Abstract
Granulocytic myeloid-derived suppressor cells (MDSC) are a MDSC subset expanded in various cancer types. As many clinical studies rely on the use of stored collections of frozen blood samples, we first tested the influence of freezing/thawing procedures on immunophenotyping and enumeration of granulocytic MDSC (G-MDSC). To identify factors involved in expansion of human G-MDSC, we then analyzed correlations between G-MDSC frequencies, clinical parameters and granulocyte-related factors in the peripheral blood of head and neck cancer patients. HLA-DR, CD14, CD33 and CD66b allowed a clear discrimination of G-MDSC from monocytic MDSC and immature myeloid cells. MDSC subsets were sensitive to cryopreservation with immature G-MDSC showing the highest sensitivity. G-MDSC frequencies were increased in advanced disease stage and associated with the level of CCL4 and CXCL8, but not with colony-stimulating factors, IL-6, S100A8/9, CXCL1 and other cytokines. Our results indicate that the frequency of MDSC, in particular G–MDSC, may be underestimated in retrospective clinical analyses using frozen blood samples. Increased G-MDSC frequencies correlate with advanced disease and increased concentrations of CXCL8, but, unexpectedly, not with growth factors (such as granulocyte colony-stimulating factor), IL-6 and CXCL1. Our data suggest that CXCL8 promotes accumulation of G-MDSC in cancer patients independent of classical colony-stimulating factors.
Keywords
Introduction
Myeloid-derived suppressor cells (MDSC) are a heterogeneous subset of immunoregulatory cells present in most patients and animals with cancer. 1 In humans, MDSC are divided into at least two or three major subsets, namely granulocytic/polymorphonuclear MDSC (G-MDSC), 2 monocytic MDSC (Mo-MDSC) 3 and immature myeloid cells (imMC) 4 (for an overview on immunophenotyping of MDSC see Greten et al. 5 and Dumitru et al. 6 ). Because substantial differences between G- and Mo-MDSC exist in mice, 1 it seems to be crucial to also investigate separately these subsets in cancer patients. Recent studies suggest that high frequencies of MDSC result in worse prognosis.7,8 A better understanding of the molecular mechanisms regulating MDSC differentiation and function is needed to assess the prognostic relevance and potential therapeutic targeting of these cells in cancer patients. 9
An important question in MDSC biology is how MDSC accumulation is regulated; potentially, it requires either one or two signals. 9 In mice and in human in vitro systems, cytokines, such as IL-6 and in particular colony-stimulating factors [granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF)], have been implicated in the expansion of G-MDSC.7,10,11 These factors may also be involved in G-MDSC expansion in human cancer patients. Therefore, we analyzed potential correlations between G-MDSC frequencies and granulocyte-related peripheral blood cytokines and growth factors. Many clinical immunomonitoring studies rely on the use of large collections of pre-stored frozen blood samples. However, granulocytes and certain subsets of MDSC may be more sensitive to cryopreservation than lymphocytes. 12 Therefore, we first tested the influence of freezing/thawing procedures on human MDSC immunophenotyping (immune monitoring). For this purpose we used a recently proposed immunophenotyping protocol, which identifies G-MDSC, Mo-MDSC and imMC within one sample by six-color fluorescence. 6
Materials and methods
Study subjects
Blood samples were collected prospectively from patients with head and neck squamous cell carcinoma (HNSCC) before oncologic therapy. Altogether, 63 patients with HNSCC of oral cavity, oropharynx, hypopharynx or larynx were included from 2008 to 2011 (Supplementary Table 1). Exclusion criteria were HNSCC in other locations, prior radiotherapy or chemotherapy, synchronous carcinoma in another location or severe concomitant systemic infectious disease. The experiments were performed according to the Helsinki Declaration and approved by the local ethics committee. Written informed consent was obtained from all subjects before sample collection.
Isolation of PBMC including MDSC
We used an established protocol for isolation of PBMC. 13 In brief, peripheral blood of HNSCC patients was drawn into 3.8% sodium citrate anticoagulant and admixed with PBS (ratio 1 : 1 v/v) before separation by density gradient centrifugation (lymphocyte separation medium, PAA Laboratories, Pasching, Austria), a standard procedure in many MDSC studies (see, e.g., Gabitass et al. 8 and Kotsakis et al. 12 ). Mononuclear cells and co-sedimenting MDSC were harvested from the interphase. PBMC were used immediately after isolation for characterization of MDSC or were frozen for later analysis.
Freeze-thaw procedure for PBMC/MDSC
Method A
First, PBMC were frozen. Freezing medium (90% FCS + 10% DMSO) was pre-chilled on ice. PBMC were resuspended in 1 ml freezing medium and kept directly on ice. The vial was transferred into a pre-chilled freezing container (cooling rate −1℃/min) and stored for up to 1 wk at −80℃. For long-term storage, vials were transferred into liquid nitrogen. Second, PBMC were thawed. RPMI 1640 (10 ml; Invitrogen, Karlsruhe, Germany) supplemented with 10% FCS (Biochrom, Berlin, Germany) and 1% penicillin–streptomycin (Invitrogen) was pre-warmed at 37℃. Frozen vials were warmed up at 37℃ in a water bath until a small lump of ice was left. PBMC were then transferred into warm medium and centrifuged (300 g, 7 min). After determination of cell numbers (Casy Model TT cell counter; Roche Innovatis, Mannheim, Germany), cells were used for MDSC characterization.
Method B
PBMC were frozen according to method A. PBMC were thawed according to method A, but pre-warmed FCS was used instead of supplemented RPMI 1640.
Method C
PBMC were first frozen by resuspending in 900 µl FCS, adding 100 µl DMSO and storing the 15 ml vial immediately at −80℃. PBMC were then thawed. Pre-warmed FCS (10 ml) was added to the frozen cells, the vial was warmed up at 37℃ in a water bath until a lump of ice was left, and centrifuged.
Flow cytometry
For characterization of MDSC subsets the following Abs were used: CD66b FITC (clone 80H3, Beckman Coulter, Krefeld, Germany), CD14 APC-Cy7 (clone MphiP9), CD33 PE (WM53), human leukocyte antigen (HLA)-DR APC (clone G46-6), CD16 PE-Cy7 (clone 3G8), CD11b PE-Cy7 (clone Mac1; CR3) and 7-aminoactinomycin D (all from BD Bioscience, Heidelberg, Germany). Cells were analyzed with BD Canto II using DIVA 6.0 software (BD Bioscience).
Peripheral blood analyses
Peripheral blood measurements included differential hemogram, C-reactive protein (CRP), measured with automated analyzers (hemogram, Sysmex XE5000; Sysmex, Norderstedt, Germany; CRP, ADVIA 1800/2400; Siemens Healthcare, Erlangen, Germany), and cytokine analysis. Serum concentrations of CCL3, CCL4 and CCL5 were determined with Bio-Plex Chemokine Assays (based on Luminex xMAP technology; Bio-Rad Laboratories, München, Germany), serum procalcitonin by immunoluminometric assay (Brahms, Henningsdorf, Germany) and serum concentrations of CXCL8, G-CSF, GM-CSF, CXCL1(R&D Systems, Wiesbaden, Germany) and S100 A8/A9 complex (Cusabio Biotech, Wuhan, China) by ELISA.
Statistical analysis
Standard descriptive statistics were used. To assess between-group differences we used non-parametric exact Mann–Whitney–Wilcoxon U test. Correlation coefficients reported are (Spearman) rank correlations. Two-sample Student’s t-tests were used for in vitro experiments. P-values are two-sided and significance level was 0.05 (using SPSS Version 16; SPSS Inc., Chicago, IL, USA).
Results and discussion
Immunophenotyping of MDSC
Similar to mice, human MDSC also contain different subsets. 1 Human MDSC are most easily analyzed in the PBMC fraction of peripheral blood subjected to density gradient centrifugation.2–4,8,13 Markers commonly used for human MDSC characterization include CD11b, CD14, CD33, CD15 or CD66b, human leukocyte antigen (HLA)-DR and, in some cases, lineage negativity.5,6
Based on previous publications, we used multicolor flow cytometry and Abs directed against CD14, CD33, CD66b and HLA-DR to phenotype MDSC, isolated with the PBMC from peripheral blood of HNSCC patients. With this set of markers we were able to clearly discriminate G-MDSC2,13 (CD66b+/CD33int/HLA-DR−) from Mo-MDSC
3
(CD33+/CD14+/HLA-DRlow) and imMC
4
(CD33int/CD66b−/HLA-DR−) (Figure 1A, left panels). According to our experience, CD11b provides no additional information for immunophenotyping of G‐MDSC if CD33, CD14 and CD66b are included. Instead, gating on CD11b+ will exclude immature G-MDSC, which lack expression of CD11b, but do express CD33.
13
CD15 is less specific than CD66b in identifying G-MDSC as, in contrast to CD66b, monocytic cells also express low levels of CD15 (data not shown).
MDSC are sensitive to cryopreservation. (A) Multicolor staining of different MDCS subsets immediately after isolation and after freezing/thawing procedure of PBMC (for procedures ‘A’, ‘B’ and ‘C see Materials and methods). First row gated on PBMC, second row gated on CD33dim HLA-DR−. (B) Frequency of CD66b+ granulocytic MDSC (G-MDSC) as percent within PBMC directly after isolation and in freeze/thawed samples and identified according to (A). Each symbol represents an individual donor. (C) Three representative patients were analyzed for CD16+ and CD16− G-MDSC in fresh and freeze/thawed blood samples.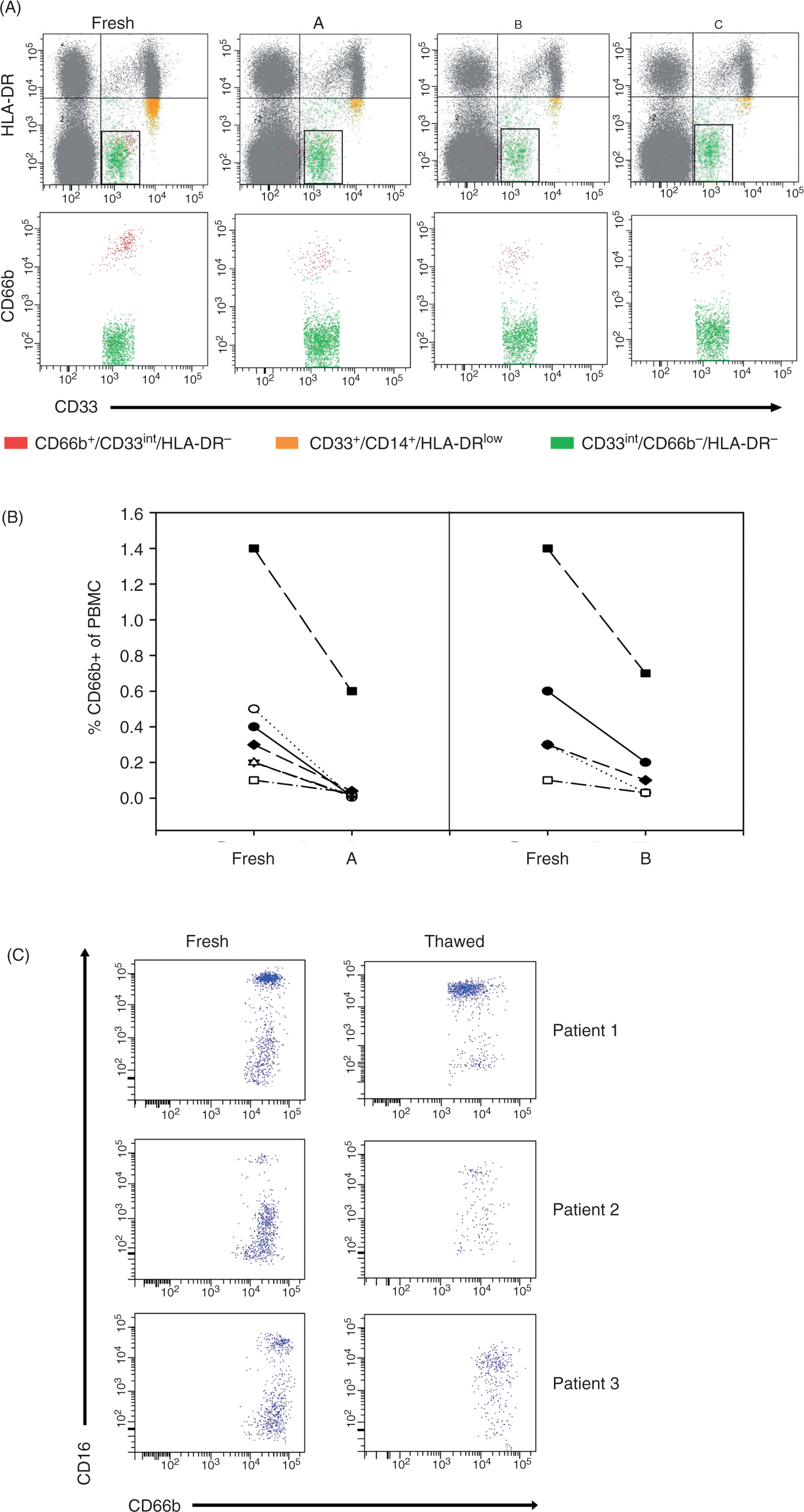
Decreased frequency of G-MDSC in thawed blood samples
The analysis and correlation of MDSC frequency, serum markers and clinical follow-up in larger clinical and explorative human studies would be very much facilitated by the possibility of using frozen stored immune cells and serum. Differential sensitivity of mature and hematopoietic progenitor cells to cryopreservation has been recognized in the field of hematopoietic stem cells transplantation. 14 However, only one study regarding the effect of freezing on MDSC frequencies in cancer patients is available at present. 12 In that study, a lineage cocktail was used, which includes CD14 and CD16. This will exclude classical CD14+Mo-MDSC and CD16+/CD15+/CD66b+G-MDSC. Consequently, and in contrast to our study, their study analyzed rare subsets of MDSCs with frequencies below 1% for all subsets and all cancer patients tested. 12 Considering the increasing interest in human MDSC biology, the usage of frozen blood samples in current clinical studies,8,15 and the paucity of information available on cryopreserved MDSCs, we first tested the effect of freezing and thawing on frequency and composition of the major MDSC subsets in PBMC of HNSCC patients. Because of the described cryosensitivity of granulocytic cells, we hypothesized that freezing of blood samples could also falsify enumeration of G-MDSC in cancer patients.
We tested three different freeze/thawing procedures and found that cryopreservation did, indeed, result in reduced frequencies of G-MDSC (Figure 1A). Frequency of CD66b+ G-MDSC was reduced by more than 50% in all samples tested (Figure 1B). Recently, we have shown that G-MDSC consist of a mixture of immature CD16− polymorphonuclear neutrophil (PMN) precursors and more mature CD16+ cells. 13 In this study, the percentage of CD16− cells within G-MDSC ranged between 0.6% and 99.5% (median 28.4%, mean 37.8 ± 29.3%, n = 24, data not shown). These immature CD16– G-MDSC were most sensitive to freezing/thawing procedures (Figure 1C). Thus, the frequency of G-MDSC and especially of their immature subset may be underestimated if frozen blood samples are analyzed. In this study we focused on the identification of factors likely to be involved in the numerical expansion of G-MDSC in human cancer patients. Therefore, functional MDSC analysis was not intended in this study. Nevertheless, it is important to note that Kotsakis et al. reported recently that MDSC functions may also be affected by cryopreservation. 12 Interestingly, other MDSC subsets were also sensitive to freeze/thaw procedures. While Mo-MDSC showed similar sensitivity compared to G-MDSC, imMC were only slightly sensitive (percentage of viable cells after freeze/thaw procedure ‘A’ or ‘B’: G-MDSC 14 ± 16% or 31 ± 14%; Mo-MDSC 29 ± 46% or 25 ± 9%; and imMC 81 ± 19 or 96 ± 6%).
G-MDSC counts are increased in advanced disease
After we had established that fresh, and not frozen, blood samples are required for reliable G-MDSC immunophenotyping and enumeration, in the second part of the study we wanted to correlate G-MDSC frequencies with clinical parameters and serum cytokines potentially involved in expansion of G-MDSC. To this end, we analyzed a cohort of patients with HNSCC from whom both G-MDSC counts (direct analysis of fresh leukocytes) and serum samples were available. The G-MDSC count ranged between 0.1% and 44.7% within the PBMC fraction (Figure 2A). G-MDSC counts were associated with disease stage (Figure 2B) and T stage (Figure 2C). The presence of lymph node metastasis (N-stage) was not associated with G-MDSC frequency (Figure 2D). Furthermore, no associations were found for histological grading, tumor localization or age (data not shown). While no study on a homogeneous large cohort of HNSCC patients was available thus far, in a study analyzing MDSC from different cancer types with emphasis on breast cancer,
16
a correlation between circulating MDSC and disease stage was also observed.
Frequency of G-MDSC in the PBMC fraction is associated with disease stage. (A) Frequency of G-MDSC within the PBMC fraction (n = 63, identification with anti-CD66b mAb). The median percentage of MDSC in the PBMC fraction was 2.7, the mean 7.2 ± 9 % (indicated with a horizontal line). (B) The G-MDSC frequency is significantly increased in advanced disease (stage III or IV, n = 51) compared with early disease (stage I or II, n = 12) (P = 0.018). This seems to be a result of an association to T stage (T1/T2 versus T3/T4) (C), not N stage (N0 versus N+) (D). (B–D) Shown are the median, percentiles (10th, 25th, 75th, 90th) and mean (dashed line) as vertical boxes with error bars, outliers as dots. P-values (Mann–Whitney) are indicated (significance level 0.05) (n = 63).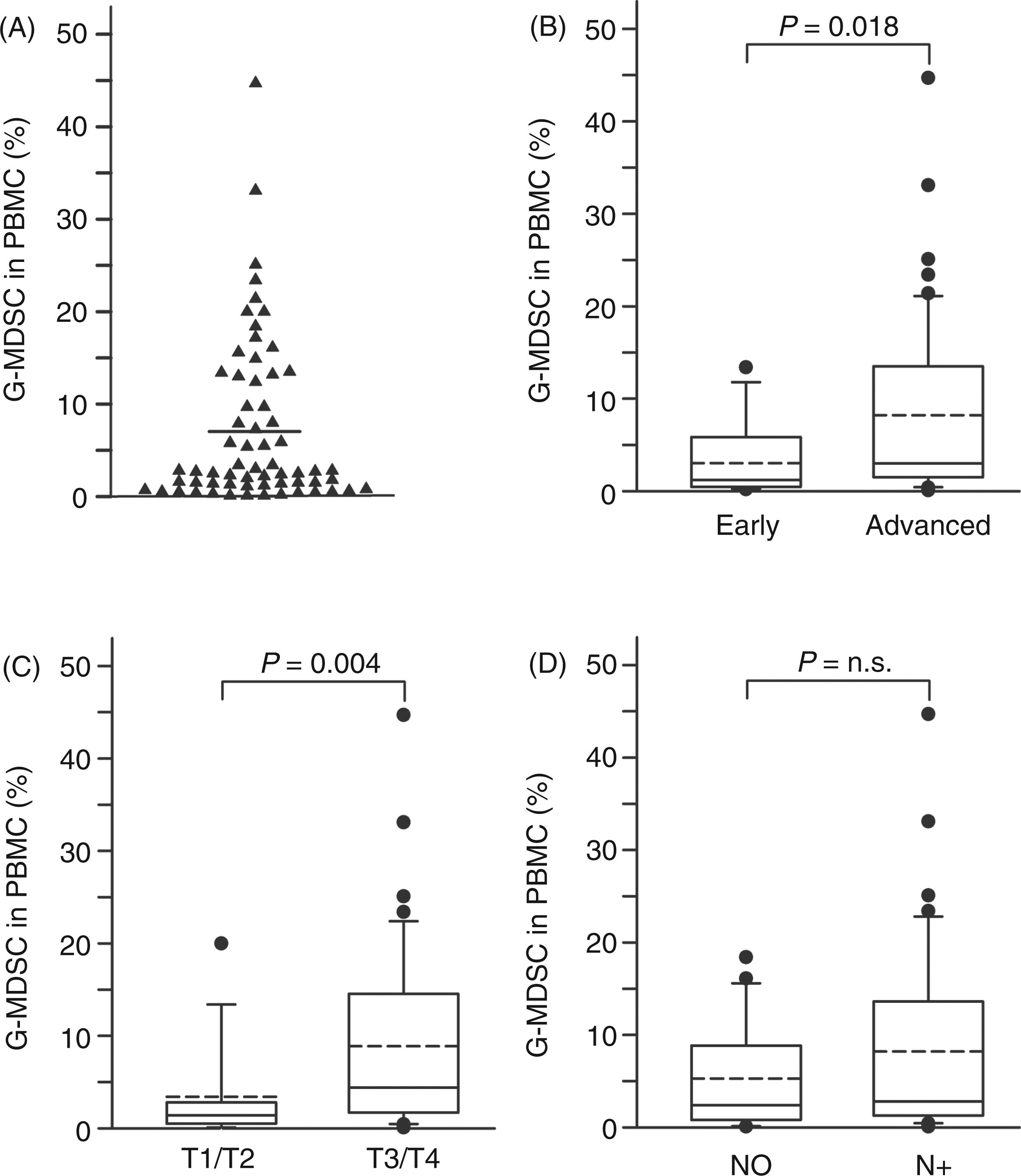
G-MDSC counts are associated with serum CXCL8 and CCL4, but not with CXCL1
HNSCC patients regularly show signs of cancer-related inflammation with increased systemic inflammatory mediators such as neutrophil-related cytokines.
17
When we analyzed PMN-related serum cytokines/chemokines, we found that G-MDSC counts were positively, and significantly, associated with CXCL8 (r = 0.523, P = 0.002) (Figure 3A) and CCL4 (r = 0.620, P = 0.001) (Figure 3B). These associations were independent of tumor stage and consistent in multivariate analysis (data not shown). No association was found between G-MDSC counts and other peripheral blood parameters, such as differential hemogram (leukocytes, neutrophil counts, neutrophil-to-lymphocyte ratio), CCL3 and CCL5 (data not shown). Interestingly, G-MDSC counts were not associated with CXCL1 in the cohort tested (Figure 3C, discussion below). S100 proteins have been implicated in the expansion of MDSC in murine models
18
and S100A9 has been suggested as a marker for human Mo-MDSC.
19
We found that HNSCC patients with high numbers of G-MDSC showed a trend toward higher expression of the S100A8/A9 complex in serum (P = 0.066, n = 54).
The G-MDSC count is associated with CCL4 and CXCL8, but not with CXCL1 and S100 A8/A9. G-MDSC count (see Figure 1 and Brandau et al.
13
) was determined by flow cytometry and plotted against levels of serum CXCL8 (A) (n = 32) , CCL4 (B) (n = 26), CXCL1 (C) (n = 27) and S100 A8/A9 (D) (n = 54). For graphical demonstration, two groups of low (below mean) and high (above mean) G-MDSC counts are shown. Box-plots and statistics as in Figure 2.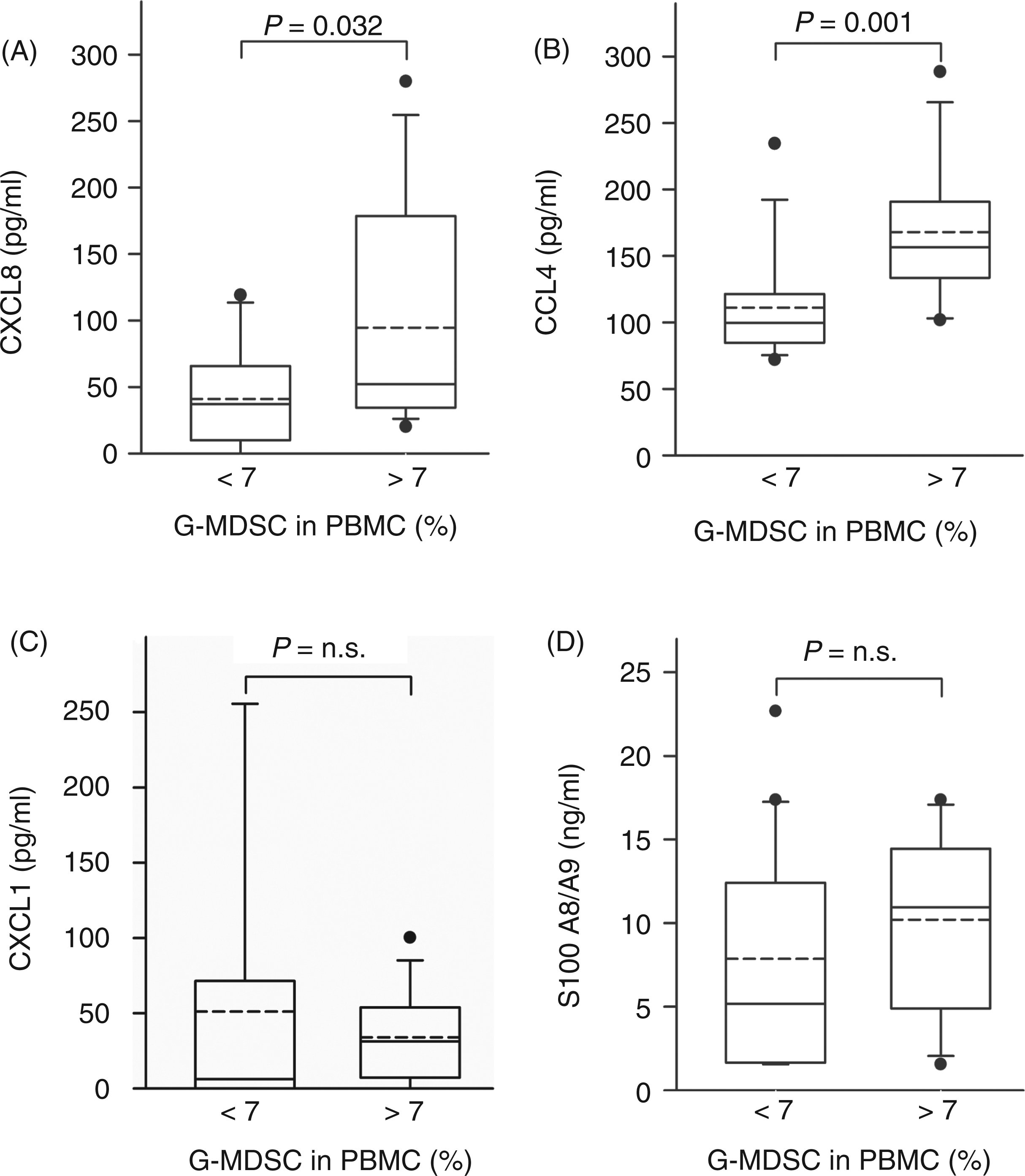
Elevated levels of CXCL8 (and possibly also S100 proteins) may derive from tumor cells or leukocytes (such as G-MDSC/neutrophils), both as a result of secretion or cell death. While studies on S100 proteins in HNSCC patients are still underway, in a former study, we have shown that head and neck cancer cells produce CXCL8 in vivo and in vitro. 17 In another study we observed no increase in production of CXCL8 by peripheral blood neutrophils from HNSCC patients compared with healthy donors. 20 However, overall neutrophil numbers and the neutrophil-to-lymphocyte ratio are increased in cancer patients.6,17 In addition, neutrophils from cancer patients showed reduced spontaneous apoptosis suggesting a longer half-live of these cells in cancer patients. This could mean that, in addition to tumor cells, neutrophils, and probably G-MDSC, also contribute to increased CXCL8 in the serum of cancer patients.
CSFs and IL-6 are not associated with G-MDSC frequencies
Colony-stimulating factors such as GM-CSF and G-CSF are typical candidates for granulocyte release into the peripheral blood in normal physiology and bacterial infection. These factors, together with IL-6, have been suggested as inducers of MDSC accumulation and expansion.7,9,10 Furthermore, CRP, a general inflammatory marker, and procalcitonin, a marker of extent bacterial inflammation, were tested for their correlation with MDSC accumulation.
GM-CSF and procalcitonin were not detectable in the majority of patients (Figure 4A) and, even if detected, no correlation with clinical parameters was found (data not shown). Increased CRP was associated with higher T stage,
17
but did not correlate with G-MDSC frequency (data not shown). G-CSF was not correlated to the G-MDSC count (r = −0.097, P = 0.522, n = 46) (Figure 4B) or differential hemogram counts (e.g. the percentage of neutrophils: r = −2.94, P = 0.115, n = 30) (Figure 4C). Also, no association was observed between serum IL-6 and G-MDSC count (Figure 4D).
G-MDSC is not associated with CSFs and IL-6. (A) GM-CSF (n = 46) was measured by ELISA and procalcitonin (n = 44) by immunoluminometric assay. The majority of patients had no detectable serum concentrations in both parameters. Shown are the number of patients in percentage for three different concentration ranges of GM-CSF (black columns) and procalcitonin (white columns). (B, C) Serum G-CSF was measured by ELISA (n = 40) and was associated neither to the G-MDSC count (B) nor to the differential hemogram counts (C). Shown (in B) are box plots according to Figures 2 and 3, and simple dot plots in (C). (D) Serum IL-6 was analyzed by ELISA (n = 33). The majority of patients had no detectable IL-6 values. No association was observed between serum IL-6 and G-MDSC count (P = n.s.). Shown are the number of patients in percent for the group of low (below mean, black columns) and high granulocytic myeloid derived suppressor cells (G-MDSC) count (above mean, white columns) for the three ranges of IL-6 values measured. NP: neutrophils; Procalc.: procalcitonin.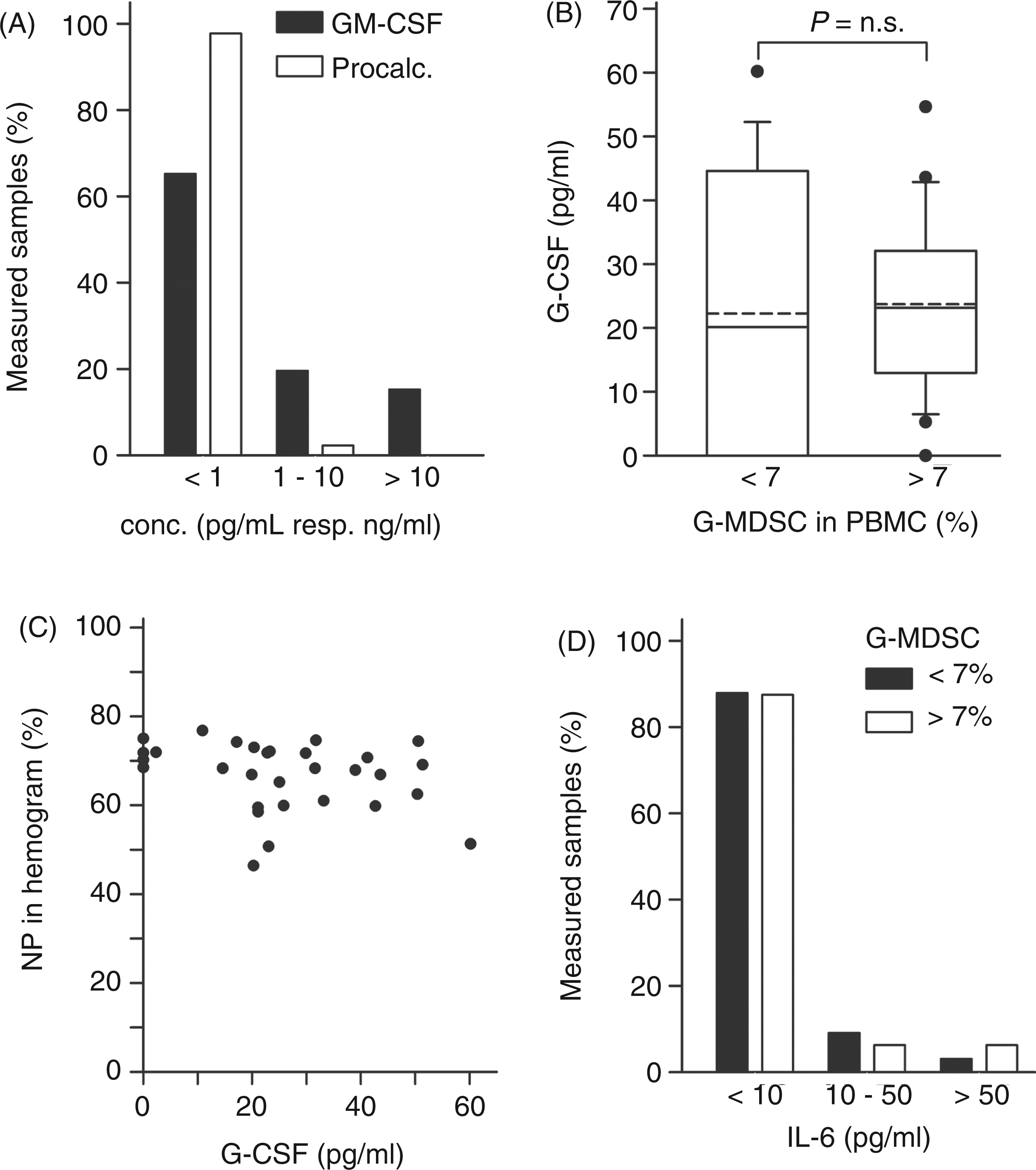
In a recent study by Gabitass et al. 8 G-CSF was not induced in the plasma of patients with gastrointestinal and other types of cancer. Partly in contrast to that, elevated G-CSF levels, as well as high MDSC counts with a high percentage of neutrophilic MDSC, were observed in glioblastoma patients. 21 However, no correlation testing was described in that study, and CXCL8 and IL-6 were not increased in glioblastoma patients.
Mobilization and recruitment of neutrophils from bone marrow to inflamed sites is mediated by CXCR2 ligands, such as CXCL1 and CXCL8, together with G-CSF.22,23 In mice, serum levels of murine CXCL1 (KC) were increased shortly after injection of human G-CSF, leading to mobilization of neutrophils. 23 Recently, G-CSF-triggered stem cell mobilization in humans was shown to induce cells reminiscent of G-MDSC. 24 In our cohort of cancer patients, G-MDSC frequencies were associated with the CXCR2-ligand CXCL8, but not with CXCL1 (Gro-α, another CXCR2 ligand) and G-CSF. As a CXCL8 homolog has not been identified in mice, it has been suggested that mouse CXCL1 is a functional homolog of human CXCL8. 25 We assume that in mice tumor-triggered G-CSF may induce expansion of G-MDSC directly or indirectly via CXCL1. By contrast, mobilization of G-MDSC in humans may be mediated independently of G-CSF via another CXCR2 ligand, namely CXCL8.
Conclusion
In accordance with other studies, which found no increase of G-CSF in the serum of cancer patients, 8 our data argue against a role for CSFs in driving accumulation of G-MDSC in HNSCC patients. No correlation of G-MDSC with CRP, procalcitonin and neutrophil-lymphocyte-ratio also argue against an accumulation of MDSC as part of a general cancer-related inflammatory process. Instead, increased G-MDSC counts rather appear to correlate with myeloid effector chemokines, such as CCL4 and CXCL8, which are produced in large amounts by activated myeloid cells also in cancer patients.26,27 These activated myeloid cells could either be MDSCs themselves 27 or non-MDSC myeloid cells, such as classical monocytes, 26 which become activated in response to serum-derived factors. It has been shown that neutrophils and G-MDSC in cancer patients exhibit a prolonged lifespan compared to their counterparts in healthy donors and if exposed to tumor-conditioned medium.13,20,28 Thus, we would like to propose that induced serum concentrations of CXCL8 mediate delayed apoptosis and increased mobilization from bone-marrow (possibly linked to a block in maturation) finally resulting in the accumulation of G-MDSC. 29 Certainly, future studies, which should use subsets of MDSC isolated directly from fresh, non-cryopreserved blood samples, are needed to further unravel the complex differentiation, mobilization and activation of these cells in human cancer patients.
Footnotes
Acknowledgements
We thank Petra Altenhoff for technical assistance and Antje Sucker (Department of Dermatology, University Hospital Essen) for advice regarding cryopreservation of cells.
Funding
This work was supported, in part, by the Deutsche Forschungsgemeinschaft (BR 2278/2-1) (S.B.), the Medical Faculty of the University Duisburg-Essen (IFORES program) (S.B.) and the Krebsgesellschaft NRW (S.B. and S.L.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
