Abstract
Neonates demonstrate functional immaturity and dysregulation of immune responses leading to systemic inflammation and enhanced apoptosis of immune cells. Thalidomide has already been proven to differentially regulate immune responses and support anti-apoptosis in immunodeficiency syndromes. Thus, it was the aim of this study to evaluate the effects of thalidomide on the cytokine response and apoptosis of neonatal immune cells. After whole blood culture and stimulation of cord and adult blood samples, the intracytoplasmic expression and the secreted amounts of IL-2, TNF-α, IFN-γ, IL-6, IL-10 and IL-8 were assessed by flow cytometry and Cytokine Bead Array. Apoptosis was detected using Annexin-V staining. Bcl-2 expression was analysed using the Cytokine Bead Array Apoptosis Kit. Exposure to thalidomide (100 µg/ml) reduced the intracytoplasmic pro-inflammatory cytokine production of neonatal monocytes and the IFN-γ production of neonatal lymphocytes. In supernatants, the addition of thalidomide resulted in reduction of TNF-α, IL-6, IL-10 and, by trend, IFN-γ. While stimulated neonatal lymphocytes exhibited susceptibility to apoptosis, thalidomide tended to diminish apoptotic cells. Bcl-2 expression tended to be increased after addition of thalidomide. The potent anti-inflammatory effects of thalidomide and its anti-apoptotic properties in cord blood immune cells provide the basis for future strategies to optimise treatment of neonatal infections and immunodeficiency syndromes.
Introduction
Neonates are prone to infectious diseases, which is to be explained by an immaturity in innate and adaptive immune responses. Innate immune responses are characterised by a reduced ability to eliminate invading microbes by chemotaxis, phagocytosis and bacterial killing.1,2 Nevertheless, neonatal innate immune cells are capable of initiating pro-inflammatory cytokine responses, thus providing the basis for an imbalance between pathogen elimination and the inflammatory response.3,4
The delicate interaction between innate and adaptive immune mechanisms is also evident in neonates. Professional antigen-presenting cells exhibit functional alterations expressing diminished co-stimulatory molecules and providing deficient assistance in T cell activation. 5 Incompletely activated neonatal T cells show deficiencies in cytokine production and proliferation with a bias towards Th2.6,7 In addition, neonatal lymphocytes are highly susceptible to apoptosis during infections (partially mediated by TNF-α), while prolonged lymphopenia has been associated with fatal outcomes of sepsis.8,9
This state of immunodeficiency in neonates may be a substantial part of a physiological maturation process of the immune system. However, an impaired balance of immune responsiveness may result in overwhelming systemic inflammatory response syndrome (SIRS) and contribute to severe organ sequelae, such as white matter damage and chronic lung disease.10,11 Therefore, recent investigations aim to find immunomodulatory drugs which may prevent or beneficially influence SIRS at innate and adaptive stages of immune responses in neonates at risk.2,12
Thalidomide is a structurally altered barbiturate, known as Contergan since the 1950s, which caused limb defects in offspring. It was re-introduced in 1997 for the treatment of erythema nodosum leprosum, chronic autoimmune diseases, multiple myeloma and, finally, solid neoplasms. 13 Apart from its potential to treat malignancies, thalidomide demonstrated distinct immunomodulatory effects predominantly reducing TNF-α expression. 14 This anti-TNF-α impact accounts for its efficacy in treating inflammatory diseases. Furthermore, modified thalidomide analogues proved to enhance Th1 cytokine responses, anti-apoptosis and the proliferation of T cells, as well as their interaction with dendritic cells. On the contrary, the monocytic cytokine response, including IL-10 expression, was inhibited.15–17 Notably, all of these features of the immune response mentioned above seem to be immature or dysregulated in neonates. 18 Thus, thalidomide might have the potential to regulate neonatal immune responses by stimulating deficient parts of the (adaptive) immune response and down-regulating hazardous pro-inflammatory responses in neonates.
Interestingly, in the case of other disease states predisposing to infections like AIDS, the immune regulating effects of thalidomide have already been proven in vivo.19,20 However, there are no data on the impact of thalidomide on the neonatal immune system. As we demonstrated previously that immunomodulators such as sirolimus or vitamin C, for example, are important tools to elucidate and influence neonatal immune responses, it was the aim of this study to evaluate the immunomodulatory effects of thalidomide on the cytokine response and apoptosis of neonatal immune cells in in vitro whole blood assays.21–23
Materials and methods
Study population
Adult whole blood samples were obtained from healthy volunteers. After informed consent of parents, cord blood samples from healthy neonates (37–42 wks of gestation) after unremarkable pregnancy and spontaneous delivery (no intrauterine infection, no pre-eclampsia, no asphyxia) were collected in lithium–heparin tubes (Sarstedt, Nürnbrecht, Germany). Two neonates born at 35 and 36 wks of gestation were included. Both of them were born prematurely because of preterm labour without signs of infection. Intrauterine infection was excluded based upon clinical assessment and laboratory values. Birth masses were within normal ranges and gender was equally distributed.
Reagents
LPS, phorbol 12-myristate 13-acetate (PMA), ionomycin and monensin were obtained from Sigma (Deisenhofen, Germany). The mouse-anti human mAbs anti-CD3 (clone CLB-T3/4.E, 1XE) and anti-CD28 (clone CLB-CD28/1, 15EB) were obtained from Sanquin Reagents (Amsterdam, the Netherlands). Sirolimus (Rapamune, Wyeth Europe Ltd, Taplow, Maidenhead, UK) and thalidomide (Fagron, Barsbüttel, Germany) were dissolved in DMSO at 10−3 M (sirolimus, thalidomide) and freshly diluted as indicated using RPMI 1640 medium. Camptothecine (Sigma, St Louis, MO, USA) was dissolved in DMSO. Vitamin C (ascorbic acid; Rotexmedica, Trittau, Germany) was dissolved to appropriate concentrations in RPMI 1640 medium only.
Culture and stimulation of cells
Heparinised whole blood was suspended in RPMI 1640 supplemented with 1% penicillin/streptomycin, 2 mM glutamine, 1 mM pyruvate and non-essential amino acids (Seromed Biochrome, Berlin, Germany) within max. 24 h to collection. Final cell concentrations (cord blood leukocytes) were 5 × 106/ml for intracellular cytokines, 1.5 × 106/ml for Cytometric Bead Array (CBA) cytokines, 1 × 106/ml for Annexin and 2.5 × 106/ml for CBA apoptosis. Incubation times were different in each experiment ranging from 2 to 96 h (see figure legends). Aliquots of whole blood were pre-incubated with sirolimus, thalidomide, vitamin C or camptothecine in concentrations as indicated, or with the same volume of dissolving agent (5% glucose, DMSO) for 1 h (2 h for VitC) in multi-well plates at 37°C, 5% CO2. To induce IL-6, IL-8 and TNF-α production in monocytes, pre-incubated whole blood cultures were stimulated with 100 ng/ml LPS. Similarly, cytokine production (IL-2, IFN-γ, TNF-α) in lymphocytes was stimulated with 3 µg/ml PMA and 3 µM ionomycin. Cells were exposed to 3 µM monensin (Sigma) during whole stimulation period, followed by fixation with 4% paraformaldehyde (Riedel de Haen, Seelze, Germany), as described previously.3,24 In order to induce cytokine release in supernatants whole blood cultures were stimulated with 1 µg/ml anti-CD3- and/or 0.5 µg/ml anti-CD28-Abs or PMA/ionomycin for 24 h. An unstimulated control was added in each experiment.
Intracellular staining of cytokines
Cells were washed in HBSS and re-suspended in a buffer consisting of HBSS, 0.1% saponin (Riedel de Haen) and 0.01 M HEPES buffer (Seromed Biochrome). Two-hundred µl aliquots of cells were added to tubes containing 0.5 µg/10 µl of mAbs (BD Pharmingen, Heidelberg, Germany) against CD3 (17A2, Cy5-conjugated), CD14 (M5E2, PC5-conjugated), IL-2 (MQ1-17H12, PE-conjugated), IL-6 (MQ2-13A5, FITC-conjugated), IL-8 (G265-8, PE-conjugated), IFN-γ (4S.B3, FITC-conjugated) and TNF-α (mAb11, FITC-conjugated). Pre-incubation with a surplus of unconjugated anti-cytokine mAbs (5 µg/10 µl; Pharmingen) served as a negative control for intracellular staining to each sample. Isotype-specific Abs were used to detect irrelevant specificity for surface molecule staining. Flow cytometric analysis was performed as described previously. 24
Extracellular cytokine detection using CBA Human Soluble Protein Master Kit and Flex Sets
After incubation for 24 h the supernatant was collected, washed and frozen at −70°C until analysis. The Flex Sets IL-2, IFN-γ, TNF-α, IL-6 and IL-10 were employed for detection of cytokines. Further procedures were performed according to the manufacturer’s protocol. In brief, a mixture of 50 µl of supernatant and 50 µl of mixed capture beads was incubated for 1 h followed by the addition of 50 µl PE-detection reagent and further incubation for 2 h. For each flow cytometric analysis a standard curve was prepared using the Flex Set Protein Standards. Each analysis comprised 1000 beads. Flow cytometric analysis was performed according to the manufacturer’s protocol using BD FACSDiva® and BD FCAP Array® software.
Apoptosis detection via Annexin-V and CBA Apoptosis Kit
Culture was performed with and without stimulation by PMA and ionomycin to assess spontaneous and activation-induced cell death. Controls consisted of 4 (−16) µM camptothecine to induce apoptosis and a sample without any additional substances as standard. After incubation, whole blood cultures were haemolysed for 20 min using haemolysis buffer (pH 8.0; NH4Cl 16,52 g, KHCO3 2 g, EDTA 0.08 g ad Aqua dest. 2000 ml).
For CBA Apoptosis Kit (active Caspase-3, PARP, Bcl-2) procedures were performed according to the manufacturer’s protocol. In brief, cells were lysed using the cell lysis buffer and exposed to proteinase inhibitor cocktail. Detritus was centrifuged and the lysate was frozen at −70°C until further analysis. A standard curve was prepared using the protein standards for each flow cytometric analysis. Each analysis comprised 1000 beads. Flow cytometric analysis was performed according to the manufacturer’s protocol using BD FACSDiva® and BD FCAP Array® software. Owing to problems with beads for active Caspase-3 and PARP by the manufacturer only results for Bcl-2 were analysed.
For Annexin, 100-µl aliquots of cells were added to tubes containing 0.5 µg/10 µl of a mAb (BD Pharmingen, Heidelberg, Germany) against CD3 (17A2, Cy5-conjugated). Isotype-specific Abs were used to detect irrelevant specificity for surface molecule staining. After surface staining the protocol of the manufacturer was applied. Flow cytometric analysis was performed according to the manufacturer’s information. A minimum of 5000 cells was investigated. Tubes without Annexin or propidium iodide served as an additional negative control.
Statistical analysis
Statistical differences between adults and neonates were tested by the Mann-Whitney U-test. For non-parametric analysis within one group the Wilcoxon Rank Sum test was applied. Data are expressed as median and range, if not otherwise indicated. The level of significance was defined as P < 0.05 in single comparisons. Statistical analyses were performed using PASW® Statistics18.0 (SPSS Inc., Chicago, IL, USA).
Results
Impaired intracellular cytokine response in neonatal lymphocytes
Before evaluating differences between thalidomide’s effects on neonatal and adult cytokine responses, the intracytoplasmic cytokine expression of neonates and adults was compared. After stimulation with PMA and ionomycin for 4 h the number of IL-2-, IFN-γ- and TNF-α-positive CD3+ lymphocytes in neonates was significantly reduced compared with adults. In contrast, the percentage of neonatal monocytes expressing IL-6 and IL-8 did not differ from adult monocytes after stimulation with LPS for 4 h, but monocytic TNF-α expression was significantly increased in neonates (Figure 1A,B).
Different cytokine expression of adult and neonatal lymphocytes and monocytes. Cord blood samples of healthy neonates (n = 13) and peripheral blood samples of healthy adults (n = 8) were incubated in multi-well plates at 37°C, 5% CO2. Cytokine production (IL-2, IFN-γ, TNF-α) in lymphocytes was stimulated with 3 µg/ml PMA and 3 µM ionomycin (A). Monocytic cytokine production (IL-6, IL-8, TNF-α) was stimulated with 100 ng/ml LPS (B). Data are presented as box plots representing median, 10th, 25th, 75th and 90th percentile of intracytoplasmatic cytokine expression. A P value of <0.05 was considered significant (Mann-Whitney U-test).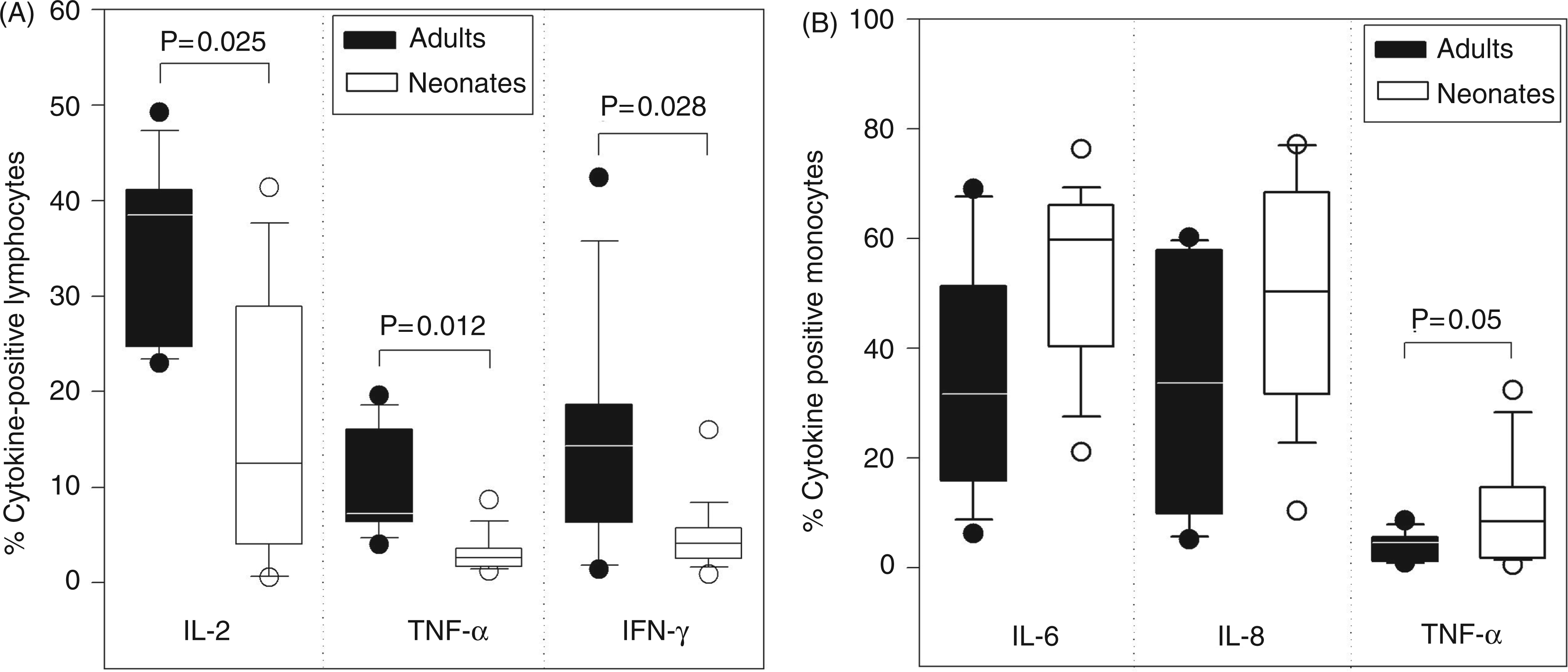
Partial anti-Th1 effect of thalidomide on intracellular cytokine response of neonatal lymphocytes
Thalidomide’s effects on the intracellular cytokine response in lymphocytes were compared to effects on adult cells. The addition of 100 µg/ml thalidomide to PMA/ionomycin-stimulated whole blood cultures reduced the IFN-γ expression of neonatal lymphocytes. Interestingly, a trend of enhancement of IL-2 expression was noted for neonatal cells (Figure 2A). Furthermore, thalidomide diminished TNF-α expression in adult lymphocytes, while the number of TNF-α-positive neonatal lymphocytes was unaffected. All effects of thalidomide were dose-dependent (Figure 2B–D). Notably, concentrations below 1 µg/ml had no effects on cytokine expression while concentrations above 1 mg/ml were cytotoxic.
Co-stimulating and anti-inflammatory influence of thalidomide on cytokine production of neonatal and adult lymphocytes. Cord blood samples of healthy neonates (n = 13) and peripheral blood samples of healthy adults (n = 8) were pre-incubated for 1 h with 100 µg/ml thalidomide or with the same volume of dissolving agent (DMSO = control) in multi-well plates at 37°C, 5% CO2. Cytokine production (IL-2, IFN-γ, TNF-α) in lymphocytes was stimulated with 3 µg/ml PMA and 3 µM ionomycin (A). Drug-concentration experiments were performed for each cytokine (n = 5; B–D). Data are presented as box plots representing median, 10th, 25th, 75th and 90th percentile of intracytoplasmatic cytokine expression or as median and range of percentage of cytokine-positive lymphocytes for drug concentration experiments. A P value of <0.05 was considered significant (Wilcoxon-Rank-Sum test).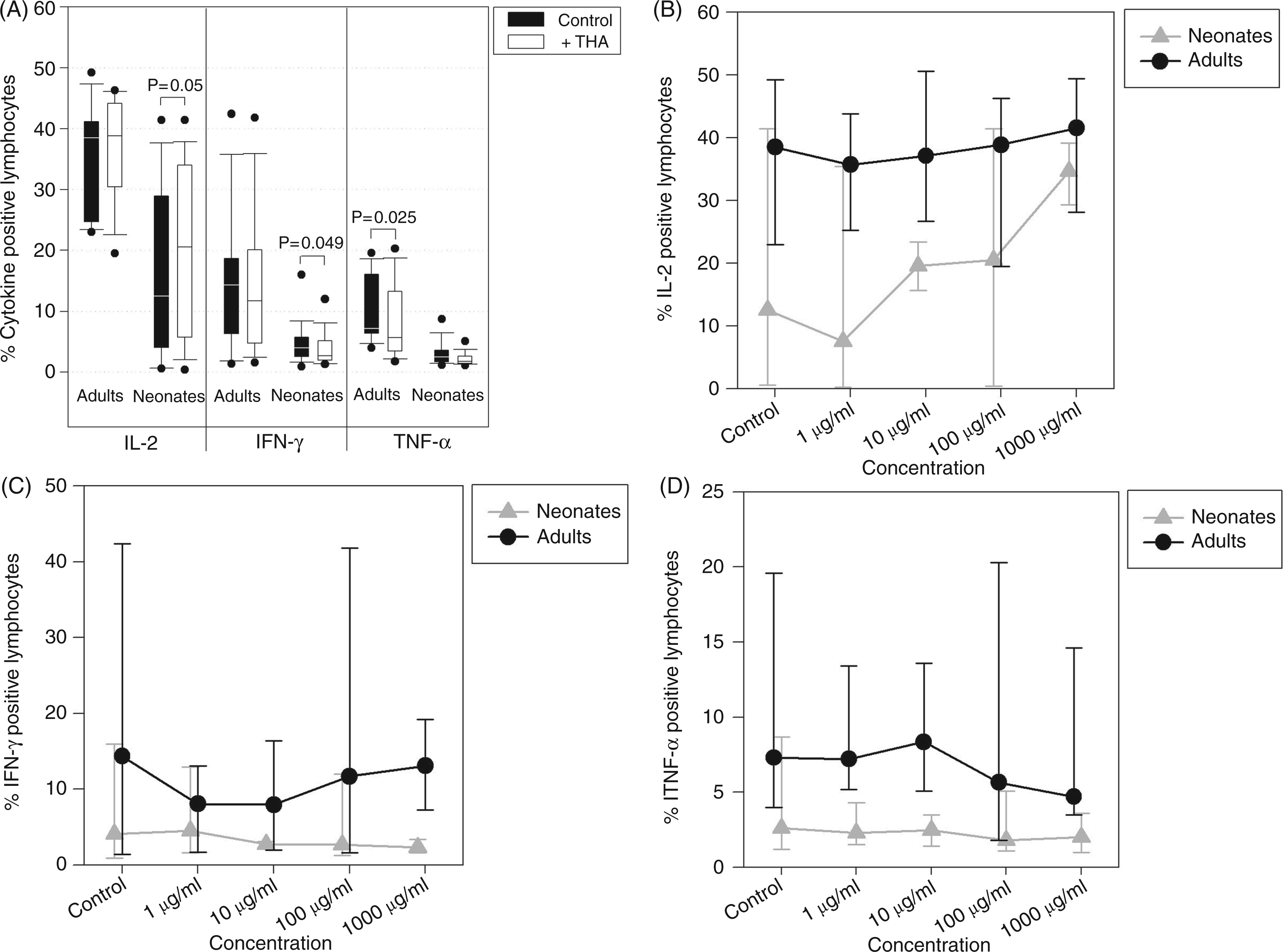
Thalidomide’s anti-inflammatory impact on cytokine release in supernatants of cord blood leukocytes
As stimulation with mAbs was reported to be deficient in neonatal whole blood cultures, it was compared firstly with PMA/ionomycin. Secondly, the effects of thalidomide on cytokine release of neonatal cells were evaluated to confirm results for intracellular cytokines.
Anti-inflammatory effect of thalidomide on cord blood leukocytes in supernatants. Cord blood samples of healthy neonates (n = 8) were pre-incubated for 1 h with 100 µg/ml thalidomide or with the same volume of dissolving agent (DMSO = control) in multi-well plates at 37°C, 5% CO2. Cytokine production in cord blood leukocytes was stimulated with 3 µg/ml PMA and 3 µM ionomycin, or 1 µg/ml anti-CD3- and/or 0.5 µg/ml anti-CD28-Abs for 24 h. Data are presented as median and range of cytokine concentration in supernatants in pg/ml. A P value of <0.05 was considered significant (Wilcoxon-Rangsum test)
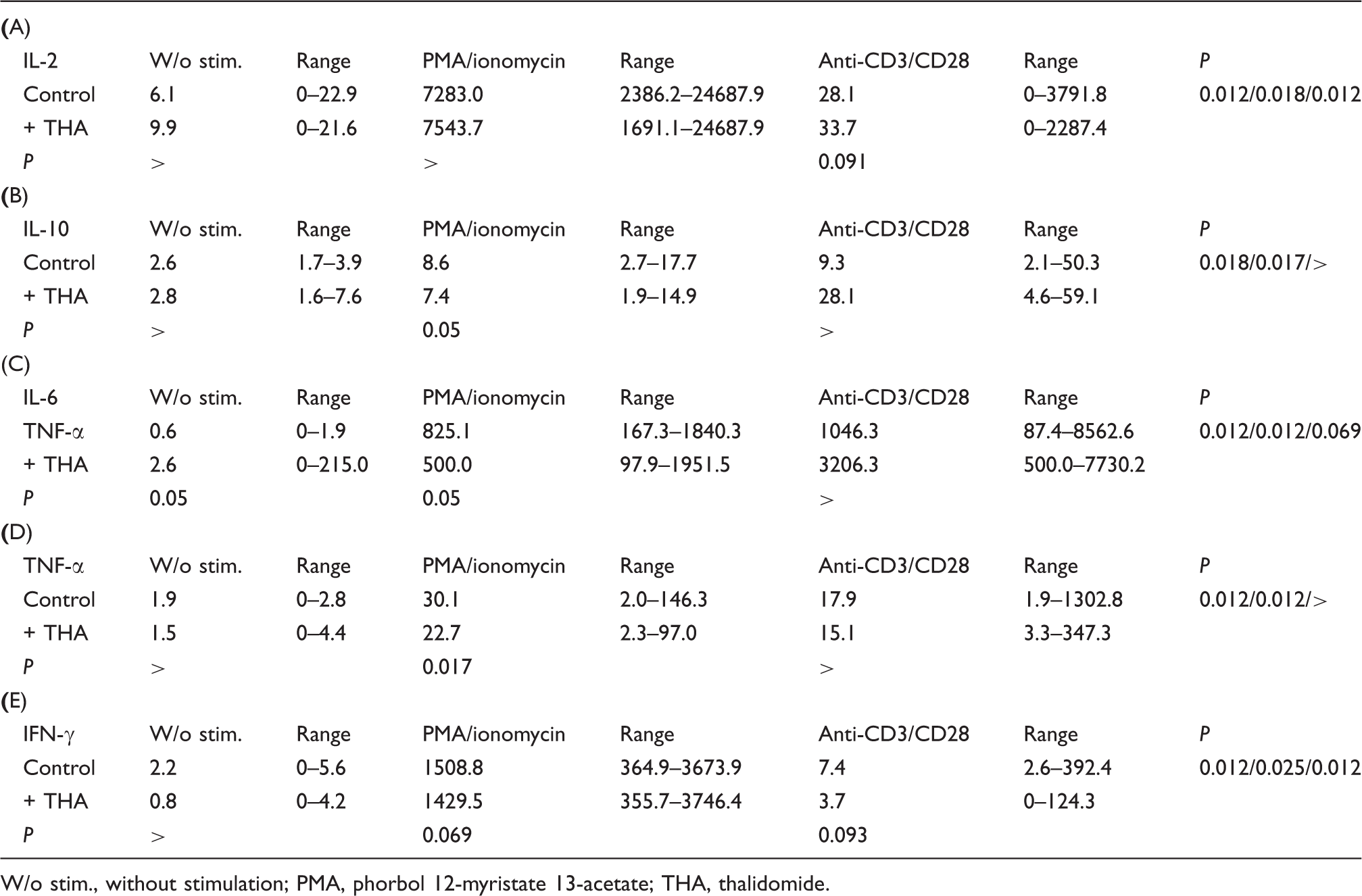
W/o stim., without stimulation; PMA, phorbol 12-myristate 13-acetate; THA, thalidomide.
The addition of 100 µg/ml thalidomide to whole blood cultures inhibited expression of TNF-α with both stimulations. Likewise, a trend was noted for an inhibitory effect of thalidomide on IL-6 and IL-10 expression in PMA/ionomycin-stimulated cultures and on IFN-γ for both stimulations (Table 1A–E). By contrast, IL-6 and IL-10 expression tended to be increased by thalidomide upon stimulation with anti-CD3/CD28.
Anti-inflammatory effect of thalidomide on neonatal monocytes
Thalidomide’s impact on the intracellular cytokine response of monocytes was analysed next. Upon addition of 100 µg/ml thalidomide a depletion of the pro-inflammatory cytokine production by LPS-stimulated neonatal monocytes was demonstrated. In particular, the production of IL-8 and TNF-α was significantly decreased. The number of TNF-α-producing adult monocytes was also inhibited (Figure 3A). With regard to dose-dependence, lower concentrations of thalidomide induced an increase in the IL-8-expression of adult monocytes, whereas neonatal cells displayed dose-dependent inhibition of pro-inflammatory cytokines (Figure 3B–D).
Selective inhibition of pro-inflammatory cytokine production of neonatal monocytes. Cord blood samples of healthy neonates (n = 13) and peripheral blood samples of healthy adults (n = 8) were pre-incubated for 1 h with 100 µg/ml thalidomide or with the same volume of dissolving agent (DMSO = control) in multi-well plates at 37°C, 5% CO2. Cytokine production in monocytes was stimulated with 100 ng/ml LPS (A). Drug-concentration experiments were performed for each cytokine (n = 5; B–D). Data are presented as box plots representing median, 10th, 25th, 75th and 90th percentile of intracytoplasmatic cytokine expression or as median and range of percentage of cytokine-positive monocytes for drug concentration experiments. A P value of <0.05 was considered significant (Wilcoxon-Rangsum test).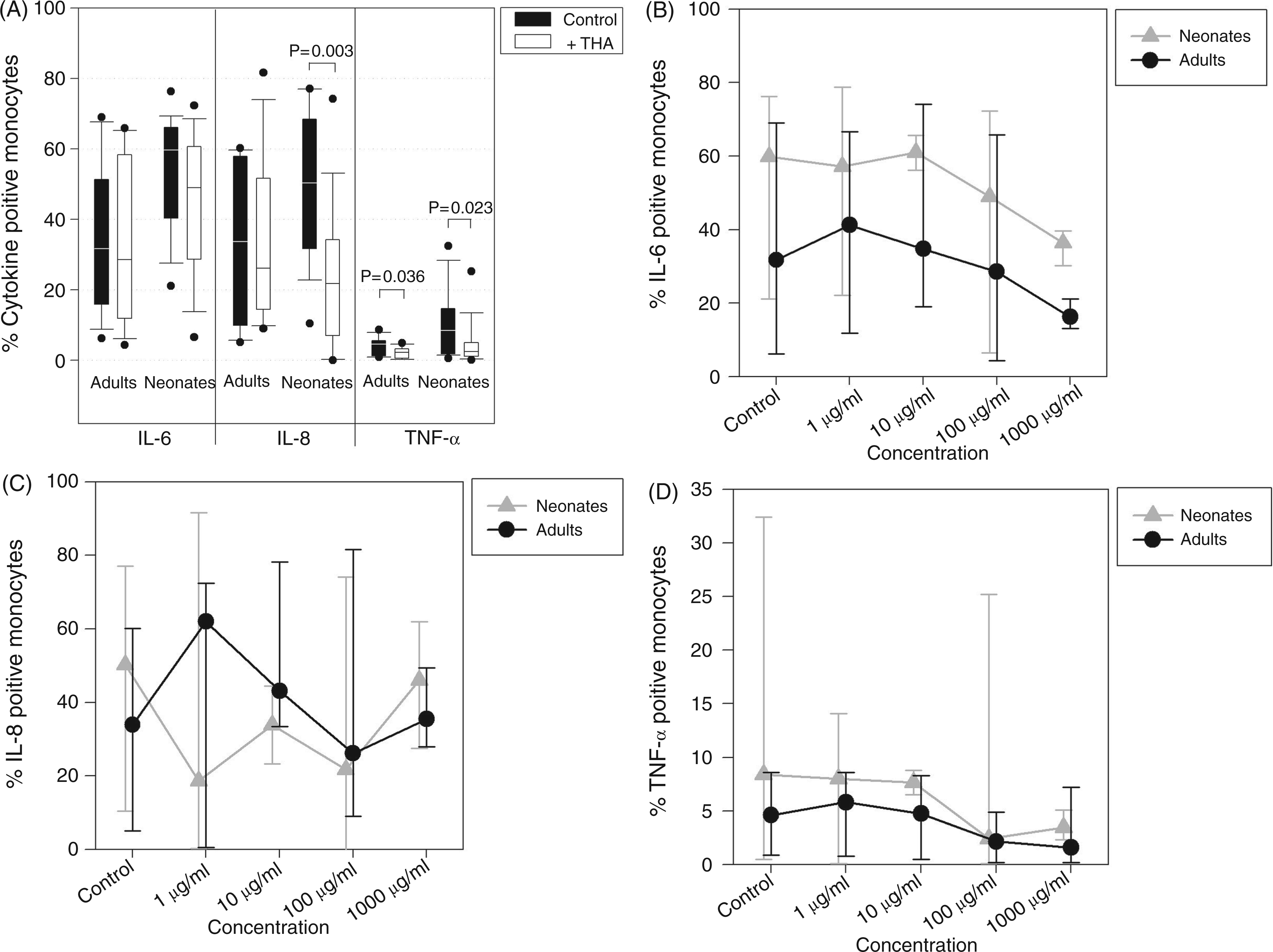
Course of apoptosis of neonatal lymphocytes in whole blood cultures
As the general course of apoptosis in whole blood cultures of cord blood was unclear, it had to be established before evaluating the effects of thalidomide. After 4 h of incubation a small proportion of neonatal lymphocytes was early in apoptosis, characterised by Annexin only. Compared with 4 h the number of apoptotic neonatal lymphocytes after 24 h without stimulation showed no major differences while increasing slowly thereafter. During the whole culture the percentage of apoptotic lymphocytes did not exceed 21% without stimulation (Figure 4A). The stimulation with PMA/ionomycin induced 23% apoptotic cells at 24 h, 47% at 48 h and 51% at 96 h (Figure 4B). Camptothecine missed the desired efficiency in induction of apoptosis (data not shown).
Time-dependent induction of apoptosis in stimulated and unstimulated neonatal lymphocytes and its modulation by thalidomide and vitamin C. Cord blood samples of healthy neonates were pre-incubated for 1 h with 100 µg/ml thalidomide or 10 ng/ml sirolimus, or for 2 h with 20 mM vitamin C or with the same volume of dissolving agent (Aqua dest. or DMSO = control) in multi-well plates at 37°C, 5% CO2. Lymphocytes were stimulated with 3 µg/ml PMA and 3 µM ionomycin or left unstimulated for 4–96 h. Data are presented as median and range of percentage of Annexin-positive lymphocytes of four identical experimental series. (A: unstimulated cells; B: PMA/ionomycin-stimulated cells).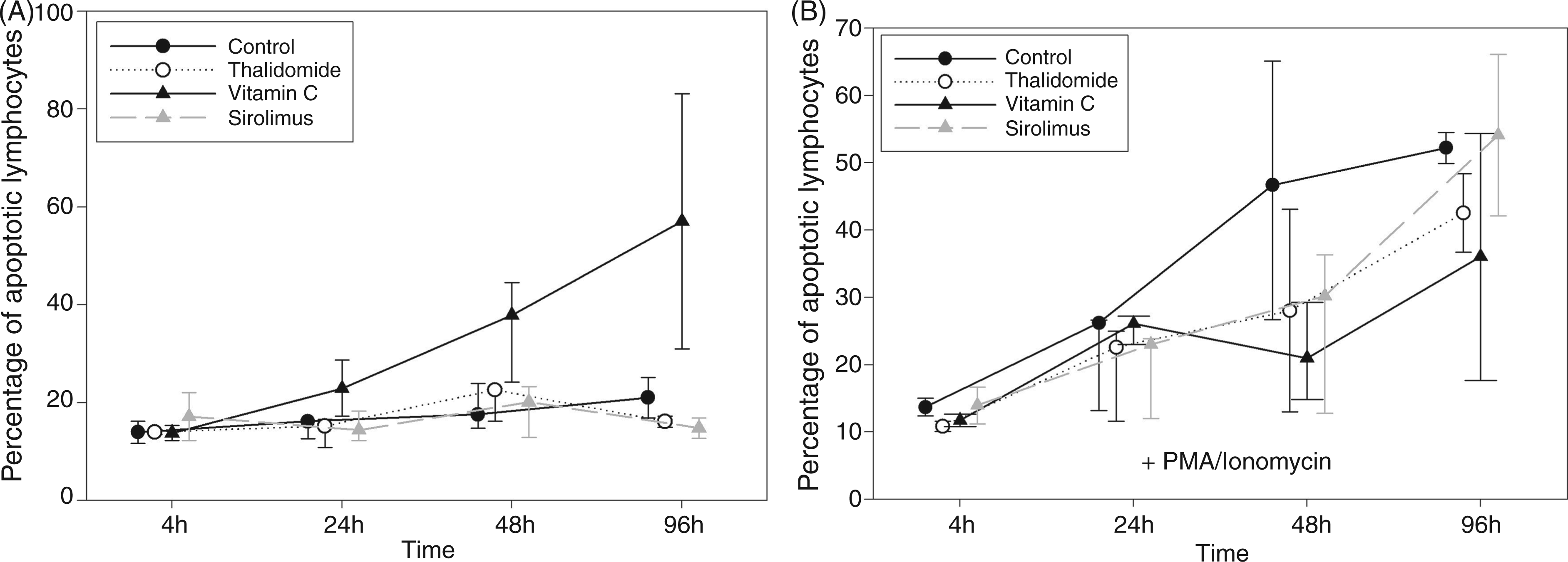
Reduction of apoptosis of stimulated neonatal lymphocytes by thalidomide and vitamin C
Thalidomide’s effects on apoptosis of neonatal lymphocytes were analysed next and compared with other apoptosis-modulating agents. The addition of 100 µg/ml thalidomide and 20 mM vitamin C resulted in a reduction of apoptosis of stimulated neonatal lymphocytes.
In particular, vitamin C demonstrated a divergent influence on apoptosis—massively increasing the number of apoptotic cells in controls without stimulation. However, vitamin C was capable of reducing the number of apoptotic cells after stimulation at 48 h and 96 h of incubation (Figure 4A,B).
Likewise, thalidomide tended to deplete apoptotic lymphocytes after stimulation with PMA/ionomycin at each point in time. Besides, thalidomide seemed to have no impact on apoptosis in unstimulated cells (Figure 4A,B). Again, concentrations below 1 µg/ml apparently failed to affect apoptosis (data not shown).
The immunosuppressive drug sirolimus was not able to modulate apoptosis in our setting.
Influence of thalidomide and vitamin C on Bcl-2 expression in neonatal cord blood leukocytes
The Bcl-2 expression was analysed to gain further insights in the anti-apoptotic effect of thalidomide. Bcl-2 was constantly expressed by stimulated neonatal cord blood leukocytes in whole blood cultures. It was also expressed at a lower rate by adult peripheral blood leukocytes.
Both 100 µg/ml thalidomide, and 10 mM and 20 mM vitamin C tended to increase the expression of Bcl-2 at 48 h of culture (P = 0.068 for each). The effect was evident at each time point for vitamin C and after 8 h for thalidomide (Figure 5A,B).
Thalidomide and vitamin C increase Bcl-2 expression in PMA/ionomycin-stimulated cord blood leukocytes. Cord blood samples of healthy neonates (n = 4) and peripheral blood samples of healthy adults (n = 4) were pre-incubated for 1 h with 100 µg/ml thalidomide or 5 µM camptothecine, or for 2 h with 20 mM vitamin C or with the same volume of dissolving agent (Aqua dest. or DMSO = control) in multi-well plates at 37°C, 5% CO2. Cord blood leukocytes were stimulated with 3 µg/ml PMA and 3 µM ionomycin for 48 h. Data are presented as box plots showing median, 25th and 75th percentile of Bcl-2 in IU/mL (A). For time series experiments cord blood leukocytes were stimulated with 3 µg/ml PMA and 3 µM ionomycin for 2–48 h after pre-incubation with 100 µg/ml thalidomide, 5 µM camptothecine or 20 mM vitamin C. Time series experiments were performed twice. Data are presented as Bcl-2 in IU/ml representing one representative experiment (B).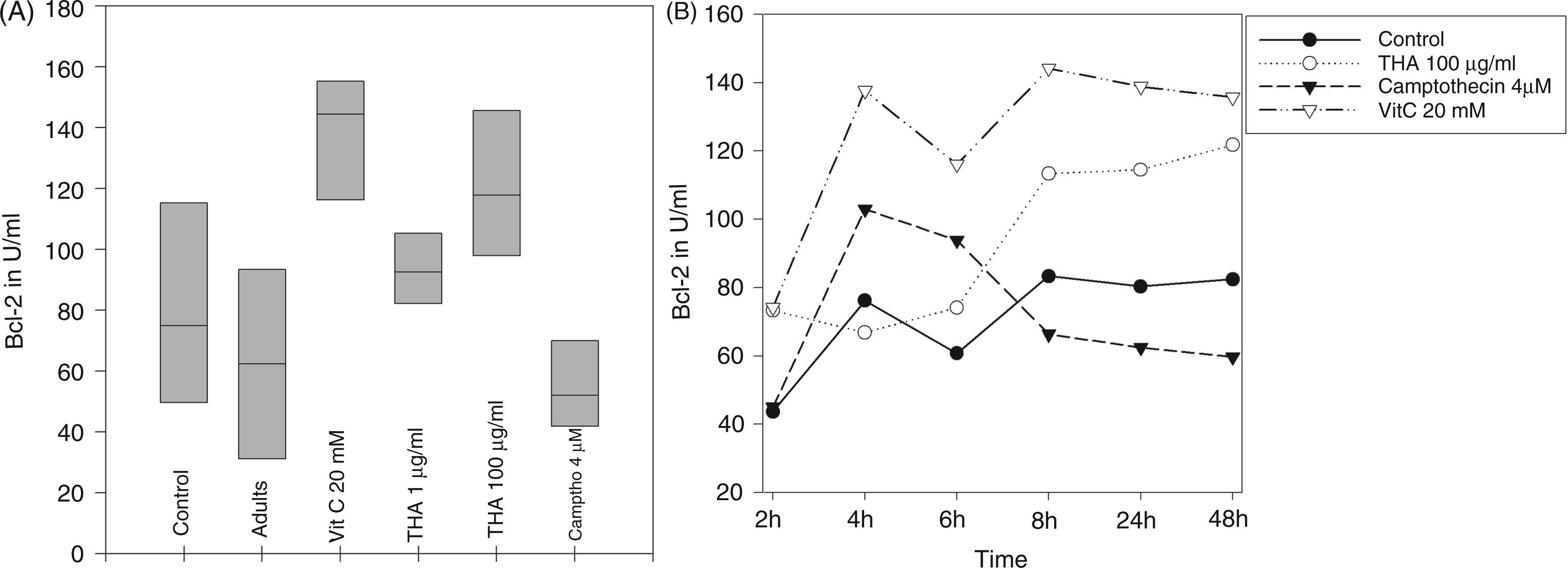
Discussion
The immunomodulatory capacity of thalidomide and its more potent analogues divided into immunomodulatory drugs (IMiDs) like Lenalidomid and SelCIDs (selective cytokine inhibiting drugs) is subject to a lot of ongoing studies for treatment of inflammatory and oncological diseases.16,25 In particular, the divergent influence of thalidomide promoting adaptive immune responses (e.g. co-stimulation of T cell proliferation or boost of Th1 responses) and inhibiting pro-inflammatory innate immune responses (e.g. down-regulation of cytokines TNF-α and IL-6) are interesting features for the evaluation of neonatal immune responses suffering from the consequences of deficiency of both adaptive and innate immunity.14,17,26,27 To the best of our knowledge there are no data on thalidomide’s effects on these functions of neonatal immune cells.
Moreover, thalidomide has been proven to function anti-apoptotically in adult cells. During neonatal sepsis severe apoptosis of lymphocytes and prolonged lymphopenia caused by high levels of TNF-α are associated with a high mortality, but data on modulation of apoptosis in neonatal lymphocytes are scarce.8,28,29
In this study we therefore investigated the effects of thalidomide on the cytokine response and apoptosis of neonatal monocytes and lymphocytes in a human in vitro whole blood assay.
Firstly, we were able to confirm that the expression of T cell cytokines is well inducible in cord blood, even if stimulated with Abs against CD3 and CD28. 30 Nitsche et al. failed to induce a cytokine response with both Abs. 31 In addition, we demonstrated that neonatal lymphocytes exhibit significantly impaired cytokine expression compared with their adult counterparts symbolising a state of immunodeficiency and probably tolerance.18,23
Thalidomide, by trend, augmented the intracellular expression of IL-2 in neonatal lymphocytes according to adult studies, although this could not hold true for cytokine expression in supernatants.15,17 On the contrary, the intracellular expression of IFN-γ was significantly reduced and the cytokine release in supernatants tended to be diminished. Decreased IFN-γ expression was demonstrated previously in adult cells by McHugh et al. and Marriot et al., while all other studies displayed a potentiation of Th1 specific cytokines.15–17,26,32 The stimulation of the Th1 response is thought to be regulated by increasing the activation of the transcription factor AP-1 and CD28 receptor signal transduction, which is deficient in neonates.27,33,34
Thalidomide’s anti-Th1 effect on neonates in our study partially objects the possible counteraction of the Th2-biased immune response of neonates by amplifying the main Th1 cytokine IFN-γ. Apart from not increasing the IFN-γ response we were able to note a reduced IL-10 expression in supernatants after PMA/ionomycin. Other typical Th1 cytokines, such as IL-12, should be analysed in future studies.14,15
Interestingly, we were not able to demonstrate any impact of thalidomide on the proliferation of neonatal cells (data not shown), although this is believed to be a co-stimulatory function in adults.15,26 However, an explanation might be the deficient IL-2 production of neonates. But, in adults, co-stimulation and proliferation by thalidomide has been achieved by exogenous activation of the CD28 receptor with release of NF-κB.27,33 Thus, the deficient signal transduction of CD28 in neonates might further explain the missing multiplier effect of thalidomide. Consequently, IFN-γ expression and already diminished proliferation would be altered again.17,31,35
Furthermore, we demonstrated a significant reduction of the expression of TNF-α on a single cell basis and in supernatants, as well as a decline in intracellular IL-8 in neonatal monocytes by thalidomide. Similarly, the IL-6 release in supernatants after PMA/ionomycin stimulation was diminished by trend. This observation is in line with studies for adult cells, but in contrast to the study of Sampaio et al., who demonstrated no effects on adult monocytes.14,36,37 The molecular basis of this inhibition of innate pro-inflammatory responses seems to be the blockage of the transcription factor NF-κB.38,39 In addition, the depletion of TNF-α is managed by faster degradation of mRNA and the inhibition of other LPS-induced cytokines by an inhibitory component of thalidomide on phosphodiesterase-4 and on cyclooxygenase-2 function.14,40,41 The anti-inflammatory effects of thalidomide on innate immune responses could provide the basis for conquering excessive cytokine release in neonatal infections or SIRS, and preventing disastrous outcomes.
Moreover, thalidomide tended to increase IL-6 and IL-10 levels in our setting after CD3/CD28 stimulation, which implies that the effect of thalidomide may be context- and, likely, cell population-dependent. Likewise, we were not able to individually assign the observed cytokine secretion and effects of thalidomide in CBA experiments to subgroups of cord blood leukocytes as a result of the whole blood culture technique, although we demonstrated cell population-specific effects in intracellular cytokine experiments. Next to lymphocytes and monocytes, neutrophil granulocytes comprising a distinct subgroup of cord blood leukocytes might have contributed to the secretion of IL-6 and TNF-α, but not IL-10.42,43 We believe that the benefits of whole blood culture (most essential factors, including accessory cells present in physiological concentrations, minimised manipulation due to isolation procedures) outweigh this handicap.44,45 Future experiments using isolated cell populations will have to elucidate whether the anti-inflammatory properties of thalidomide is cell-type dependent.
In a final step we evaluated the course of apoptosis in cord blood cultures and its manipulation by thalidomide in a small experimental series, which may limit the generalisability of our results. We noted resistance to apoptosis in unstimulated neonatal lymphocytes, even after 96 h of whole blood culture. This might be explained by findings of Wasiluk et al., which demonstrated reduced production of FasR and FasL in neonates. 46 We were able to further illustrate an enhanced expression of Bcl-2 in neonatal cord blood leukocytes in comparison to adult cells. High levels of Bcl-2 are considered a survival factor (e.g. in recent thymic emigrants). 47 Likewise, the deficient IL-2 production of neonatal lymphocytes failing to prime the apparatus of activation-induced cell death might explain reduced apoptosis. 48 The resistance to apoptosis counters observations by Wechsler-Reya et al. and Tuaeva et al., but supports findings by Fritzsching et al. for cord blood regulatory T cells. Following stimulation apoptosis was induced in cord blood lymphocytes.7,49,50
In our setting, camptothecine failed to induce apoptosis in the Annexin assay, but decreased Bcl-2 in neonatal cells after 8 h of CBA assay. Though resistance to apoptosis might be one cause for this deficiency, a reduction of Bcl-2 does not need to result in apoptosis. The Annexin assay scans for stronger signs of apoptosis, which camptothecine might not be able to induce in neonatal cells, as previously demonstrated in other cell lines.51,52
Thalidomide tended to inhibit apoptosis after 48 h of cord blood culture, which was apparent at every time point of culture. Sampaio et al. displayed a similar anti-apoptotic influence on monocytes in vivo during treatment of erythema nodosum leprosum, while Aseffa et al. were not able to demonstrate any impact on apoptosis.53,54 Aside from increasing the intracellular Bcl-2 concentration by trend and significantly decreasing pro-apoptotic TNF-α production, the augmentation of the intracellular IL-2 expression might further explain this anti-apoptotic effect. Vitamin C behaved similarly in apoptosis assays providing the basis for future studies. 2 Interestingly, inhibition of CD34+ stem cells by lenalidomide upon up-regulation of a negative regulator of cell cycle has already been demonstrated. 55 Although providing opportunities for prolonged inflammation by supporting survival of all leukocytes, thalidomide’s anti-apoptotic impact on neonatal cells could improve lymphocyte death in neonatal SIRS and circumvent lymphopenia. 46
In summary, our data indicate potential inhibition of apoptosis of lymphocytes and inhibition of pro-inflammatory cytokine production of monocytes by thalidomide next to an anti-Th1 effect, and a slight increase of IL-2 expression. Together with a fortification of cytotoxic activity of CD8 T cells, the activation of natural killer cells and the amelioration of the interaction of T cells and dendritic cells, we provide the basis for future studies of thalidomide as a positive immune modulator in neonatal SIRS and states of immunodeficiency.17,56 Notably, long-term effects of thalidomide regarding the (early) development of the immune system remain unclear, although single case reports do not report severe immune alterations. 57 Official health surveys in the patient cohort of thalidomide-affected newborns of the 1950s and 1960s are missing. 58
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there are no conflicts of interest.
