Abstract
Despite NK cells being originally identified because of their ability to kill tumor cells in vitro, only limited information is available on NK cells infiltration of malignant tumors, especially in humans. NK cells infiltrating human colorectal carcinomas (CRCs) were analyzed to identify their potential protective role in an antitumor immune response. The expression and function of relevant molecules were analyzed from different sources, comparing tumor-associated NK cells (TANKs) with autologous peripheral blood NK cells (PB-NKs) from CRC patients—the latter in comparison with PB-NKs from normal donors. TANKs displayed a profound alteration of their phenotype with a drastic reduction of NK cell receptor expression. Co-culture experiments showed that CRC cells produce modulation in NK phenotype and functionality. Moreover, PB-NKs from CRC patients also exhibited an altered phenotype and profound defects in the ability to activate degranulation and IFN-γ production. For the first time, TANK and PB-NK cells from CRC patients have been characterized. It is shown that they are not capable of producing relevant cytokines and degranulate. Taken together, our results suggest that NK cells from CRC patients present alterations of phenotype and function therefore supporting the progression of cancer.
Introduction
NK cells are part of the innate immune system and have diverse biological functions, including the ability to recognize and kill a variety of tumor cells without prior sensitization.1,2 Human NK cells can be divided into two main functional subsets based on the intensity of CD56 expression. 3 Although there is some degree of functional overlapping, CD56dim NK cells have a potent cytotoxic function, whereas CD56bright NK cells are thought to have immunoregulatory properties.3–6 Recent data support the notion that CD56bright and CD56dim NK cells represent different stages of maturation, with CD56dim NK cells being the more differentiated type.7,8 NK cells are regulated by a balance of activating and inhibitory signals from cell surface receptors.9–11 The inhibitory signals are mediated mainly by human leukocyte antigen class I (HLA-I)-binding receptors, including killer cell Ig-like receptors (KIRs), CD94/NKG2A and leukocyte Ig-like receptor B1 (LILR-B1). 12 Activating signals are determined by a wide array of receptors, including NKG2D, DNAX accessory molecule-1 (DNAM-1), natural cytotoxicity receptors (NCRs) (NKp30, NKp44, NKp46), CD94/NKG2C and KIRs with activating intracellular domains.9,11 Additionally, the intermediate-affinity FcγRIIIA receptor (CD16) mediates Ab-dependent cellular cytotoxicity (ADCC). 13 Engagement of specific combinations of activating receptors on NK cells dictates qualitatively-distinct responses and can lead to synergistic effects for activation function.14,15 When appropriate inhibitory receptor/HLA-I ligand interactions occur, the receptors deliver negative signals that inhibit NK-mediated killing. However, in the absence of such interactions, as it may occur in tumor or virus-infected target cells, activating receptors are allowed to deliver triggering signals, resulting in target cell killing. Several of the activating receptors mentioned above are important in tumor cell recognition. However, different studies have reported reduced expression of these molecules and consequent weakened NK cell activity in human cancers.16–25
Colorectal carcinoma (CRC) is one of the most common malignancies and a major cause of cancer death worldwide. 26 The development and course of CRC are influenced by factors such as microsatellite instability and inflammatory mediators, for example. 27 Inflammation can promote tumor progression and metastasis formation. However, the innate and adaptive arms of the immune system can protect the host against tumor development through mechanisms of immunosurveillance. 28 The role of NK cells in solid tumors and, in CRC in particular, is not well understood, particularly with regard to whether NK cells contribute to immunosurveillance. 29 Halama and coworkers 30 recently performed a detailed quantification of infiltrating NK and T cells, and quantified the corresponding chemokine/cytokine composition in primary CRC tumors, adjacent normal mucosa and liver metastases. They determined that despite high local chemokine levels, NK cells were generally scarce within CRC tissues, independently of HLA-I expression. On the contrary, adjacent normal mucosa contained significantly higher numbers of NK cells. Corresponding T-cell numbers varied substantially and were positively correlated with higher chemokine levels.
Cetuximab (Erbitux®, Merck, Whitehouse Station, NJ, USA), an anti-epidermal growth factor receptor (EGFR) mAb, has been shown to increase the median survival of CRC patients. 31 Several mechanisms may contribute to the antitumor activity of Cetuximab, including the direct inhibition of EGFR tyrosine kinase activity 32 and mediation of ADCC, performed mainly by NK cells. EGFR expression is not the only determinant of Cetuximab response, as recent findings reported that patients with K-RAS point mutations are resistant to the treatment.33,34 In addition, only 40% of patients with wild-type K-RAS respond to Cetuximab therapy; this observation implies that, for the remaining 60% of patients, other mechanisms are involved in resistance to Cetuximab and require further investigation.
Here, we have examined the activating and inhibiting receptor repertoire and function of peripheral blood NK cells (PB-NKs) and tumor-associated NK cells (TANKs) in CRC patients to identify their potential protective role in antitumor immune response and MAb therapy.
Materials and methods
Patients and healthy donors
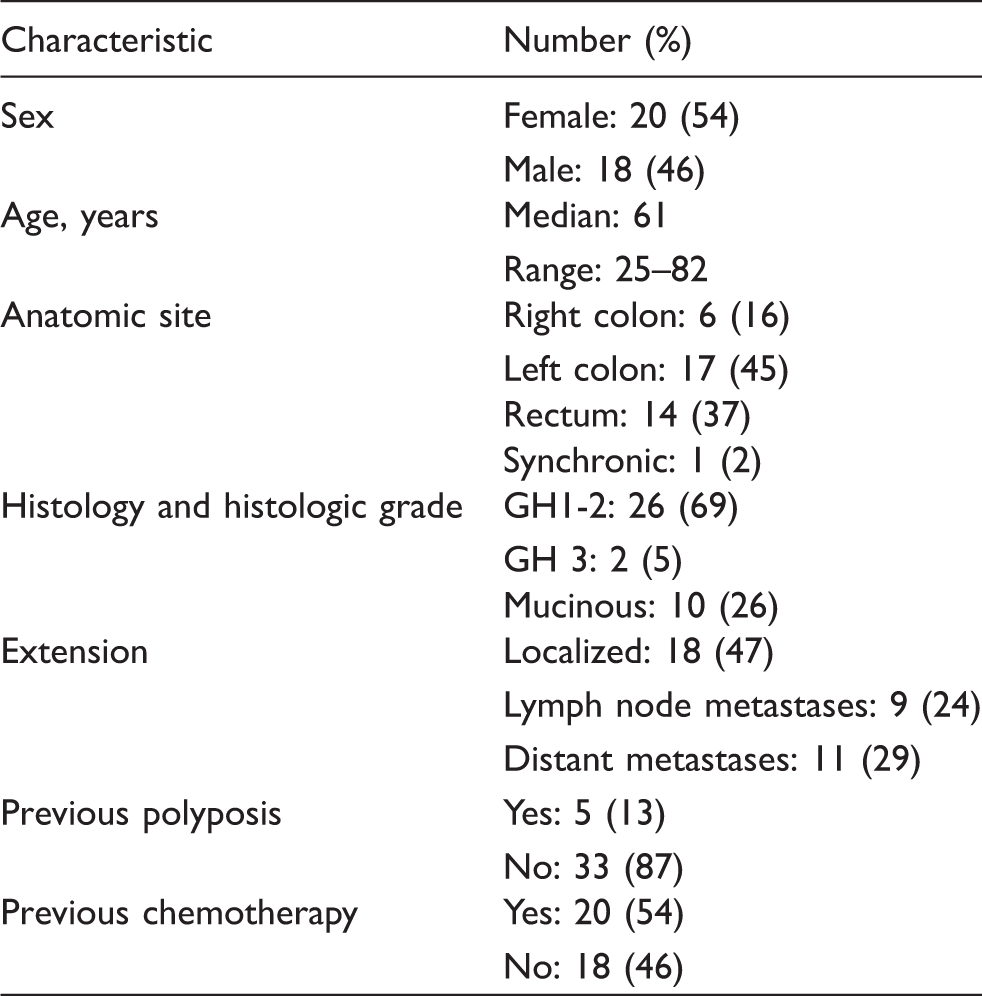
Clinical and histologic characteristics of colorectal carcinoma patients.
Preparation of single-cell suspensions
CRC specimens were incised immediately after removal and split into sagittal parts, half of which were paraffin-embedded to perform confirmatory pathological analysis and the other half processed for single cell isolation. Tumor and non-tumoral tissue (obtained at >5 cm from the tumor) were processed for single cell isolation by mincing the tissues with scissors. Mechanical dissociation was performed in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) with 10% FCS (Natocor, Córdoba, Argentina). Cell suspensions were filtered through a 70 -µm cell strainer (BD Labware, Mountain View, CA, USA), washed with 1X PBS (Invitrogen) and tissue-associated mononuclear cells were isolated by Ficoll-(Sigma, St Louis, MO, USA)-Hypaque (BerliMed S.A., Madrid, Spain) density gradient centrifugation. Similarly, PBMCs were isolated using Ficoll-Hypaque density gradient. Cells were directly analyzed by flow cytometry.
mAbs and flow cytometry
Analysis of cell surface markers in single-cell suspensions was performed by multicolor flow cytometry. NK cells were defined as CD3−CD56+ cells within lymphocyte gate, delimited by CD45+ cells. We used the following mAbs in a direct immunofluorescence assay: PerCP (Peridinin-chlorophyll-protein complex)-conjugated anti-CD45, FITC (Fluorescein isothiocyanate)-conjugated anti-CD3, APC (Allophycocyanin)-conjugated anti-CD56, PE (R-Phycoerythrin)-conjugated anti-NKG2D, anti-NKp30, anti-NKp44, anti-NKp46, anti-CD16, anti DNAM-1, anti-CD94, anti-CD161, anti-CD158a/h, anti-CD158b, anti-IFN-γ, anti-CD107a (BD PharMingen, San Diego, CA, USA) and PE-conjugated anti-NKG2A (R&D Systems, Minneapolis, MN, USA). Negative controls included directly labeled isotype-matched irrelevant mAbs. Flow cytometry data were analyzed using Cellquest Pro software (BD Biosciences, Franklin Lakes, NJ, USA).
Cell lines
The K562 (HLA-I-/-) cell line was derived from a patient with chronic myeloid leukemia and was obtained from the American Type Culture Collection (ATCC-CCL-243).The K562 cells were cultured in RPMI supplemented with 10% FCS. The human colorectal adenocarcinoma cell lines HT-29 and DLD-1 (Dukes´ C) were also obtained from the ATCC. Both cell lines were grown with DMEM (Invitrogen), supplemented with 10% heat-inactivated FCS, 2 mM
CD107a mobilization assay
PBMC from CRC patients or normal donors, pre-activated or not with 1000 U/ml IL-2 (Preprotech, Distrito Federal, Mexico), were used as effectors in a CD107a mobilization assay. K562 cells were used as target. To detect spontaneous degranulation, a control sample without target cells was included. An effector/target (E/T) ratio of 10:1 (106 effector cells: 105 target cells in a volume of 1 ml) was used. Anti-CD107a (PE-conjugated) was added in each well (3 μl/well) before incubation for 6 h. Cells were then washed twice in PBS and stained with MAbs (FITC-conjugated anti-CD3 and APC-conjugated anti-CD56) for flow cytometry analysis. Surface expression of CD107a was assessed in NK cell populations.
IFN-γ production
PBMC from CRC patients or normal donors, pre-activated or not with IL-2 (1000 U/ml) were used as effectors in an intracellular IFN-γ production assay. K562 cells were used as a stimulus. To detect spontaneous IFN-γ production, a control sample without target cells was included. An E/T ratio of 10:1 (106 effector cells: 105 target cells in a volume of 1 ml) was used. To inhibit protein transport, GolgiStop (BD Biosciences, San José, CA) was added to each well (1μl) 1 h after the start of the co-culture. Effectors and targets were then co-incubated at 37°C for 5 h. Cells were then washed (Perm/Wash, BD Biosciences), fixed and permeabilized (Cytofix/Cytoperm, BD Biosciences), and, finally, stained with PE-conjugated anti-IFN-γ. NK cell populations were stained with mAbs (FITC-conjugated anti-CD3 and APC-conjugated anti-CD56) for flow cytometry analysis, and intracellular expression of IFN-γ was assessed.
Cytotoxicity and ADCC assays
PB-NKs from CRC patients or normal donors were assayed in a calcein release assay for their ability to kill target tumor cells in the presence of Cetuximab. Briefly, EGFR-expressing HT-29 cells were used as target and labeled with calcein-acetyoxymethyl (Calcein-AM; Molecular Probes, Invitrogen). The cytotoxicity assay was performed using 10:1 and 20:1 E:T ratios (5 × 104 or 1 × 105 PBMC: 5 × 103 HT29 cells), in triplicate, with 1 µg/ml Cetuximab or control MAb (Rituximab). Three replicate wells for spontaneous (only target cells in RPMI medium with 10% FCS) and maximum release (only target cells in medium plus 1% Triton X-100) were also measured. After incubation at 37°C in 5% CO2 for 4 h, supernatants were analyzed by fluorimetry at 485/535 nm using a DTX880 spectrophotometer (Beckman Coulter, Brea, CA, USA) to measure cell death (Calcein release). The percentage of specific lysis was calculated as: (experimental fluorescence – spontaneous fluorescence)/(maximum fluorescence – spontaneous fluorescence) × 100.
Co-culture experiments
PBMC (1 × 107) from normal donors activated with 100 U/ml IL-2 were cultured at a 5:1 E:T ratio in the presence of CRC cells HT-29 or DLD-1 (2 × −106), in a final concentration of 1 × 106 PBMC/ml. Experiments were also performed adding transwell membrane (BD Falcon™, Franklin Lakes, NJ, USA), to test cell contact influence or supplementing with 5 ng/ml hu-recombinant TGF-β (R&D Systems). After 5 d of co-culture, the phenotype of NK cells was analyzed by flow cytometry. NK cells modulated by HT-29 or DLD-1 cells were tested for C107a expression and IFN-γ production after incubation with K562 cells, as explained above.
Statistical analysis
Statistical analysis was performed with GraphPad Prism5 software using the Mann-Whitney t tests for unpaired groups and one-way ANOVA with Dunn’s multiple comparison for multiple comparison analysis.
Results
TANKs from CRC patients displayed a lower proportion of activating surface receptors
TANKs were isolated from surgically resected cancer specimens (n = 12) and compared with NK cells from normal mucosa (MANKs) (n = 9) and with PB-NK from the same, or other, CRC patients (n = 38) (Table 1). After density gradient separation, only mononuclear cells were collected and analyzed for NK cell content. While there were no differences in the CD3+ T-cell percentages between CRC tissue and autologous PBMCs (Figure 1A), the percentage of NKs, identified as CD3−CD56+ cells isolated from CRC tumors, was consistently lower (3.5% ± 4.2) than the peripheral blood counterpart (17.4% ± 10.3; P < 0.001) (Figure 1B). CD3−CD56+ dot plots obtained from analysis of TANK and MANK samples displayed a unique homogeneous population in which it was not possible to distinguish dim and bright subsets. Moreover, no differences were observed in the CD56+ mean fluorescence intensity (MFI) comparing TANKs and MANKs (P > 0.05) (Figure 1C).
(A) Proportion of CD3+ cells in total lymphocytes population of CRC patients. PB, peripheral blood (gray dots; n = 24) and tumor-associated lymphocytes (black dots; n = 9); ns, non significant differences. (B) Proportion of CD3−CD56+ (NK) cells in total lymphocytes population (PB-NK: gray dots; n = 8), normal mucosa (MANKs: white dots; n = 9), and tumor (TANKs: black dots; n = 11). ***: P < 0.001. (C) CD56 mean fluorescence intensity (MFI) on NK subset from MANKs (white bar; n = 7) and TANKs (black bar; n = 10).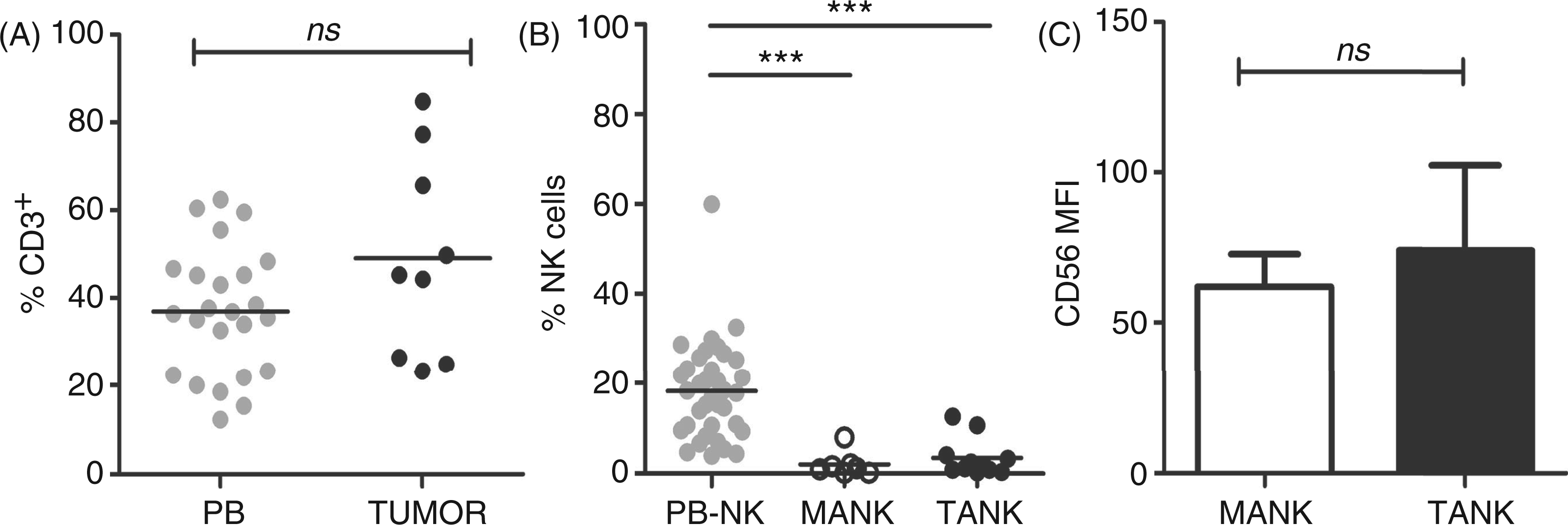
Furthermore, TANKs displayed a peculiar surface molecular pattern. We determined the frequency of NK cells expressing activating and inhibitory receptors. Among the activating ones we studied NCRs NKp30, NKp44 and Nkp46; the C-type lectin receptor NKG2D; Fc Receptor CD16; CD161; and DNAM-1. We also studied HLA-I-specific inhibitory receptors, including the KIRs CD158a/h (2DL1/DS1) and CD158b (KIR2DL2/DL3), and the lectin-like CD94-NKG2A heterodimer. A representative phenotypic pattern of NK cells isolated from the three different tissues is depicted in Figure 2A. Most of the CD3−CD56+ NK tumor-infiltrating cells displayed significantly lower levels of surface CD16, NKG2D, NKp30, NKp46, CD161 and DNAM-1, in agreement with similar observations in lung23,24 and breast cancer.
25
It is noteworthy that NKp44 is usually induced in PB-NK cells upon cytokine activation and its expression ex vivo has been detected previously in NK cells derived from inflamed tissues.
35
Accordingly, TANK and MANK cells displayed increased levels of NKp44. Moreover, evaluation of KIR CD158b and CD158a/h showed significantly lower values in TANKs compared with PB-NKs (for both receptors P < 0.01), as was previously described in tumor-associated NKs from lung cancer patients (Figure 2B).
22
NKG2A did not present differences between TANKs and PB-NKs (data not shown). We also performed paired tests between the data of NK receptor expression obtained from tumor samples and autologous peripheral blood. We observed that for most activating receptors analyzed in paired tests, expression levels were lower in TANKs than in autologous PB-NKs (Supplementary Figure 1). Interestingly, MANKs showed non-significant intermediate values between PB-NKs and TANKs for NKG2D, NKp30, NKp44, NKp46, DNAM-1 and CD158a/h. MANKs were equivalent to PB-NKs for the activating receptor CD161. The receptors CD16, CD94 and CD158b were also significantly diminished in MANKs as in TANKs compared with PB-NKs (Figure 2B).
(A) Representative contour plots of the receptors NKG2D, NKp30, DNAM-1 and CD161 from autologous mononuclear cells gating on the CD3−CD56+ population isolated from peripheral blood (PB-NK), adjacent mucosa (MANK) and tumor (TANK). Samples were comparatively analyzed by three-color flow cytometry using mAbs specific for indicated markers. (B) Analysis of NK cell receptors expression in peripheral blood of patients (PB-NK: gray dots), non-tumoral distant mucosa (MANK: white dots) and intra-tumoral tissue (TANK: black dots). Mean is represented by horizontal bar; *P < 0.05; **P < 0.01; ***P < 0.001.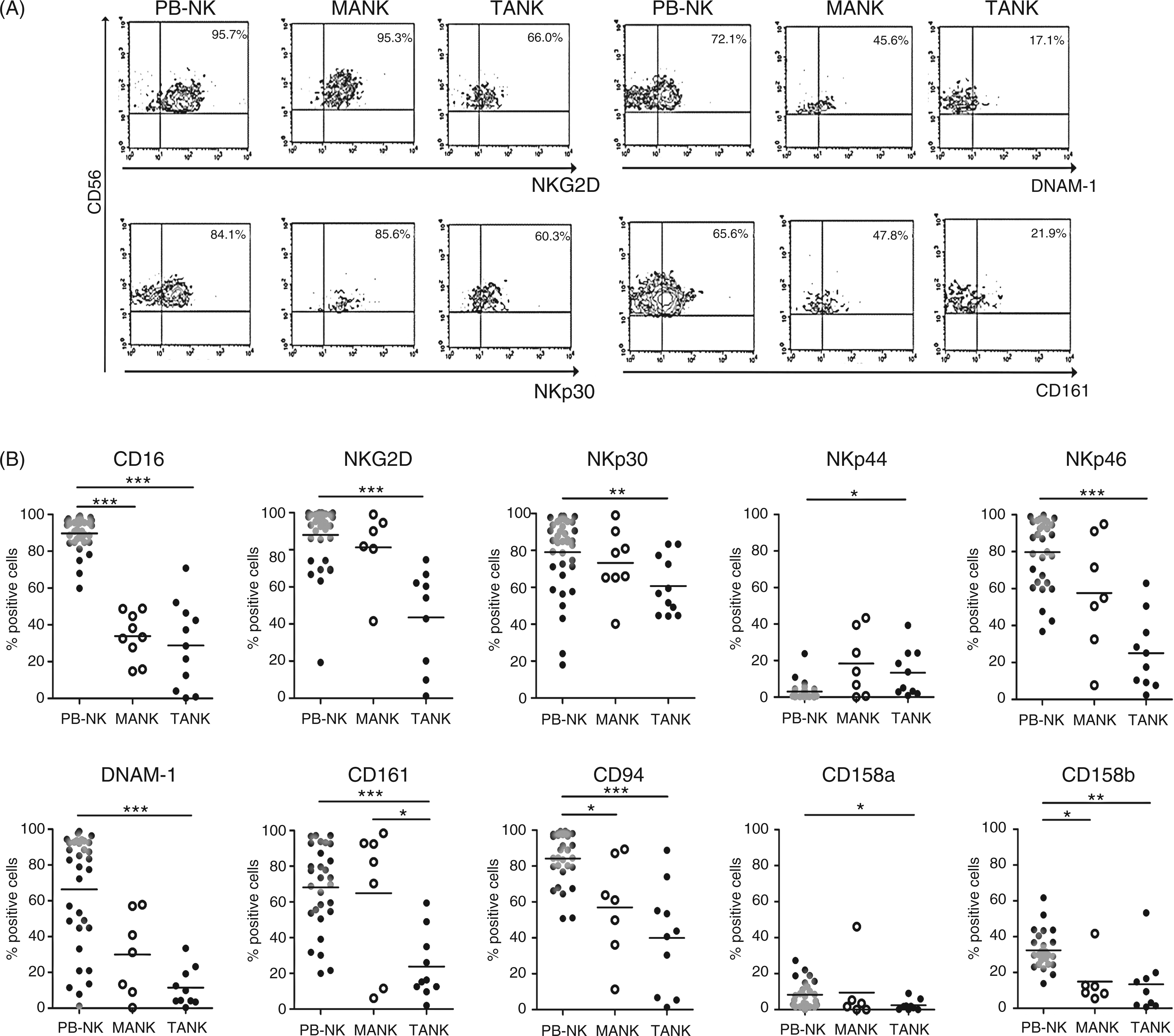
Altered phenotype was induced by colorectal tumor cells
The lower proportion of activating receptors in TANKs may indicate that NK cells present in the CRC microenvironment display an altered phenotype. We investigated whether tumor cells allow the selection of NK cells with an altered phenotype or induce the phenotypic alterations. To test this hypothesis, we co-cultured PBMC from normal donors (n = 3) with HT-29 or DLD-1 CRC cell lines in the presence or absence of a transwell membrane (n = 2), and, in one case, in the presence of hu-recombinant TGF-β (Supplementary Table 1). The phenotype of NK cells was determined after 5 d of co-culture. The results of these experiments were variable between normal donor samples and were affected by tumor cell lines used. The percentages of DNAM-1 - and CD161-positive cells was diminished in most cases after tumor cell co-culture. The expression of CD16 was not affected by HT-29 cells, but it was negatively modulated in presence of DLD-1 cells in 2/3 experiments. For most receptors, HT29 and DLD-1 caused different effects in their modulation (Figure 3A). For NKG2D and NKp30, we could only observe lower values of MFI, maintaining similar percentage values relatives to controls (Supplementary Figure 2). However, NKp46, CD94, CD158a/h and CD158b expression was not affected (data not shown). Transwell experiments revealed that DNAM1 down-regulation appeared to be dependent on physical contact between NK and tumor cells. On the contrary, lower expression of CD161 was not dependent on NK and tumor cell contact. In most cases, TGFβ described as a potent immunosupressive molecule produced by many cancer cells,36,37 was not the only cause of such decreased expression. In this sense, the reduction in receptor expression was not as relevant as that produced by tumor cell contact (Figure 3A).
(A) Percentage of NK cell receptor CD16, DNAM-1 and CD161 expression after 5 d co-culture with CRC cell lines, analyzed on IL-2-stimulated PBMC obtained from 3 normal donors (represented as a circle, square and triangle). Co-cultures were performed in the presence of HT29 cells, DLD-1 cells, DLD-1 cells with transwell membranes (+CM-DLD) and DLD-1 cells with 5 ng/ml hu-recombinant-TGF-β. (B) Representative density plots of INF-γ production (left panel) and degranulation (right panel) of NK cells targeting the K562 cell line in the presence or absence of IL-2. NK cells had been co-cultured previously for 5 h with CRC cells. The percentage of responding cells for each treatment is indicated and they represent one from three independent experiments.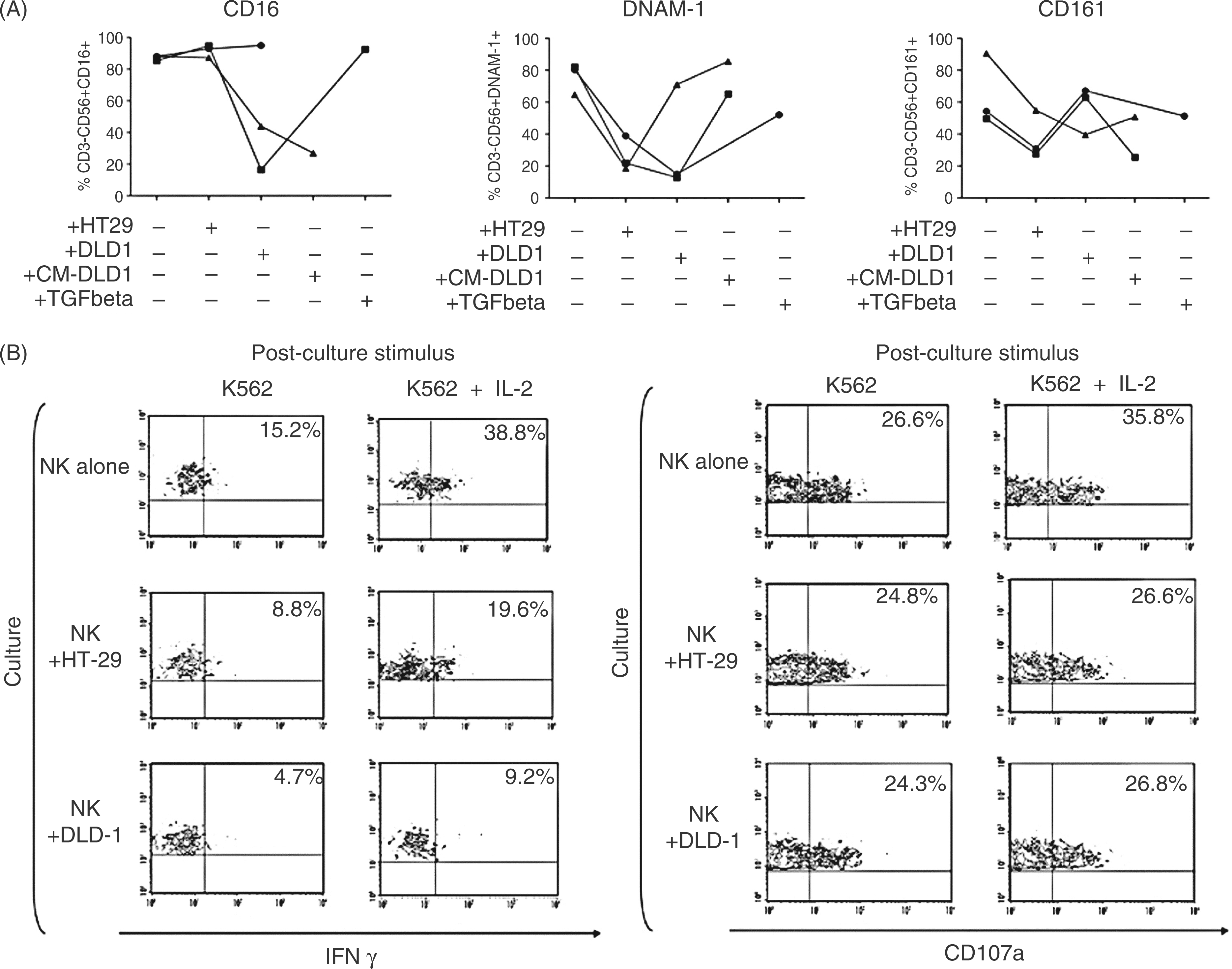
We also wanted to investigate if the altered phenotype of TANKs, characterized by the reduced expression of several activating receptors, was related to poor NK cell functionality. As functional studies of infiltrating NK cells are challenging because of the scarce cell numbers available, we used the NK cells derived from the co-cultures with HT-29 and DLD-1 cell lines as TANK approximations to evaluate NK cell function in the tumor microenviroment.
Degranulation and IFN-γ production were assessed by flow cytometry on the basis of cell-surface mobilization of CD107a and intracellular staining of IFN-γ respectively. Effector NK cells were modulated with HT-29 or DLD-1 cells, and the K562 cell line was used as a target. We observed a strong decrease in the ability of modulated NK cells to produce IFN-γ. These cells also presented lower degranulation activity in the presence of IL-2 (Figure 3B).
Phenotypic deregulation in PB-NKs from CRC patients
PB-NKs from CRC patients were evaluated at the time of surgery (n = 36). The results were compared with those obtained for PB-NKs from normal donors (n = 36). The percentage of circulating NK cells was similar in the CRC patients (18.2% ± 10.8, range 3.9–59.9%) compared with NK cells of normal donors (16.6% ± 11.9, range 4.2–54.9%) (Figure 4A). As it refers to the proportion of the CD56bright or CD56dim NK subsets, no differences were observed (P > 0.05) (Figure 4B).
(A) Proportion of CD3−CD56+ cells in total lymphocyte population, in peripheral blood from normal donors (n = 36, gray dots) and CRC patients (n = 36, black dots). ns, non-significant differences; (B) proportion of CD56dim in total NK population, in peripheral blood from normal donors (gray dots) and CRC patients (black dots). (C) Phenotype of peripheral blood NK cells from normal donors (gray dots) compared with CRC patients (black dots). NKG2D, NKp30, DNAM-1, CD94 and CD158a/h presented significant differences. **P < 0.01, ***P < 0.001. (D) Degranulation assay against K562 cells was determined by CD107a labeling and FACS analysis. Unstimulated or IL-2-stimulated PBMC from normal donors (n = 6; gray bars) or CRC patients (n = 9; black bars) were used as effector against K562 cells at an E:T ratio of 10:1. Bars give the mean values of CD107+ proportion on CD3−CD56+ NK cells. **P < 0.01. (E) IFN-γ production assay against K562 cells was determined by intracellular IFN-γ labeling and FACS analysis. Unstimulated or IL-2-stimulated PBMCs from normal donors (n = 10; gray bars) or CRC patients (n = 10; black bars) were used as effector cells against K562 cells at an E:T ratio of 10:1. Bars give the mean values of IFN-γ producing CD3-CD56+ NK cells. **P < 0.01. (F) ADCC assay against EGFR expressing HT-29 cells determined by fluorometric assay. PBMCs from normal donors (n = 5; gray bars) and CRC patients (n = 5; black bars) were used as effector cells at an E:T ratio of 10:1 and 20:1. Inset: CD16 (Fcg RIII) is expressed at the same level in normal donors and patients NK cells.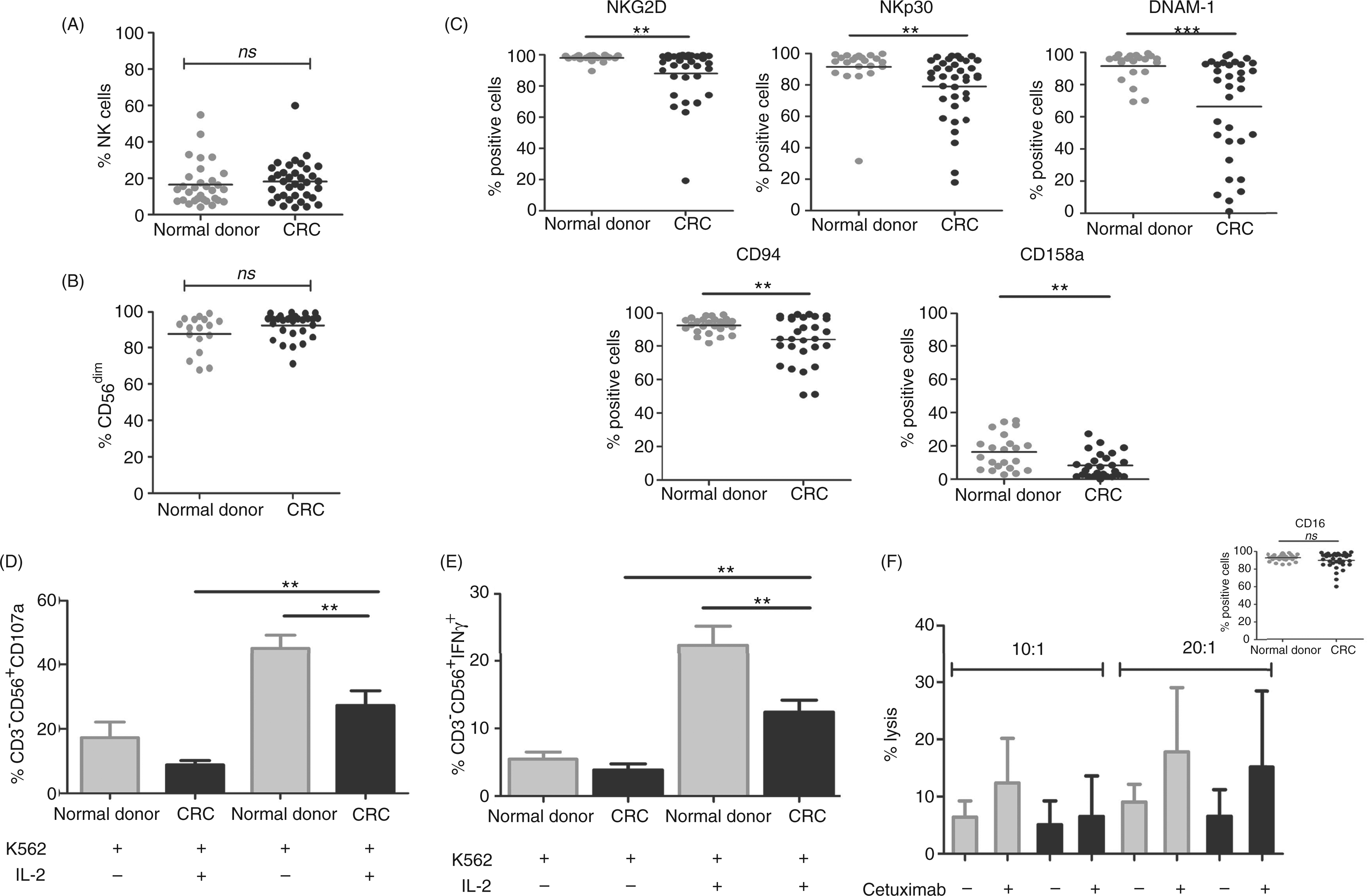
However, when we compared the receptors repertoire, we found that NKG2D, NKp30 and DNAM-1 activating receptors, and CD94 and KIR 158 a/h were significantly decreased in CRC patients (Figure 4C).
PB-NKs from CRC patients exert low degranulation activity and are less competent in cytokine production
By means of the CD107a assay we analyzed the degranulation activity of NK cells isolated from CRCs. These studies indicated that PB-NK from CRC patients display significantly lower degranulation activity than normal donors, against the NK-susceptible K562 cell line, even when NK cells were stimulated with IL-2 (27.3% ± 13.6 vs 45.2% ± 10.0; P < 0.01) (Figure 4D).
Also, when the same NK cell subsets were analyzed for their ability to produce IFN-γ, a deficient cytokine production was observed in PB-NK from CRC patients compared with PB-NK from normal donors, even when effector cells were pre-activated with IL-2 (12.4% ± 5.9 vs 22.4% ± 8.9, P < 0.01) (Figure 4E).
These results suggest that NK cells from CRCs are not fully competent in relevant degranulation and cytokine release.
PB-NKs from CRC patients exert normal cytolytic activity mediated by Cetuximab
We measured the ability of PB-NK cells to mediate ADCC against the HT-29 cell line (10:1 and 20:1 E:T ratio). We observed an enhancement of cytotoxicity in the presence of 1µg/ml of Cetuximab. Although lower responses were observed in patients as compared with normal donors, these differences were not significant, which is coherent with similar levels of CD16 expression (Figure 4F).
Discussion
In the present study we show that CRC tumor-associated NK cells are present in low amounts. Our data indicate that TANKs display a particular phenotype, characterized mainly by a low proportion of activating receptors. Peripheral blood NK cells from CRC patients also have a deregulated phenotype and they are poorly capable of degranulation and release cytokines, but they maintain Cetuximab-mediated ADCC activity.
It has been proposed that immunosurveillance escape might be the seventh hallmark of cancer. 38 Intricate interactions between tumor and immune cells take place within the tumor microenvironment. These complex, and often unknown, processes result in tumor destruction (elimination phase), equilibrium phase or tumor growth (escape phase) by sculpting immunogenic cancer cells. Experimental and clinical evidence suggests that the edition process dictates the immune response within the tumor microenvironment. NK cells play a crucial role in tumor immunosurveillance 39 and prognosis of CRC patients is dependent on the local immunological tumor microenvironment. 40 The scarcity of NK cells in CRC tumors and normal adjacent mucosa was recently determined by immunohistochemistry. 30 That work was published while we conducted this study in which we obtained similar results by flow cytometry. Our objective was to characterize the NK cells phenotype from different samples of CRC patients and to understand their potential functionality. In this article we examined activating and inhibitory NK cells receptors. We determined that TANKs displayed an altered phenotype, characterized by low expression of the activating receptors CD16, NKG2D, NKp30, NKp44, NKp46, CD161 and DNAM-1. Remarkably, in most of the samples analyzed, NK cells present in normal mucosa showed intermediate values between peripheral blood and tumor. However, MANKs displayed lower levels of expression of CD16, CD94 and CD158b receptors than TANKs. This similarity could be attributable to the fact that NK cells residing in tissues may have altered phenotypes, regardless of whether the tissue is normal or tumor. Surprisingly, KIR receptors were also under-expressed in cancer tissue which, as other authors have described, 41 could indicate a state of hyporesponsiveness. Several mechanisms could explain the down-regulation of NK receptors, such as chronic ligand exposure or cytokine-induced down-modulation, among others. In order to evaluate the role of tumor cells in NK phenotype modulation, we co-cultured NK cells from normal donors over 5 d with CRC cells and we observed that co-cultured NK cells displayed TANK-like altered receptor expression. Co-culture experiments showed CD16, DNAM-1 and CD161 down-modulation. Particularly, for DNAM-1, down-regulation was reversed in transwell assays, suggesting that the mechanism responsible implies cell contact between NK and tumor cells. Interestingly, HT-29 and DLD-1 cells promoted different changes in receptor expression, which leads us to speculate that both cell lines present differences in their ligand expressions. It would be interesting in future studies to characterize such differences, both for cell lines and tumor samples. Even though NKG2D and NKp30 presented a diminution of MFI values, both receptors maintained normal percentages of positive cells after co-culture. The results of functional studies showed a strong decrease in IFN-γ production, but a moderate down-modulation of degranulation activity. This contrast could be explained by normal percentages of NKG2D- and NKp30-positive cells intimately associated with degranulation capacity.42–44 Also degranulation may be more easily induced than IFN-γ production owing to a lower activation threshold. In a recent study 25 a more drastic reduction of INF-γ production than CD107a expression for a particular NK receptor profile in breast cancer was also described.
Not only did TANKs show a different phenotype, but we also observed a reduced expression of certain activating receptors, such as NKG2D, NKp30 and DNAM-1, in peripheral blood of CRC patients. This pattern was also determined in different cancer types, such as acute myeloid leukemia and metastatic melanoma. 45 This altered phenotype in peripheral blood was associated with a lower capacity to stimulate IFN-γ secretion and to degranulate. Unlike NK from co-cultures, PB-NKs presented a reduction in NKG2D- and NKp30-positive cells, which could explain the poor capacity to degranulate. Therefore, we suggest that the altered receptor repertoire in TANKs, as well as the lower lytic activity and production of pro-inflammatory cytokines in PB-NKs, reflects an impairment of NK cell activity toward tumor cells that may contribute to cancer progression.
However, we observed that despite their peculiar phenotype, PB-NKs from patients retained the same in vitro Cetuximab-mediated ADCC activity as those from normal donors. This was probably owing to the fact that the CD16 receptor was normally expressed in PB-NKs from CRC patients. This implies that during Cetuximab treatment, PB-NKs may have the capacity to mediate, at least in part, the antitumor activity of this MAb. TANKs are scarce and only a low percentage of them display normal values of CD16 expression, which would imply a deficient Cetuximab-mediated ADCC at the tumor site. Still, normal ADCC exerted by PB-NKs could be a relevant mechanism for the elimination of circulating tumor cells.
Funding
This work was supported by grants from the Fundación Sales, Fundación P. Mosoteguy, Fundación Cáncer (FUCA), Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) and Fundación María Calderón de la Barca, Argentina. M. Bianchini, J. Mordoh, and E.M. Levy are members of CONICET. Y.S. Rocca, M.P. Roberti and J.M: Arriaga are fellows of CONICET.
Footnotes
Acknowledgements
We are grateful to the members of the Servicio de Hemoterapia of the Instituto A. Fleming for providing blood samples of healthy donors.
Conflict of interest statement
The authors declare that there are no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
