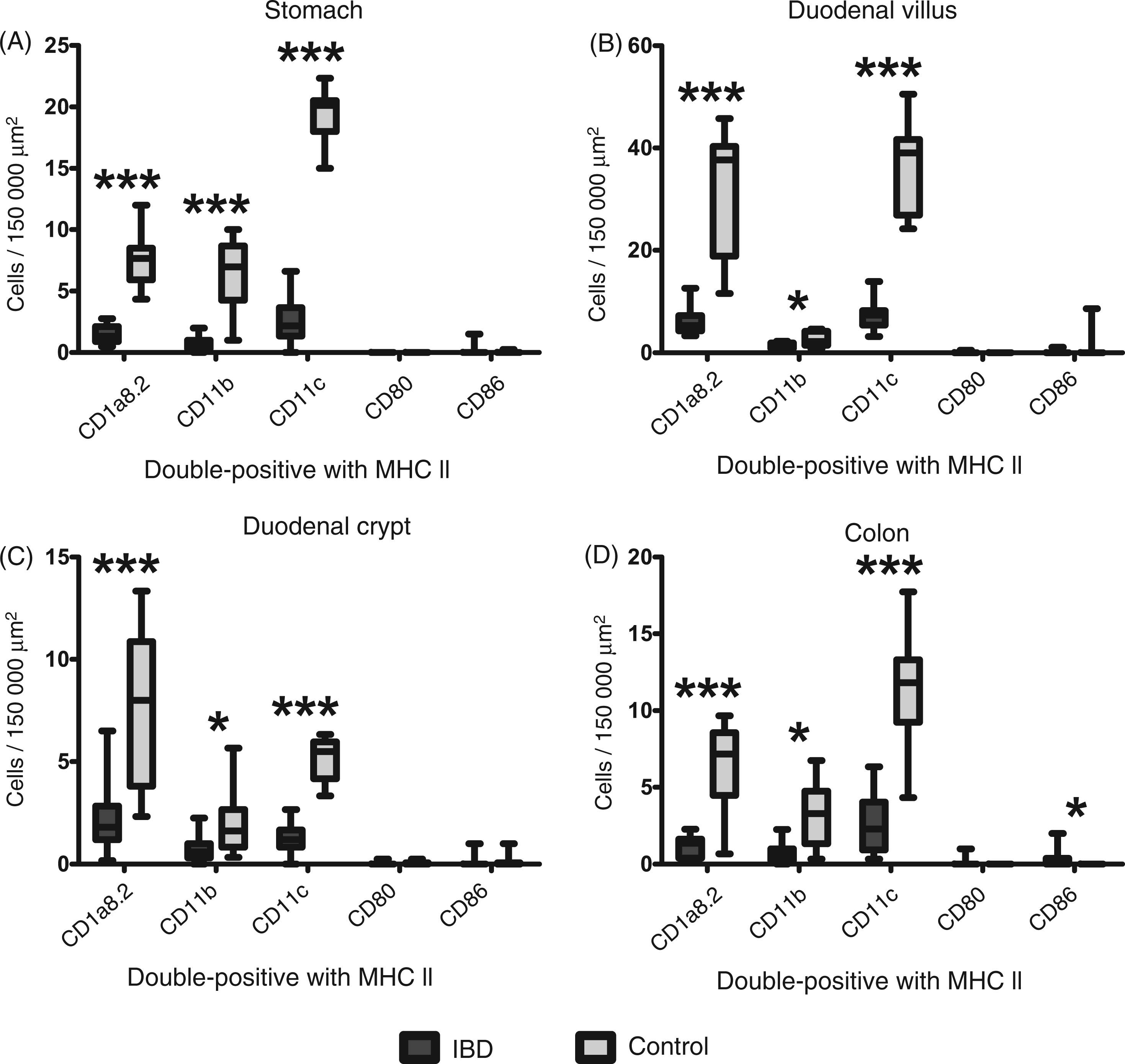
In contrast, a mild, but significant, increase in colonic CD86+ LP DC-like cells was present in dogs suffering from IBD (P = 0.01732). All dogs with increased numbers of CD86+ DC-like cells revealed colitis histopathologically (n = 8). However, 10 dogs (nos 2, 5, 6, 8, 10, 11, 17–19, 21) showed colonic inflammation without the presence of CD86+ DC-like cells within the LP. Furthermore, no differences between dogs with IBD and healthy controls were seen concerning the amounts of CD86+ DC-like cells within the LP of the stomach and duodenum, and CD80+ LP DC-like cells in all locations. Regarding Spearman’s rank correlation coefficient, there was a significant positive correlation between numbers of CD86+ LP DC-like cells and WSAVA scores in the colon of dogs with eosinophilic (P = 0.0011), but not in those with lymphoplasmacytic IBD (P = 0.7963) nor in IBD disregarding their histopathological type (P = 0.0812).
Finally, numbers of DC-like cells within the LP did not show any differences between canine eosinophilic and lymphoplasmacytic IBD.
Discussion
DCs induce and orchestrate immune responses at many sites of the body, and act as the sentinels of intestinal homeostasis and oral tolerance. 31 Accordingly, defects in DC function may be involved in the development of chronic intestinal inflammation due to exaggerated responses to commensal bacteria. 32 Therefore, the aim of the study was to analyse canine gut DCs in the steady state and in IBD.
Dogs used as controls lacked any clinical or pathomorphological signs of gastrointestinal disorders. However, slight histopathological alterations were present within the gut of these animals. According to a previous study, 33 these findings were interpreted as background variations of the normal intestinal mucosa, especially owing to the absence of clinical or macroscopical abnormalities. In contrast, dogs with IBD revealed significantly increased WSAVA scores reflecting their gastrointestinal pathology.
Interestingly, intestinal MHC II expression was not only present on lymphocytes and mononuclear cells resembling DCs or macrophages, but was also detected on enterocytes. This is similar to a previous study in dogs, 34 and suggests that enterocytes are potentially involved in antigen uptake and presentation. In canine IBD, MHC II expression was upregulated in the gut mucosa. This is in accordance with previous observations 24 and may be a consequence of general inflammatory processes within the intestinal mucosa.
Immunofluorescence of CD1a, CD11b, CD11c, CD80 and CD86, which are reported to be expressed on canine DCs, revealed positive medium-to-large-sized cells resembling phagocytes with obvious cytoplasmic projections that were double-positive for MHC II. Such cytoplasmic projections are the most characteristic morphological features of DCs if compared with macrophages. 29 Additionally, MHC II is classically found on professional APCs. 35 Therefore, we suggested them to be canine DCs. However, we cannot totally exclude an overlap with macrophages as they showed many phenotypic similarities in vitro, such as expression of MHC II, CD1, CD11b, CD11c, CD80 and CD86. 29 In spite of that, CD1a, CD11c, CD80 and CD86 are described as reliable markers of DCs in vivo.15,20,21 In contrast to the in vitro situation, in vivo expression of CD11b seems to be more related to macrophages than DCs. 15 Nevertheless, we defined double-positive phagocytes with clear cytoplasmic projections as DC-like cells in this study, as those markers are described on canine DCs in vivo and/or in vitro.
In dogs, the CD1 locus contains eight CD1A genes of which only three (CD1A2, CD1A6 and CD1A8) reveal complete sequences without indications of pseudogenes. 30 While transcripts of CD1A2 are only described in canine thymocytes, both CD1A6 and CD1A8 are transcribed in canine lymphoid and non-lymphoid tissues and two related canine CD1a8 proteins exist. 30 Owing to the lack of information about different CD1a proteins in the canine gut, we used Abs specific for CD1a6, CD1a8.1 and CD1a8.2 in this study. While CD1a6 was absent from the canine gut, CD1a8.2 was expressed in both LP and structured GALT parts of all control animals (n = 10). In contrast, CD1a8.1 was only detected in PPs of seven dogs. This is similar to a previous study, in which CA9.AG5 Abs recognised CD1a8.1 in canine thymocytes of approximately 60% of investigated individuals. 30 The presence of CD1a8.1+ DC-like cells in SEDs may indicate a special DC subset in canine PPs that might be particularly involved in antigen uptake beneath the FAE. Interestingly, CD1a8.1 was further detected on DC-like cells in the gut LP and MLNs of only four dogs, all of which revealed CD1a8.1 expression in SEDs simultaneously. Although this may indicate functional differences of CD1a8.1+ DC-like cells between individual dogs, further studies are needed to clarify its effect on mucosal immunity.
In the steady state, canine intestinal DC-like cells in the LP predominantly expressed CD1a8.2, CD11c and MHC II, while those expressing CD11b were present in lower quantities. This phenotype is consistent with previous reports on extraintestinal canine DCs evaluated in neoplastic disorders, healthy and diseased skin, and in vitro studies.15,16,21 Furthermore, LP DC-like cells in the canine gut lacked expression of both CD80 and CD86. This is similar to humans in which LP APCs, mostly supposed to be macrophages, revealed either low or absent expression of these molecules under steady state conditions.36–38
Functionally, phagocytosed and processed peptides are linked to MHC II within maturing DCs followed by their presentation to T cell receptors. 35 Additionally, CD1 represents a conserved family of glycoproteins in mammals relevant for capturing microbial and self lipid antigens with MHC-independent presentation to T cells. 39 In contrast to molecules mainly involved in antigen presentation, CD11b and CD11c are well known β2-integrins that belong to a family of adhesion molecules found on cellular surfaces and represent parts of different complement receptors (CRs). 40 Both CR3 (CD11b/CD18) and CR4 (CD11c/CD18) are present on immature DCs, 41 and they can bind to several ligands, including the intercellular adhesion molecule 1, fibrinogen, as well as bacterial, parasitic or fungal products.42,43 Additionally, CR3 and CR4 are able to bind the complement fragment iC3b and therefore mediate uptake of opsonised microbes and apoptotic cells, 44 which further inhibits DC maturation and down-regulates CR3 and CR4 expression by DCs. 41 In contrast, CD80 and CD86 serve as co-stimulatory molecules that are important for antigen presentation in T cell areas of lymphoid organs and are therefore upregulated on mature DCs. 45 Therefore, the expression of CD1a, CD11b and CD11c in addition to the absence of co-stimulatory molecules suggests canine intestinal DC-like cells to be immature cells in the steady state that are specialised for peripheral uptake of antigens, and possibly involved in mediation of tolerance via clearance of microbes and apoptotic cells.
The expression of CD11c and MHC II by intestinal DCs represents a widely conserved phenotype that is reported in humans, mice, cattle and sheep, although DCs are occasionally subdivided based on the expression of additional markers, such as CD103 and chemokine receptor CX3CR1 in mice.14,46–48 Additionally, CD1a is found on porcine immature DCs 49 and is further known as a specific marker for human 50 and canine 21 Langerhans cells. Although CD11b+ DCs are described in mice, sheep and pigs,51,52 double expression of CD11b and MHC II is also reported on canine macrophages. 53 Therefore, this co-expression found on canine intestinal DC-like cells may either indicate their monocyte origin or might suggest an overlapping phenotype with macrophages.
Beside the LP, high amounts of DC-like cells were present in organised parts of the canine GALT generally indicating their essential role in antigen presentation and initiation of immune responses including the induction of tolerance. In canine PPs, we found CD1a8.2+, CD11c+ and, sparsely, CD11b+ DC-like cells in SEDs beneath the FAE. Additionally, CD11c+ DC-like cells were visible in IFRs and germinal centres. The expression of MHC II and CD11c is also reported for DCs in murine, human and ovine PPs, whereas those in mice, sheep and pigs are further subdivided based on the presence of CD11b.51,54–57 In MLNs, the phenotypes of DC-like cells were quite similar to those in PPs except for a slight increase in CD11b+ cells. Although these CD11b+ DC-like cells in MLNs may have originated from other sites than PPs as CD11b was only scattered in SEDs, further studies are needed to elucidate possible migration of distinct DC subsets between different compartments of the canine GALT.
In contrast to the LP, CD80+ and CD86+ DC-like cells were present in both PPs and MLNs. These cells revealed obvious extensions in contact with surrounding lymphocytes suggesting them to be interdigitating DCs involved in antigen presentation. This is similar to lymphoid organs in mice, 58 and shows canine PPs and MLNs, rather than the LP, as the main site of antigen presentation in the context of co-stimulatory signals.
Additionally, the numbers of canine LP DC-like cells were quantified. Concerning their vertical distribution, numbers of both CD1a8.2+ and CD11c+ DC-like cells were significantly increased in small intestinal villi compared with crypts. Similar differences of CD11b+ DC-like cells were only present within the jejunum. An equal distribution of T cells was described in previous studies, suggesting a stronger exposition of the apical intestinal surfaces to luminal antigens.59–61 Therefore, the increased numbers of DC-like cells in villi may reflect a high antigenic load in the apical region leading to increased antigen sampling by DCs. Regarding their horizontal distribution, there was a slight decline of CD1a8.2+ and CD11c+ DC-like cells from duodenal to ileal crypts, as well as of those expressing CD11c from duodenal to ileal villi. This is in accordance with a recent investigation of canine Tregs, 33 whereas no horizontal differences are described for the intestinal expression of CD3, CD4 or CD8.59,61 The decline of DC-like cells and Tregs may be due to changes in the composition of luminal antigens, as food proteins are progressively destroyed along the small intestine, while amounts of bacteria increase from duodenum to colon. 62 However, further studies are needed to clarify possible differences between food proteins and commensal bacteria concerning their interaction with DCs.
Furthermore, we detected DC-like within the LP cells showing cytoplasmic projections between epithelial cells into the intestinal lumen. DCs exhibiting similar projections are also reported in mice expressing both CD11c and MHC II. 63 Additionally, murine CX3CR1+ cells with transepithelial dendrites are described, 47 and recent studies suggest they represent a specific non-migratory cellular subset rather than classical DCs migrating to the MLNs. 64 Nevertheless, the presence of transepithelial processes indicates direct sampling of luminal antigens by DCs that is also confirmed in vitro. 65 In addition to mice, DCs with transepithelial processes are also present in the jejunum and colon of rabbits (MHC II+) 66 as well as in the porcine jejunum (CD16+ CD11b+). 52 Therefore, transepithelial projections in dogs suggest direct sampling of luminal antigens by canine DCs. This is in addition to antigen uptake across the FAE of PPs, which might be due to the high density of DC-like cells in apical SEDs. Interestingly, CD1a8.2+ MHC II+ transepithelial dendrites were also present within the FAE of PPs. Therefore, further studies are needed to investigate whether the uptake of antigens in PPs is only supported by specialised epithelial cells with a microfold apical surface (M cells), or if it is also mediated by DCs with trans-cellular projections penetrating M cells within the FAE, as recently described in mice. 67
Breakdown of intestinal homeostasis, including a failure of oral tolerance and altered interaction with commensal bacteria, is suggested to be crucial for the pathogenesis of both human and canine IBD.13,68 At the intestinal border, DCs act as the watchman of both the induction of immune response to pathogens and maintenance of tolerance. 31 In mice, it is suggested that conditioning of DCs in the steady state in the context of retinoid acid and TGF-β is essential for the expansion and homing of Tregs in the intestinal mucosa. 4 Otherwise, CD80 and CD86 increase in inflamed mucosa of mice suffering from experimental colitis and disease manifestation can be prevented by blocking of CD80 in vivo.69–71 In human IBD, DCs are recruited to the inflamed mucosa and reveal upregulation of several co-stimulatory molecules, including CD40, CD80, CD83 and CD86.37, 72–74 Those DCs further show upregulation of TLR2 and TLR4, increased production of pro-inflammatory cytokines and enhanced response to LPS.32,75 Concerning β2-integrins in human IBD, CD11a was decreased on peripheral blood lymphocytes in a study on Crohn's Disease and Ulcerative Colitis, whereas CD11b was similar to healthy individuals. 76 Additionally, CD11c expression on blood DCs of IBD patients is reported to be equal to controls. 37 Within the intestinal mucosa, CD11a, CD11b and CD11c were upregulated in the colon, but not in the ileum of both Crohn's Disease and Ulcerative Colitis patients in one study. 77
Interestingly, CD1a8.2+, CD11b+ and CD11c+ DC-like cells were significantly decreased in the LP of diseased dogs. This is similar to a recent study on CD11c in canine IBD 25 and paediatric patients with Crohn's Disease, 78 and indicates DCs to be involved in the pathogenesis of canine IBD. In the steady state, maintenance of intestinal homeostasis mainly depends on regulatory capacity, but also requires a distinct inflammatory activity to ensure integrity of the intestinal border and to prevent its penetration by intestinal microbiota.5,79,80 Generally, a decline of mucosal DCs may result in lower capacity of antigen recognition and presentation. Therefore, their reduction in canine IBD could enhance disturbances of the commensal microflora—as described for both human and canine IBD13,81—by shrinking the inflammatory activity of the gastrointestinal mucosa. Additionally, expression of CD1a, CR3 and CR4, but absence of CD80 and CD86, suggest canine intestinal DCs to be tolerogenic in the steady state, although this has to be elucidated by additional analyses. Thus, the reduction of mucosal DCs further implicates a loss of tolerogenic capacity in canine IBD that may trigger chronic inflammation. Despite this, functional studies are needed to evaluate possible markers for canine inflammatory DCs, which would allow investigation of whether this downregulation of molecules illustrates a phenotypic change from tolerogenic to inflammatory subsets, rather than a general reduction of mucosal DCs in canine IBD.
Furthermore, there was a slight upregulation of CD86+ DC-like cells in the colonic mucosa of dogs suffering from IBD, which correlated with the presence of colitis. This is similar to human IBD and murine experimental colitis, and might indicate an activation of colonic DCs. Interestingly, numbers of CD86+ DC-like cells were positively correlated with histopathological scores only in dogs with eosinophilic IBD. However, CD86 expression in IBD was similar to controls in other than colonic localisations. As already known, the number of luminal bacteria in dogs progressively increases from duodenum to colon. 62 Additionally, abnormal interactions between the mucosal immune system and commensal microbiota are suspected to trigger both human and canine IBD.13,81 Therefore, the increase of CD86+ DC-like cells in the colonic LP of diseased dogs may reflect excessive exposure to luminal bacteria and might further indicate a pathogenetic role of DCs in canine IBD.
In summary, we showed that the canine gut hosts a complex network of DC-like cells in both structured and diffuse parts of the GALT that might be involved in active sampling of luminal antigens. Their phenotypic characterisation constitutes the basis for further studies concerning their function in gut homeostasis and pathogenic conditions. Furthermore, the shift of intestinal DC-like cells in diseased dogs implicates their pathogenetic significance for canine IBD.
Footnotes
Funding
Johannes Junginger received a scholarship from the German National Academic Foundation.
Conflict of interest
The authors declare that there is no conflict of interest.
Acknowledgements
The authors thank Bettina Buck, Petra Grünig, Kerstin Rohn, Danuta Waschke, Ines Blume and Klaus-Peter Kuhlmann for excellent technical assistance. Additionally, we are grateful to Dr Martin Beyerbach (Department of Biometry, Epidemiology and Information Processing, University of Veterinary Medicine Hannover, Germany) for statistical assistance; Prof. Dr Christiane Pfarrer, Dr Nina Hambruch and Dr Jan-Dirk Häger (Department of Anatomy, University of Veterinary Medicine Hannover, Germany) for providing the fluorescence microscope; the colleagues of the Small Animal Clinic (Duisburg-Rheinhausen, Germany) for obtaining endoscopic biopsies of diseased dogs; and Vanessa Herder PhD, Florian Hansmann PhD, Maximilian Iseringhausen and Eva Christin Junginger for their helpful discussions and comments on the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
