Abstract
Hepcidin has been found to be the key regulator of iron metabolism that leads to
hypoferremia during inflammation. Recent work has shown that equine hepcidin is
predominantly expressed in the liver of horses. In this study, hepcidin gene expression
was determined in the liver and bone marrow of six healthy horses after
Introduction
Hepcidin is a 25-amino acid peptide synthesized mainly in mammalian, fish and bird liver in response to iron store and inflammation. 1 – 4 It is also expressed in other tissues, e.g. cerebral cortex, spleen, kidney, heart, bone marrow, muscle and intestinal tissues. 4 – 7 Recently, hepcidin real-time PCR relative-quantification in healthy horses showed that hepcidin transcripts is highly abundant in liver, although lower level expression is present in other tissues. 8
Hepcidin was originally identified as a disulfide-rich peptide with activity against some Gram-positive and Gram-negative bacteria, as well as yeasts.9,10 Further studies demonstrated the role of hepcidin in iron metabolism in mammalian species.1,2,3 Hepcidin binds to the iron exporter ferroportin (Fpn), which is the only known iron exporter in vertebrates. 11 This association induced Fpn internalization and degradation11,12 and inhibition of the iron influx into plasma from the ferroportin-rich tissues. 3 The resulting hypoferremia, considered part of the innate immune system, 9 is a common response to systemic infections or generalized inflammatory disorders, 13 primarily serving as a host defense mechanism limiting iron availability to invading microorganisms.1,13
Many studies have focused on the role of hepcidin in innate immunity and control of iron metabolism mainly during experimental and/or clinical models of acute inflammation. The expression of hepcidin mRNA increased during systemic inflammation induced by LPS in several species.1,2,4,5,7,14 As endotoxins (LPS) play a pivotal role in many pathophysiologic states and diseases in horses, a series of studies has been performed using an inflammation-model with LPS infusion in horses. 15 – 20 Therefore, in the current study we documented the expression of hepcidin mRNA in the equine liver and bone marrow during an experimental model of endotoxemia in horses and, to our knowledge, this is the first report to describe the expression of hepcidin in horses with inflammation experimentally induced by LPS infusion.
Materials and methods
Materials
Animals
All experiments were carried out according to the Unesp-Univ Estadual Paulista,
Institutional Animal Care and Use Committee (108-A/2007). Six adult Quarter horses mares,
not pregnant, 6–9 years old and mean body weight (BW) 425 (± 20) kg, were used in this
study. Mares were housed in individual stalls and received the same food, water
Induction of endotoxemia
Six mares received an intravenous infusion of 30 ng/kg BW of LPS (
Physical examination
A clinician (J.P.O.F.) examined all animals for clinical signs of endotoxemia, such as fever, restlessness, muscle fasciculation, lethargy, yawning, hyperemic mucous membranes, respiratory and heart rates, intestinal motility and behavioral evidence of abdominal pain or discomfort (e.g. pawing, rolling, sweating, looking at flanks) during the study.17,18,20 The physical charts ofall studied horses classified the clinical signs according to a previously standardized clinical score (CS) that included 4 grades: 0 = normal, 1 = mild response, 2 = moderate response, and 3 = severe response.17,18,20 These assessments were performed as follows: at baseline (sampling time: 0 h immediately before LPS infusion), 2, 4, 6, 8, 10, 12, 16, 18, 20, 24, 30, 36, 42, 48, 60, 72, 96, 120, 144, 168, 192, 216 and 240 h PI.
Samples collection
Blood samples were collected after physical examination into vacuum tubes with sodium heparin (iron analysis) (BD Vacutainer®, Franklin Lakes, NJ, USA) and tubes with EDTA [BD Vacutainer®; white blood cell counts (WBC), PVC and plasma fibrinogen concentration]. The WBCs were performed at 0 h before and at 6, 24, 48, 72, 168, and 240 h after LPS infusion. Samples were kept on ice until processing.
Based on prior studies in humans 5 and rats 7 , liver and bone marrow biopsies were performed immediately prior to LPS infusion (0 h, baseline) and at 6 and 18 h after challenge with LPS. Liver biopsies were obtained from the right, 14th intercostal space in a line drawn from the point of the shoulder to the tuber coxae. Before taking the biopsies, a 25 cm 2 area was shaved and disinfected with povidone-iodine 10%. All animals were sedated with 1 mg/kg BW of xylazine hydrocloride and 2 ml local anaesthesia (2% lidocaine). Liver biopsies were taken using a 14 G Tru-cut biopsy needle (Biomedical®, Florence, Italy). Immediately after harvesting, all samples were frozen in liquid nitrogen and stored at −80°C until processing. Bone marrow biopsies were performed immediately after liver biopsies. The sternal region was shaved and disinfected with povidone-iodine 10%, and local anaesthesia was performed as previously described. Bone marrow biopsies were taken using an 11 G × 5 cm bone marrow needle (LangMed®, Rio de Janeiro, Brazil). Needle and stylet were inserted through the skin, periosteum, cortex and advanced into the marrow cavity. The stylet was removed and a syringe with heparin (5000 IU) was attached and bone marrow samples were obtained. Samples were kept on ice until processing.
Blood analyses
Heparinized blood samples were centrifuged and plasma was obtained and stored at −80°C until analysis. Plasma was thawed and iron determination was performed in duplicate using a semi-automated chemistry analyzer SB-190 (Celm®, Barueri, Brazil) with the Ferro Cromazurol reagent. Blood samples with EDTA were processed immediately after blood collection. The PCV was determined by centrifugation using the microhematocrit method and plasma fibrinogen concentration was determined by a heat-precipitation refractometry method. 23 An automated hematology analyzer Hemascreen® 18 (Ebran®, Sao Paulo Brazil) was used to perform WBC.
RNA purification, reverse transcription and q-RT-PCR
Total RNA was isolated from the liver and bone marrow samples using the RNeasy® Mini Kit and QIAamp® RNA Blood Mini Kit, respectively, following the manufacturer’s instructions. The relative purity and quality of the isolated RNA was determined by Nanodrop® 2000 Spectrophotometer (Thermo ScientificTM, Wilmington, DE, USA) and the ratio of A260–A280 nm exceeded 1.8 for all preparations. To ensure the complete removal of traces of genomic DNA, 1 µg of total RNA was incubated with RQ1 RNase-Free DNase. First-strand cDNA synthesis was performed with 500 ng of total RNA per 60 µl of reaction using random hexamers and ImProm-IITM Reverse Transcription System, following the manufacturer’s instructions.
Primer sets used in the qRT-PCR amplifying an 81 bp (base pair) fragment of the equine
hepcidin mRNA (GQ_253624) were an 81 bp fragment of the equine IL-6 mRNA (ECU_64794) and
an 86 bp fragment of the
Isolation of PBMCs
Peripheral blood from four Quarter horse mares determined to be healthy by the methods
described previously, was used in the
Challenge of monocyte cultures with LPS
The mononuclear cell suspension (4 × 106 cells/ml) was added to each 40 mm
well of a sterile, 24-well polystyrene plate and incubated for 2 h at 37°C in a 5%
CO2 atmosphere. After incubation, non-adherent cells were gently removed by
washing the plates three times with 37°C RPMI-1640 medium. After adherence, monocyte
cultures were supplemented with 1 ml of the complete tissue culture medium (CTCM;
RPMI-1640 medium containing 2 mM L-glutamine, 10% heat-inactivated bovine serum, 20 mM
HEPES and 40 µg/ml gentamicin) and incubated for up to 18 h at 37°C, in a 5%
CO2 atmosphere with 1 µg/ml of the same LPS used in the
RNA purification, reverse transcription and q-RT-PCR were performed as described above in order to measure the relative quantification of hepcidin and IL-6 mRNA in LPS-stimulated equine monocytes.
Statistical analysis
Statistical analysis was performed using repeated measures of ANOVA and the Tukey
post-test for all parameters. The statistical analysis was performed with SAS software 9.2
(SAS Institute, Cary, NC) to determine the statistical significance between the means of
multiple time-points measured and the mean of baseline values. Baseline values refer to
the values before endotoxin infusion, with each animal serving as its own control in this
respect. A
Results
Clinical data
In response to the endotoxin infusion, all horses were depressed, showed muscle
fasciculations, intestinal hypomotility and had evidence of mild-to-moderate abdominal
pain, such as pawing, sweating and looking at flanks. Except for one horse, all the other
horses became anorectic within 1 h after endotoxin infusion had been stopped. Horses had a
significant increase in clinical score from 1 to 6 h ( Mean clinical score (A), rectal temperature (B),
heart rate (C) and respiratory rate (D) of the horses given intravenous infusion of
30 ng/kg LPS (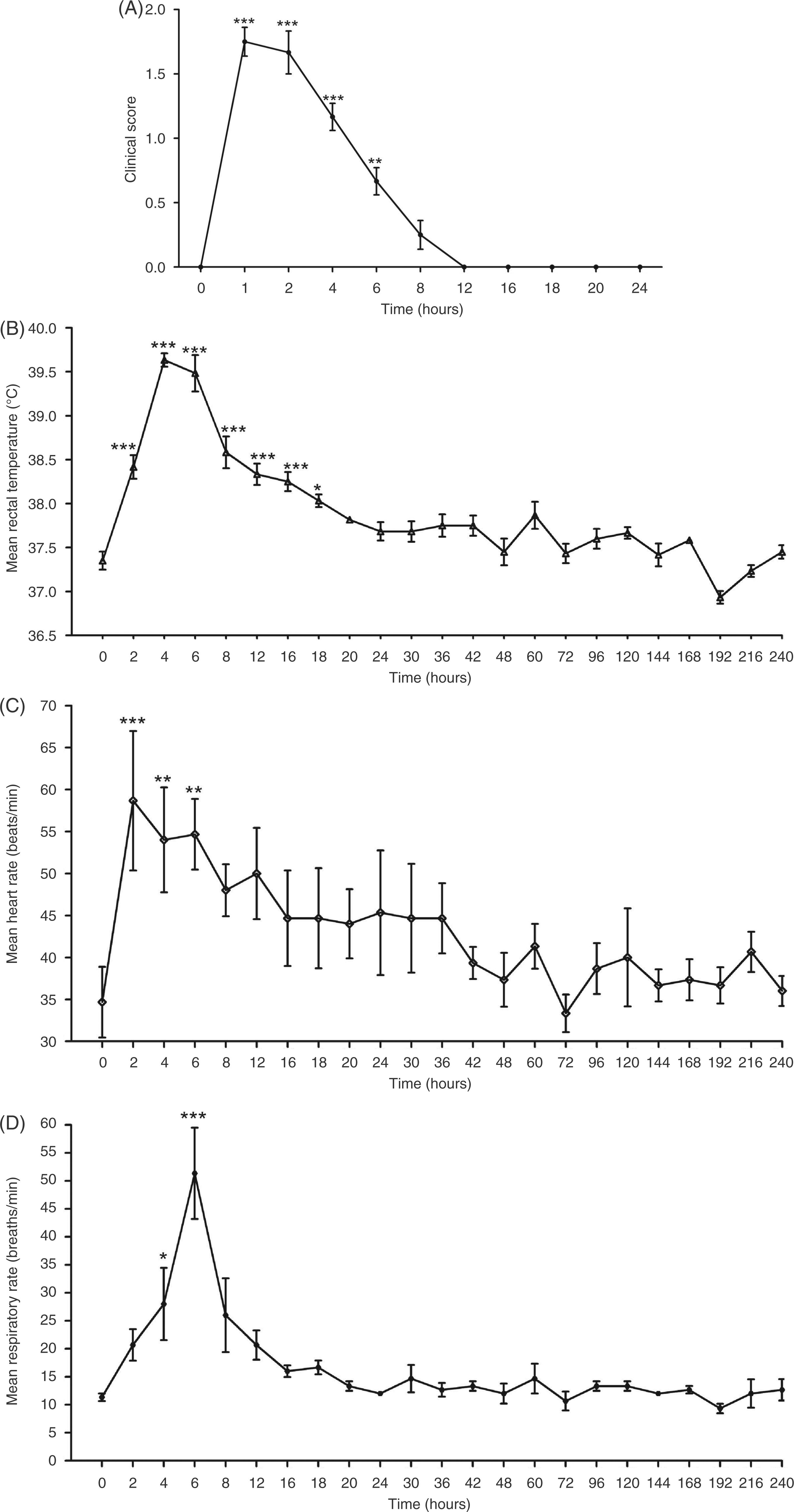
Compared with baseline values, rectal temperature was statistically higher from 2 to 18 h
PI (
Time-course analysis of hematological variables in horses injected with LPS
Mean WBC was significantly higher than control time at 24 ( Mean
white blood cells counts (WBC), neutrophil and lymphocyte counts (A) and plasma iron
concentration (B) of the horses given of 30 ng/kg LPS IV (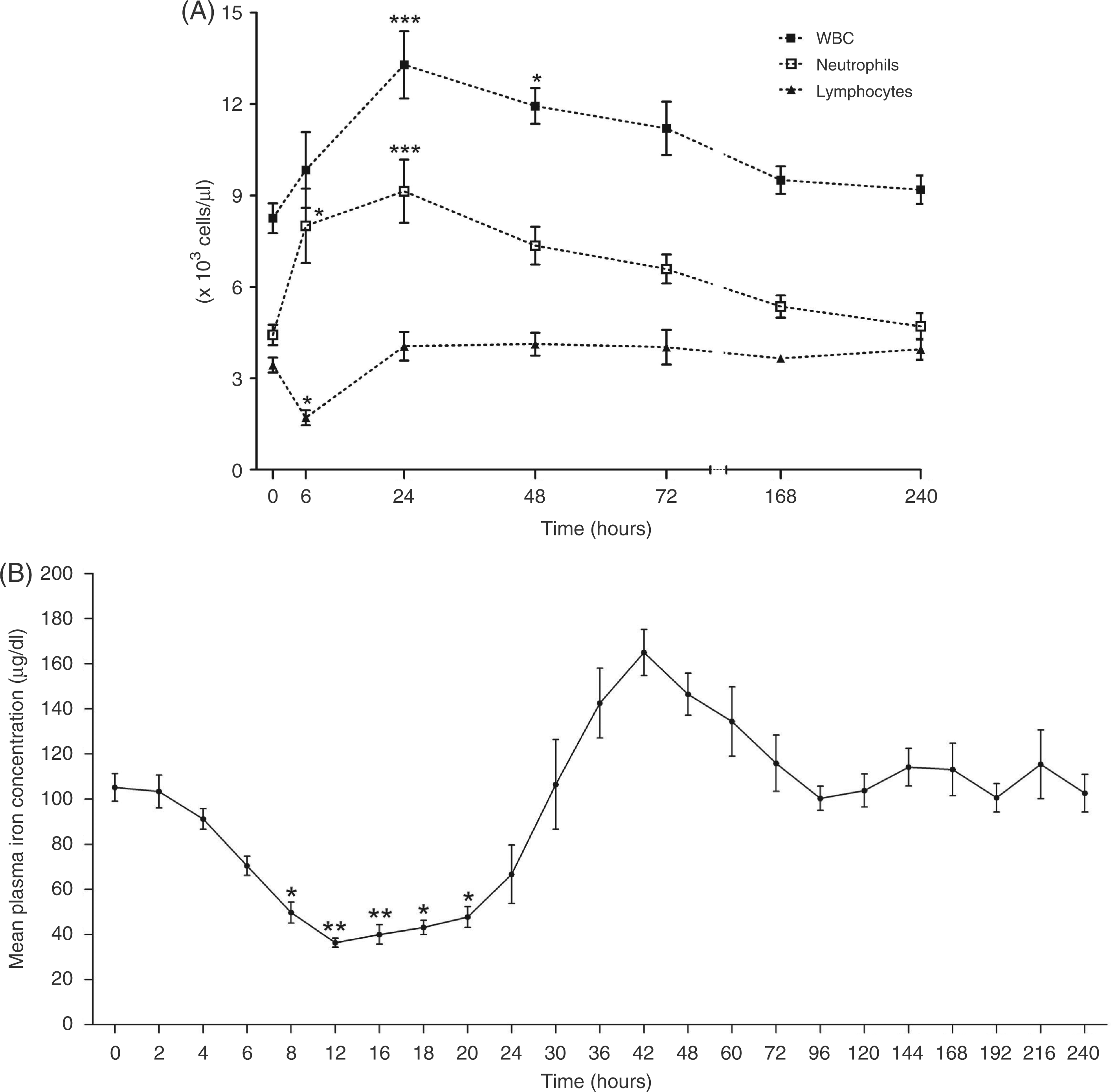
Plasma iron concentration was significantly decreased from the pre-infusion LPS level
from 8 to 20 h PI (
Hepcidin mRNA expression induced by LPS
The main goal of this study was to investigate the effects of LPS on the expression of
hepatic hepcidin mRNA expression. Six h after the end of LPS infusion, hepcidin mRNA
expression increased 7.8 times ( Relative
hepcidin (A) and Il-6 (B) gene expression over time following intravenous infusion
of 30 ng/kg LPS (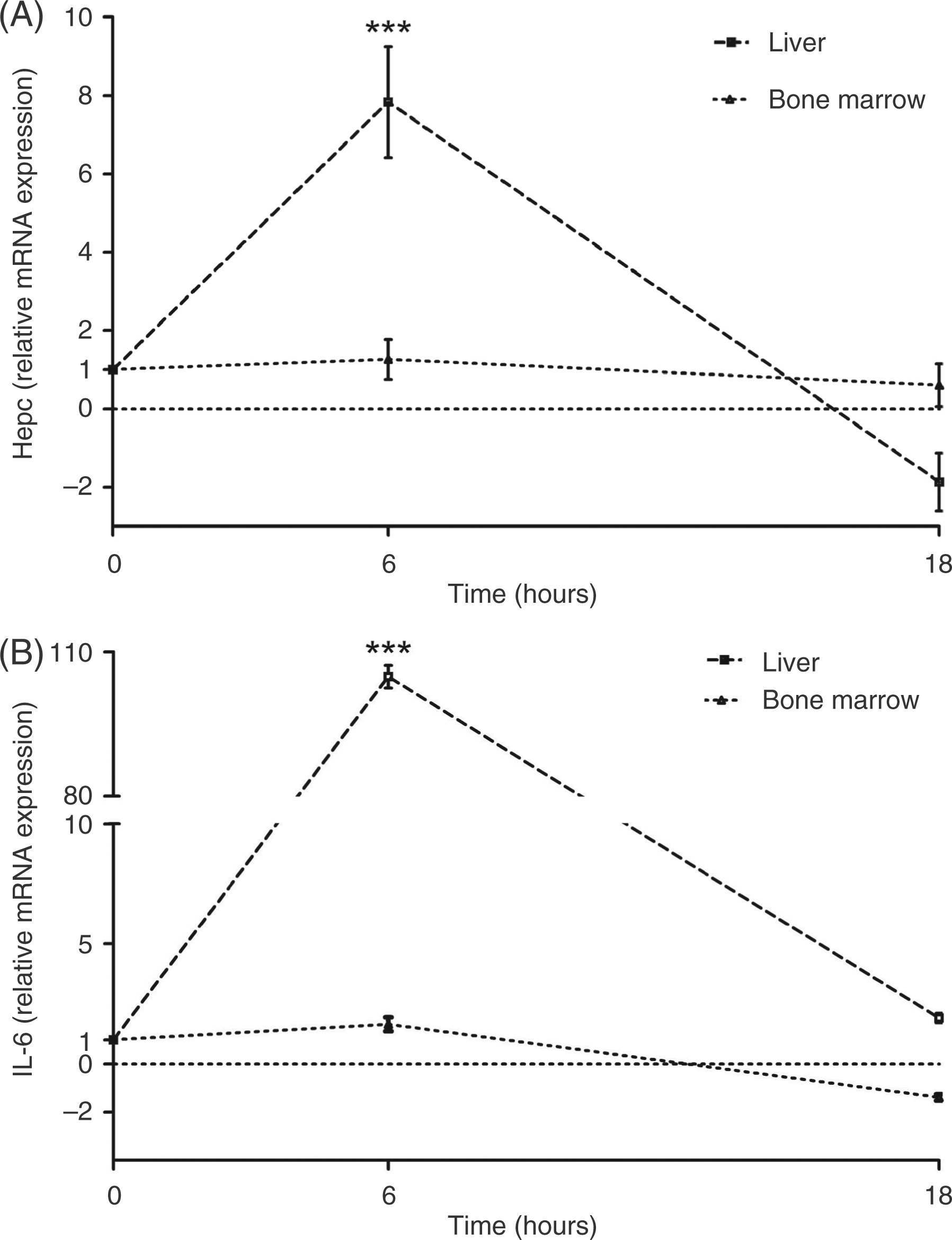
We also evaluated hepcidin and IL-6 mRNA expression in LPS-stimulated equine monocytes.
We observed that hepcidin ( Relative hepcidin (A) and Il-6 (B) gene expression
LPS-stimulated equine monocytes (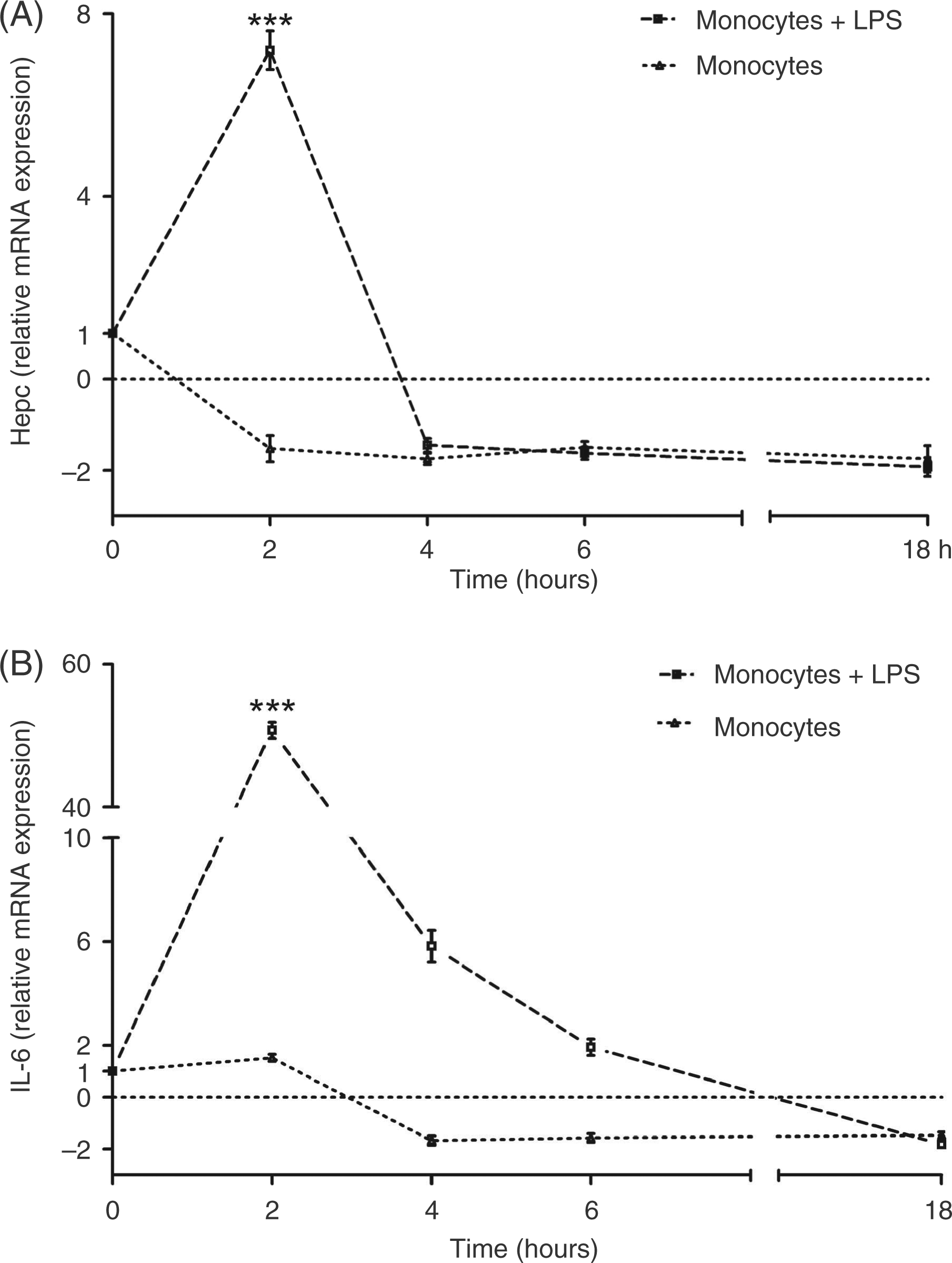
Hepcidin and IL-6 expression in monocyte cultures incubated for 2, 4, 6 and 18 h in CTCM without LPS was biologically insignificant (<two-fold).
Discussion
We previously showed that hepcidin mRNA is highly expressed in liver of the healthy horses. 8 To verify the role of hepcidin in equine iron metabolism, we investigated hepcidin mRNA expression in horses with experimental systemic inflammation induced by LPS infusion. Endotoxin concentrations were not measured in this study; however, it has been previously shown that endotoxemia develops in horses injected with a similar dose of LPS.16,17 The LPS-model of inflammation used here promoted a reliable and consistent inflammatory response, as all horses developed signs of endotoxemia, as indicated by tachycardia, tachypnea, muscle fasciculation, abdominal pain, depression and the cessation of intestinal sounds. These results were similar to previous reports that described clinical findings in horses receiving different concentrations of LPS infusion.17,18,20,22,26 Fever of up to 39.5°C observed in all horses at 4 h after LPS induction has been considered as a characteristic response of horses to LPS challenge16,18,20,27 and provides evidence of systemic inflammation. 28 This increase in body temperature was previously described in response to LPS and is induced by cytokines, mainly TNF-α, IL-1 and IL-6.16,17,29
Herein, WBC counts were evaluated 6 h after LPS infusion, and a significant increase of neutrophils, significant decrease of lymphocytes and a significant increase of WBC relative to baseline values were observed. At 24 h after LPS infusion, marked leukocytosis and neutrophilia were observed. After 48 h, WBC count, neutrophil and lymphocyte count were within the normal range in all horses. 25 These findings are consistent with those of previous studies,17,18,22 which observed leukocytosis and neutrophilia at 6 h after LPS infusion. Neutropenia has been described prior to 6 h in other studies, but WBC counts were not performed prior to 6 h PI in our study. The lymphopenia observed in this study was also observed in horses and in other species during endotoxemia 29 – 31 and is probably caused by a decrease in lymphoid cell production by bone marrow or as a result of migration of activated lymphocytes to the lymphatic tissue. 20
The concentrations of fibrinogen and iron have been used to diagnose and monitor
inflammatory disease in horses;
28
both were considered acute-phase reactants.
30
, The concentration of plasma
fibrinogen increased significantly 12 h after intravenous,
27
and 24 h after intra-peritoneal, infusion
of LPS in horses.
30
Peritoneal fibrinogen also increased significantly after intra-peritoneal infusion of LPS in
horses,
20
indicating an increased inflammatory response to LPS; however, in the current study, no
significant increase in plasma fibrinogen concentration was seen at any of the time- points
measured. On the other hand, the hypoferremia peak (12 h PI) was a sensitive indicator of
acute inflammation compared with the fibrinogen concentration in horses with endotoxemia
induced by LPS. A previous, retrospective study comparing serum iron and plasma fibrinogen
in horses with naturally acquired inflammation demonstrated that hypoferremia was a better
indicator of acute, sub-acute and chronic systemic inflammation than fibrinogen
concentration and that iron concentration may be preferable for detecting natural acute
inflammation in horses.
28
Hypoferremia has also been observed in other inflammation models using an
injection of Freund’s complete adjuvant or turpentine oil in horses.
32
–
34
Horses submitted to
experimental inflammation in a model using
Persistent hypoferremia has been observed during inflammatory processes and is a cause of anemia of chronic disease in mammalian species.1,14 In the present study, hypoferremia was rapid and transitory and did not cause anemia of inflammation in horses. In one previous report, no changes were observed in the PCV values of horses given LPS (30 ng/kg BW of LPS, IV) when compared with controls during the 8 h measured after LPS infusion. 16 These findings were similar to those reported herein, but are not in agreement with a study in horses where the mean PCV value increased significantly 1 h after LPS infusion (200 ng/kg BW diluted in 1 l of 0.9% NaCl administered over 30 min) and remained elevated throughout the experiment period (6 h), 18 or other studies of equine endotoxemia that verified that PCV increased above baseline values during endotoxemia in horses.15,36
Cytokines are an integral component of the adaptive and innate immune responses. Three of
the most intensively studied cytokines, IL-1, IL-6 and TNF-α share common actions in the
host defense against invading pathogens. An
In addition, IL-6 release was dramatically induced within 3 h after LPS injection and
urinary hepcidin peaked within 6 h, followed by a significant decrease in serum iron in
humans.
5
Therefore,
the increase of the hepatic hepcidin mRNA expression observed in horses 6 h after LPS
infusion may have been stimulated by IL-6, as describe in other species.5,7,13,37 In rats, the marked increase (9.5-fold) in
the expression of liver hepcidin mRNA occurred 4 h after LPS injection, while the
hypoferremia peak was observed 2 h later.
7
The decrease in hepcidin mRNA to levels
lower than in controls at 24 h after LPS administration in the horses is similar to what has
previously been reported in rats.
7
The expression of hepcidin mRNA increased significantly in the bone
marrow of swine challenged with
In summary, to our knowledge, this report is the first to describe the effects of LPS infusion on the expression of hepcidin mRNA in horses. We conclude that LPS infusion up-regulated hepatic hepcidin mRNA expression causing rapid hypoferremia. This suggests that hepcidin plays a key role in iron metabolism and the innate immune system of horses. In addition, plasma iron concentration was a sensitive indicator of clinically apparent endotoxemia in the horses. Studies have shown that LPS infusion up-regulates gene expression of IL-6 in horses 19 and that hepcidin mRNA is induced by IL-6 in humans and rats.5,7,14 Therefore, we speculate that similar to those species, hepcidin could act as a type II acute-phase protein in horses. Further studies are required to fully understand the effects of endotoxins on the expression of hepcidin and to confirm the role of cytokines in the up-regulation of hepcidin expression in horses.
Footnotes
Acknowledgements
This study was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo - FAPESP (07/07344-6, 07/05008-9). The last author has a research grant from Conselho Nacional de Desenvolvimento Científico e Tecnológico CNPq. The authors acknowledges Dr. Ana Paula Bordon Graciani for helping with monocyte culture procedure.
Conflict of interest
The authors have no financial conflict of interest.
