Abstract
Chronic periodontal diseases are characterised by a dysregulated and exaggerated inflammatory/immune response to plaque bacteria. We have demonstrated previously that oral keratinocytes up-regulate key molecular markers of inflammation, including NF-κB and cytokine signalling, when exposed to the periodontal bacteria Porphyromonas gingivalis and Fusobacterium nucleatum in vitro. The purpose of the current study was to investigate whether α-lipoic acid was able to abrogate bacterially-induced pro-inflammatory changes in the H400 oral epithelial cell line. Initial studies indicated that α-lipoic acid supplementation (1–4 m
Introduction
Periodontitis is a ubiquitous chronic inflammatory disease that destroys the supporting structures of teeth; the most common form is chronic periodontitis which, if untreated, results in the breakdown of soft tissues and bone, leading, ultimately, to tooth loss. The disease-initiating factor is plaque bacteria present in a biofilm at, and below, the gingival margin; 1 however, the subsequent tissue damage that characterises this disease is the result of an aberrant and exaggerated host response within susceptible individuals. 2 The plaque biofilm is a complex ecosystem containing a wide diversity of bacteria, two Gram-negative anaerobes Porphyromonas gingivalis and Fusobacterium nucleatum, which are widely regarded as key to disease progression. 3 Fusobacterium nucleatum interacts with other early colonising bacteria enabling a ‘bridge’ for the development of a more mature and pathogenic biofilm 4 that is colonised subsequently by strict anaerobes, including P. gingivalis, which expresses a range of virulence factors and is strongly associated with periodontal disease pathogenesis. 5
An aberrant host immune response has been reported in periodontitis, with neutrophils from patients demonstrating hyperactivity/reactivity 2 and their excessive production of reactive oxygen species (ROS) is believed to be partly responsible for the local tissue damage, exacerbated by reduced local tissue antioxidant defences. 6 The mechanisms whereby neutrophils are recruited to, or cleared from, the periodontal tissues may also have an important bearing on the aetiology of this disease. The sulcular and junctional epithelia, in common with similar epithelia at other sites in the body, were originally thought to simply provide a passive physical barrier, protecting the host from the external environment. More recently, however, periodontal epithelium has been shown to be a key orchestrator of the inflammatory response to a colonising biofilm and certain bacteria present within. 7 Indeed, recent work has demonstrated that periodontitis-associated bacteria, such as P. gingivalis and F. nucleatum, can initiate keratinocyte pro-inflammatory responses resulting in elevations in cytokine transcript and protein production in vitro. 8 Central to the pro-inflammatory response is NF-κB, a key REDOX-sensitive transcription factor activated by increases in intracellular oxidative stress caused by a wide range of factors, including bacterial stimulation,8,9 resulting in downstream changes in gene expression and cytokine production.
α-Lipoic acid (ALA) (also known as α-lipoate or thiocytic acid) is a naturally-occurring di-thiol present in a wide range of food substances, including meats, such as liver, kidney and heart, as well as vegetables, such as spinach and broccoli. Recently, ALA has attracted considerable attention owing to its varied antioxidant actions. ALA is able to reduce levels of oxidative stress by (i) direct free radical scavenging and (ii) regeneration of other intracellular antioxidants,10,11 most notably glutathione—a key antioxidant in maintaining cellular REDOX status. 12
Owing to its ability to boost levels of intracellular glutathione, ALA is able to regulate NF-κB activation. 13 ALA has undergone extensive clinical trials and has been shown to have efficacy in the treatment of several chronic inflammatory diseases, for example diabetes and cardiovascular disease.14,15 ALA therefore represents a potential candidate for modulation of NF-κB and the associated pro-inflammatory activity of periodontal epithelium. Thus, the aim of this study was to investigate the effect of ALA on the pro-inflammatory response of oral keratinocytes exposed to periodontal bacteria by analysing NF-κB activation, gene expression and cytokine production.
Materials and methods
Bacterial culture and suspensions
Escherichia coli LPS (serotype 026:B6; Sigma, Poole, UK) was reconstituted with cell culture growth media (DMEM; Invitrogen, Paisley, UK) to produce a stock solution (250 µg/ml) which was stored at −20°C prior to use. Porphyromonas gingivalis (ATCC33277, isolated from the human gingival sulcus) and F. nucleatum (ATCC10953, isolated from inflamed human gingivae) were grown in broth culture at 37°C anaerobically, as described previously. 8 Cell suspensions were centrifuged, and the bacterial pellet washed three times using PBS, heat-inactivated (100°C for 10 min) and suspended in PBS to give a final concentration of 4 × 108 cells/ml. Heat inactivation was confirmed by plating and culturing experiments. Bacterial suspensions were stored at −20°C prior to use for keratinocyte exposure.
Cell culture
Details of cell culture methods were as described previoulsy.8,16 Cells were grown in a range of plastic-ware, including flasks, 96-well plates and Petri dishes (including some containing multi-well glass slides). H400 cells (an immortal cell line derived from an oral squamous cell carcinoma of the alveolar process in a 55-year-old patient) were cultured in DMEM (Invitrogen) supplemented with 10% FCS (Labtech, Ringmer, UK), 4 m
ALA
A 0.06 M ALA (Sigma) stock solution was prepared in 0.029 M sodium hydroxide. The stock solution was stored at −20°C in aliquots prior to use
Effect of ALA on cell adherence
Cells were pre-incubated with ALA (0.0625–4 m
Effect of ALA on NF-κB nuclear translocation in LPS stimulated keratinocytes
Escherichia coli LPS, a validated activator of NF-κB nuclear translocation in various cell types, was used as a known positive control prior to testing relevant periodontal pathogens in our test system. H400 cells cultured on glass slides within Petri dishes were pre-incubated with ALA (0.25–2 m
Effect of ALA on keratinocytes stimulated with periodontal bacteria
H400 cells were pre-incubated for 24 h in 0.5 m
Immunocytochemistry of NF-κB translocation
Cell monolayers were fixed using dry acetone for 10 min at room temperature (20–25°C), air dried and stained by immunohistochemistry. The staining protocol employed a MAb to the NF-κB p65 subunit (clone F-6, diluted 1 : 100; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and followed a biotin–streptavidin immunoperoxidase technique (StrAviGen, Biogenex, San Ramon, CA, USA) as reported previously. 8 The resulting bound peroxidase was visualised using 3,3′-diaminobenzidine reagent for 5 min and counterstained with Meyer’s haematoxylin before mounting in Xam. Slide-washing and dilution of reagents were performed using 0.01 M PBS, pH 7.6. Positive- and negative-staining controls were included, which consisted of replacement of the NF-κB monoclonal with Ki67 (clone MM1, diluted 1 : 100; NovacastraTM, Vision Biosystems, Newcastle, UK) as a positive control and replacement of NF-κB Ab with PBS or a MAb with irrelevant specificity, but the same immunoglobulin isotype as a negative control.
Quantification of NF-κB translocation was performed on cell monolayers using a microscope (Leica, Wetzlar, Germany) fitted with an eyepiece graticule (10 × 10 0.01 mm2 squares) and viewed at 100 × magnification. Analysis using Hunting curves indicated that counting of 30 randomly selected fields provided representative cell counts. For cells to be classified as demonstrating NF-κB activation (positive), nuclear staining for p65 was required with no residual cytoplasmic staining evident. The total and mean numbers of positive cells were determined to calculate the percentage of cells demonstrating NF-κB activation.
High content analysis of NF-κB nuclear translocation
H400 cells were cultured in 96-well plates (Corning, Amsterdam, The Netherlands) to semi-confluence and either pre-incubated with 0.5 m
Semi-quantitative RT-PCR analysis
Details of primer sequence and semi-quantitative RT-PCR conditions. All DNA sequences are shown in the 5' to 3' orientation.
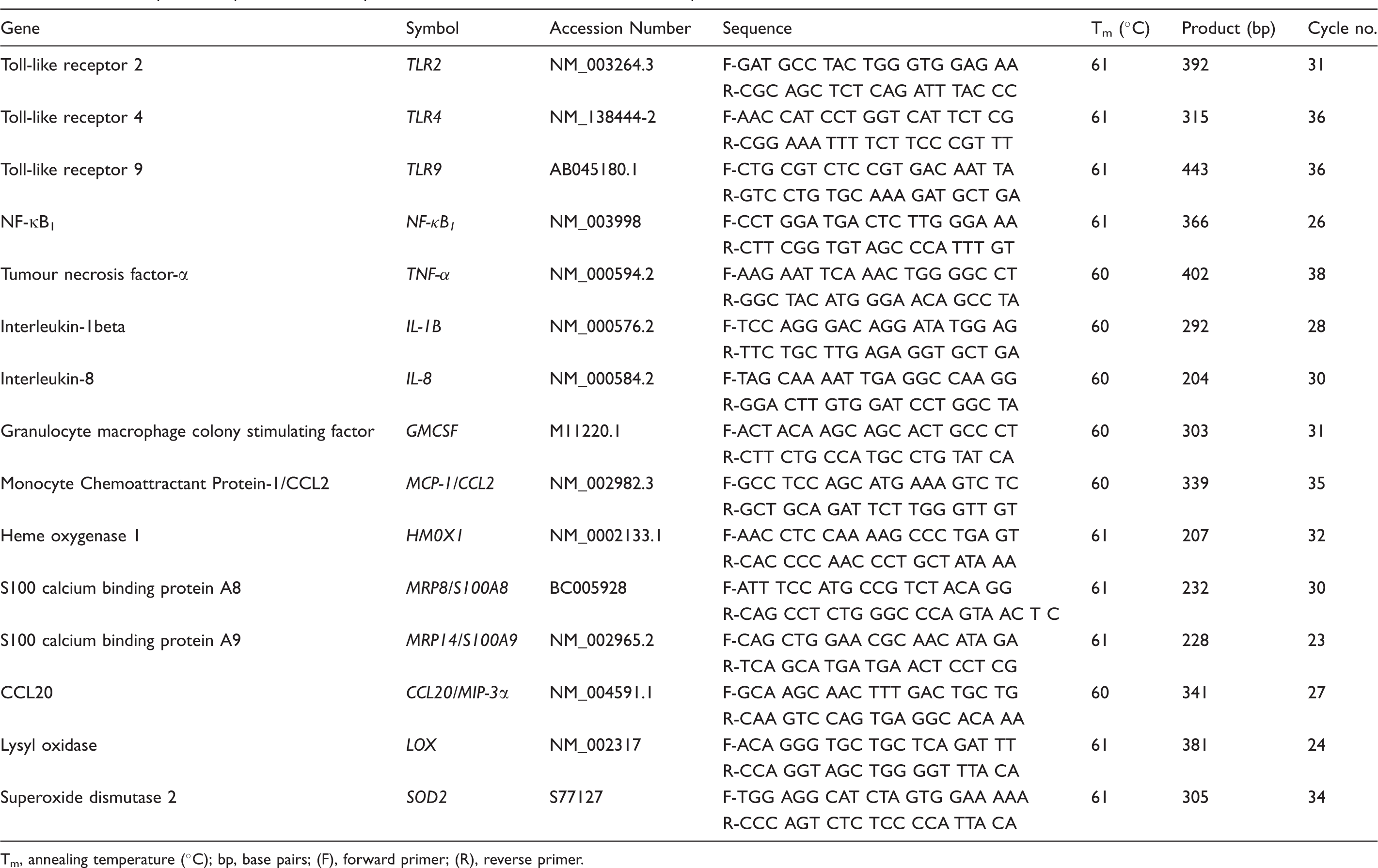
Tm, annealing temperature (°C); bp, base pairs; (F), forward primer; (R), reverse primer.
ELISA
A commercially-available sandwich ELISA kit (IDS Ltd, Boldon, UK) enabling sensitive and specific detection/quantification was used to determine levels of IL-8 present in cell culture media. Culture media was filtered using a 0.2 micron filter (Millipore, Watford, UK) to remove cell debris prior to analysis. One hundred microlitres of standard or sample was added to wells and processed following the manufacturer’s protocol. Absorbance was read at 450 nM (Spectrophotometer, Jenway, Stone, UK). Known IL-8 concentrations were used to generate a standard curve from which levels of IL-8 from the experimental samples (pg/ml) were determined. The detection limit for the assay was 25 pg/ml.
Results
Determination of ALA concentrations to be used in experiments using periodontal bacteria
As ALA culture supplementation has been reported previously to modulate NF-κB activation
18
and initial experimentation indicated an ability to disrupt cell adhesion, dose–response experiments were performed to identify suitable concentrations for use with the H400 cell line. Data demonstrated that with increasing concentrations of ALA, adherent cell numbers decreased dose-dependently and ALA concentrations greater the 0.5 m H400 cells incubated with (A) control solution (DMEM only; negative control) and (B) 0.5 m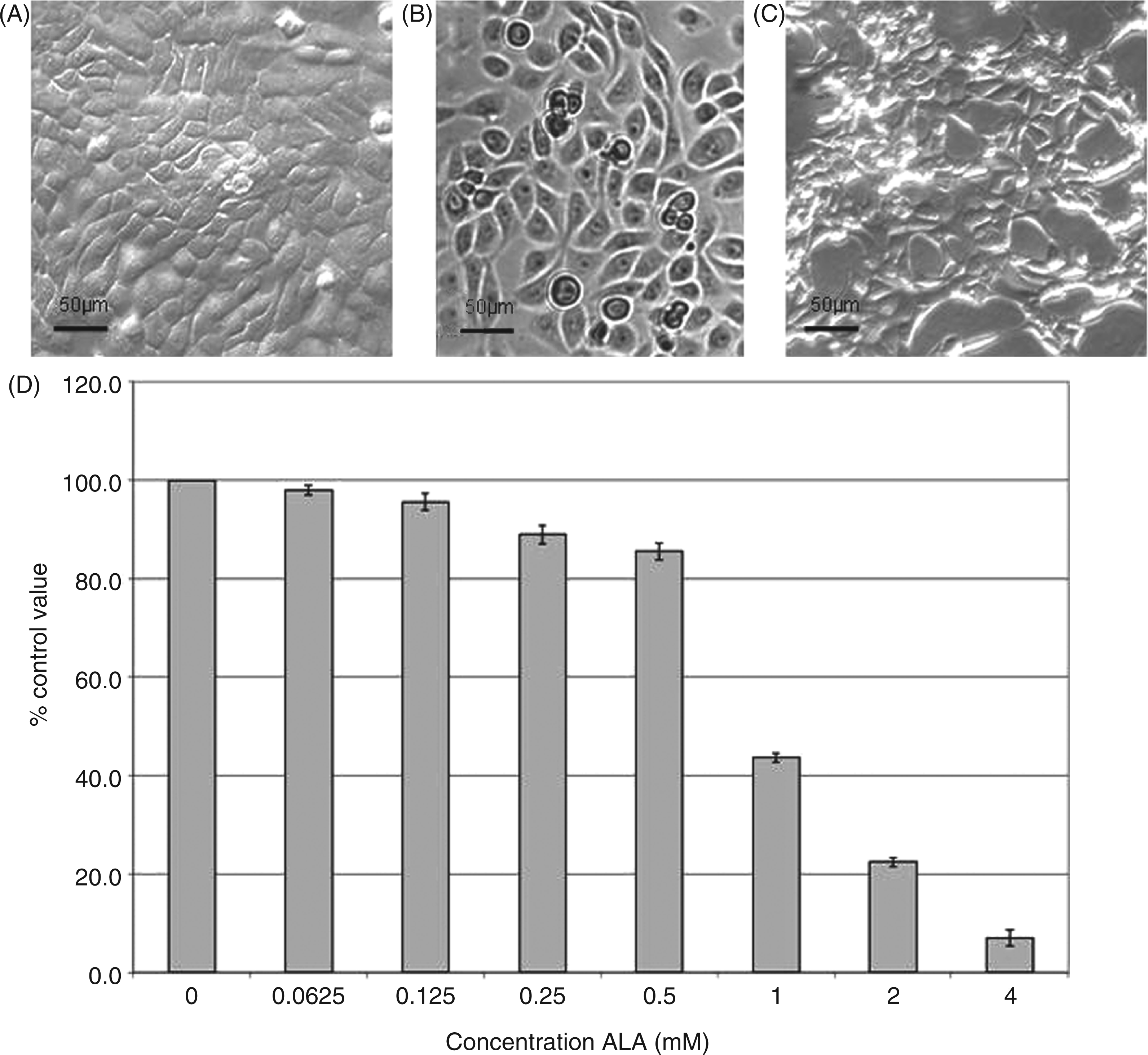
To determine the effect of ALA on NF-κB translocation under simulated pro-inflammatory conditions in H400 cells, cultures were pre-incubated with a range of ALA concentrations (0–2 m Analysis of H400 cells pre-incubated with a range of α-lipoic acid concentrations for 24 h prior to stimulation with E. coli LPS (1 h; 10 µg/ml) and immunocytochemically assayed for NF-κB translocation. (A) Images of stained cultures including (i) negative-staining control (primary Ab replaced with PBS), (ii) vehicle (DMEM) containing no ALA, (iii) 0.25 m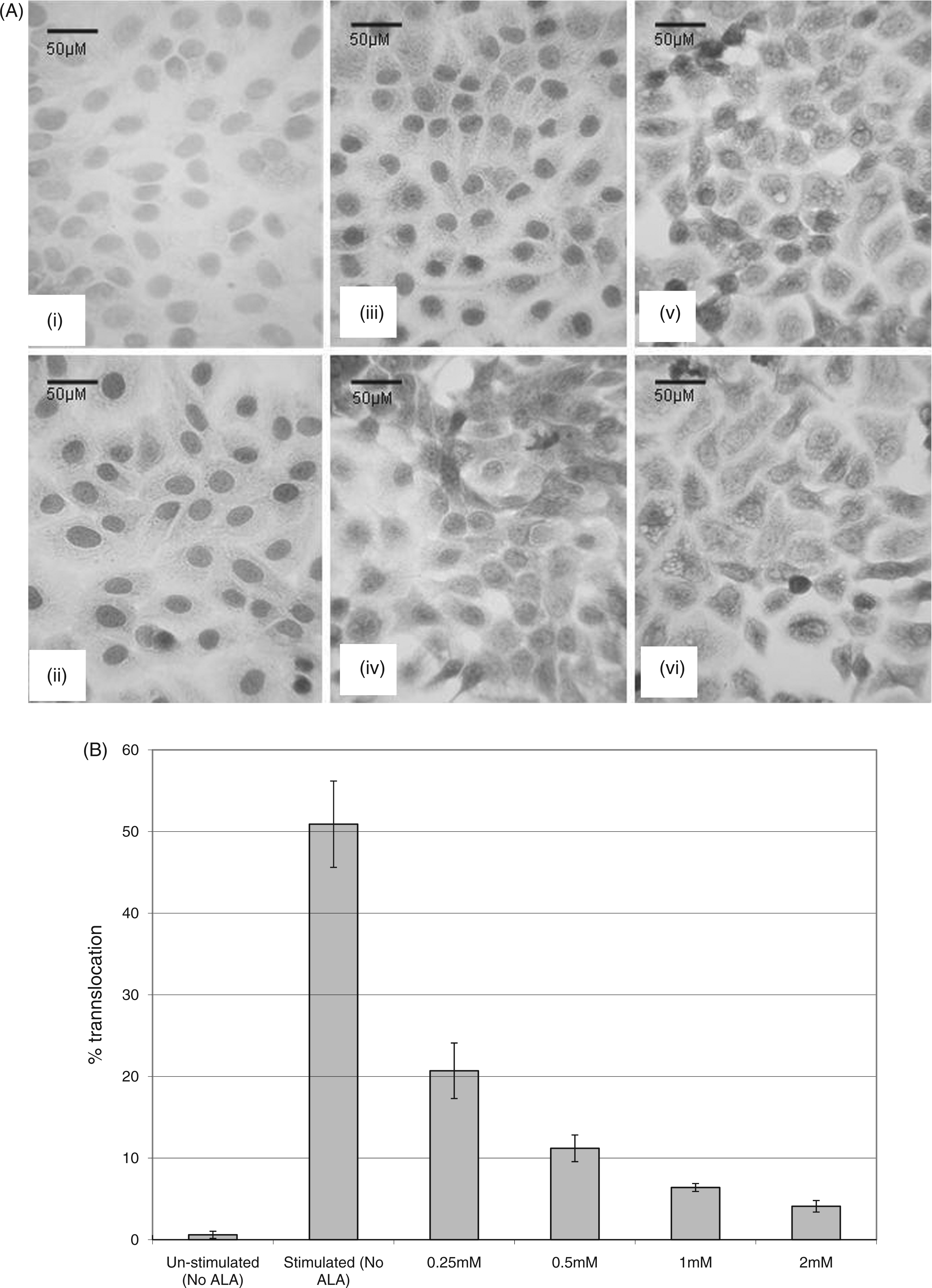
NF-κB nuclear translocation and expression regulation by periodontal bacteria and ALA
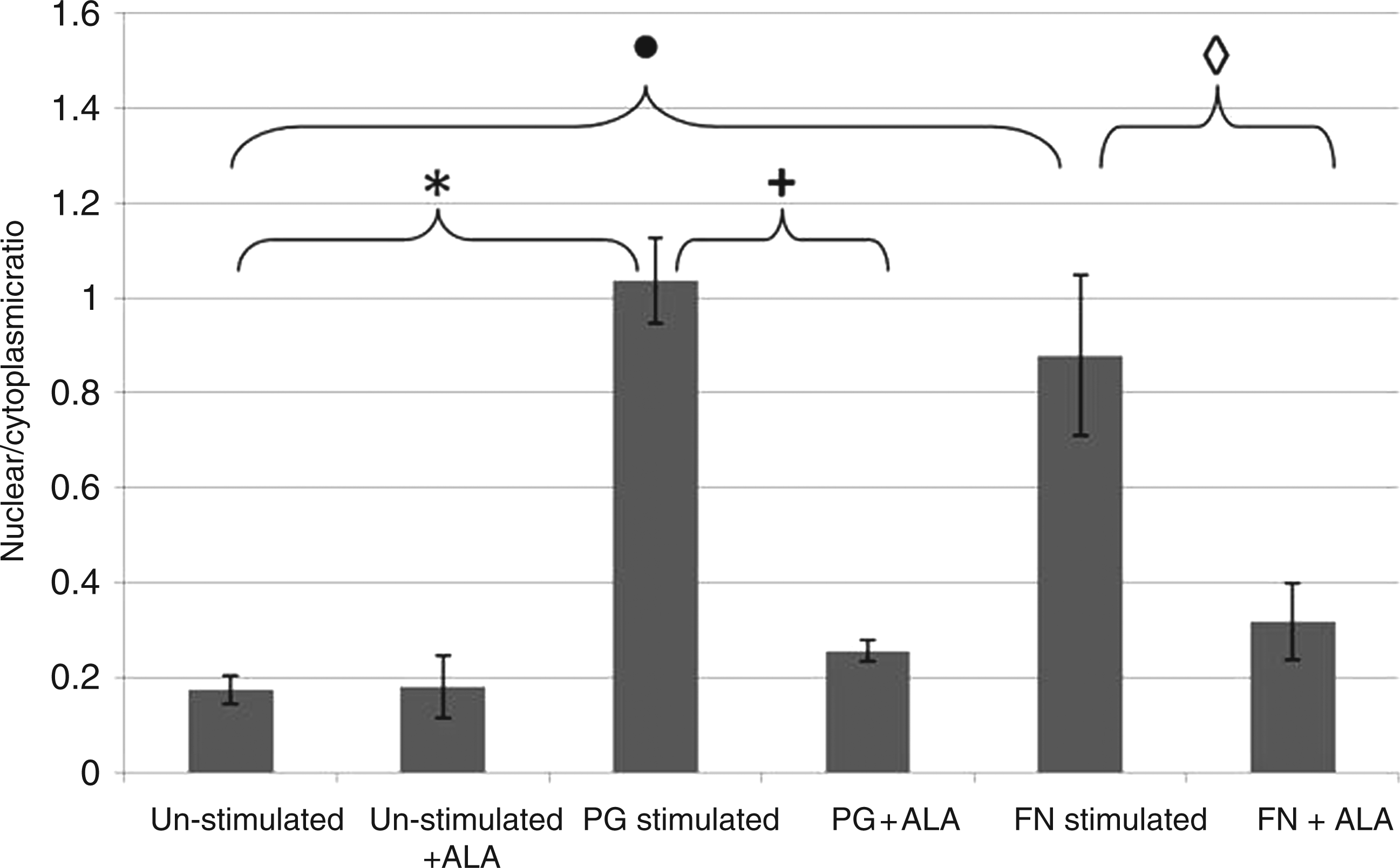
To determine the kinetics of NF-κB nuclear translocation in H400 cells, using the periodontal bacterial stimuli P. gingivalis and F. nucleatum, high content analysis was performed. Initial experiments aimed to identify a suitable time-point for assay of NF-κB activity in H400 cells exposed to periodontal bacteria, which that demonstrated NF-κB translocation appeared to peak at different time-points for P. gingivalis (∼60 min) and F. nucleatum (∼100 min) (Figure 3). Subsequent experiments utilised 60 min of exposure for both P. gingivalis (∼100% stimulation) and F. nucleatum (∼84% stimulation) to enable comparison between bacteria and consistency with work published previously.
8
These experiments demonstrated that NF-κB translocation was stimulated significantly by both P. gingivalis (P = 0.0016) and F. nucleatum (P = 0.02; Figure 4) compared with un-stimulated controls. Furthermore, cells pre-incubated with ALA (0.5 m NF-κB translocation kinetics for H400 cells exposed to P. gingivalis and F. nucleatum determined by high content analysis over a 240-min time period. Levels of translocation are shown as nucelear:cytoplasmic ratios (mean ± SD, n = 3). High content immunocytochemical analysis of NF-κB in H400 cells exposed to P. gingivalis or F. nucleatum ± α-lipoic acid (n = 3, mean ± SD). Cultures were pre-incubated with 0.5 m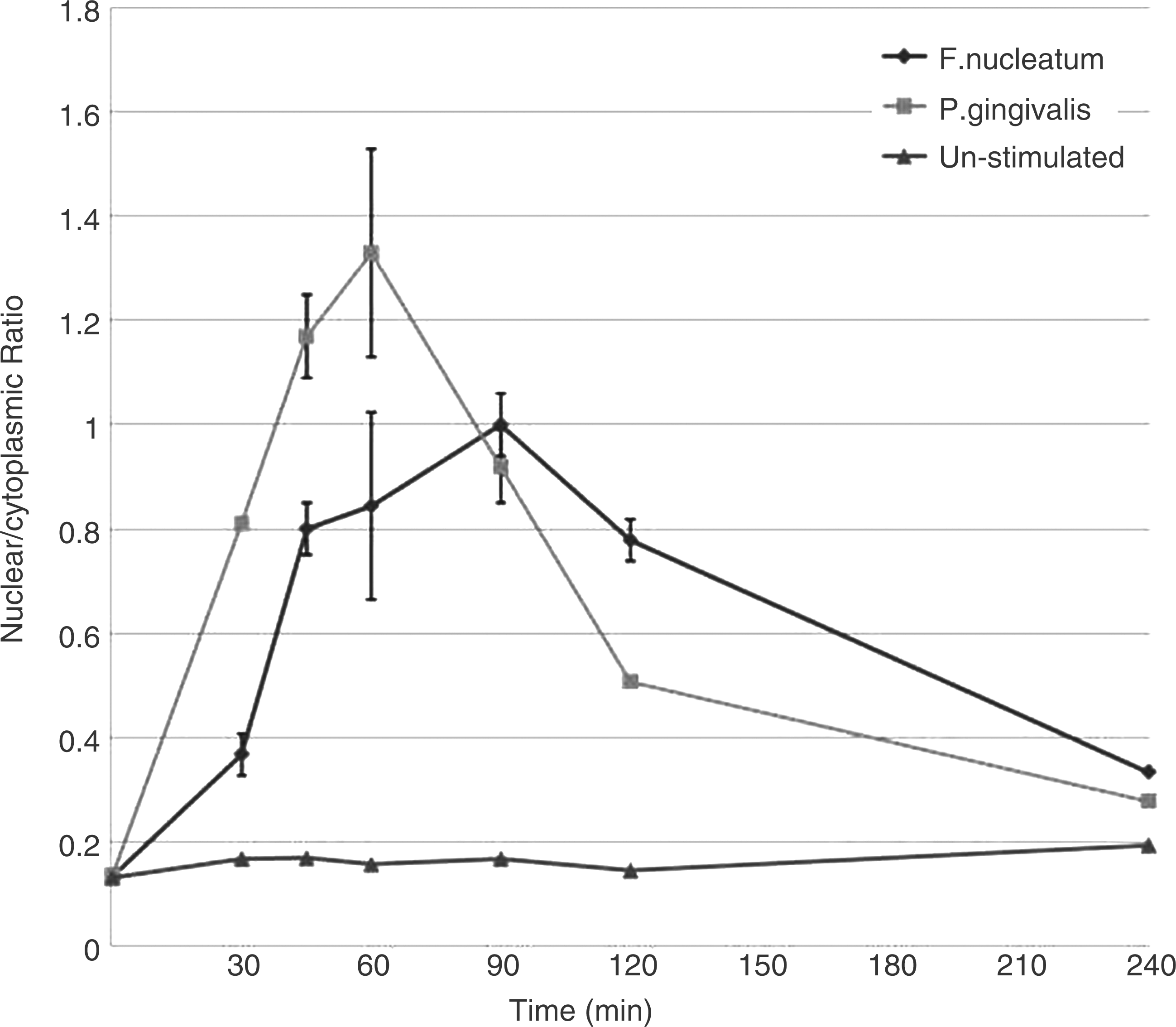
Semi-quantitative RT-PCR was performed subsequently to assess transcript level regulation (Figure 5) in genes identified previously as being associated with the inflammatory response, bacterial recognition (TLR), epithelial protection, antioxidant defence and tissue repair, as well as transcripts which we have identified previously as being regulated differentially by P. gingivalis and F. nucleatum.
8
IL-8, GM-CSF, TNF-α and IL-1β demonstrated increased levels of gene expression in H400 cells when exposed to both P. gingivalis and F. nucleatum when compared with un-stimulated controls. Pre-incubation with ALA (0.5 m Gene transcription changes as determined by sq-RT-PCR analysis in H400 cells exposed to P. gingivalis (1 h), F. nucleatum (1 h) or media (negative control) and pre-incubated with 0.5 m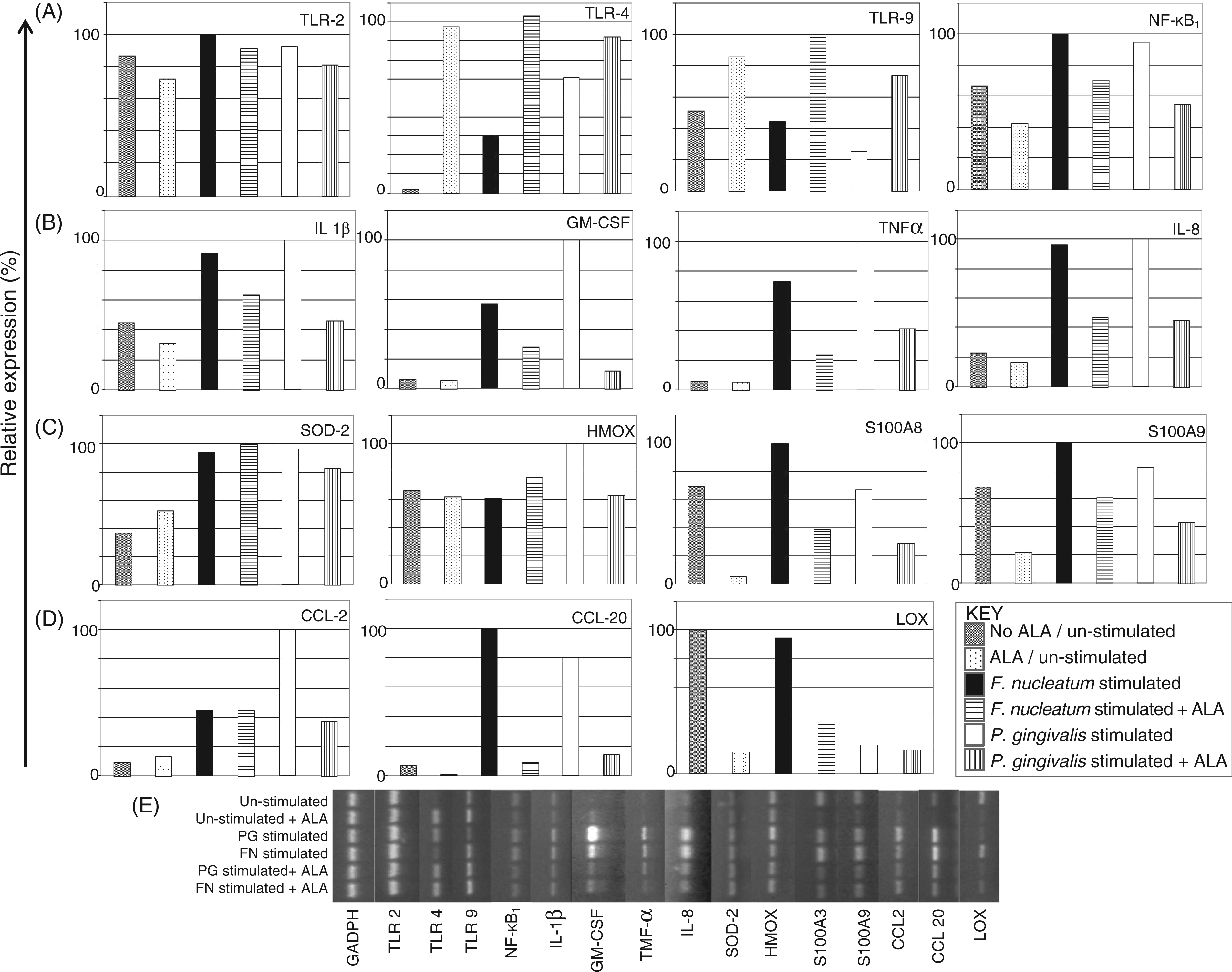
While transcriptional regulation in H400 cells by ALA was demonstrated further confirmation that these changes translated to effects on functional protein levels was investigated. Subsequently, analysis of the levels of pro-inflammatory cytokine, interleukin (IL-8), a surrogate marker of NF-κB activation demonstrated that stimulation with P. gingivalis and F. nucleatum increased secreted protein levels of IL-8, while pre-incubation of cells with ALA significantly reduced these stimulated levels (Figure 6).
Secreted IL-8 levels (pg/ml) as determined by ELISA in H400 cell cultures pre-incubated with 0.5 m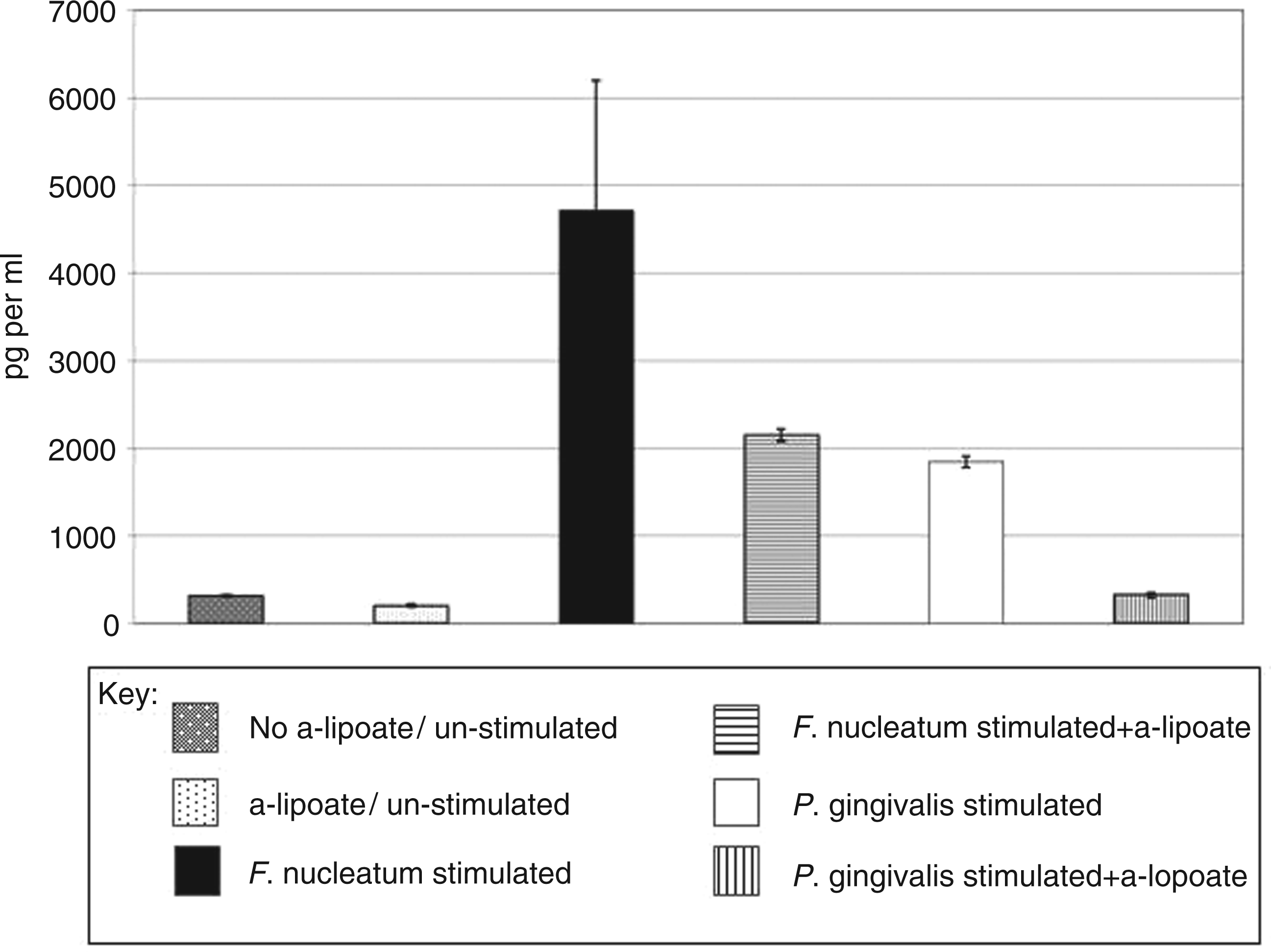
Discussion
Current evidence regarding periodontal disease pathogenesis indicates that the substantive tissue damage seen in patients is a result of a dysregulated, non-resolving inflammation in response to the plaque biofilm 22 associated with oxidative stress 23 and elevated neutrophil reactive oxygen species release 2 characteristic of the periodontitis phenotype. Emerging data indicate that, initially, the oral epithelium detects the presence of periodontal bacteria 7 and, plays an important role in orchestrating the subsequent pro-inflammatory response. The regulation of epithelial responses therefore provides a point for potential therapeutic intervention and management of this disease. The NF-κB signalling pathway is a key molecular regulator of the cellular inflammatory response and is REDOX sensitive; therefore, subsequent changes in intracellular oxidative stress levels regulate pathway activation and result in downstream sequelae, including cytokine production. 24 This pathway therefore provides an ideal target for modulation of the inflammatory response and, as the natural di-thiol ALA is reported to boost intracellular antioxidant capacity, it has been identified as an ideal candidate for modulating epithelial-driven inflammation. This study investigated the effects of ALA on key stages in periodontal pathogen stimulated epithelial cell activation, including bacterial component recognition via TLRs, NF-κB signalling, transcriptional responses and cytokine production. The expression of three key TLRs were investigated and data demonstrated that ALA pre-treatment may result in increases in keratinocyte expression of these molecules which, hypothetically, may have translated to increases in the cells’ sensitivity and ability to detect bacteria. Conversely, however, epithelial cell pre-incubation with ALA with subsequent bacterial stimulation resulted in reduced levels of NF-κB activation, suggesting that increases in TLR transcript levels are not directly translated to increases in inflammation owing to ALA’s ability to modulate activation of NF-κB. These findings are in agreement with data from previous studies which have analysed a range of other cell types and have shown that ALA pre-treatment reduces the initiation and propagation of the inflammatory response.25–28
ALA is important in regenerating intracellular antioxidants, including glutathione, which is central to maintaining cellular REDOX status.10–12 Mechanistically, it has been shown that ALA is internalised into the cell and converted to its more active antioxidant form, dihydrolipoic acid (DHLA). 29 Preliminary experiments have demonstrated that incubation of ALA with H400 cells enhances its antioxidant capacity, as determined using an enhanced chemiluminescence assay 30 (data not shown), indicating that H400 cells are able to metabolise ALA to its more active antioxidant form and suggesting that our observations are likely explained by ALA boosting intracellular antioxidant levels in H400 cells.
Interestingly, LOX gene expression appeared to be reduced following exposure to F. nucleatum and P. gingivalis. LOX is a copper-dependent enzyme important in epithelial function, development and repair, and functions by cross-linking elastin and collagen. 31 In periodontitis breakdown of these tissues is associated with disease progression. Therefore, reduction in expression of LOX post P. gingivalis and F. nucleatum stimulation may play a role in the pathogenic mechanism induced by these bacteria and conceivably lead to the periodontal tissue breakdown and frustrated tissue healing characteristic of this disease. LOX gene expression was also reduced in the presence of ALA; this may be due to the copper-chelating properties of ALA, an essential co-factor in the action of this enzyme. 32 ALA has been used in a range of chronic inflammatory diseases and reports in other systems, including skin 25 and gastric ulcers, 33 have suggested that ALA enhances healing. Further studies are therefore needed to investigate functional protein production in response to ALA supplementation.
Genes that have a role in cellular protection, neutrophil recruitment and activation (MRP-8/S100A8 and MRP-14/S100A9) were potentially reduced in bacterially-stimulated cells owing to pre-incubation with ALA. While these data indicate that ALA supplementation may reduce cell protection mechanisms following bacterial challenge, these reductions may also result in reduced neutrophil chemotaxis and activation subsequently modulating the pro-inflammatory response. In addition, recent work 34 has demonstrated that the increased expression of these molecules associates with ROS-mediated transcription pathway activation, therefore their down-regulation in response to ALA further supports its anti-inflammatory action. Pre-incubation of the H400 cells with ALA resulted in a potential decrease in transcript levels of NF-κB which may equate to reduced cellular levels of the translated protein which potentially diminishes the activation potential of this pathway. In further support of this, pre-incubation of H400 cells with ALA prior to stimulation with P. gingivalis and F. nucleatum potentially reduced protein transcription levels of the key neutrophil recruiting and activating cytokine, IL-8. As this molecule is implicated in the induction and propagation of a variety of chronic inflammatory responses the ability of ALA to modulate IL-8 production may have important ramifications for periodontal disease management. Pre-incubation of oral epithelial cells with ALA has now, for the first time, been shown to modulate periodontal bacterial induced NF-κB activation, pro-inflammatory gene expression and cytokine production. It is, however, interesting to note that ALA does not abrogate the pro-inflammatory response, but rather attenuates it. Indeed, complete blocking of this key first-line host response to bacterial challenge may be detrimental as it may render the host susceptible to overwhelming bacterial invasion. Therefore, this characteristic of ALA has potentially important clinical implications; the aim of any new anti-inflammatory therapeutic agent would be to appropriately reduce exaggerated levels of inflammation in periodontitis patients and facilitate resolution of inflammation with minimal collateral tissue damage.
Our immunocytochemical and high-throughput data suggest increased levels of NF-κB translocatory activation with P. gingivalis compared with F. nucleatum stimulation. Thus, the finding of significantly higher epithelial secretion of IL-8 after F. nucleatum stimulation compared with P. gingivalis stimulation appears contrary to the NF-κB data. The possible reasons for this disparity in data are unclear; however, it is possible that: (i) NF-κB activation does not directly correlate with IL-8 levels, for example levels of activated transcripts may be significantly induced, but this may not translate to increased protein levels; and (ii) the kinetics of activation of NF-κB likely influence IL-8 levels, which were measured 24 h after bacterial challenge. There is little published data on the kinetics of induction of NF-κB over a 24 h time period. It is clear from our data that a different temporal activity profile exists for NF-κB activity when stimulated by the two bacteria, with nuclear translocation being highest for P. gingivalis at 60 min but lower than F. nucleatum at 90 min and 120 min after. Such differences in temporal profile may explain why higher IL-8 levels are detected following F. nucleatum stimulation. In addition, while our data and that of others indicate that nuclear translocation of NF-κB peaks at about 1 h after stimulation and is recycled by 4 h, it is not clear as to when nuclear translocation can be re-stimulated. It may, therefore, be feasible that different NF-κB activation profiles exist for F. nucleatum and P. gingivalis stimulation within the 24-h time period, which may also explain the lack of correlation between 60-min NF-κB nuclear translocation and IL-8 levels determined at 24 h.
ALA has been used widely for managing a range of chronic inflammatory diseases, including those which associate with periodontitis, such as diabetes; 35 however, its potential in managing periodontal disease has not been proposed previously. This study investigated the effects of this natural di-thiol on periodontal pathogen-stimulated oral epithelium in terms of NF-κB activation, gene expression changes and cytokine production, utilising a previously well-characterised epithelial cell culture system.8,16 Data indicated the ability of periodontal bacteria (F. nucleatum and P. gingivalis) to stimulate a pro-inflammatory phenotype which was subsequently modified by pre-incubation of H400 cells with ALA. These data suggest that ALA supplementation, either via local or systemic delivery, may provide utility in managing periodontitis patients.
Footnotes
Funding
This research was part-funded by the Oral and Dental Research Trust.
Acknowledgements
The authors would like to thank Imagen Biotech, Manchester, UK, for the high content analysis and Professor Stephen Prime (University of Bristol, UK) for his kind donation of the H400 cell line.
