Abstract
CpG-oligonucleotide (ODN)-induced TLR9 activation exerts anti-inflammatory effects. TREM-1 is a DAP12-associated receptor, which is up-regulated in response to LPS-mediated TLR4 activation, and plays an essential role in innate immune response by augmenting the production of pro-inflammatory chemokines and cytokines. TREM-1 up-regulation resulted in a grave outcome in animal models, and in patients with sepsis and rheumatoid arthritis, while its soluble form (sTREM-1) exerted anti-inflammatory effects. We hypothesized that CpG-ODN regulates membrane TREM-1 expression and sTREM-1 shedding. The effect of CpG-ODN-induced TLR9 activation on TREM-1 expression and shedding was studied in mouse peritoneal macrophages and the mouse macrophage cell line RAW 264.7. While TREM-1 expression was not altered by CpG-ODN alone, stimulation with both LPS and CpG-ODN significantly abrogated TREM-1 LPS-induced up-regulation. Moreover, CpG-ODN-induced TLR9 activation either alone or in combination with LPS resulted in a significant increase of supernatant sTREM-1. The release of sTREM-1 was correlated positively with MMP-9 activity and was inhibited by chloroquine. These results suggest (i) a novel CpG-ODN-induced TLR9 pathway for the regulation of macrophage TREM-1 expression and MMP-9-mediated TREM-1 shedding; and (ii) a novel mechanism for an anti-inflammatory effect of CpG-ODN through abrogation of LPS-induced membrane TREM-1 up-regulation and increased MMP-9-mediated TREM-1 shedding.
Keywords
Introduction
The TLR family of pattern recognition is largely responsible for mediating macrophage activation by exogenous pathogens and is associated with the pathogenesis of infectious and autoimmune diseases. 1 TLR4 is a membrane surface receptor activated by the Gram-negative bacterial cell envelope component LPS, while TLR9 is localized in the endoplasmic reticulum and recognizes microbial unmethylated CpG-DNA or synthetic CpG-oligonucleotide analogs containing a CpG-oligodeoxynucleotide (ODN). Both TLR4 and TLR9 have been shown to be involved in the activation of innate immunity in infectious and autoimmune diseases, such as rheumatoid arthritis (RA).2,3 TLR9 activates macrophages through activation of metalloproteinase (MMP)-9 via NF-κB signaling pathway. 4 CpG-ODN-induced TLR9 activation has been shown to have a therapeutic benefit in cancer, infectious diseases and allergy. 3 Moreover, arthritis is aggravated in TLR9 knock-out mice 3 and TLR9 deficiency leads to disease exacerbation in various lupus mouse models. 5 Thus, TLR9 agonists may be used as effective targeted immune modulators as was previously shown in vaccine adjuvants, and therapies in cancer and infectious diseases. 3
Triggering receptor expressed on myeloid cells-1 (TREM-1) is a DAP12-associated receptor expressed on monocytes, macrophages and neutrophils,6–8 that functions as an amplifier of the TLR4-mediated inflammatory response.9,10 TREM-1 is up-regulated by TLR4 activation and induces pro-inflammatory cytokine, such as TNF-α, IL-1b, IL-8 and GM-CSF, release, as has been shown in human and mouse model of sepsis. 10 A soluble form of TREM-1 (sTREM-1) has been shown to be released during human sepsis. Soluble TREM-1 might pose an anti-inflammatory effect following an infection, as suggested by its inverse correlation with TNF-α and IL-1β level in murine sepsis. Recently, TREM-1 up-regulation was demonstrated in synovial fluid and cells of patients with rheumatoid arthritis (RA), as well as in the murine model of RA (collagen-induced arthritis; CIA). 11 In addition, TREM-1-derived peptides, which are designed to inhibit the interaction between TREM-1 and its unknown ligand, has been shown to be protective in animal models of endotoxemia and sepsis,12,13 as well as inflammatory bowel disease. 14 Furthermore, it was suggested that sTREM-1 shedding is generated through MMP-mediated proteolytic cleavage of membrane anchored TREM-1. 15 Taken together, TREM-1 plays a major role in infectious and autoimmune inflammatory diseases, and sTREM-1 exerts anti-inflammatory and protective effects, although its mode of protective action has not been fully explored.
In this study we sought to determine the effect of CpG-ODN-induced TLR9 activation on the expression of mouse macrophage surface TREM-1 expression and release of its soluble form. Using murine peritoneal macrophages and a macrophage RAW 264.7 cell line, we were able to demonstrate that CpG-ODN binding to TLR9 abrogates the effect of LPS on macrophage TREM-1 expression, whereas sTREM-1 level increases in response to CpG-ODN alone or in combination with LPS. We demonstrate that CpG-induced TREM-1 shedding is mediated by increased MMP-9 enzymatic activity and inhibited by chloroquine (CQ). Our results suggest a novel TLR9 mediated pathway for the regulation of membrane TREM-1 expression and sTREM-1 shedding which provide new insight to the mechanism of CpG-ODN as an immune modulator and the potential implications of sTREM-1 as an anti-inflammatory molecule.
Materials and methods
Peritoneal macrophage purification
C57BL/6 J (6–8-wk-old) mice were injected i.p. with 4% thioglycollate to induce chemical sterile peritonitis. Four d after the induction peritoneal cavities were lavaged with sterile RPMI 1640 (Biological Industries, Beit Haemek Ltd, Israel) culture medium. Hypotonic lysis of erythrocytes following 2 washes with PBS and adherence of the cells in a 12-well plate (1 × 106 cells/ml) for 90 min. Non-adhered cells were removed by washing the cells with medium RPMI 1640 containing 10% FBS. The cell monolayers were routinely found to contain >95% macrophages.
Cell culture
RAW 264.7 macrophage cell-line was obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were maintained in RPMI 1640 with 10% FBS, penicillin and streptomycin. Viability was assayed using trypan blue dye exclusion, and was typically >95%. Cells were plated 5 × 105 cells/ml in 12-well plates and treated as follows.
Macrophages stimulation
The adhered macrophages were treated with LPS (L880, Escherichia coli; Sigma (Saint Louis, Missouri, USA)) CpG-ODN (Type A ggGGTCAA
FACS
Peritoneal macrophages or macrophages RAW cells were incubated at 4°C for 40 min with anti-mouse TREM-1-phycoerythrin (PE)-conjugated (FAB1187P; R&D Systems (Minneapolis, MN, USA)) or anti-mouse CD11b-FITC-conjugated (Jackson). After washing, cell were resuspended in 300 µl of flow buffer and subjected to flow analysis using a Becton Dickinson flow cytometer (Becton Dickinson). Surface expression of TREM-1 or CD11b was expressed as mean fluorescence intensity (MFI).
ELISA
Conditioned media were centrifuged at 1000 g for 10 min to obtain cell-free supernatants and stored at –20°C. The soluble TREM-1 level was measured in the conditioned mediums collected from the treated macrophages and analyzed by ELISA according to the manufacturer's instructions (R&D Systems). The sTREM-1 level was established in a sandwich assay by comparison with standard curve according to the manufacturer's recommendation. All measurements were performed in duplicate.
MMP-9 gelatin zymography
A gelatin zymography assay system was employed in order to identify MMP-9 activity. Proteins samples of treated macrophages culture medium were loaded and electrophoresed in 7.5% SDS-PAGE containing 200 µg/ml gelatin (Sigma). Gels were washed with 2.5% Triton-X100 for 1 h and incubated at 37°C for 20 h in a reaction buffer containing 0.2% Brij 35 (Sigma), 5 mM CaCl2, 1 mM NaCl and 50 mM Tris at pH 7.4. Then, the gels were stained with Coomassie Brilliant Blue R-250 (0.2%) in 40% methanol and 10% acetic acid. MMP-9 activity was detected as appeared clear bands.
Statistical analysis
The results are expressed as mean ± standard deviation (SD). Variables were compared using an independent t-test. The correlation between variables was evaluated with Spearman's rank test (double-side) and correlation curve test. A P-value of ≤0.05 was considered statistically significant.
Results
CpG-ODN abrogates the effect of LPS on macrophage TREM-1 expression
The effect of CpG-ODN-mediated TLR9 activation on macrophage TREM-1 was studied using mouse peritoneal macrophages and the mouse macrophage cell line RAW 264.7. Mouse peritoneal macrophages and RAW 264.7 cells were plated into wells (5 × 105 cells/well) and stimulated for 18 h with LPS (50–500 ng/ml) or CpG-ODN (0.5, 1 or 3 µM), or both LPS (for peritoneal cells 200 ng/ml and RAW cells 500 ng/ml) with CpG-ODN (1 and 3 µM), or CpG-ODN negative control (1 and 3 µM). As shown previously, LPS induced a significant increase in membrane TREM-1 expression compared with untreated cells (Figure 1A , C) (for peritoneal cells MFI = 229 ± 19.4 vs 99.35 ± 5.4, respectively, P < 0.001 and for macrophages cell line MFI = 97 ± 7.56 vs 28 ± 2.95, P < 0.001 respectively). In addition, membrane expression of TREM-1 was not significantly altered by CpG-ODN alone, as shown previously (Figure 1A, C; for peritoneal cells MFI = 75.19 ± 3.3 and for macrophages cell line MFI = 24.52 ± 2.1).
16
However, we were surprised to find that macrophage activation with both LPS and CpG-ODN (1 or 3 µM) significantly abrogated the up-regulatory effect of LPS on TREM-1 expression (for peritoneal cells MFI = 86.03 ± 8.6 vs 229 ± 19.4, P < 0.005, and for RAW cells MFI = 55.7 ± 5.68 vs 97 ± 7.56, P < 0.05 respectively) (Figure 1). Figure 1B displays a representative FACS analysis of four experiments analyzing surface TREM-1 expression on peritoneal LPS and CpG-activated macrophages alone or in combination after 18 h stimulation. The TREM-1 surface expression in the macrophages was compared with CpG-ODN negative control and resulted in no effect on the expression of TREM-1. Peritoneal macrophages that expressed TREM-1 also expressed CD11b molecule (data not shown). Consequently, CpG-ODN-induced TLR9 activation strongly inhibited the effect of LPS on the surface expression of TREM-1.
The inhibitory effect of CpG-ODN on TREM-1 expression in macrophages. Peritoneal macrophage cells (A) and macrophages RAW 264.7 cell line (C) were treated with either CpG-ODN (0.5–3 µM) or LPS (50–500 ng/ml), or both for 18 h and the expression of surface TREM-1 was analyzed using FACS. The cells were incubated with anti-TREM-1 PE-conjugated Ab (black bars) or anti-IgG2A PE-conjugated (grey bars) (for peritoneal macrophages n = 3 *P < 0.001 and **P < 005; for RAW 264.7 macrophages n = 5 *P < 0.001 and **P < 0.05). (B) A representative histogram of three independent experiments analyzing TREM-1 expression in peritoneal macrophages. Bars represent mean ± standard errors.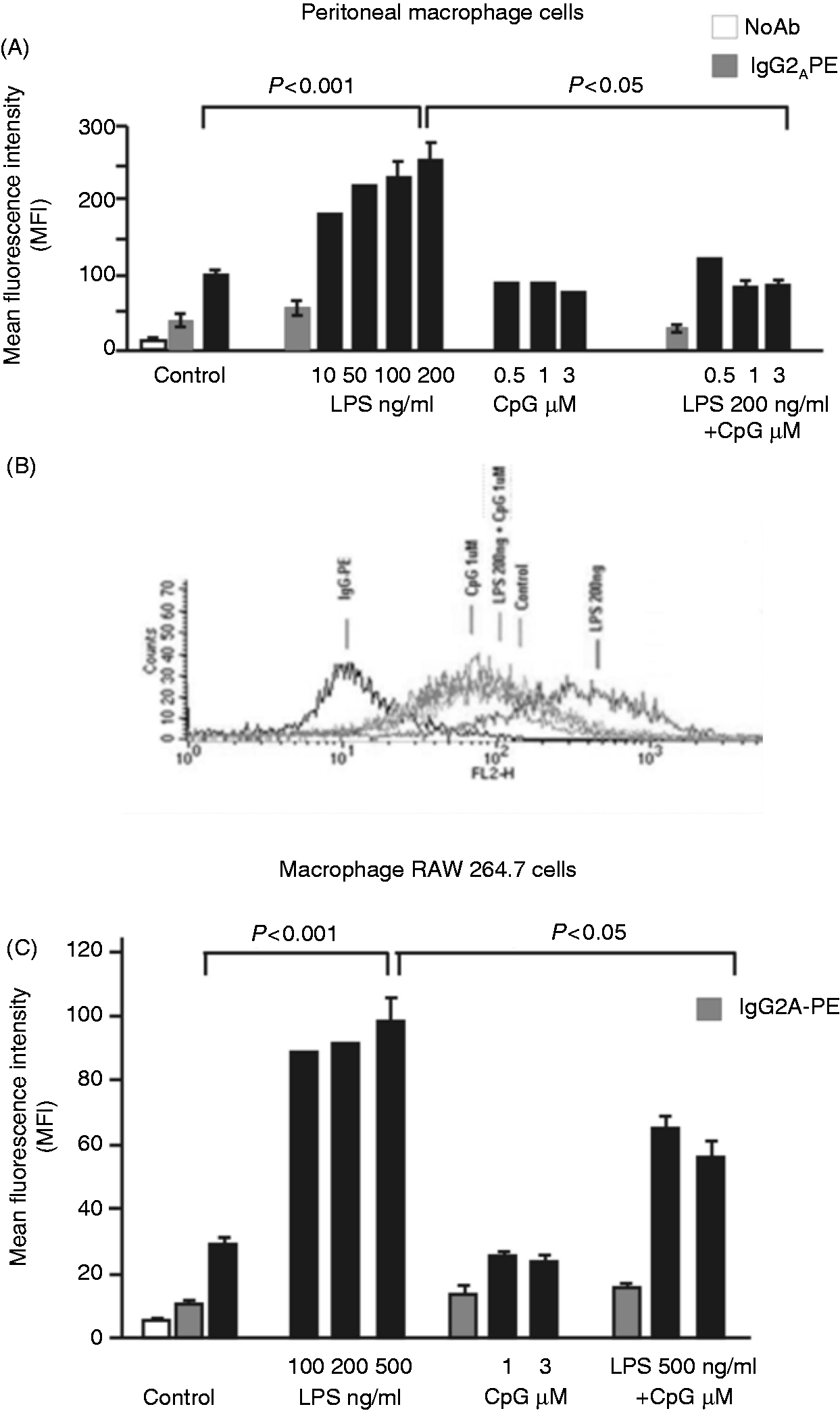
CpG-ODN induces elevation of sTREM-1 level
In addition to TREM-1 surface expression, we assayed macrophage supernatant sTREM-1 level of the treated cells. Supernatants were collected 18 h after the macrophages were treated, and analyzed for sTREM-1 level (Figure 2A, B). CpG-ODN and LPS, each separately, induced a significant increase of supernatant sTREM-1 level in both cell types compared with untreated cells or CpG-ODN negative control (for peritoneal macrophages LPS = 39.1 ± 5.12 pg/ml and CpG-ODN = 33.17 ± 5.9 vs control = 17.5 ± 3.5, P < 0.001; and for RAW cells LPS = 48.5 ± 4.36 pg/ml and CpG-ODN = 16.2 ± 2.74 vs control = 6.89 ± 1.4, P < 0.001). Moreover, macrophage activation by LPS-and CpG-ODN together induced a significantly higher supernatant level of sTREM-1 than with LPS or CpG-ODN separately (for peritoneal cells 54.43 ± 6.12 pg/ml, and RAW cells = 56.4 ± 5.36, P = 0.05). All together, our results suggest that CpG-induced TLR9 activation abrogates the effect of LPS on macrophage TREM-1 expression that parallels with an increase in the level of sTREM-1.
CpG-ODN treatment increased sTREM-1 release. (A) Peritoneal macrophage cells. (B) Macrophage RAW 264.7 cells were treated with LPS (200 ng/ml or 500 ng/ml, respectively) or CpG-ODN (1 and 3 µM), or both for 18 h. The release of sTREM-1 was analyzed by ELISA. For peritoneal cells n = 4, *P < 0.001, **P = 0.05 and for RAW 264.7 macrophages n = 5, *P < 0.001, **P < 0.05, ***P = 0.05. Bars represent mean ± standard errors.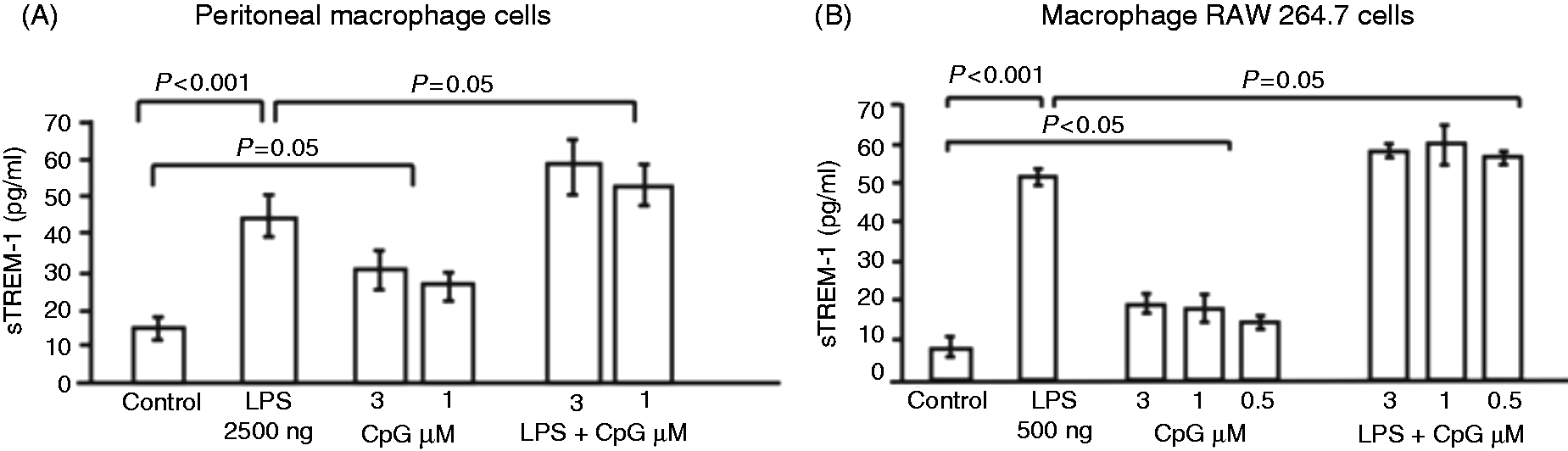
CpG-ODN-induced TLR9 activation and MMP-9 activity are associated with TREM-1 shedding
As the effect of CpG-ODN on macrophage RAW cells was similar to the peritoneal macrophage cells, the next experiments were conducted on RAW 264.7 cells only.
As shown previously in murine macrophage RAW 264.7 cells, CpG-ODN-induced MMP-9 expression and activation is mediated by TLR9 via NF-κB pathway and inhibited by CQ, a TLR9 and MMP-9 inhibitor.17,18 In addition, a proteolytic cleavage of membrane-anchored TREM-1 by general MMPs was suggested in several studies to be the mechanism for TREM-1 release.
15
Thus, we aimed to explore whether the effect of CpG-ODN on MMP-9 activity is correlated with the release of sTREM-1. RAW 264.7 cells were pretreated with CQ 2.5µg/ml for 2 h followed by treatment with LPS, CpG-ODN or both, and the supernatant sTREM-1 level was analyzed using an ELISA kit. CQ pretreatment significantly inhibited sTREM-1 release from the cells treated with CpG-ODN 1 µM or 3 µM compared with cells treated with CpG-ODN only (P < 0.05). The effect of CQ was also significantly observed in cells treated with both LPS and CpG-ODN, as shown in Figure 3A and summarized in Table 1. Macrophage pretreatment with CQ did not change membrane TREM-1 expression (data not shown). Our results suggest a MMP-9 mediated shedding of membrane TREM-1 in response to CpG-ODN- or LPS-induced macrophage activation. In order to confirm this observation, MMP-9 activity was assayed by means of a zymography method. Supernatant of LPS and CpG-ODN-activated macrophages, with or without CQ pretreatment was assayed for MMP-9 activity. As shown in Figures 3B and 3C, CQ (2.5 µg/ml) macrophage pretreatment significantly reduced MMP-9 activity (P < 0.05). Similarly, CQ pretreatment significantly reduced MMP-9 activity in macrophages stimulated with LPS and CpG combination (P < 0.05; Figure 3B). MMP-9 activity was determined using densitometry analysis and fold activity was measured per each experiment and compared with the control, which was defined as fold 1. The activity of MMP-9 was found to be significantly associated with sTREM-1 level, as shown in Figure 3D. Shedding of sTREM-1 correlated positively with MMP-9 activity (r = 0.914, P < 0.005), suggesting that TLR9-induced TREM-1 shedding is mediated by MMP-9 activation.
MMP-9 is associated with TREM-1 shedding. Macrophage RAW 264.7 cells were treated with LPS (500 ng/ml) or CpG-ODN (1 and 3 µM), or both in the presence or absence of CQ for 18 h. The conditioned media of the treated cells were collected and analyzed (A). The release of sTREM-1 using ELISA kit (B, C). MMP-9 activity densitometry. n = 3, *P < 0.05, bars represent mean ± standard errors (D). Correlation curve analysis of the 3 independent experiments for sTREM-1 and MMP-9 activity (r = 0.914; p<0.005). Chloroquine inhibited CpG-ODN induced sTREM-1 released.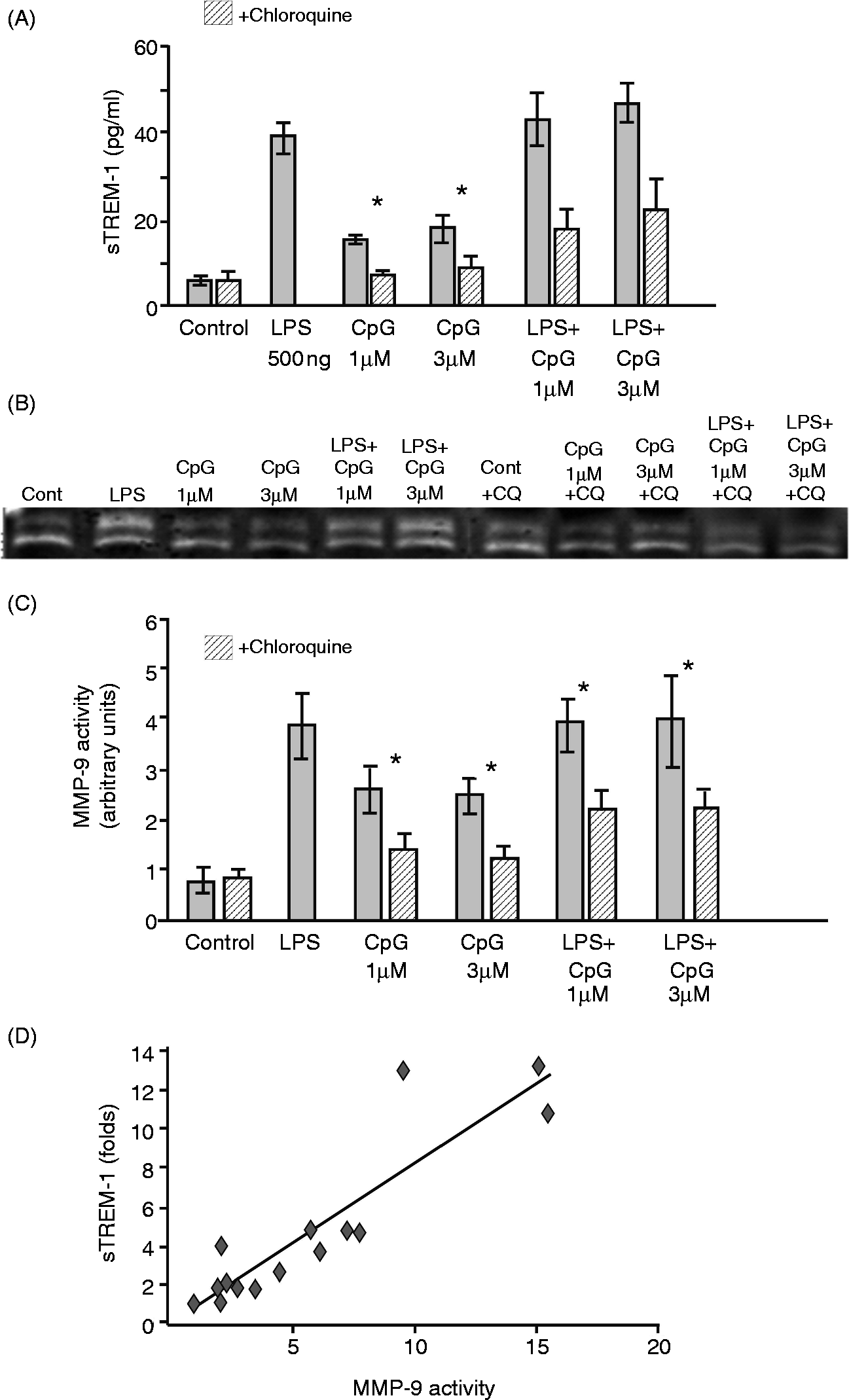

Discussion
Macrophage TREM-1 up-regulation and shedding of sTREM-1 were shown previously to be the result of LPS-mediated TLR4 activation and correlated with innate immune activation in sepsis, as well as in non-infectious inflammatory conditions.9,13 In the present study, we demonstrate, for the first time, that CpG-mediated TLR9 activation abrogates the effect of LPS on macrophage surface TREM-1 expression while it augments the effect of LPS with respect to sTREM-1 shedding, as well as increasing the sTREM-1 level by itself. Moreover, we were able to show that TREM-1 shedding is correlated with MMP-9 activation. Thus, our results suggest a novel TLR9-mediated pathway for the regulation of macrophage TREM-1 surface expression and shedding.
Previous studies have shown that TREM-1 up-regulation occurs though TLR4 activation while other TLRs have no effect on TREM-1 expression. 12 Various studies have addressed the integration of TREM-1 with other TLRs. 19 It has been shown previously that stimulation of monocytes with CpG-ODN had no effect on the expression of TREM-1 gene, as well as no effect on cell-surface TREM-1 expression. 16 However, we demonstrate that CpG-ODN-induced TLR9 activation exerts a bimodal effect on TREM-1. Whereas TLR9 activation does not change macrophage surface TREM-1 expression, it induces a MMP-9-associated increase in sTREM-1 level (Figures 1–3). Moreover, CpG-ODN inhibits the effect of LPS on TLR4-mediated surface TREM-1 up-regulation, as well as augmenting the effect of LPS-induced increase of sTREM-1 level. Macrophage surface TREM-1 up-regulation was associated with a grave outcome and increased mortality in animal models of sepsis, as well as in infectious diseases in humans, and binding of its unknown ligand was shown to synergize with the effect of LPS by amplifying the production of pro-inflammatory cytokines.9,10 However, TREM-1/3 deficiency resulted in increased mortality in a mouse model of Pseudomonas aeruginosa-induced pneumonia, and increased local and systemic cytokine production, as well as decreased neutrophil infiltration of the airways owing to inhibition of neutrophil transepihelial migration. 20 Thus, TREM-1 plays a pivotal role in the innate immune response against infectious insults.
The CpG ODNs are known to be potent immunomodulators and induce highly effective protective immunity in a number of infectious diseases, including tularemia, 21 listeriosis, 22 Leishmania 23 and Burkholderia mallei or Burkholderia pseudomallei infections.24–27 Similarly, other studies have demonstrated a protective effect of CpG-ODN on the outcome of sepsis in a murine model of hemorrhagic shock with a subsequent Staphylococcus aureus (methicillin-sensitive Staphylococcus aureus) pneumonia, 28 as well as in an animal model of acute polymicrobial sepsis. 29 In addition, soluble TREM-1 might pose an anti-inflammatory effect following an infection, as suggested by its inverse correlation with TNF-α and IL-1β level in murine sepsis. 9 Our results on the effect of CpG-ODN on LPS-mediated TREM-1 cell-surface expression and the effect of CpG-ODN on sTREM-1 level suggest a novel pathway to ameliorate the deleterious effect of LPS in acute bacterial infection, and might suggest a protective effect of CpG-ODN in human infectious conditions, such as sepsis and pneumonia.
TREM-1 up-regulation was demonstrated in synovial cells, and sTREM-1 level was shown to be elevated in synovial fluid and serum of patients with RA as well as in a murine model of RA (CIA).2,3 Blockade of TREM-1 using a recombinant adenovirus encoding extracellular domain of TREM-1 or a synthetic TREM-1 antagonistic peptide were shown to ameliorate arthritis in the murine model of CIA.11,30 Thus, our results of sTREM-1 up-regulation induced by CpG-ODN-mediated TLR9 activation suggests a novel pathway by which TREM-1 might play a protective or anti-inflammatory role in autoimmune inflammatory disorders, such as RA, in addition to its protective effects in infectious diseases.
Taken together, our findings on the inhibitory effect of CpG-ODN on LPS-mediated up-regulation of macrophage surface TREM-1 coupled with an increase in sTREM-1 level suggest a novel anti-inflammatory mechanism of TLR9. Establishing the anti-inflammatory effects of CpG-ODN via the regulation of TREM-1 might prove a new way of ameliorating inflammatory disorders in which exogenous or endogenous stimuli of TLR4 play a role.
MMPs are well known mediators of cell migration, proliferation, leukocyte influx and respondents to numerous pathological stimuli. 31 Macrophage production of MMPs is induced by pro-inflammatory stimuli, such as cytokines, chemokines and bacterial wall components.32,33 In addition, Gomez-Pina et al. 15 have demonstrated that human monocyte TREM-1 shedding is mediated by MMPs. We were able to show that CpG-ODN-induced macrophage TREM-1 shedding is mediated through MMP-9 activation. MMP-9 enzymatic activity was assessed in response to LPS and CpG-ODN macrophage activation with and without pre-treatment with CQ. The inhibitory effect of CQ on the activity and expression of MMP-9 correlated positively with sTREM-1 level. CQ inhibits both TLR9 and MMP-9, and our finding that shedding of sTREM-1 is inhibited by CQ supports the involvement of MMP-9 activation in TREM-1 shedding. Thus, our results suggest a novel MMP-9-mediated mechanism for macrophage TREM-1 shedding. However, CQ-induced reduction of sTREM-1, as shown in Table 1, did not reach the baseline (control) level, thus suggesting a possible another mediator that plays a role in the shedding of TREM-1.
In conclusion, our results suggest a novel mechanism for the regulation of macrophage TREM-1 expression mediated through CpG-ODN binding to TLR9 and a possible anti-inflammatory mode of action of CpG-ODN through MMP-9-mediated sTREM-1 shedding. Further studies are required to elucidate the biological significance of the regulatory effect of CpG-ODN on TREM-1 and might pave a road toward a novel therapeutic approach for the treatment of inflammatory human diseases.
Footnotes
Funding
This work was funded by the Research Authority, Beilinson Hospital, Rabin Medical Center, Israel.
