Abstract
Swine building exposure causes inflammatory reactions that appear to be attenuated with prolonged periods of contact. The mechanisms behind this adaptation to a dusty and endotoxin-rich environment are poorly understood. Our aim was to compare levels of selected inflammatory mediators in swine farm workers at times with differences in exposure. Participants had blood sampling done before and after each of three work shifts—two in winter and one in summer. Before one of the winter visits they had avoided pulmonary exposure to the swine buildings by wearing respiratory protection for 4 d. The other visits were done after non-protected periods of work. Protein and mRNA concentrations were measured in blood. Mixed models were used for the statistics. During summer higher concentrations of mRNA to IL-8, lymphocyte function-associated antigen 1 and bactericidal/permeability-increasing protein (BPI) were observed. BPI mRNA increased only over the work shift after the unprotected winter period (P = 0.039). BPI decreased from elevated levels across the shift after use of respiratory protection (P = 0.003), but was unchanged during the other two visits. The findings suggest possible roles for these proteins in adaptation to the swine building environment after repeated exposures.
Keywords
Introduction
Tolerance, which develops with continued exposure to bacterial endotoxin, is a well-known phenomenon in biology. In humans, tolerance has been described with endotoxin release into the bloodstream during infection, 1 after injection 2 and after inhalation. 3 Such an adaptation may be important in occupations with exposure to endotoxin-rich bioaerosols, such as the cotton industry and confined animal farming operations (CAFOs). The work-related health effects and the adaptation to these are commonly ascribed to endotoxin in bioaerosol-exposed occupations despite the abundance of other inflammogenic agents in the bioaerosol.
The mechanisms regulating response to endotoxin are complex and not understood in detail. Important for the host response is the type and dose of endotoxin, type of co-exposure, the balance between extracellular proteins, such as LPS binding protein (LBP), bactericidal/permeability-increasing protein (BPI), soluble CD14 and TLR4, and intracellular factors, such as kinases. Downstream of these are the concentrations of numerous proteins, such as TNF-α and its receptors; interleukins; and adhesion molecules, etc. which are responsible for the type and size of the physiologic response. It is still an enigma whether the organism is also able to respond and adapt with tolerance to microbial associated molecular patterns (MAMPs) other than endotoxin via similar pathways.
Because the environment inside CAFOs is rich in endotoxin and other MAMPs the responses of humans exposed to it have been subject to several studies since initial reports of detrimental health effects of these environments in the 1980s. 4 Adaptation during regular swine house work has been described previously.5,6 Recently, our group found that adaptation was partly lost during the low-exposure summer season 7 or after use of respiratory protection. 8
As part of a study on effects of different work conditions on lung function and inflammation among swine building workers, we investigated plasma levels and white blood cell mRNA expression of selected proteins known to be involved in the response to inhaled endotoxin or swine dust. Our aim was to identify differences in selected mRNAs and some of their proteins known to relate to work in the swine houses on three ordinary workdays preceded by different work conditions.
Regarding effects of work shifts, we hypothesized that the mRNA expression of proteins involved in inflammation would be up-regulated during a work shift after a period of respiratory protection in the high exposure winter season compared with smaller, or no, changes after a period of unprotected work. We questioned if—during unprotected work—the lower exposure in summer would be sufficient to maintain the adaptation associated with repeated exposures in winter.
Regarding effects of adaptation to repeated exposures, we hypothesized that lower systemic levels of pro-inflammatory cytokines, such as TNF-α and its receptors, and a balance in favor of BPI instead of LBP and CD14 in adapted workers compared with less adapted workers would be associated with an attenuated expression of genes of the down-regulated proteins. Further, we hypothesized that there would be down-regulation of TLR4 mRNA and possibly changes in the concentrations of mRNA of the proteins IL-6 and IL-8 with inflammatory capacities, and IL-10 with anti-inflammatory capacities, as well as the adhesion molecule lymphocyte function-associated antigen 1 (LFA-1).
Materials and methods
Study population and design
Details on the population are described elsewhere. 7 In brief, swine farm workers (operators, as well as owners) within 150 km of Quebec City were visited twice during winter and once during summer. All swine farms included at least one mechanically ventilated finishing building. Smokers and workers suspected of lung diseases or with symptoms of airways infection in the seven days preceding the visits were excluded. Health examinations took place in the morning immediately prior to the first entry of the worker into the animal house and in the afternoon at the end of the work shift (preferably at least 6 h later).
At one of the two visits during winter the workers were requested to abstain from exposure to swine and swine houses for at least 4 d. In case this could not be achieved they agreed to wear N95 respirators (3 M, St Paul, MN, USA) during all entries into swine houses for protection of the airways. These visits are referred to as visits after respirator use. Correct use of the respirators was demonstrated and the fit checked by one of the investigators during a visit to the farm. At the two other visits the workers were examined after ordinary periods of work on the swine farm without any use of respiratory protection of at least 4 d. At all of the visits the workers answered questions on respiratory protection, medication and work in the previous 7 d.
Complete spoken and written information on the study was given prior to the first evaluation and a signed informed consent to participate obtained. The study was approved by the institutional Ethics Committee at the Institut Universitaire de Cardiologie et de Pneumologie de Québec (IUCPQ).
Blood samples
Venous blood samples were taken before and after work at each visit in standard K3 EDTA-coated tubes. One tube was kept at room temperature for white blood cell (WBC) count. At the end of each clinical evaluation the remaining tubes were centrifuged for 10 min at 1200 g. The plasma was removed carefully with a pipette and put on dry ice. After removal of the plasma, approximately 500 μl of the WBC layer (buffy coat) was mixed with 4 ml Trizol Reagent (Invitrogen, Burlington, ON, Canada). After mixing, 1-ml aliquots were added to polypropylene tubes and placed on dry ice. Upon return to the laboratory after the final examination of the day, WBC counts were performed and the remaining samples stored at −80℃ for later analyses. The concentration of CD14 was measured with a chemiluminescence ELISA kit from R&D (R&D Systems, Minneapolis, MN, USA). BPI and LBP were measured with ELISA kits from Hycult (Hycult Biotechnology, Uden, the Netherlands). According to the manufacturers, the mean limits of detection were 125 pg/ml for CD14 and 250 pg/ml for BPI. No limit of detection was given for LBP.
mRNA extraction and transcription
Buffy coats were thawed on ice for 10 min and vortexed before 200 μl chloroform (C-2432, 99% Molecular Biology grade; Sigma-Aldrich Canada Ltd, Oakville, ON, Canada) was added. Centrifugation at 12,000 g for 15 min (MicromaxRF; ThermoIEC, Needham Heights, MA, USA) allowed the collection of the aqueous phase (on top) containing the total RNA. Pipetting of this layer was done very carefully so that proteins and genomic DNA of the organic phase would stay at the bottom of the tube. The RNA was precipitated by adding 500 μl of a 2-propanol solution (I9516-25ML, 99% Molecular Biology grade; Sigma-Aldrich Canada Ltd, Oakville, ON, Canada) to every 200 μl of aqueous phase, followed by incubation on ice for 10 min. Qiagen RNeasy MinElute Cleanup Kit (Cat. No 74 204; Qiagen, Mississauga, ON, Canada) was used to purify and concentrate the RNA. The 700 μl mixture (aqueous phase and 2-propanol) was loaded on the top of the columns followed by an 8000 g 15 s centrifugation (MicromaxRF; ThermoIEC). Once the flow-through discarded, 500 μl of Buffer RPE was added and the columns were centrifuged again using the same parameters. This last step was repeated with 500 μl of 80% ethanol. After changing the collection tubes to new ones, they were spinned for 5 min at 21,000 g in order to completely dry out the ethanol from the silica-gel membrane. Finally, elution of the bound RNA was done by adding 20 μl RNase free water followed by a 1-min centrifugation at 21,000 g. The concentration of the samples was determined by mixing 2 μl of eluted RNA in 68 μl of a 10 mM Tris-HCl solution. The mixture was transferred in plastic 70 μl UV cuvettes (759220; Sigma-Aldrich Canada Ltd., Oakville, Ontario) and read by a GeneQuantPro spectrophotometer (TBiochrom US, Holliston, MA, USA) using a 260 nm/280 nm wavelength ratio. All RNA samples were adjusted to a 100 ng/μl standard concentration before reverse transcription to cDNA.
iScript cDNA Synthesis Kit (170-8891; Bio-Rad, Mississauga, ON, Canada) was used to obtain cDNA. All reactional mixtures and PCR cycles were done following the manufacturer’s recommendations (500 ng/μl RNA in a 20 μl total volume, followed by 5 min at 25℃, 30 min at 42℃ and 5 min at 85℃). The obtained cDNA was then diluted using a 1:4 ratio (cDNA:H20) and stored at −20℃ until necessary for quantitative PCR reactions.
Quantitative PCR
QuantiTect® Primer Assay (Qiagen) were used because their sequence is specifically designed to cross exon/exon boundaries in order to prevent co-amplification of genomic DNA (which may compromise assay specificity and dynamic range). The following primers were used: IL-6 (QT00083720), IL-8 (QT00000322), IL-10 (QT00041685), TNF-α (QT01079561), LBP (QT00027293), ITGAL (synonyms: LFA-1, integrin, Ag CD11A) (QT00034006), TLR-4 (QT0167023) and BPI (QT00096649). GAPDH (QT00079247) was used as a housekeeping gene. Reactional mixtures were performed directly on 96-well plates (HSP-9655 Bio-Rad) and contained the following (per sample): 12.5 μl iQ SYBR Green SuperMix 2X (170-8882, Bio-Rad), 2.5 μl of primer (Qiagen), 2.0 μl of cDNA (diluted 1:4 after reverse transcription) and 8.0 μl of nuclease free water. Wells were then covered by optical flat cap strips (TCS-0803; Bio-Rad). The PCR program was as follows: 94℃ for 3 min, 94℃ for 20 s, 55℃ for 40 s, plate read, 72℃ for 40 s, step two repeated 40 times and then for the melting curve from 30–94℃ read every 1℃, hold 1 s and finally 72℃ for 5 min. RT-PCR was performed on a DNA Engine Opticon 2 (Bio-Rad, Mississauga, Ontario).
Statistical methods
The Friedman test was used to compare time spent with swine and between tests at each visit. Student’s t-test was used to compare characteristics between subgroups of the sensitivity analysis. The main outcome variables—the protein and mRNA levels—were analyzed with a linear mixed model with repeated measurements on each worker. The factors were time (two levels: morning and afternoon) and visit (three levels: each of the three visits). We considered subjects as random block effects in the model. The statistical approach was to perform a multivariate repeated measures design (doubly multivariate data) with a fixed factor linked to the visits and the other fixed factor to the time. The covariance structure specified direct product structures by taking the Kronecker product of an unstructured matrix (modeling covariance across the observations at the three visits) with an additional covariance matrix (modeling covariance across time). The model used a general Kenward-Roger approximation for the denominator degrees of freedom. The variance assumptions were verified using the Brown and Forsythe's variation of Levene's test statistic. The univariate normality assumptions were verified with the Shapiro-Wilk tests. The multivariate normality was verified using the Mardia’s test. If necessary, logarithmic transformation of the data was used to achieve these assumptions. P-Values ≤0.05 are reported. If applying the Bonferroni correction on the 13 mediators or ratios being tested the statistical significance should be 0.05/13 = 0.0038. The repeated measures analyses were conducted using PROC MIXED and TYPE = UN@AR(1) in the statistical package SAS v9.1.3 (SAS Institute Inc., Cary, NC, USA). SPSS v. 13.0 was used for the remaining analyses (SPSS, Chicago, IL, USA).
Results
Characteristics of the study population.

Time [mean (SD)] of work with swine and between clinical tests from morning to afternoon clinical examination.
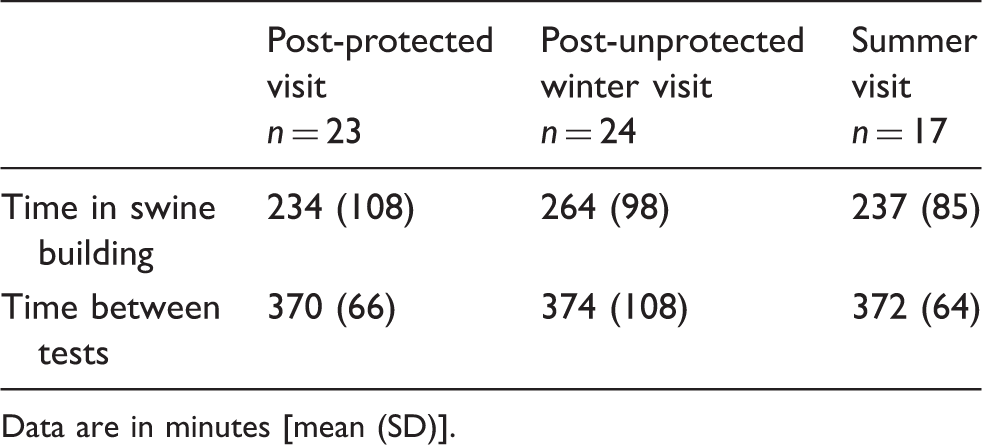
Data are in minutes [mean (SD)].
Overall mRNA expression in the buffy coat and protein concentration in plasma of the measured inflammatory mediators during the three farm visits. The data are from the mixed model with data for three visits using back-transformed concentrations accounting for missing data and covariance structure. mRNA concentrations are expressed as ratios to GADPH. Protein concentrations are geometric means (pg/ml). BPI/LBP and LBP/CD14 ratios are based on ratios from subjects with data on both concentrations used for the calculations.
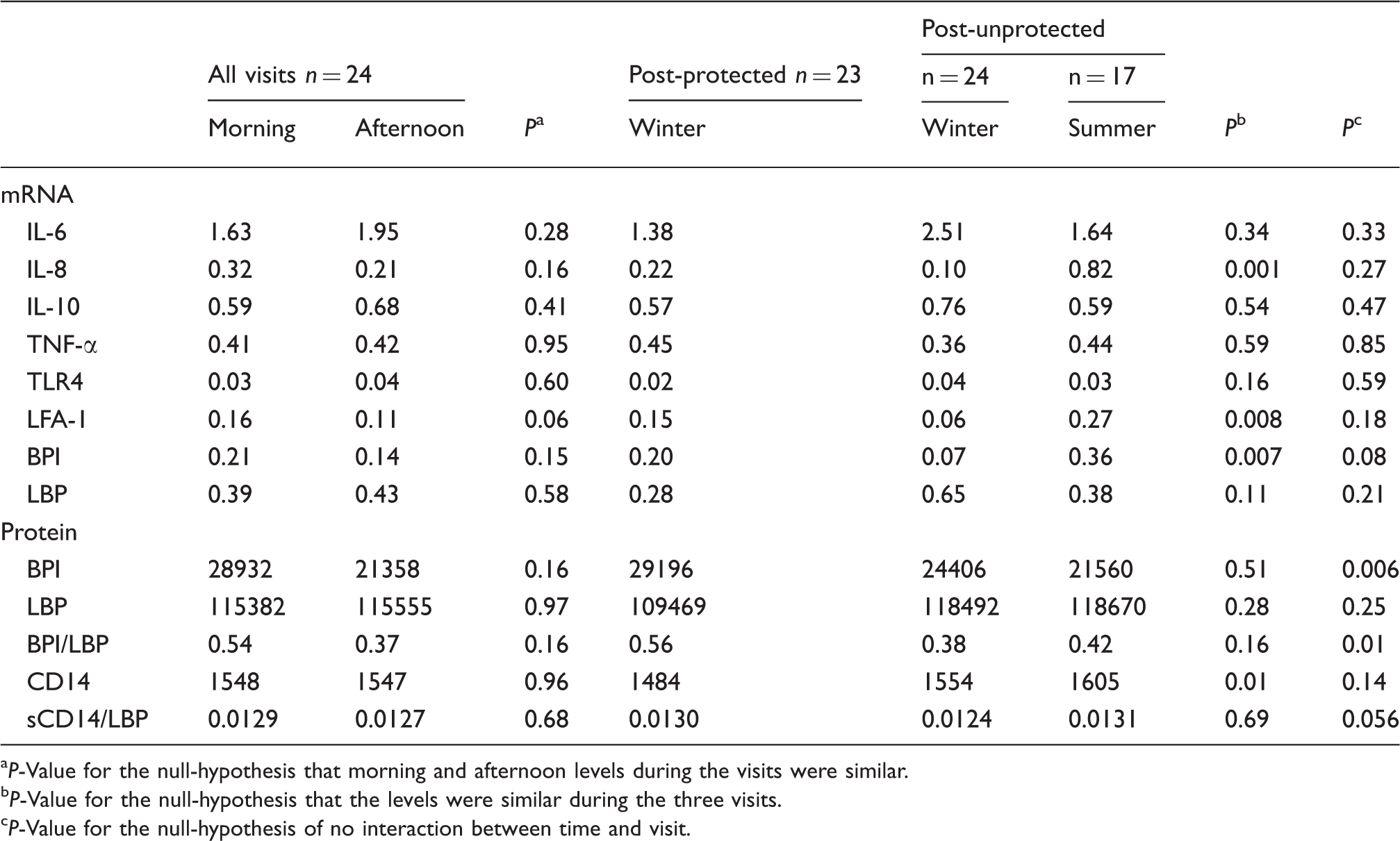
P-Value for the null-hypothesis that morning and afternoon levels during the visits were similar.
P-Value for the null-hypothesis that the levels were similar during the three visits.
P-Value for the null-hypothesis of no interaction between time and visit.
mRNA expression in the buffy coat and protein concentrations in plasma of the measured inflammatory mediators before and after the work shift during the three farm visits. The data are from the mixed model with data for three visits using back-transformed concentrations accounting for missing data and covariance structure. mRNA concentrations are expressed as ratios to GADPH. Protein concentrations are geometric means (pg/ml). BPI/LBP and LBP/CD14 ratios are based on ratios from subjects with data on both concentrations used for the calculations.
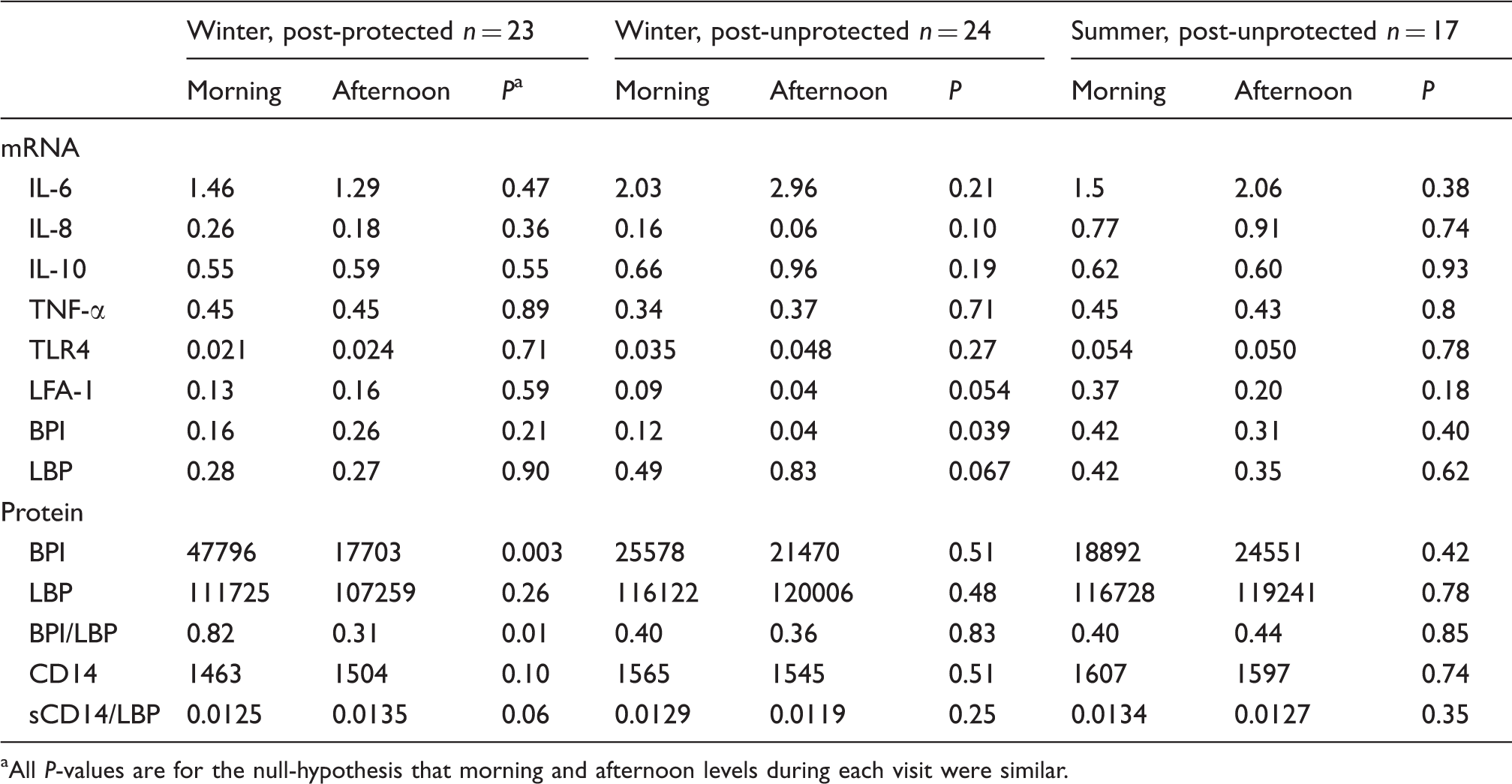
aAll P-values are for the null-hypothesis that morning and afternoon levels during each visit were similar.
Differences in the levels of expression of mRNA between the visits (Table 3) were observed for IL-8, LFA-1 and BPI. The expression of mRNA for IL-8 and LFA-1 was higher during summer than during any of the winter visits (LFA-1 mRNA; see Figure 1). BPI mRNA was lowest during the winter visit preceded by regular exposure. After the period with respiratory protection, BPI mRNA expression was more than twice that of the ordinary work period and during summer the expression was more than four times higher than during ordinary work in winter. BPI mRNA was the only mediator for which not only different levels, but also different patterns, of kinetics were observed between visits (Table 4 and Figure 1). A tendency to increase across the work shift after the respirator use and a decrease of 67% (P = 0.039) after the ordinary work period were observed (P = 0.0059 for an interaction between time and visit). No cross shift changes were observed during the summer visit.
Changes in selected mediators during the three farm visits. White bars: morning prior to work with swine. Grey bars: afternoon after work with swine. Horizontal lines: mean on the day of visit. W: winter visits; S: summer visit; +: respirator use prior to visit; -: ordinary unprotected work prior to visit. a: P ≤ 0.01 for difference in mean levels during the three visits; b: P ≤ 0.01 for difference in levels across the work shift; c: P ≤ 0.01 for difference in cross shift change during the three visits.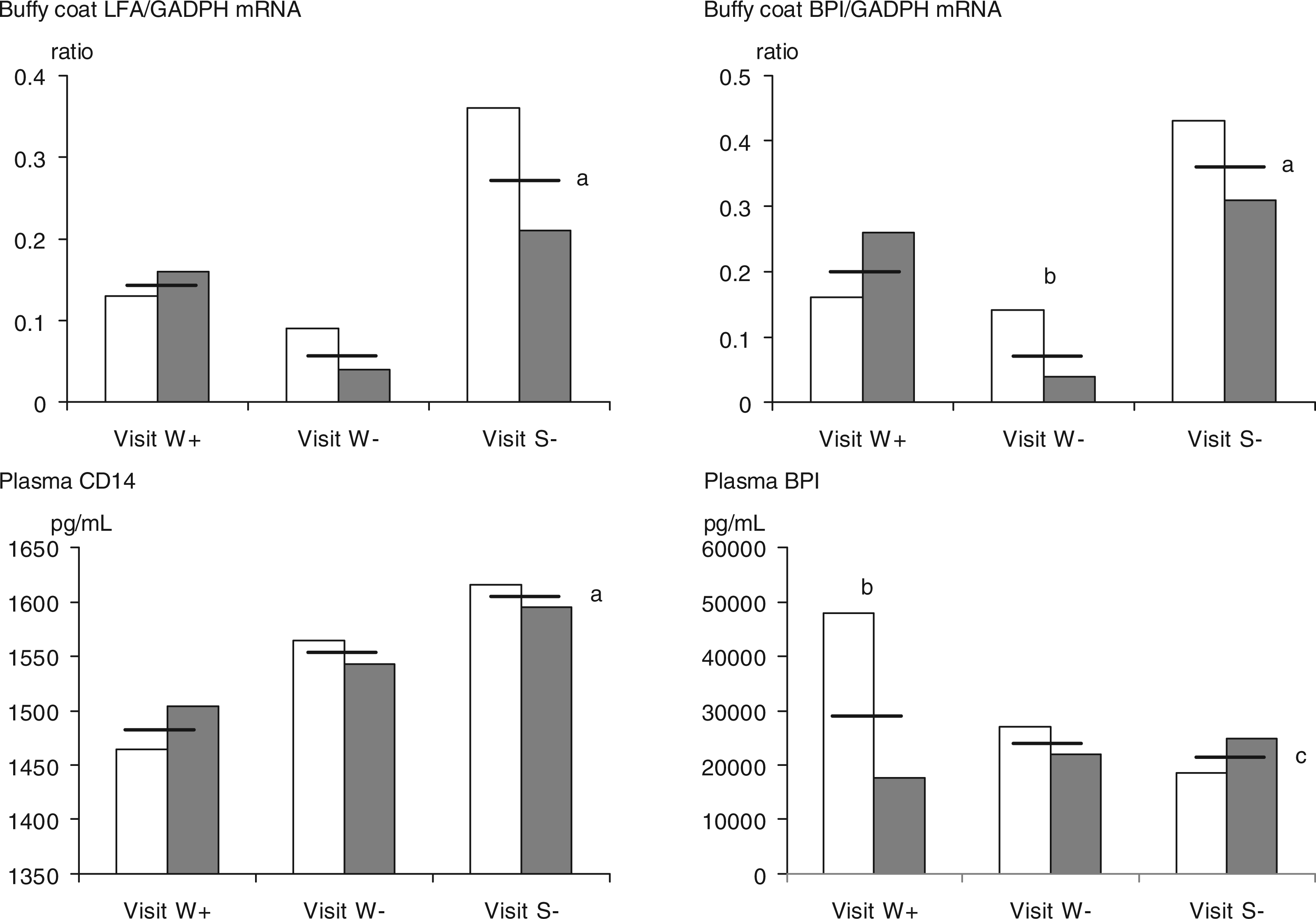
Of the plasma proteins assessed only CD14 was observed to differ in concentration between visits (P = 0.0102) with concentrations being higher during summer than during the two winter visits, and particularly higher than at the visit after respirator use (Figure 1). An interaction between time and visit appeared for plasma BPI (P = 0.006) caused by cross shift declines in this protein during the winter visits opposed by an increase during summer (Figure 1). As a result, differences in the cross shift changes of the BPI/LBP ratio appeared (P = 0.0104). During work after respirator use BPI/LBP decreased 62% (P = 0.01), whereas it did not change during the two other visits. Likewise the LBP/CD14 ratio behaved differently during the visits with a 7% decrease during work after respirator use (P = 0.06) contrasting the absence of cross shift changes during the other two visits.
A subgroup analysis was performed on the workers that adhered most stringently to the protocol by selecting those workers that had at least 6 d with respirator use or who were off work. The 17 workers in this subgroup did not differ significantly in demographic or work characteristics from the remaining workers (data not shown), and the findings were robust to this selection. Thus, in the subgroup the differences between visits in IL-8 mRNA (P = 0.007), LFA-1 mRNA (P = 0.0001) and BPI mRNA (P = 0.007) remained, with highest levels during summer. For plasma CD14 the difference between visits with lowest levels during the visit after respirator use remained close to statistical significance (P = 0.052). Regarding the visit*time interaction for plasma BPI it was somewhat weakened (P = 0.044).
Discussion
The study was successful in identifying mediators involved in inflammatory reactions in swine building workers that were differentially expressed across a work shift depending on season and/or the use of respiratory protection in the days prior to the work days. Three of the eight mediators for which mRNA expression in the buffy coat was measured showed greater expression during summer than during the two winter visits. This included—in addition to mRNA of IL-8 and LFA-1—the mRNA of BPI, which is particularly interesting considering that the protein level in plasma of BPI was highest during the post-respirator use visit during winter and not during summer.
It is tempting to speculate on the patterns observed in the mRNA expression across the work shift of these mediators, even though these patterns did not reveal any statistically significant cross shift changes. Thus, BPI mRNA exhibited opposite cross shift changes during the visit after use of respiratory protection where it tended to increase compared with tendencies to decrease during work after the non-protected periods summer and winter. Despite these tendencies, the plasma BPI decreased only across the work shift after respirator use. 8 A possible interpretation of this could be that BPI loss was greater at swine building exposure after respirator use than after the non-protected work periods, thus increasing demand for de novo synthesis of BPI. Such systemic BPI loss could be caused by different mechanisms, of which consumption from contact with endotoxin or post-translational modifications are two possibilities.
BPI is a major constituent of neutrophils, but is also expressed in human mucosal epithelia. 9 It has a high affinity for LPS and can bind significant amounts of endotoxin in the inflamed lung and antagonize the effects of LBP. LBP binds LPS in the lungs and can potentiate the response to LPS by transfer to CD14. LBP in the lungs is important in the response to inhaled LPS 10 and, although it may be produced locally, 11 increased plasma levels have been observed with lung injury. 12 Owing to the size of LBP, systemically produced LBP probably plays a minor role in the pulmonary reaction to inhaled LPS. Accordingly, we observed no cross shift changes in LBP but, owing to the changes in BPI concentration, the BPI/LBP ratio decreased dramatically during the work shift at the visit after respirator use. The BPI/LBP ratio is an important determinant of the response to LPS as BPI, when binding to LPS, inhibits an inflammatory response. Down-regulation of the organism’s response to LPS results from a higher BPI/LBP ratio, but will also result if LBP is less able to deliver LPS to CD14 because of a low sCD14/LBP ratio. 13 The opposite, i.e. a tendency for an increase in sCD14/LBP across the work shift, was observed only after the respirator use period (P = 0.06). Together these observations suggest greater potential for an inflammatory reaction to the abundant endotoxin in the swine building environment after just a few days of use of respiratory protection or absence from work. Our findings support the—mostly anecdotal—reports of more symptoms among such workers at return to work after holidays.
As, in mice, the transcriptional activation of BPI is controlled by TLR4 and other TLRs, it was surprising that no change in TLR4 mRNA was observed. 14 TLR4 is also the receptor on cell membranes that binds to the LPS–sCD14 complex delivered by LBP 15 and has been shown to be important in the response to LPS. 16 It is possible that any changes in TLR4 mRNA expression took place earlier in the work day and were no longer detectable at the time of our second blood sample.
The mRNA expression in blood leukocytes and plasma protein levels which we assessed in this study do not necessarily reflect local changes in the same mediators, e.g. in the airways exposed to the organic dust components of the swine buildings. Whether such local effects, which could have been studied in nasal and bronchial lavage if these had been included in the study, result in systemic changes that can be assessed in blood samples depend on their size and localization. Even if the effects caused by breathing during work in swine buildings was unlikely to result in systemic changes in inflammatory mediators, the inclusion of lavage could have improved our understanding of the mechanisms of our observations. However it was not feasible to perform lavage twice during the three visits of the study.
LFA-1 is an adhesion molecule important for neutrophil adhesion and is expressed on almost all immune cells. Basit et al. demonstrated that LFA-1 is important in neutrophil recruitment into the alveolar spaces after inhaled LPS and that with lack of LFA-1 this recruitment was lowered with more than 50%. 17 Conceivably among the workers of this study, neutrophil recruitment would be greatest during summer when LFA-1 mRNA expression was highest. However, our findings, indicated the opposite, with a tendency of lower blood neutrophil concentrations during summer than during winter. 7
TNF-α is a pro-inflammatory cytokine that increases in the lungs following endotoxin 18 and swine dust inhalation. 19 This increase is not followed by a systemic increase, 20 and plasma TNF-α was found to drop after swine house exposure in adapted and highly exposed workers. 7 The same was seen for one of its receptors, TNFRII, which is known to increase after i.v. endotoxin administration. 21 IL-6, a cytokine downstream of TNF-α known to increase 10-fold in serum of naïve subjects exposed to swine house work, 22 does not even double in adapted workers.7,23 IL10 is a cytokine with endotoxin tolerance-inducing properties that is produced by monocytes after endotoxin challenge, 24 but which has not been studied in swine farmers. Surprisingly, no effects of the working conditions tested in this study on the mRNA of TNF-α, IL-6 or IL-10 expression in plasma were observed.
In short-term studies of LPS delivered to the airways, bronchial lavage levels of mediators such as TNF-α and IL10 have been shown to increase with little change in systemic levels of the same mediators. 25 In studies of subjects chronically exposed to LPS via the airways little is known about the effects on systemic levels of mediators and whether such systemic changes are caused by excessive production in the lungs or by production elsewhere, for example in blood leukocytes. LPS was measured at similarly higher levels during the two winter visits than during summer,7,8 yet none of the mediators were observed at higher levels during winter. Rather, the mediators tended to be lower during summer and particularly so for CD14 and mRNA of LFA-1 and BPI (Table 3). This could suggest that LPS was not the important driver of the responses to the work environment. However, as the exposure to dust and gases followed the pattern for LPS and was also higher during winter, the factor that drove up the responses during summer would have to be temperature or humidity. We believe that this is an unlikely explanation for our findings and that the explanation is more likely to be adaptive mechanisms in the pulmonary and/or immune systems.
The major strengths of this study were the repeated measurements of the workers at the same time of day, thus eliminating many potential confounders, and the use of several mediators known to be involved in inflammatory reactions. Major drawbacks were the lack of bronchial lavage and the use of only two time points for blood sampling. The kinetics of mRNA expression differs from one mediator to another and the time window used in this study may not be right for several of the mediators chosen. The monocyte production of IL-10 for example is slow, with mRNA increases detectable 7 h after endotoxin challenge. 24 Our findings do not preclude a late rise in IL-10 mRNA (or other mRNAs) production that appears after the end of the work shift.
Furthermore, the use of the entire buffy coat with all its different cell types probably leads to the detection of high household gene expression from the bulk of cells not involved in immune reactions and only a limited range of increased of lowered expression compared with the household gene expression. With sufficient sensitivity of the assay this should not hamper our results. During airways inflammation the locally produced mediators may change little in their systemic production and, as discussed previously, we may have missed such local changes in the mRNA expression of mediators as we may have missed events occurring earlier or later than at the time of the second blood sample of the visits. Additional weaknesses of the study related to the limited number of participants and their adherence to respirator use protocol have been discussed elsewhere. 7
Conclusions
In this group of swine farm workers different concentrations of mRNA to IL-8, LFA-1 and BPI were observed between three visits preceded by different working conditions. In addition, differential work shift changes were observed in BPI mRNA and BPI itself. The findings suggest important roles for these proteins in adaptation to the swine building environment during repeated exposure.
Footnotes
Acknowledgements
We thank Serge Simard for advice and development of the statistical models.
Funding
This study was funded by the Fonds québécois de la recherche sur la nature et les technologies in collaboration with the Institut de Recherche Robert-Sauvé en Santé et Sécurité du Travail (2004-EN-100314). Jakob Bønløkke was a CIHR Strategic Training Fellow in PHARE, research funded by the CIHR Strategic Training Program and Partner Institutes.
Conflicts of interest
The authors have no conflicts of interest to declare.
