Abstract
The prototypic long pentraxin PTX3, a soluble pattern recognition receptor, plays an important role in innate defense against selected pathogens by favoring their elimination and the initiation of protective responses. PTX3 has notably beneficial effects in mice infected with Aspergillus fumigatus and Pseudomonas aeruginosa. Cystic fibrosis (CF), a severe inherited autosomal recessive disease, is characterized by recurrent lung infections, especially by these two pathogens. We thus hypothesized that the status of PTX3 may be altered in CF patients. Level and integrity of PTX3 were analyzed in the sputum samples from 51 CF patients and 7 patients with chronic obstructive pulmonary disease (COPD). The levels of PTX3 were increased in serums from CF patients, but low in their respiratory secretions. PTX3 concentrations in sputum samples were dramatically lower in CF patients than in COPD patients. The low concentration of PTX3 resulted from a proteolysis cleavage by elastase and A. fumigatus proteases. Interestingly, the N-ter domain of PTX3, involved in protection against A. fumigatus, is preferentially degraded by these proteases. These results indicate that the selective proteolysis of PTX3 in the CF lung may explain, in part, the recurrent lung infections by PTX3-sensitive pathogens in CF patients.
Introduction
Innate immune cells recognize microbes via a limited number of pattern recognition receptors (PRRs). PRRs recognize highly conserved motifs (pathogen-associated molecular patterns), expressed exclusively by microbes. PRRs are either cell-associated or soluble. 1 Cell-associated PRRs can be subdivided into two groups: (i) endocytic PRRs [such as scavenger receptors and C-type lectin receptors (CLR)], involved in the binding and internalization of microbes; and (ii) signaling PRRs (i.e. members of the TLR, NOD-like receptor and RIG-I-like receptor families) involved in immune cell activation.
Soluble PRRs, including mainly pentraxins and the soluble CLRs collectins and ficolins, facilitate the clearance of microbes by phagocytes. Pentraxins, a family of evolutionarily conserved proteins characterized by a pentameric structure, 2 are classified into two groups depending on the primary structure of the protomer: (i) the short pentraxins C-reactive protein (CRP) and serum amyloid P (SAP) (produced by hepatocytes); and, (ii) the long pentraxins, that differ from the short pentraxins for the presence of an unrelated N-terminal domain. 2 PTX3, the prototypic long pentraxin, is expressed by numerous cell types, including myeloid cells, endothelial cells and epithelial cells, but not by hepatocytes, upon stimulation with PRR agonists or pro-inflammatory cytokines. 2 PTX3 is also constitutively expressed by neutrophils. 3
Increased levels of circulating PTX3 have been reported in various infections, including sepsis and septic shock, meningococcal disease, tuberculosis and dengue infection.4–7 In all these situations, the levels of PTX3 are correlated with disease severity and have a prognostic value. PTX3 is involved in the resistance against a variety of fungi,8,9 bacteria10,11 and viruses. 12 PTX3 binds selected microbes, such as Aspergillus fumigatus and Pseudomonas aeruginosa, and facilitates their clearance by phagocytes.8,10,13 The protective role played by PTX3 in immunity has been evidenced in Ptx3−/− mice, which are susceptible to invasive pulmonary aspergillosis owing to a defective recognition of A. fumigatus conidia by phagocytes and the initiation of an inappropriate Th2-biased immune response. 8
Cystic fibrosis (CF) is a severe inherited autosomal recessive disease caused by mutations in the chloride channel CF transmembrane conductance regulator (CFTR) gene. 14 CF patients suffer from recurrent lung infections by the two opportunistic pathogens A. fumigatus and P. aeruginosa. Infections are one of the main causes of morbidity and mortality in CF. The local innate immune system in CF patients is profoundly impaired, as illustrated by an ineffective mucociliary clearance 15 and a dysregulated inflammatory response.16–18
Studies reported that Ptx3−/− mice are susceptible to A. fumigatus and P. aeruginosa infections, and that PTX3 protects mice against infections by these two pathogens.8,10 The aim of this study was thus to evaluate whether a qualitative or quantitative defect of PTX3 may contribute to the increased susceptibility of CF patients to some PTX3-sensitive opportunistic pathogens.
Materials and methods
Reagents
Recombinant PTX3, monoclonal anti-PTX3 Abs (clones MNB4 and 16B5) and polyclonal anti-PTX3 Ab have been described previously. 19 Recombinant SAP and CRP, and anti-SAP mAb were from Millipore (Bedford, MA, USA). The polyclonal anti-CRP Ab was from Abcam (Cambridge, MA, USA). Recombinant PTX3 and SAP were biotinylated, according to the manufacturer’s instruction (Pierce, Rockford, IL, USA). Recombinant neutrophil elastase (HNE), the HNE specific inhibitor Ala-Ala-Pro-Ala-chloromethylketone (AAPA-CMK), proteinase 3 (PR3) and cathepsin G (CatG) were from Merck (Darmstadt, Germany). The CatG inhibitors α-chymotrypsin and Ala-Ala-Phe-chloromethylketone (AAF-CMK), HNE inhibitor Ala-Ala-Pro-Val-chloromethylketone (AAPV-CMK) and PR3 inhibitor were kindly provided by Dr Korkmaz (Tours, France). The protease inhibitors pefabloc SC, aprotinin, leupeptin, E-64, antipain, phosphoramidon, EDTA-Na2, bestatin, chymostatin and pepstatin were from Roche (Mannheim, Germany). PMSF and GM6001 were from Sigma-Aldrich (St Louis, MO, USA) and Millipore, respectively. LPS (Escherichia coli serotype O111:B4) was from Sigma-Aldrich, IL-1β from Myltenyi Biotec (Bergisch Gladbach, Germany) and TNF-α from Immunotools (Friesoythe, Germany).
Sputum and serum collection
Demographic and clinical characteristics of CF and COPD patients.
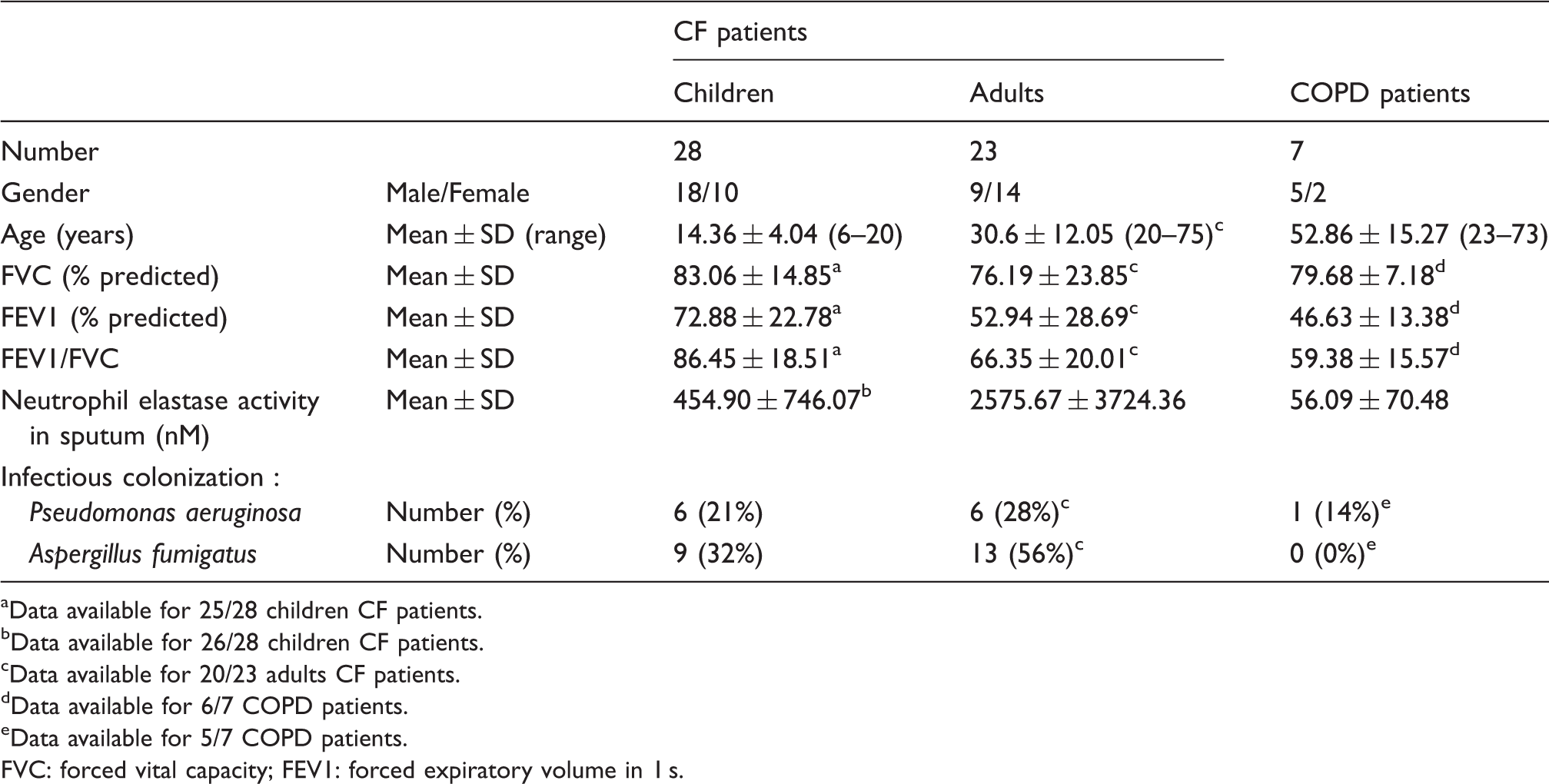
Data available for 25/28 children CF patients.
Data available for 26/28 children CF patients.
Data available for 20/23 adults CF patients.
Data available for 6/7 COPD patients.
Data available for 5/7 COPD patients.
FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s.
Culture of A. fumigatus
Conidia of A. fumigatus strain CBS 113.26 were obtained from 5-d cultures on yeast extract–peptone–dextrose agar plates or on Aspergillus minimal medium (to induce synthesis of proteases) by scraping off the mycelium in sterile water. After filtration through 28-µm diameter pore-size filters, conidia were pelleted by centrifugation (1500 g, 5 min), washed and enumerated. Culture filtrates were stored at –20°C.
Epithelial cell culture
The human epithelial cells IB3-1 (derived from a CF patient with a ΔF508/W1282X genotype 20 ) and S9 (derived from the IB3-1 cell line in which the CF phenotype was corrected 21 ) were maintained in LHC-8 culture medium (Invitrogen, Carlsbad, CA, USA) supplemented with 5% FCS (Biowest, Nuaillé, France) and cultured in fibronectin-coated flasks. Primary human bronchial epithelial cells were maintained in bronchial epithelial cell growth medium (both from Lonza, Verviers, Belgium). Epithelial cells were stimulated with LPS, IL-1β or TNF-α at the indicated concentrations.
Isolation and activation of human neutrophils
Peripheral blood neutrophils were isolated from CF patients and healthy donors, as reported previously. 3 Neutrophils (2 × 106 cells/ml) were cultured in RPMI 1640 medium containing 2% FCS (to maintain cell viability) or no FCS (to induce cell apoptosis 22 ) and supplemented with antibiotics (all from Lonza). Cells were stimulated with LPS, IL-1β plus TNF-α or A. fumigatus conidia at the indicated concentrations. Airway neutrophils from CF patients were isolated using a protocol adapted from Corvol et al. 23 Briefly, sputums were incubated for 30 min at 37°C with an equal volume of a 200 µg/ml EDTA–170 U/ml trypsin solution (Life technologies, Saint Aubin, France). After filtration on a 40 -µm diameter filter, sputums were centrifuged at 300 g for 5 min at 4°C. Neutrophils were isolated by depleting CD14+ and MHC-II+ cells (Miltenyi Biotec); the purity of neutrophils, based on CD66b expression, was >99% (data not shown). Cell death was evaluated by Annexin V–propidium iodide labeling.
Quantification of PTX3 by ELISA
PTX3 was quantified by ELISA, as described previously, 3 using the anti-PTX3 mAb MNB4 as a capture Ab and the anti-human PTX3 pAb as the detection Ab. Culture supernatants were incubated 2 h at 37°C, and sputum and serum samples overnight at 4°C. Recombinant PTX3 was used as a standard.
Elastase activity assay
HNE activity was determined as described previously. 24 Briefly, the chromogenic substrate Suc-Ala-Ala-Pro-Val-pNa (1 mM final concentration) (Sigma-Aldrich) was mixed with serial dilutions of sputum samples. Recombinant HNE was used as a standard. Plates were incubated in a 37°C warmed microplate reader (Molecular Devices, Sunnyvale, CA, USA). The absorbance at 405 nm was read every minute for 30 min. The change in OD/min in samples was converted to nM elastase.
Western blotting analysis of protein proteolysis
Biotinylated or non-biotinylated PTX3, SAP and CRP were incubated at 37°C for the indicated times in CF or COPD sputum samples (diluted 1:3 in PBS), or in A. fumigatus culture filtrates, containing or not containing protease inhibitors. In other experiments, pentraxins were incubated with HNE, CatG or PR3, with or without their respective inhibitors. The pentraxin integrity was analyzed by Western blotting. Briefly, samples were separated electrophoretically on a 4–20% gel (Bio-Rad, Hercules, CA, USA) in reducing conditions, and then transferred to an Immobilon membrane (Millipore). After saturation, membranes were incubated with 3 µg/ml anti-PTX3 pAb, anti-PTX3 mAbs (clones 16B5 or MNB4), anti-SAP mAb, anti-CRP pAb or with peroxidase-conjugated streptavidin (diluted 1:10,000; GE Healthcare, Buckinghamshire, UK). After washing, membranes were incubated with 1 µg/ml peroxidase-conjugated anti-rabbit IgG, anti-rat IgG, anti-goat IgG or anti-mouse IgG Abs (Invitrogen) when appropriate. Bound Abs were detected by chemiluminescence (Thermo Scientific, Rockford, IL, USA).
Statistical analysis
Data are shown as mean ± SD or mean ± SEM, and comparisons between groups or individuals were carried out using one-tail Mann–Whitney test or one-tail Wilcoxon matched pairs test respectively. Correlation of HNE activity and PTX3 degradation (scored from 0, multimeric PTX3, to 10, totally degraded) was assessed using the non-parametric Spearman rank correlation assay (correlation coefficient: r). Statistics were calculated using Prism software (GraphPad Software, La Jolla, CA, USA). A P-value <0.05 was considered statistically significant.
Results
Analysis of PTX3 expression in CF patients
PTX3 was quantified by ELISA in the serums from CF and COPD patients, and from healthy volunteers. The levels of PTX3 were significantly higher in the serums from CF patients (1.93 ± 1.55 ng/ml; mean ± SD, n = 51) than in the serums from healthy participants (1.16 ± 0.51 ng/ml; n = 27) (Figure 1A). PTX3 was also elevated in COPD patients (2.03 ± 1.06 ng/ml; n = 7) (Figure 1A).
Status of PTX3 in CF serum and sputum samples. (A, B) Quantification of PTX3 by ELISA in serum samples (A) and sputum samples (B) from 51 CF patients (23 adults + 28 children) and 7 COPD patients. The levels of PTX3 were also determined in serum samples from 27 healthy subjects (A). Bars represent means; *P < 0.05, **P < 0.005. (C) Individual analysis of PTX3 in serum and sputum samples from 51 CF patients (23 adults + 28 children) and 7 COPD patients. Bars represent means; *P < 0.05, ***P < 0.0001.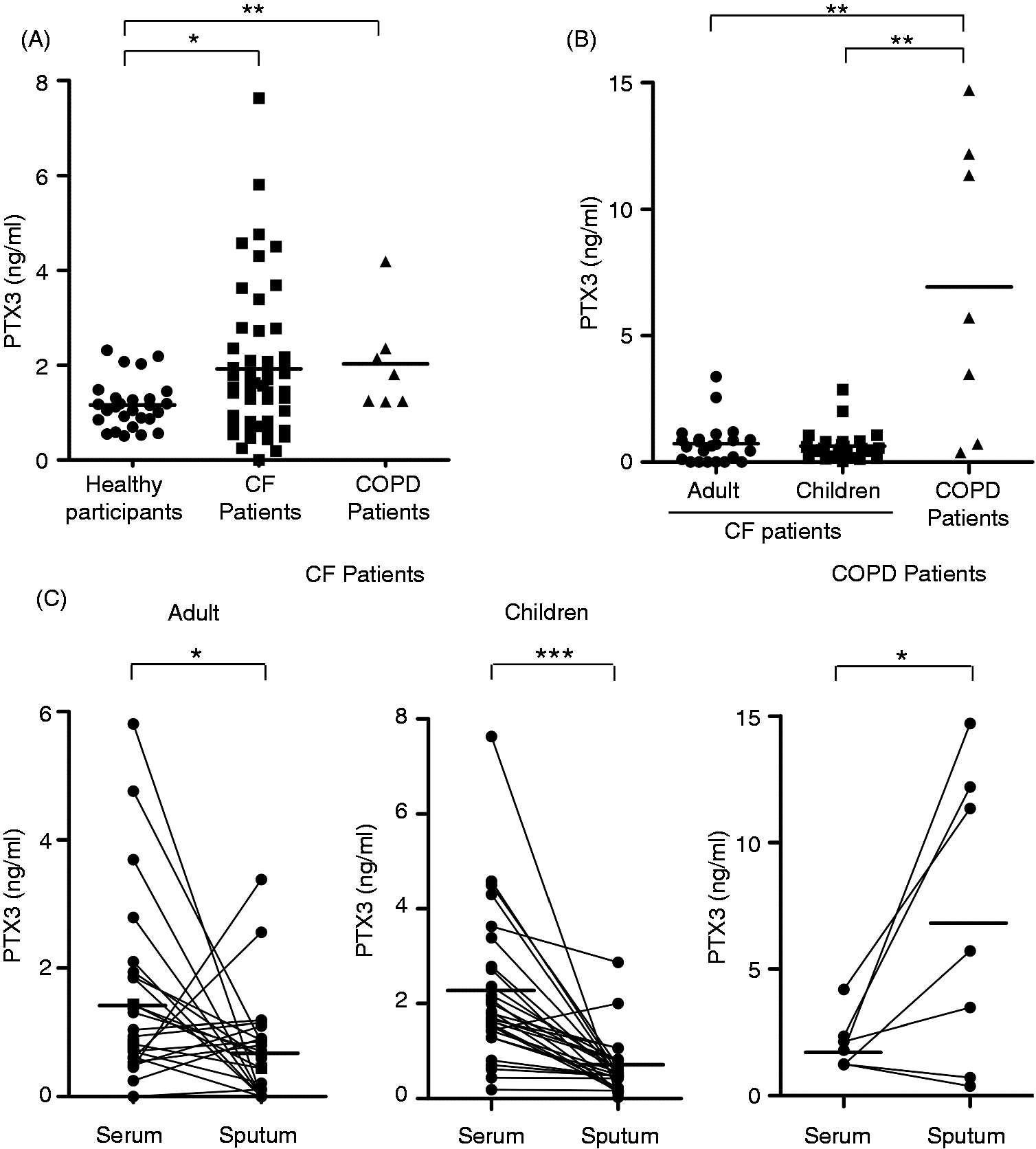
As CF primarily affects the respiratory tract, we also quantified PTX3 in sputum samples. Surprisingly, the concentrations of PTX3 were lower in the sputum samples from adults (0.73 ± 0.83 ng/ml; n = 23) and children CF (0.63 ± 0.59 ng/ml; n = 28) than in their serums (1.57 ± 1.46 ng/ml and 2.22 ± 1.58 ng/ml, respectively) (Figure 1B). Individual analysis revealed that, in most cases, PTX3 was lower in sputum samples than in serums in CF patients (Figure 1C). In contrast, the levels of PTX3 were higher in the sputum samples (6.94 ± 5.81 ng/ml, mean ± SD, n = 7) than in the serums (2.03 ± 1.06 ng/ml) of COPD patients (Figure 1C).
PTX3 is degraded in CF sputum
The low concentration of PTX3 in CF sputum samples may result from a local degradation or from a reduced production. We first analyzed by Western blotting the integrity of PTX3 in CF sputum samples (Figure 2A). In 44 of 51 samples, either the 90-kDa dimeric (as illustrated in lanes 1, 4 and 5), the 50-kDa monomeric (lanes 2–5) or both forms of PTX3 (lanes 4 and 5) were undetectable. In 43 of 51 samples, immunoreactive bands with apparent molecular masses lower than 40 kDa, that may correspond to degraded forms of PTX3, were observed (Figure 2A and data not shown). In contrast, monomeric and dimeric forms of PTX3 were detected in 6/7 COPD patients, with only 4/7 samples exhibiting immunoreactive bands with a molecular mass lower than 40 kDa (Figure 2A and data not shown). Recombinant PTX3 and PTX3 in saliva of healthy participants gave rise to 90- and 50-kDa immunoreactive bands (Figure 2A). To confirm the degradation of PTX3 in CF sputum samples, biotinylated PTX3 (bPTX3), allowing discrimination from endogenous PTX3, was added to sputum samples. A degradation of PTX3 was observed as early as 15 min of incubation, and no immunoreactive PTX3 was detected after 30 min (Figure 2B). In contrast, bPTX3 was not degraded, or degraded more slowly in COPD sputum samples (Figure 2B and data not shown).
PTX3 is degraded in sputum samples from CF patients. (A) Western blotting analysis of PTX3 integrity in six representative sputum samples from adult CF patients, three representative sputum samples from COPD patients and one representative saliva sample from healthy donors. (B) bPTX3 (50 ng) was incubated in CF or COPD sputum samples at 37°C for the indicated times and the integrity of bPTX3 was analyzed by Western blotting. Data are representative of one of five experiments using sputum samples from different CF and COPD patients in each experiment. (C) Western blotting analysis of SAP and CRP integrity in three representative sputum samples from adult CF patients and two representative sputum samples from COPD patients. (D) bSAP and bCRP (50 ng) were incubated for 2 h at 37°C in CF and COPD sputum samples. bSAP and bCRP integrity was analyzed by Western blotting. Recombinant PTX3, SAP and CRP (A, C) and biotinylated PTX3, CRP and SAP (A, D) were used as controls.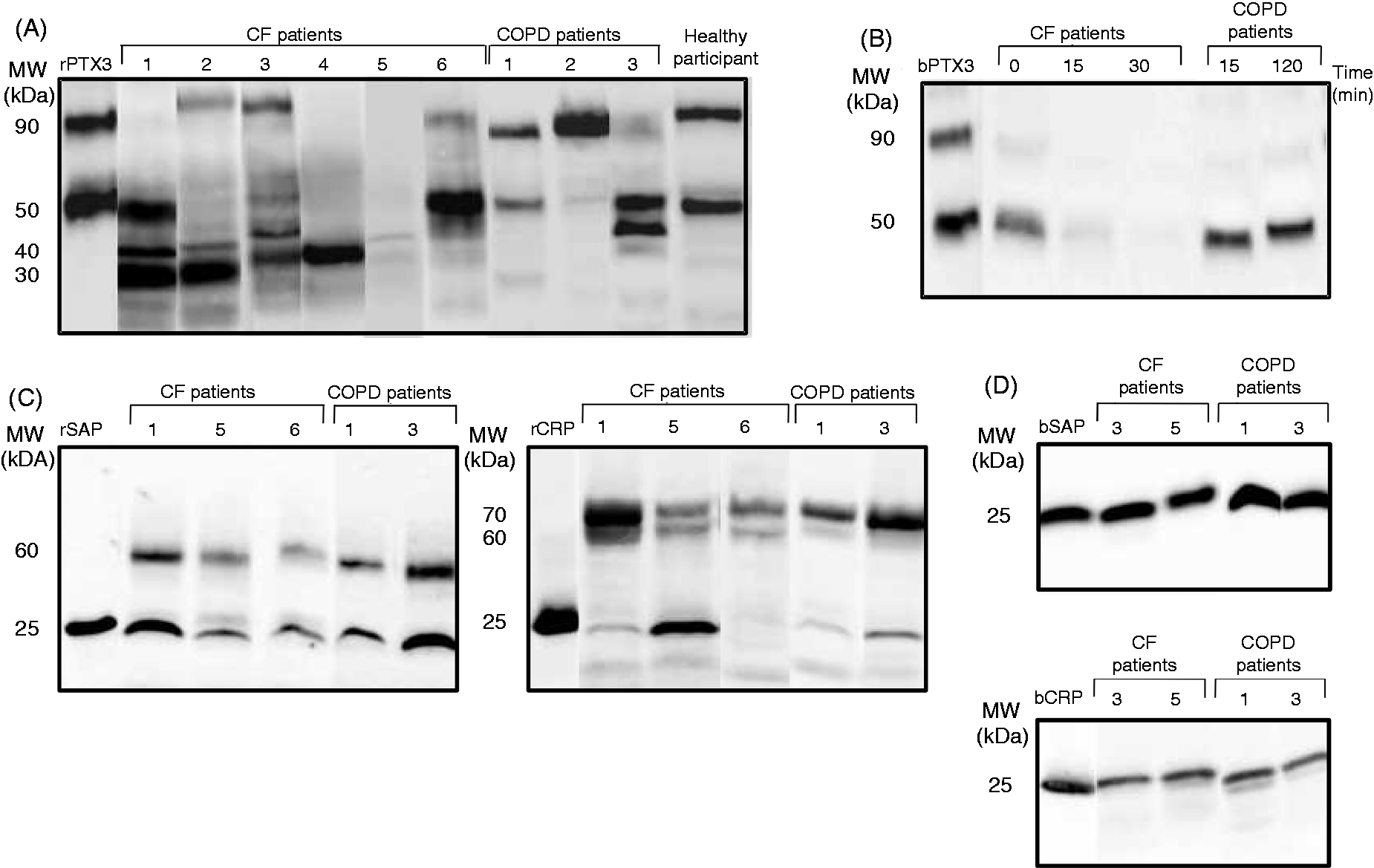
We investigated whether the short pentraxins CRP and SAP may be also altered in respiratory secretions from CF patients. Multimeric forms of SAP and CRP were detected in CF and COPD sputum samples (Figure 2C), with no bands with a molecular weight lower than 25 kDa. Accordingly, biotinylated SAP (bSAP) and biotinylated CRP (bCRP), added in CF or COPD sputum samples, remained intact (Figure 2D).
In a second set of experiments, we analyzed the capacity of pulmonary epithelial cell lines and neutrophils from CF patients to produce PTX3. The epithelial cell lines IB3-1 and S9 produced similar amounts of PTX3 in response to LPS or pro-inflammatory cytokines (Figure 3B, left). As a control, and in agreement with a previous study,
25
we observed that human primary bronchial epithelial cells produce PTX3 in response to IL-1β ± TNF-α, but not to LPS (Figure 3B, right panel, and data not shown). Peripheral blood neutrophils from CF patients and healthy participants (viability >95%) produced equivalent amounts of PTX3 upon activation (Figure 3A, left). We next evaluated the ability of neutrophils isolated from the airways of CF patients to secrete PTX3. Results showed that no PTX3 was detected in the supernatants of neutrophils isolated from CF sputums and stimulated with LPS or IL-1β ± TNF-α (data not shown). Interestingly, Western blotting analysis showed that neutrophils, freshly isolated from CF sputums, contained PTX3 (Figure 3A, right panel). A similar Western blotting profile is observed in peripheral blood neutrophils from healthy subjects after in vitro stimulation (Figure 3A, right panel).
The production of PTX3 is not altered in cells from CF patients. (A) Peripheral blood neutrophils isolated from CF patients (n = 6) and healthy participants (n = 4) were stimulated for 24 h with or without 100 ng/ml LPS, 20 ng/ml IL-1β + 40 ng/ml TNF-α or heat-inactivated A. fumigatus conidia (conidia per cell ratio = 5:1). The levels of PTX3 were determined by ELISA (mean ± SEM) (left). Western blotting analysis of cell-associated PTX3 in non-stimulated neutrophils isolated from CF airways (lanes 1 and 2) and in peripheral blood neutrophils from healthy participants after 24 h activation with 100 ng/ml LPS (lanes 3 and 4) (right). (B) The IB3-1 and S9 cell lines were stimulated for 24 h with LPS, IL-1β or TNF-α, at the indicated concentrations (left). Primary human bronchial epithelial cells were stimulated for 24 h with 10 µg/ml LPS or 200 ng/ml IL-1β plus 400 ng/ml TNF-α (right). The levels of PTX3 were determined by ELISA (mean ± SEM of three independent experiments).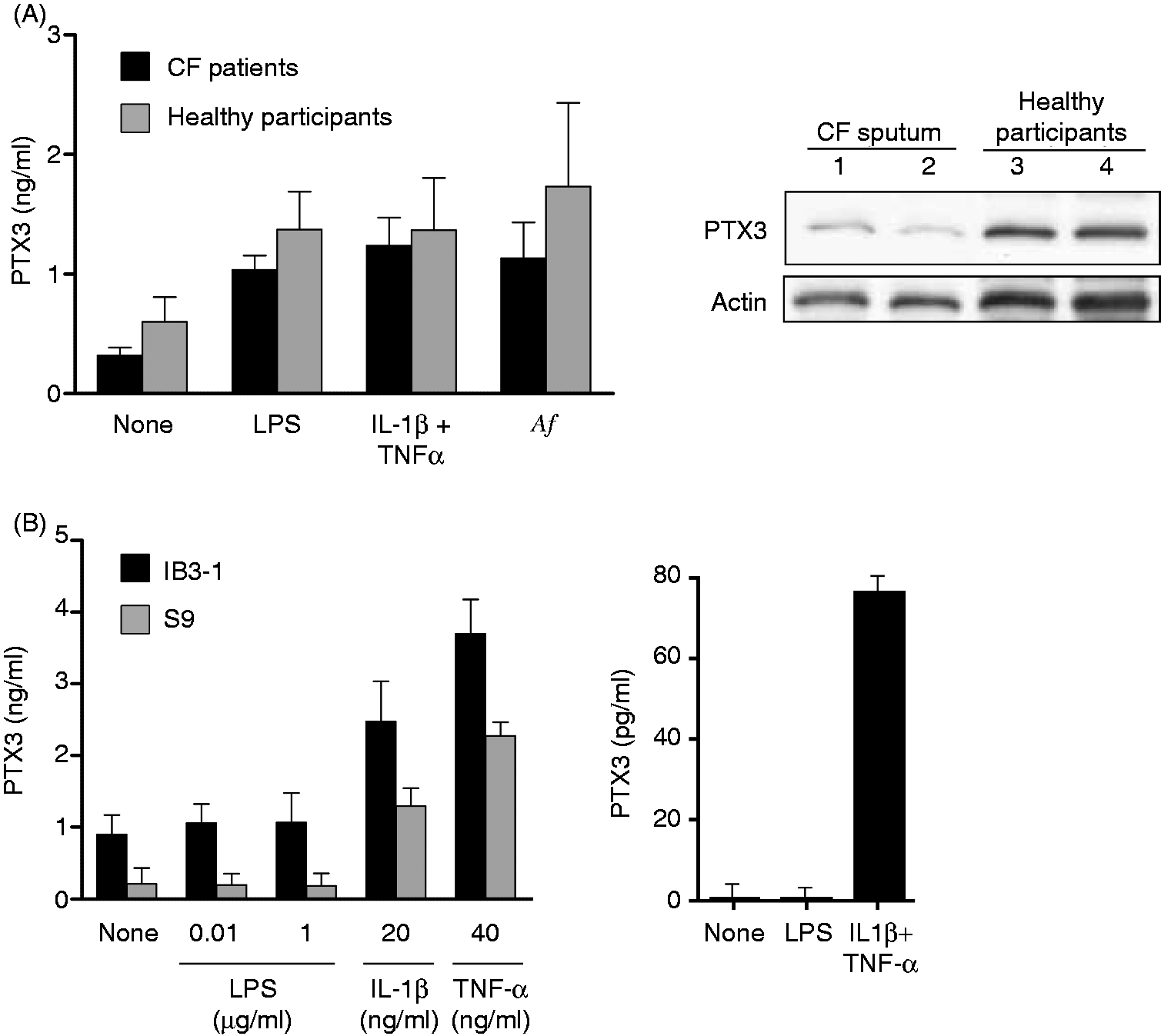
Serine proteases in CF sputum degrade PTX3
We investigated the nature of the proteases responsible for the local degradation of PTX3 in CF patients. The integrity of bPTX3 in CF sputum samples, containing or not containing protease inhibitors, was analyzed by Western blotting. Only the serine protease inhibitors PMSF and pefabloc protected bPTX3 from proteolysis (Figure 4). In contrast, the cysteine protease, metalloprotease, amino-peptidase, aspartate protease and chymotrypsin inhibitors did not protect PTX3 from degradation (Figure 4).
PTX3 is degraded by serine proteases present in CF sputum samples. bPTX3 (50 ng) was incubated for 1 h at 37°C in CF sputum samples containing or not containing proteases inhibitors. The integrity of PTX3 was analyzed by Western blotting. The results are representative of three experiments.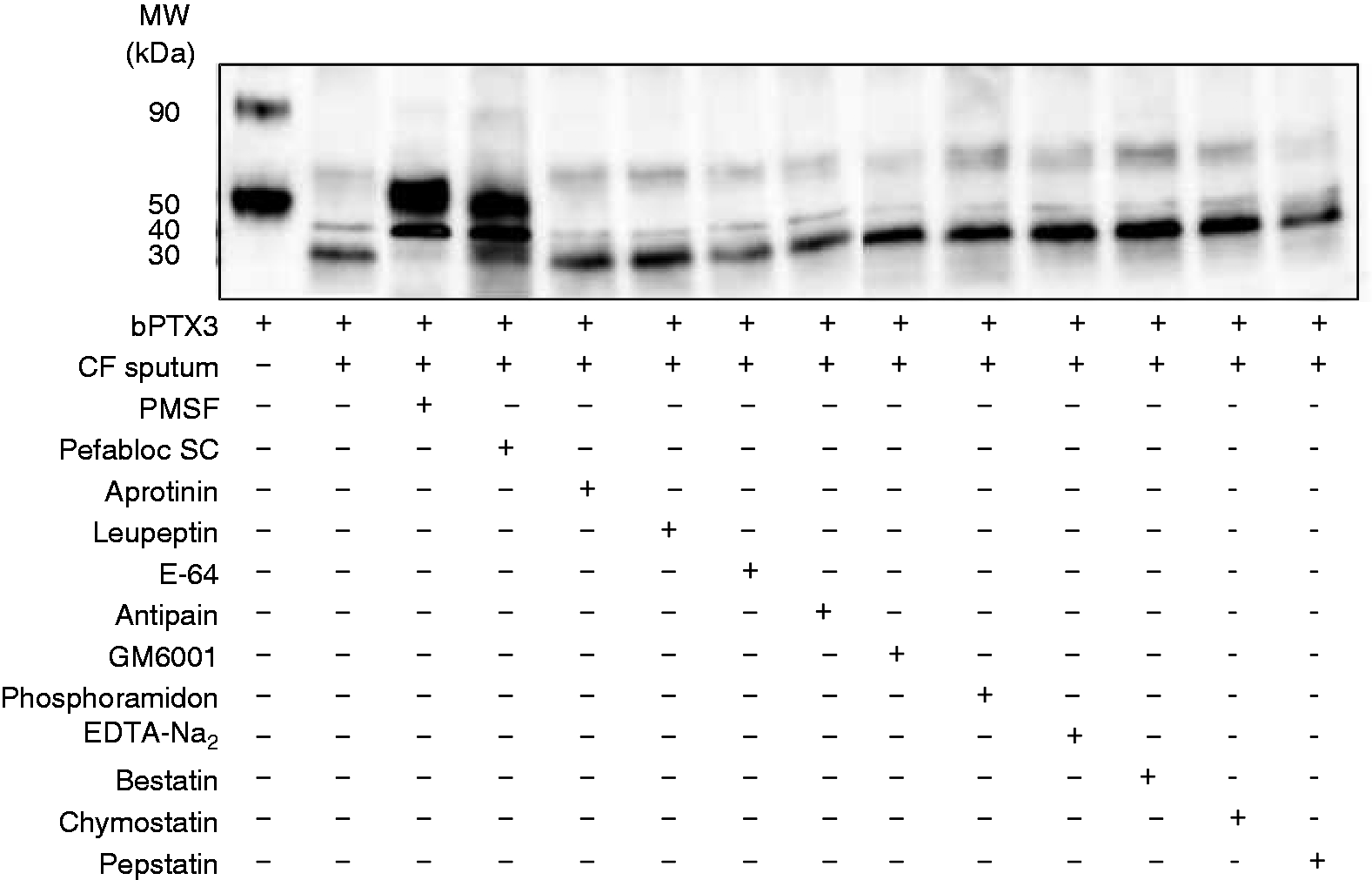
Neutrophil elastase is involved in the degradation of PTX3 in CF sputum
Neutrophils have a unique arsenal of serine proteases.
26
Moreover, activated and dying neutrophils, which persist in the lung of CF patients,
27
may release the serine proteases HNE, CatG and PR3 into the respiratory tract. We thus hypothesized that neutrophil-derived proteases may be involved in the degradation of PTX3 in CF patients. The specific HNE inhibitors, but not CatG and PR3 inhibitors, prevented the degradation of PTX3 in CF sputum samples (Figure 5A). As a control, these inhibitors protected recombinant PTX3 from the degradation by HNE, CatG and PR3 (Figure 5B, Supplementary Figure 1A). The following support a role for HNE in the degradation of PTX3 in CF patients: (i) HNE activity was detected in sputum samples from CF adults (Figure 5C and Table 1) and in the supernatants of dying neutrophils (Supplementary Figure 2A), as reported previously;
28
and (ii) we observed a significant correlation between the HNE activity and the level of endogenous PTX3 degradation in CF sputum samples (Figure 5C; Spearman correlation coefficient, r = 0.507; P = 0.016). In agreement with the absence of degradation in CF sputum samples, HNE, CatG and PR3 did not degrade SAP and CRP (Supplementary Figure 1B).
Correlation between HNE activity and PTX3 degradation in CF sputum samples. (A) Specific HNE (0.5 mM AAPA-CMK or AAPV-CMK), CatG (1 µM α-Chym or 0.5 mM AAF-CMK) or PR3 (60 µM) inhibitors were added to CF sputum samples 15 min before addition of 50 ng bPTX3. (B) Recombinant HNE, CatG and PR3 (125 mM) were incubated or not incubated with their specific inhibitors (0.5 mM AAPA-CMK, 0.5 mM AAPV-CMK, 0.5 mM AAF-CMK, 1 µM α-Chym or 60 µM PR3 inhibitors) for 15 min at 37°C before the addition of 50 ng recombinant PTX3 (rPTX3). (A, B) After a further 30 min incubation at 37°C, the status of PTX3 was analyzed by Western blotting. Results are representative of one of four independent experiments. (C) The activity of HNE was correlated with the integrity of PTX3 in adult CF sputum samples using the Spearman rank correlation assay. Degradation of PTX3 (determined by Western blotting) was scored from 0 (no degradation) to 10 (totally degraded). Correlation coefficient, r = 0.507, P = 0.016, n = 23.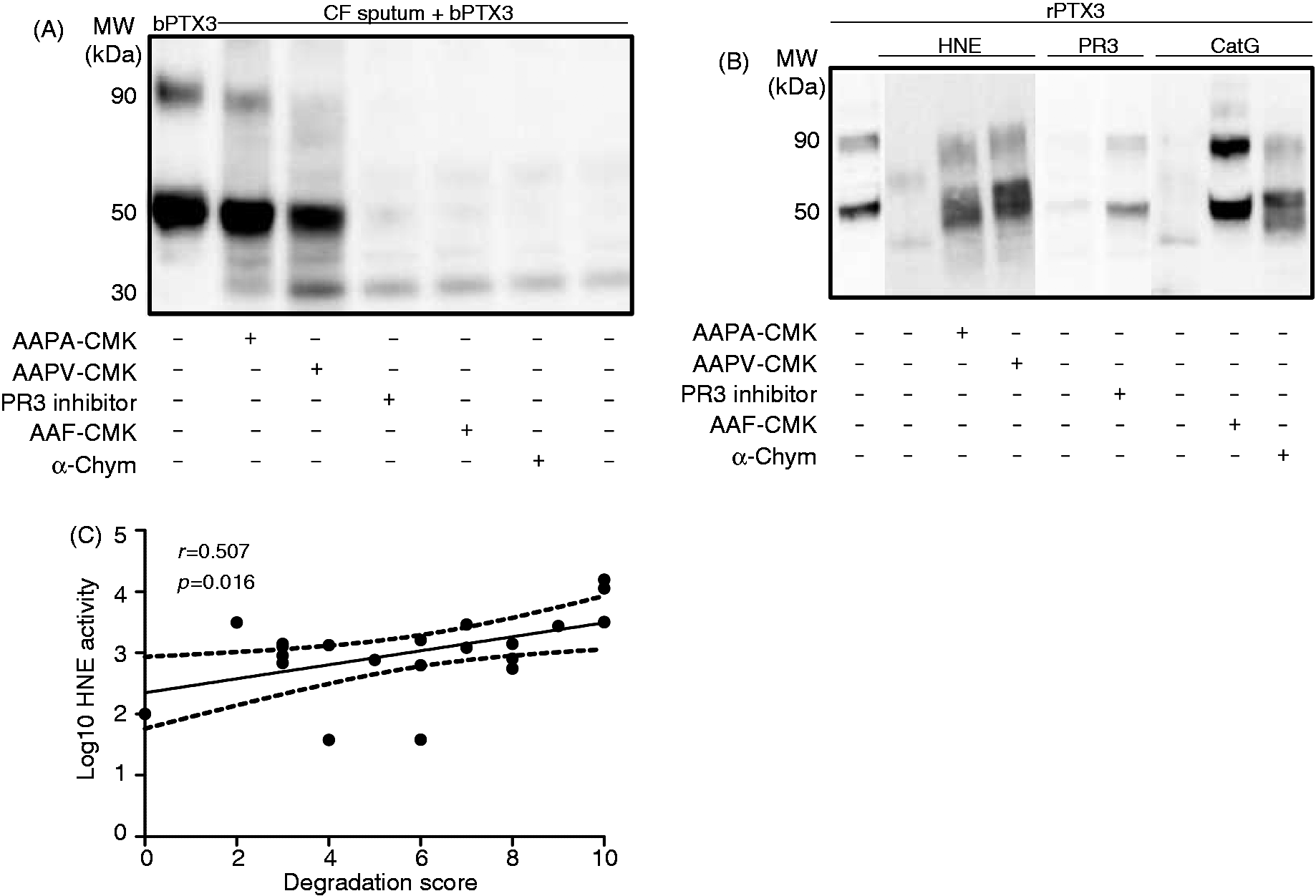
Proteases secreted by A. fumigatus can degrade PTX3
Most CF patients are chronically infected, especially by A. fumigatus—one of the most frequent fungal colonizers in CF patients.
29
Results showed that PTX3 was degraded rapidly (<30 min) in A. fumigatus culture filtrates (Figure 6A). A. fumigatus produces three main endoproteases:
30
the alkaline protease Alp, the aspartic protease Pep and the metalloprotease Mep. The degradation of PTX3 in A. fumigatus culture filtrates was inhibited by the serine protease inhibitors PMSF, antipain and chymostatin, but not by the aspartic-, metallo-, cysteine- and aminopeptidase protease inhibitors (Figure 6B). Moreover, A. fumigatus culture filtrates did not induce the proteolytic cleavage of SAP and CRP (data not shown).
Proteases secreted by A. fumigatus (Af) degrade PTX3. (A) Recombinant PTX3 (50 ng) was incubated in A. fumigatus culture filtrates for the indicated times at 37°C. (B) Protease inhibitors were added to A. fumigatus culture filtrates for 30 min at 4°C before addition of 50 ng PTX3 for further 30 min incubation at 37°C. (A, B) The status of PTX3 was evaluated by Western blotting using a specific polyclonal anti-PTX3 Ab. Recombinant PTX3 (rPTX3) was used as a control. Results are representative of three experiments.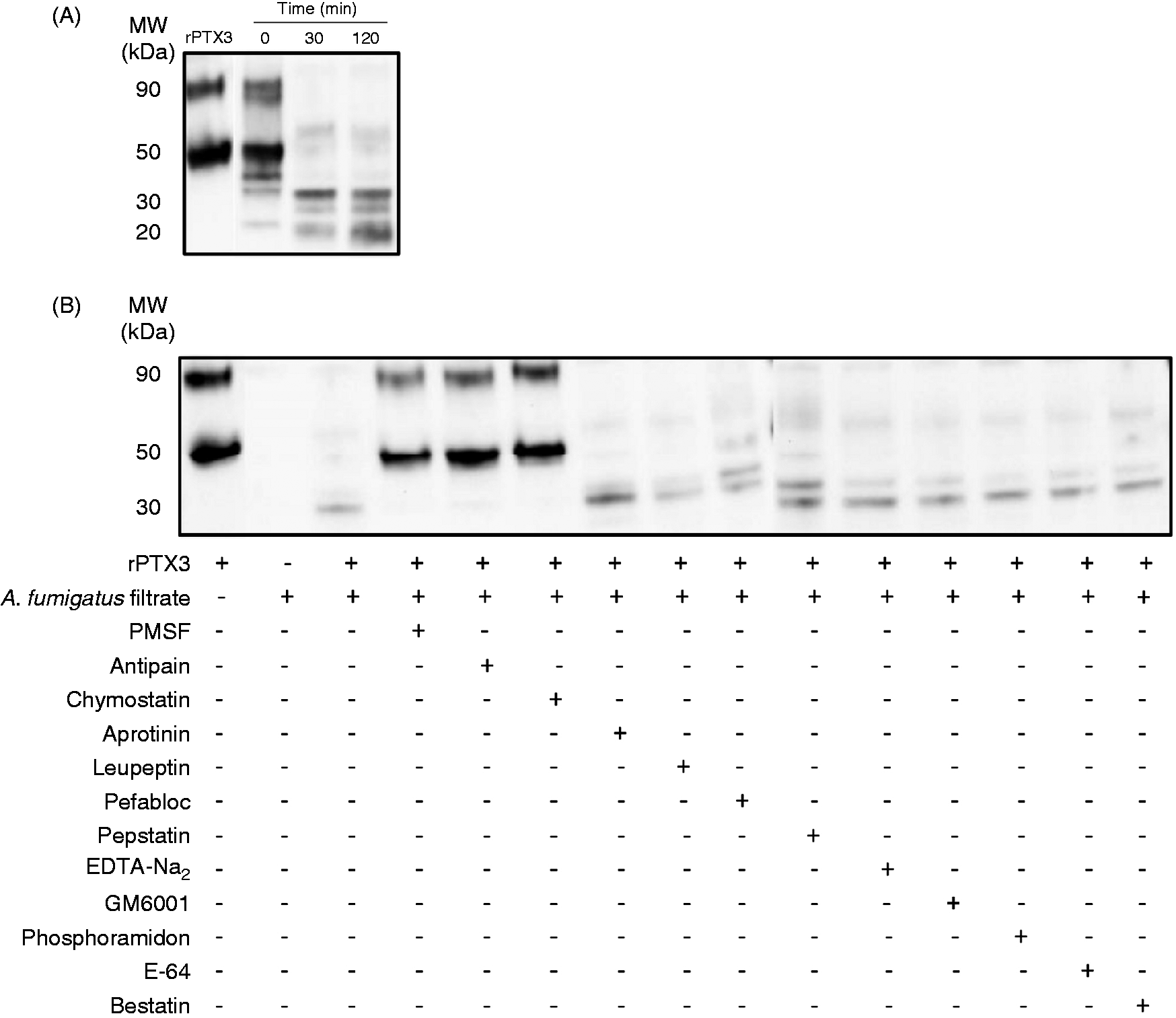
HNE and A. fumigatus serine proteases degrade the N-ter domain of PTX3
SAP and CRP were resistant to proteolysis by A. fumigatus and neutrophil proteases, suggesting that the common C-ter domain of pentraxins is preserved. We therefore analyzed the degradation of the N-ter and C-ter domains of PTX3, by using the C-ter and N-ter specific mAbs 16B5 and MNB4, respectively. Interestingly, HNE (Figure 7A) and proteases secreted by A. fumigatus (Figure 7B) rapidly degraded the N-ter domain of PTX3 while, in contrast, a longer incubation time was required to induce the degradation of the C-ter domain. A similar profile of PTX3 degradation was observed with proteases released by dying neutrophils (Supplementary Figure 2B).
HNE and A. fumigatus proteases degrade the N-ter domain of PTX3. (A) Recombinant PTX3 (50 ng) was incubated with 250 nM HNE for 2 h at 37°C. (B) Recombinant PTX3 (50 ng) was incubated at 37°C in A. fumigatus culture filtrates during the indicated times. (A, B) The status of PTX3 was analyzed by Western blotting using the anti-PTX3 monoclonal Abs 16B5 or MNB4. Results are representative of three independent experiments. Recombinant PTX3 (rPTX3) was used as a control.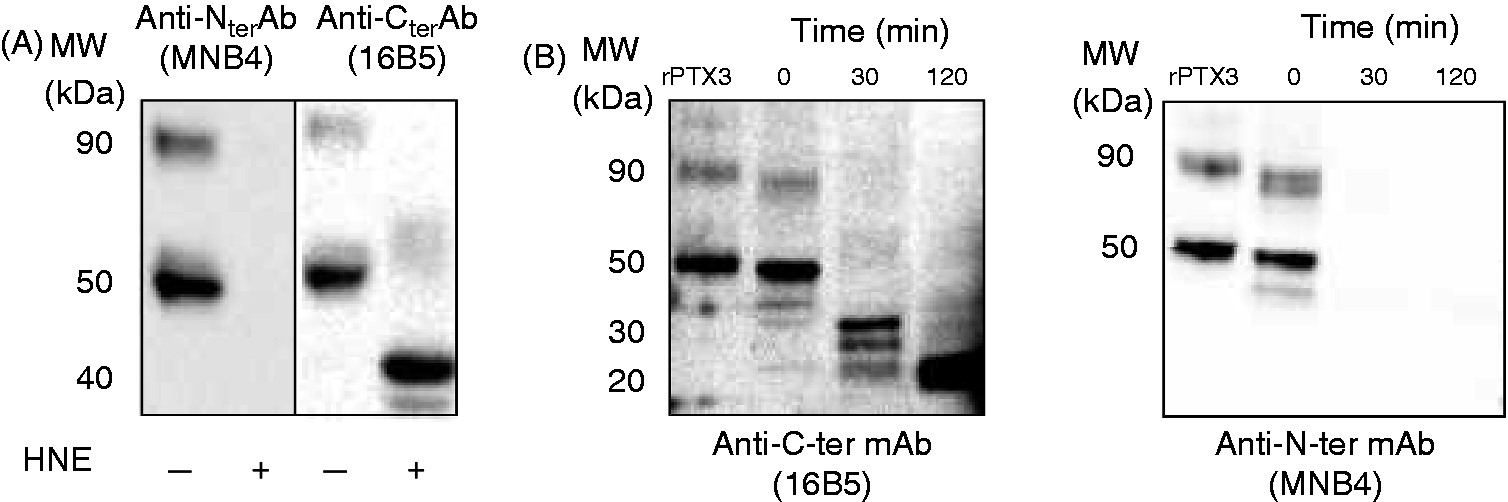
Discussion
PTX3 plays a pivotal role in the resistance against selected pathogens, especially A. fumigatus and P. aeruginosa.8,10 CF is characterized by chronic lung infections, prominently with these two pathogens. We thus hypothesized that the expression and/or function of PTX3 may be altered in CF patients. Results showed that endogenous PTX3 is degraded locally in CF patients and that its proteolysis can be mediated by elastase and microbial serine proteases.
The level of circulating PTX3 increases rapidly during infection and inflammation, especially in severe sepsis and septic shock. 6 PTX3 is now recognized as an early and robust indicator of shock in severe meningococcal infection 7 and a prognostic factor in patients with bacteremia. 31 We report that PTX3 is also elevated in CF patients. However, no correlation was evidenced between the levels of PTX3 and the clinical and biological parameters of CF patients.
CF patients suffer from chronic lung infections, suggesting that PTX3 may be elevated in the lung. Surprisingly, we observed that (i) the concentrations of PTX3 were, in most cases, lower in the sputum than in the serum of CF patients, and (ii) that the low levels of PTX3 in the airways of CF patients were consecutive to a proteolysis by serine proteases and did not result from a defective production.
HNE, present in an active form in CF sputum, plays a major role in the degradation of PTX3. Previous studies reported that HNE is involved in the degradation of soluble PRRs, such as complement proteins and the collectins SP-A and SP-D.32,33 HNE also cleaves opsonin receptors (such as CR1) expressed by phagocytes, resulting in an impaired phagocytosis of pathogens and persistence of infection. 34 However, PTX3 appears the most sensitive to serine proteases, as its degradation occurs within minutes. HNE is also involved in inflammation through the upregulation of CXCL8 35 which, in turn, increases the release of HNE. 36 The cleavage of the phosphatidylserine receptor by HNE impairs the clearance of apoptotic cells, 27 resulting in a vicious circle of neutrophil accumulation and excessive HNE activity in the airways. We also observed that serine proteases secreted by A. fumigatus degrade PTX3. Innate immune molecules, such as the complement proteins, are degraded by the A. fumigatus alkaline protease Alp1. 37 A. fumigatus proteases also degrade collagen, elastin and fibrinogen (reviewed in Abad et al. 38 ), a process involved in inflammation and tissue remodeling. However, owing to the lack of specific inhibitors, we were unable to identify the A. fumigatus serine protease(s) involved in the degradation of PTX3. We also observed that Scedosporium apiospermum, another major colonizer of the CF patient airways, 39 produces proteases able to degrade PTX3 (personal observation).
In contrast to CF patients, the levels of PTX3 were higher in the sputum samples than in the serums in COPD patients, used as a control group of patients with lung infection. Even though COPD patients exhibited lung infection associated with exacerbation, the degradation of PTX3 and the HNE activity in their sputum samples were reduced compared with CF patients. A recent study reported that COPD is associated with a reduced pulmonary interstitial expression of PTX3 and that PTX3 expression in small airways, correlated significantly with FEV1. 40 However, and in agreement with our results, Pizzichini et al. 41 reported increased levels of PTX3 in the sputum samples from COPD patients compared with controls.
In contrast to PTX3, the short pentraxins CRP and SAP were not degraded in CF sputum samples and in the A. fumigatus culture filtrates. Moreover, in our experimental conditions, and in contrast to a previous study,42,43 CRP and SAP were not degraded by HNE. The reason for such discrepancy is not clear. We observed that PTX3 is degraded rapidly by proteases. Thus, we cannot exclude that a lengthy incubation is required to degrade the short pentraxins. PTX3 differs from CRP and SAP for the presence of an unrelated N-ter domain. 2 Interestingly, the N-ter domain of PTX3 was rapidly and preferentially degraded by HNE and A. fumigatus proteases while, in contrast, a longer incubation time was required to induce the degradation of the C-ter domain. This result is in agreement with the fact that the short pentraxins are less sensitive to proteases than the long pentraxin PTX3. Immunoreactive bands with apparent molecular weights lower than 40 kDa were detected in the sputums of CF patients but not after incubation with HNE, suggesting that, in CF patients, both HNE and microbial proteases cooperate to degrade PTX3. The N-ter domain of PTX3 contains the binding site to A. fumigatus conidia, 44 suggesting that the preferential degradation of this domain may represent an important physiopathological process involved in the lung colonization by selected pathogens (such as A. fumigatus) and their persistent colonization in the lung consecutive to an impaired clearance. Previous studies suggested that the biology of lung neutrophils and macrophages can be modified in CF patients.45,46 No PTX3 was detected in the supernatants of in vitro activated neutrophils isolated from CF sputums. Interestingly, Western blotting analysis revealed that PTX3 remained detectable in neutrophils freshly isolated from CF sputums. This result has to be analyzed in light of previously published results 3 showing that a part of the preformed stock of PTX3 remains associated with activated neutrophils, even after a lengthy stimulation. These results suggest that neutrophils isolated from CF airways have already secreted PTX3 and are unresponsive, in terms of PTX3 release, to a further in vitro stimulation. Concerning macrophages, we did not isolate enough cells to perform in vitro experiments. We observed that bronchial epithelial cells from CF patients also produce PTX3 upon stimulation, as reported for epithelial cells isolated from healthy participants. 25 Collectively, these data suggest that the capacity to produce PTX3 is not impaired in CF patients compared with healthy subjects.
PTX3 has important roles in the resistance to a variety of microbes, mainly via its capacity to facilitate their phagocytosis8,10,12 and to favor the initiation of protective immune responses.8,47 In vivo studies indicate that PTX3 protects mice from A. fumigatus pulmonary infection by reducing fungal burden and by increasing survival.9,47 PTX3 reduces colonization and inflammation in models of aspergillosis and P. aeruginosa lung infection in Ptx3-/- mice.8,10 A protective role of PTX3 was also found associated with a specific haplotype in CF patients. 48 Our study confirms that a defective activity of PTX3, as observed in CF patients, may have profound consequences on the local immunity.
In conclusion, this study shows that (i) the selective proteolysis of PTX3 in the lung may represent a pivotal mechanism for recurrent lung infections in CF patients, and (ii) suggests that PTX3-based therapeutic strategies have to take account of its selective and rapid degradation. These results also suggest that the local degradation of PTX3 is a hallmark of CF.
Footnotes
Acknowledgements
We sincerely acknowledge nurses from the University Hospital of Angers for their help in sample collection. We are grateful to Dr Korkmaz (Inserm Unit 618, Tours, France) for providing the neutrophil serine protease specific inhibitors.
Funding
This work was supported by institutional grants from the University of Angers and from Inserm and by grants from the Agence Nationale pour la Recherche (project Pentraximmune) and from the French cystic fibrosis association ‘Vaincre la mucoviscidose’. YH received a fellowship from the Conseil Général du Maine et Loire and from the French cystic fibrosis association ‘Vaincre la mucoviscidose’ (Grant RF20110600452).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
