Abstract
The mouse monoclonal antibody (mAb) WN1 222-5 recognizes a carbohydrate epitope in the inner core region of LPS that is shared by all strains of Escherichia coli and Salmonella enterica and is able to neutralize their endotoxic activity in vitro and in vivo. Immunization of mice with mAb WN1 222-5 yielded several anti-idiotypic mAbs one of which (mAb S81-19) competitively inhibited binding of mAb WN1 222-5 to E. coli and Salmonella LPS. After immunization of rabbits with mAb S81-19, the serological responses towards LPS were characterized at intervals over two years. Whereas the serological response against the anti-idiotype developed as expected, the anti-anti-idiotypic response against LPS developed slowly and antibodies appeared after 200 d that bound to E. coli LPS of the R3 core-type and neutralized its TNF-α inducing capacity for human peripheral mononuclear cells. We describe the generation of a novel anti-idiotypic antibody that can induce LPS core-reactive antibodies upon immunization in rabbits and show that it is possible, in principle, to obtain LPS neutralizing antibodies by anti-idiotypic immunization against the mAb WN1 222-5. The mimicked epitope likely shares common determinants with the WN1 222-5 epitope, yet differences with respect to either affinity or specificity do exist, as binding to smaller oligosaccharides of the inner core was not observed.
Introduction
Infections frequently cause a systemic inflammatory response syndrome called sepsis triggered by stimuli of innate immune responses.1,2 The recognition of LPS (endotoxin) of Gram-negative bacteria is by far the most potent stimulus1,3– 5 and, therefore, neutralization of LPS for the treatment of sepsis has been the subject of intensive research for decades.1,2,6
Lipopolysaccharides are glycophospholipids constituting the outer leaflet of the outer membrane and in wild-type bacteria such as Escherichia, Salmonella, Klebsiella and Pseudomonas, consist of a lipid A, a core oligosaccharide (core-OS) and an O-polysaccharide (O-PS). 7 The membrane embedded lipid A is the endotoxic principle of LPS. 8 The O-PS is structurally very heterogeneous giving rise to a large number of serotypes within a species. 7
Enterobacteria contain a second outer membrane polysaccharide called the enterobacterial common antigen (ECA). The polysaccharide of ECA consists of trisaccharide repeating units that are membrane-anchored by either phosphoglycerolipids (ECAPG) or the LPS core-OS (ECALPS). 9 – 11 A soluble cyclic form has also been discovered10,12 and its biosynthesis investigated.13,14 Antibodies that are specific for the polysaccharide domain of ECA cross-react between different enterobacteria but clinical trials have failed to show a protective effect against sepsis. 15
In contrast, antibodies against the O-PS are generally protective against a homologous strain in animal models of septic shock and, therefore, antibody therapy is regarded as a valid concept against Gram-negative sepsis, in principle.
16
However, translation into clinical application is precluded by the high specificity for a single serotype. A broader medical application would only be achievable with cross-reactive antibodies neutralizing LPS of the most clinically prevalent, if not all O-serotypes. Several antibodies to lipid A or inner parts of the core-region have also been reported to protect animals in models of sepsis. Their epitopes have been claimed to be 3-deoxy-α-
The mAb WN1 222-5 is unique in its ability to bind E. coli LPS and bacteria of a large collection of blood, fecal and urinary isolates independently of the serotype. 22 Importantly, this antibody also neutralizes LPS toxicity in vitro and is protective in various in vivo models of endotoxic shock. 22 – 24 Using whole LPS and a number of neoglycoconjugates containing core-OS from all E. coli core types (R1 to R4 and K-12), Salmonella enterica and the mutant strain E. coli J-5, we have identified parts of an epitope important for high-affinity binding of the neutralizing mAb WN1 222-5. 25 Immunization of mice and rabbits with such neoglycoconjugates did not lead to the formation of mAb WN1 222-5-type antibodies (unpublished observation), indicating the recognition of a conformational epitope and reflecting the difficulties generally encountered to obtain such cross-reactive antibodies.
It has been observed that antibodies (Ab2, anti-id) that are specific for another antibody's (Ab1, idiotype) combining site may induce antibodies (Ab3, anti-anti-id) upon immunization that resemble Ab1 in terms of specificity. 26 Thus, if Ab1 was a carbohydrate-specific antibody, it is possible, in principle, to achieve a structural or functional mimicry of carbohydrates by antibodies. Such an anti-idiotypic mimicry of carbohydrates has been reported, including successful production of antibodies that cross-react with the original antigen. 27 – 29 A structural investigation has revealed the molecular basis of such mimicry and that functional mimicry can be achieved even if only a subset of contact residues at the combining site are retained between the different interactions. 30 – 32 This concept has been transferred even to peptides and such mimics, called mimotopes, can be used in vaccines to achieve T-cell help in the generation of carbohydrate-specific antibodies. 33 – 40 The immunization with anti-idiotypic antibodies has been reported to induce a protective immune response also against LPS. 41 – 45
The potential of anti-carbohydrate antibodies to serve as template immunogens for the generation of anti-carbohydrate antibodies in principle led us to investigate whether this approach could be successfully exploited for the generation of LPS- neutralizing antibodies and be used to overcome the general difficulty in obtaining cross-reactive antibodies by immunization with core-OS. As mAb WN1 222-5 is so far the only example combining these properties, we have chosen it to immunize mice to generate an anti-idiotypic antibody and characterized the anti-anti-idiotypic response in rabbits. Such an approach may also be important for resolving non-typhoidal salmonellosis, which is a serious health threat for human immunodeficiency virus (HIV)-infected individuals.46,47 Vaccination against LPS has been suggested as an option for treatment; 48 however, aberrant formation of antibodies against the O-PS of S. enterica has been reported to prevent binding of protective antibodies and neutralization of the bactericidal activity of serum. 49 This may be overcome by redirecting the immune response towards the core-OS.
Materials and methods
Bacterial LPS and LPS-conjugates
Lipopolysaccharides of E. coli wild-type strains O4, O6, O12, O15, O18 and O111 were prepared by phenol-water extraction; LPS of S. enterica sv. Minnesota (S. minnesota) strain R60 (rough mutant containing the complete core) and E. coli rough mutants of core type R1 (strain F470), R2 (strain F576), R3 (strain F653), R4 (strain F2513) and K-12 (strain 3110) were obtained by extraction of bacteria with phenol/chloroform/light petroleum ether and the deacylated LPS (LPSdeac) after partial chemical degradation as described. 50 – 52 Rough LPSdeac was conjugated to bovine serum albumin (BSA) as described. 53 In addition, mutants of the E. coli strains were constructed which were unable to synthesize the ECA; details will be described elsewhere. These mutants were used for SDS-PAGE and Western blots. Klebsiella pneumoniae strain i28/94 and the ECA-specific mAb 898 were kind gifts of D. S. Hansen (Statens Serum Institute, Copenhagen, Denmark) and Prof. D. Bitter-Suermann (Institute of Medical Microbiology, Hannover, Germany), respectively.
Immunization of mice with WN1 222-5
The mouse mAb WN1 222-5 recognizes an epitope in the inner core region of S. enterica and all E. coli core types, independent of whether an O-antigen is present or not. 22 BALB/c mice were immunized with this mAb by the following two protocols. Firstly, three mice were injected i.p. on day 0 with native mAb (150 µg in 150 µl of PBS) emulsified with an equal volume of Freund's complete adjuvant. On day 21, another 150 µg of mAb WN1 222-5 in 150 µl of phosphate buffered saline (PBS) containing as a cross-linking reagent 0.00625% of glutardialdehyde (WN1 222-5-GA) were emulsified with an equal volume of Freund's incomplete adjuvant and injected subcutaneously (s.c.) at four different sites. 54 On day 42 and 63, one mouse received 150 µg of WN1 222-5-GA i.p. and was boosted on day 137 i.v. with 30 µg of WN1 222-5 F(ab)2 fragments in 200 µl of PBS. The mouse was boosted again on days 176 (i.v.), 177 (i.p.) and 178 (i.p.) with 30 µg each of WN1 222-5 F(ab)2 fragments in 200 µl of PBS. Two days after the last injection, the animal was exsanguinated and the spleen was removed for fusion (number S81).
In the second protocol, only native protein without cross-linker was used following a published protocol. 55 Three BALB/c mice were injected on day 0 s.c. at four different sites with mAb WN1 222-5 (50 µg in 125 µl of PBS) emulsified with an equal volume of Freund's complete adjuvant. On day 28, another 50 µg of mAb WN1 222-5 in 100 µl of PBS emulsified with an equal volume of Freund's incomplete adjuvant were injected i.p. One mouse was boosted with 30 µg each of WN1 222-5 F(ab)2 in 200 µl of PBS on days 130 (i.v.), 238 (i.v.), 239 (i.p.) and 240 (i.p.). Two days after the last injection, the animal was exsanguinated and the spleen removed for fusion (number S83).
Fusion and hybridoma production
Spleen cells were isolated and fused with X63 Ag8.65 myeloma cells in a ratio of 2:1 according to conventional protocols. After selection in HAT medium, culture supernatants were tested for reactivity with WN1 222-5 F(ab)2 by enzyme-linked immunosorbent assay (ELISA; see below). From fusion S81, two mAbs were obtained, S81-10 and S81-19; from fusion S83 three mAbs were obtained, S83-1-16, S83-1-23 and S83-11. Monoclonal antibodies were cloned three times by limiting dilution and isotyped using a commercial isotyping kit (Bio-Rad, Hercules, CA, USA). Purification was achieved by affinity chromatography using protein G (Amersham, Biosciences, Freiburg i. Br., Germany) or protein L (Bind™ L, Fluka, Buchs, Switzerland).
Isolation of F(ab)2 fragments
The F(ab)2 of mAb WN1 222-5 (IgG2a) were prepared as described.
56
Briefly, mAb WN1 222-5 (0.6 mg/ml in 0.1
Enzyme-linked immunosorbent assay with F(ab)2
Using ELISA, we determined the titer of S81-19 against WN1 222-5 and of rabbit anti-anti-idiotypic antisera against S81-19. Of these mAbs F(ab)2, fragments were coated (100 and 200 ng/well) at 4°C for 16 h in carbonate buffer (50 m
After three washings, 50 µl Fc-specific horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG or goat-anti-rabbit IgG (both from Dianova, Hamburg, Germany) diluted in PBS–CB 1:1000 and 1:2000, respectively, was added and incubation continued for 1 h at 37°C. Goat anti-mouse IgG was detected by addition of 100 µl of aqueous ready-to-use solution of 3,3′,5,5′-tetramethylbenzidine (TMB substrate, Uptima Interchim, Monluçon, France) after three washings with PBS. The reaction was stopped after 30 min at 37°C by the addition of 100 µl of 1
For detection of goat-anti-rabbit IgG azino-di-3-ethyl-benzthiazolinsulfonic acid (ABTS) substrate instead of TMB was used. The substrate solution was freshly prepared by dissolving ABTS in 100 m
Enzyme-linked immunosorbent assay with LPS
The titers of rabbit anti-anti-idiotypic antisera (diluted 1:100 in PBS-C) against LPS were determined by ELISA. Microtiter polyvinyl plates (96-well, Falcon 3911; Becton Dickinson, Heidelberg, Germany) were coated for 16 h at 4°C with 250 ng/well of LPS diluted in PBS, pH 7.2. The coated plates were washed four times with PBS and blocked for 1 h with PBS-C under gentle agitation at 37°C. The remaining steps were as described above, omitting BSA from all buffers and a solution of ABTS/H2O2 in citrate buffer as substrate, but the HRP-conjugated goat anti-rabbit IgG was diluted 1:500 in PBS-C in this assay.
Biotinylation of mAb WN1 222-5 and mAb S81-19
Sulfo-succinimidyl-6-(biotinamido) hexanoate (NHS-LC biotin, Pierce, Rockford, IL, USA) was dissolved at a concentration of 5 mg/ml in DMSO and added to a 1 mg/ml solution of mAb WN1 222-5 in 50 m
Monoclonal Ab S81-19 was biotinylated using non-spacered NHS-Biotin (Pierce). The NHS-biotin was dissolved in DMSO (5 mg/ml) and 13 µl were added to 450 µl of a 0.9 mg/ml mAb S81-19 solution (molar ratio 1:50) followed by incubation for 1 h at 20-22°C and dialysis as described above. The activity of the biotinylated mAb was confirmed by ELISA with F(ab)2 of WN1 222-5 as antigen using anti-IgG (Fc) as secondary antibody (1:500). The titers were identical to non-biotinylated mAb.
Inhibition of mAb S81-19 binding to immobilized WN1 222-5 by LPS
To study whether LPS and mAb S81-19 bind competitively to WN1 222-5 we have performed competition ELISA in which the plates (Maxisorb, Becton Dickinson) were coated with F(ab)2 of WN1 222-5 (300 ng/cup in PBS) over 2 d at 4°C. The coated plates were washed four times with PBS and then blocked for 1 h with PBS-C. Twenty µg of LPS in 30 µl PBS-CB were transferred to V-shaped microtiter plates (96-well, Nunc) serially diluted (1:2) and an equal volume of biotinylated mAb S81-19 diluted in the same buffer was added to each cup. The mAb concentration was adjusted to give an OD450 between 0.5 and 1.0 without the addition of inhibitor. Fifty µl of the mixture were transferred to coated ELISA plates and incubation was continued for 1 h at 37°C. After three washings with PBS, HRP-conjugated streptavidin (Dianova) diluted 1:2000 in PBS-CB was added and incubation was continued for 1 h at 37°C. After three washings in PBS the remaining steps were as described using TMB as substrate.
Inhibition of binding of mAb WN1 222-5 to LPS by S81-19
The binding of mAb WN1 222-5 to LPS was competitively inhibited with anti-idiotypic mAb or F(ab)2 thereof. Polyvinyl plates (96-well Falcon 3911, Becton Dickinson) were coated with LPS (100 ng/cup in 50 µl PBS) for 16 h at 4°C. The coated plates were washed 4 times with PBS and then blocked for 1 h with PBS-C. Serial two-fold dilutions of inhibitor protein in PBS-C (30 µl) were mixed in V-shaped microtiter plates (96-well, Nunc) with an equal volume of biotinylated mAb WN1 222-5 diluted in the same buffer. The mAb concentration was adjusted to give an OD405 between 0.5 and 1.0 in ELISA in the absence of inhibitor. Fifty µl of the mixture were transferred to coated ELISA plates and incubation was continued for 1 h at 37°C. After three washings in PBS, HRP-conjugated streptavidin (Dianova) diluted 1:2000 in PBS-C was added and incubation was continued for 1 h at 37°C. After three washings in PBS, the remaining steps were done as described using ABTS as substrate. For testing mAb S81-19 and F(ab)2, the highest inhibitor concentration was 50 n
Inhibition of binding of WN1 222-5 to deactivated lipopolysaccharide (LPSdeac)-BSA glycoconjugates by S81-19
The binding of mAb WN1 222-5 to LPSdeac-BSA glycoconjugates was competitively inhibited with anti-idiotypic mAb. Maxisorb microtiter plates (96-well, U-bottom, Nunc) were coated with LPSdeac-BSA glycoconjugates in carbonate buffer (50 m
Immunization of rabbits with S81-19
Schedule for the immunization of rabbits with anti-idiotypic antibody S81-19. Immunizations were initiated injecting 100 µg of each immunogen followed by injections of 150 µg each at later timepoints. Immunogens were dissolved in 2 ml for the first three immunizations and in 0.5 ml for the final injections and were divided into equal aliquots for the different injection routes

Injection into the inguinal and axillary lymphnodes; bCFA, complete Freund’s adjuvant; IFA, incomplete Freund’s adjuvant.
Isolation of IgG from rabbit sera
Rabbit serum (2 ml) was dialyzed against Tris-HCl buffer (10 m
SDS-PAGE and Western blots
To analyze the reactivity of antisera taken from immunized rabbits on different days in blots, LPS (100 to 200 µg) was applied to one large slot (12.5 cm) and separated by SDS-PAGE on a 5% stacking and 15% separating gel at a constant voltage of 150 V. The gels were transferred for 16 h onto polyvinylidene difluoride (PVDF) membranes (pore size 0.45 µm, Millipore, Schwalbach, Germany) by tank blotting (Bio-Rad). Before use, the membranes were wetted in methanol for 10 s, after which they were washed in distilled water for at least 5 min. Following transfer, the blots were cut into stripes (0.5 cm width) and placed in Mini-incubation trays (Bio-Rad). The following steps were performed at 20–22°C. After blocking in blotting buffer (50 m
For an analysis of LPS from ECA+- and ECA−-strains of E. coli in silver stained gels and the reactivity with mAb 898 or polyclonal anti-anti-idiotypic-antiserum in Western blots, the amount of LPS loaded per lane was 2 µg, 10 µg and 20 µg, respectively. For these experiments the membranes were not cut and AP-conjugated goat-anti mouse IgG (heavy and light chain specific, Dianova, diluted 1:1000) or goat-anti-rabbit IgG was used as secondary antibody, respectively.
Stimulation of human mononuclear cells (MNC) with LPS
Mononuclear cells were isolated from the heparinized blood of healthy adult donors by Ficoll-Isopaque density gradient centrifugation. 59 Isolated MNC were washed in PBS and cultured in RPMI 1640 containing 10% fetal calf serum (FCS), 100 U/ml penicillin and 100 mg/ml streptomycin (Biochrom, Berlin, Germany). Cells [1×106/ml] were stimulated in triplicates with 1 ng/ml of various LPS types. In blocking experiments, isolated IgG of rabbit sera (up to 400 µg/ml) obtained after immunization with anti-idiotypic mAb S81-19 was mixed with LPS before the stimulation of cells for 30 min. After 4 h, supernatants were collected and the content of TNF-α was quantified by ELISA (Biosource).
Results
Generation and characterization of monoclonal anti-idiotypic antibodies against mAb WN1 222-5 in mice
We have compared two protocols for immunizing BALB/c mice with the intention of inducing the formation of anti-idiotypic antibodies against mAb WN1 222-5. In one protocol, a combination of intact Ig and F(ab)2 was used, while in the other the Ig molecules were cross-linked by glutardialdehyde derivatization.
54
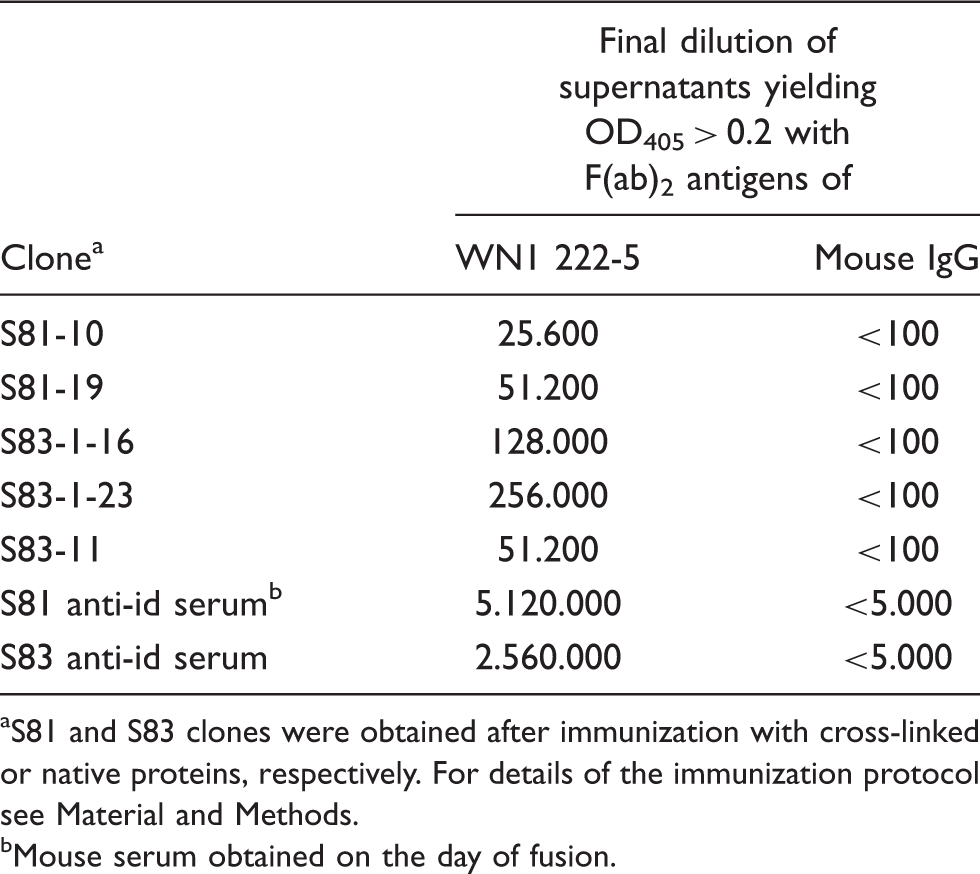
Both immunizations yielded anti-idiotypic antibodies against mAb WN1 222-5 with a titer of several millions in ELISA using immobilized F(ab)2 (Table 2). The titers against normal mouse IgG as antigen were below 5,000. From these mice, five mAb were obtained, two after immunization with cross-linked Ig and three after immunization with the native proteins. The titers of these clones and of the sera taken on the day of fusion determined by ELISA are shown in Table 2. The clone S81-19 (IgG1) bound in ELISA at a very low concentration of 70 p Binding curves of anti-idiotypic mAb S81-19 to immobilized F(ab)2 of mAb WN1 222-5. ELISA plates were coated with 50 (triangles), 200 (circles) or 800 ng/cup (squares) F(ab)2. Each data point represents the average of quadruplicate measurements with confidence values not exceeding 10%. Binding in ELISA of hybridoma supernatants to F(ab)2 of WN1 222-5 and normal mouse IgG S81 and S83 clones were obtained after immunization with cross-linked or native proteins, respectively. For details of the immunization protocol see Material and Methods. Mouse serum obtained on the day of fusion.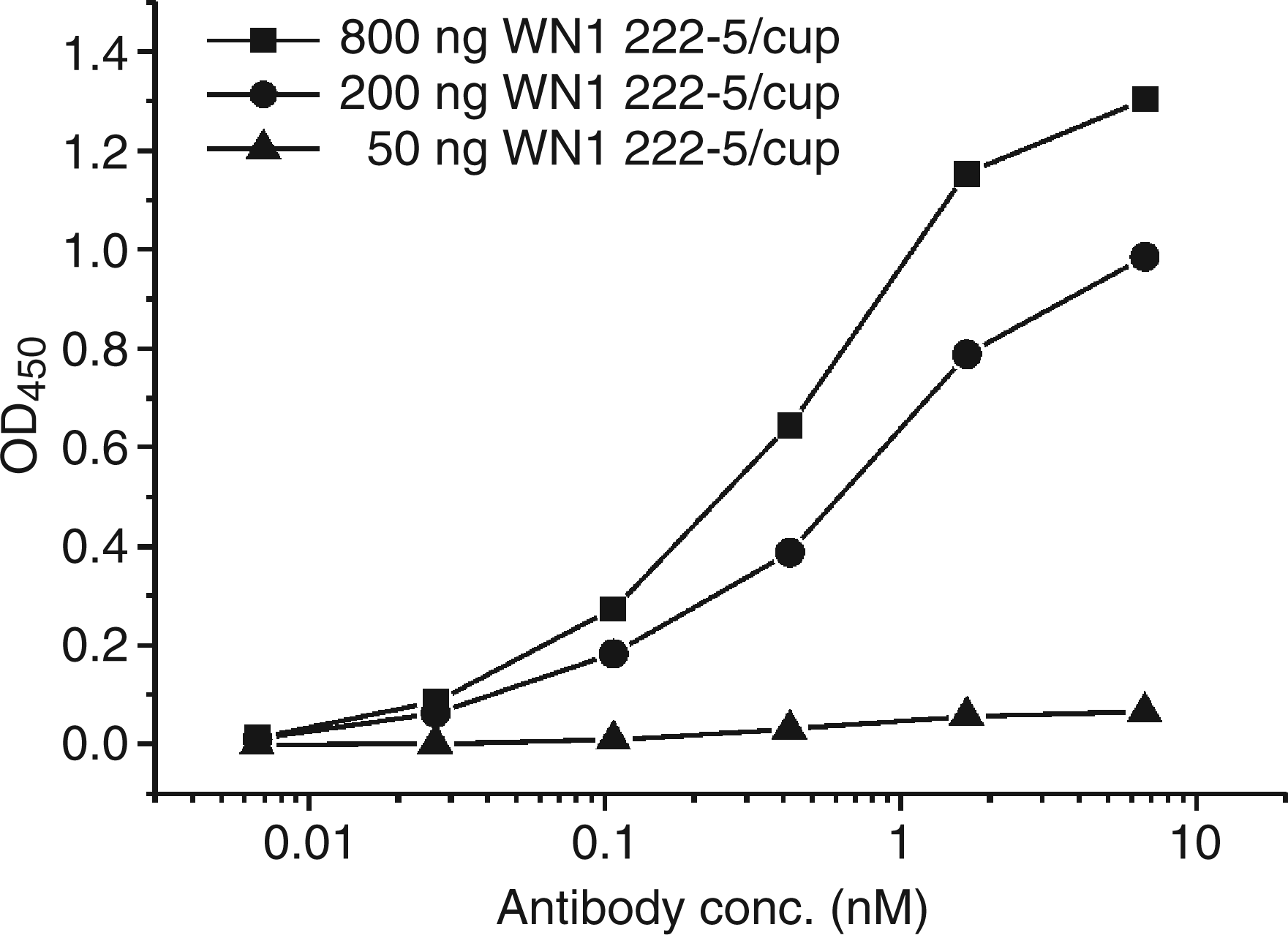
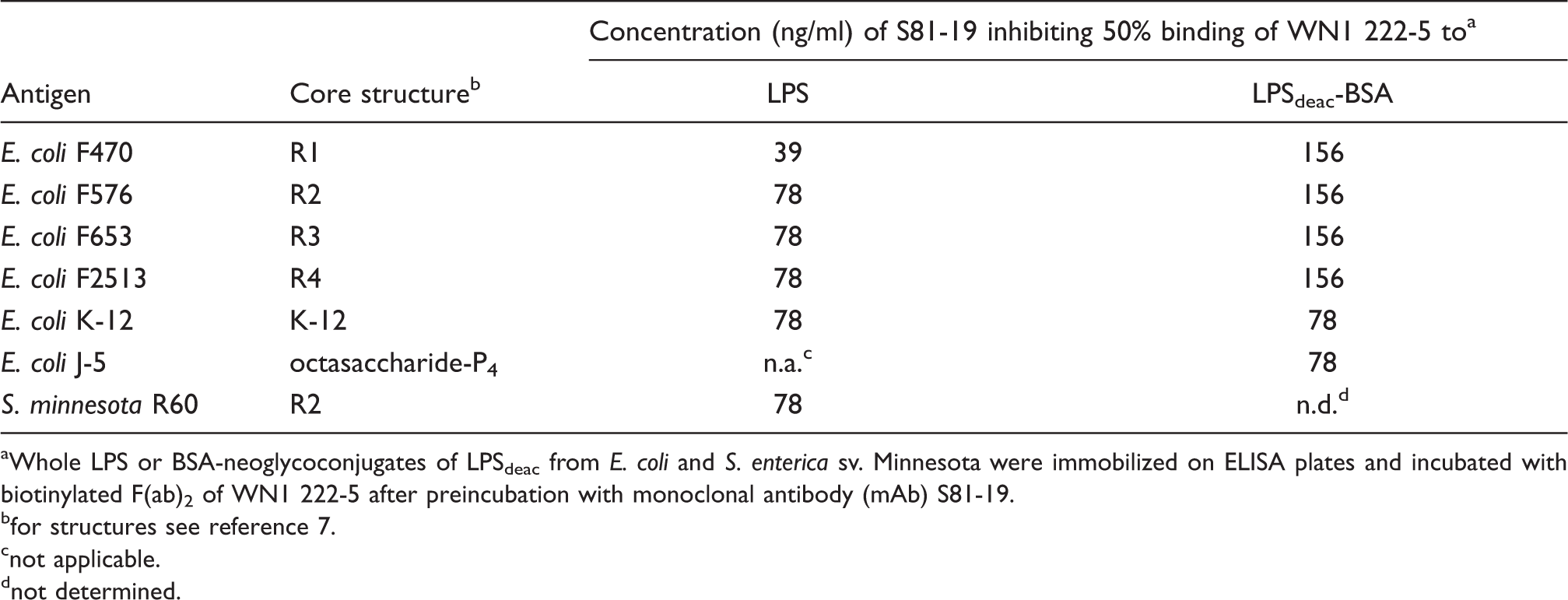
Next, we tested the ability of five anti-idiotypic mAbs to inhibit the binding of mAb WN1 222-5 to LPS ligands. Only mAb S81-19 was able to inhibit the binding to E. coli rough LPS of all core types (R1 to R4 and K-12) and S. minnesota strain R60 (Table 3). The same was observed for neoglycoconjugates containing rough LPSdeac as carbohydrate ligands (Table 3). In this assay, we have also included a neoglycoconjugate of a tetraphosphorylated octasaccharide which we had isolated from the mutant E. coli J-5 and identified as the smallest ligand bound with high affinity by mAb WN1 222-5.
25
The concentrations of S81-19 resulting in 50% inhibition of mAb WN1 222-5 binding ranged between 39 and 156 ng/ml (0.26–1.0 n Inhibition of binding of biotinylated anti-idiotypic mAb S81-19 to immobilized WN1 222-5 F(ab)2 fragments by the following LPS: S. minnesota R595 (Re-LPS, stars), S. minnesota R5 (RcP--LPS, squares), E. coli J-5 (RcP+-LPS, circles), E. coli R1 (diamonds), E. coli R2 (triangles down), E. coli R3 (triangles up) and E. coli R4 (pentagons). Each data point represents the average of triplicate measurements. Inhibition of mAb WN1 222-5 binding to LPS structures by anti-idiotypic mAb S81-19 Whole LPS or BSA-neoglycoconjugates of LPSdeac from E. coli and S. enterica sv. Minnesota were immobilized on ELISA plates and incubated with biotinylated F(ab)2 of WN1 222-5 after preincubation with monoclonal antibody (mAb) S81-19. for structures see reference 7. not applicable. not determined.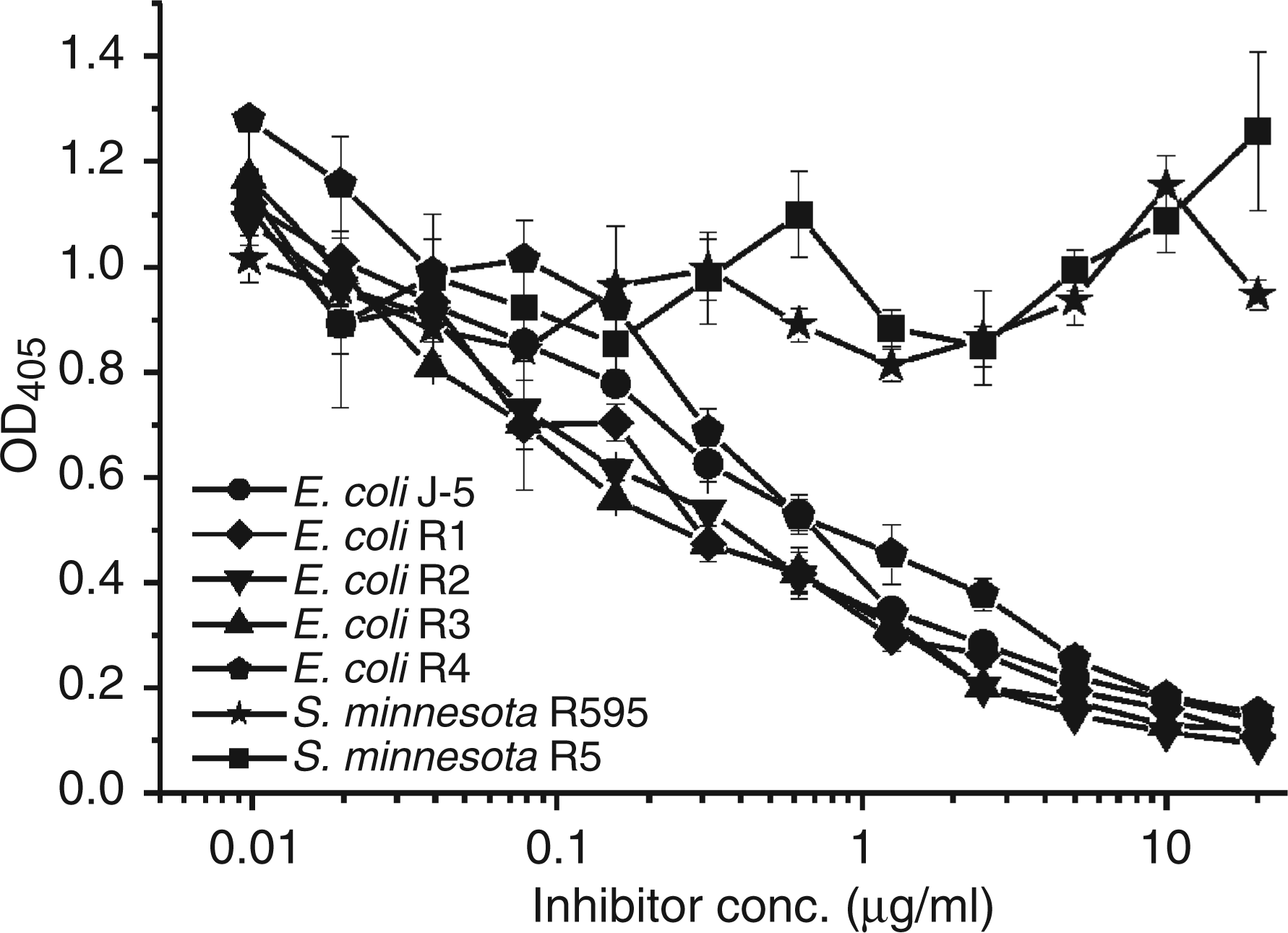
According to these results, S81-19 was considered potentially anti-paratopic (Ab2β) and used for further immunizations aiming at the induction of anti-anti-idiotypic antibodies 26 in rabbits.
Generation and characterization of anti-anti-idiotypic antibodies against mAb S81-19 in rabbits
Immunization of rabbits was achieved with either F(ab)2 alone or by a combination of IgG and F(ab)2 of S81-19. To ensure that pepsin cleavage did not affect the binding site, we compared the ability of mAb and F(ab)2 to inhibit the binding of mAb WN1 222-5 to all E. coli core types (Figure 3). Both preparations yielded identical inhibition curves. After immunization of four rabbits (K436-439) with S81-19 F(ab)2, the immune response was then followed over 728 d by taking serum samples of each individual animal at 12 time points and analyzing the reactivity in ELISA and blots. In dot blots, all different core types were stained (data not shown) whereas in Western blots, i.e. after separation by SDS-PAGE, only the core-OS of the R3-type was stained, in addition to a high molecular mass polysaccharide with a typical banding pattern (Figure 4) in all core types apart from the E. coli R3 LPS. This polymer was identified as ECA using the anti-ECA mAb 898 (Figure 4).
61
As this reactivity was observed for E. coli but not for an ECA-containing LPS preparation of K. pneumoniae i28/94, the antiserum did not contain antibodies against the ECA but against an epitope composed of the E. coli core with attached ECA.
Comparison of S81-19 mAb (open symbols, dashed lines) and F(ab)2 (filled symbols, straight lines) in their ability to inhibit the binding of biotinylated WN1-222-5 to E. coli R1 (squares), R2 (circles), R3 (stars), R4 (diamonds) and K-12 (hexagons) LPS. Each data point represents the average of triplicate measurements. Western blot after SDS-PAGE of E. coli LPS (20 µg each) strains F470 (R1), F576 (R2) F653 (R3), F2513 (R4), and W3110 (K-12) applied to lanes 1 to 5, respectively, and K. pneumoniae i28/94 (200 µg) applied to lanes 6 and 7. Lanes 1 to 6 were immunostained with serum (diluted 1:100) of rabbit 437 taken 576 d after the first injection and lane 7 was stained with 5 µg/ml ECA-specific mAb 898.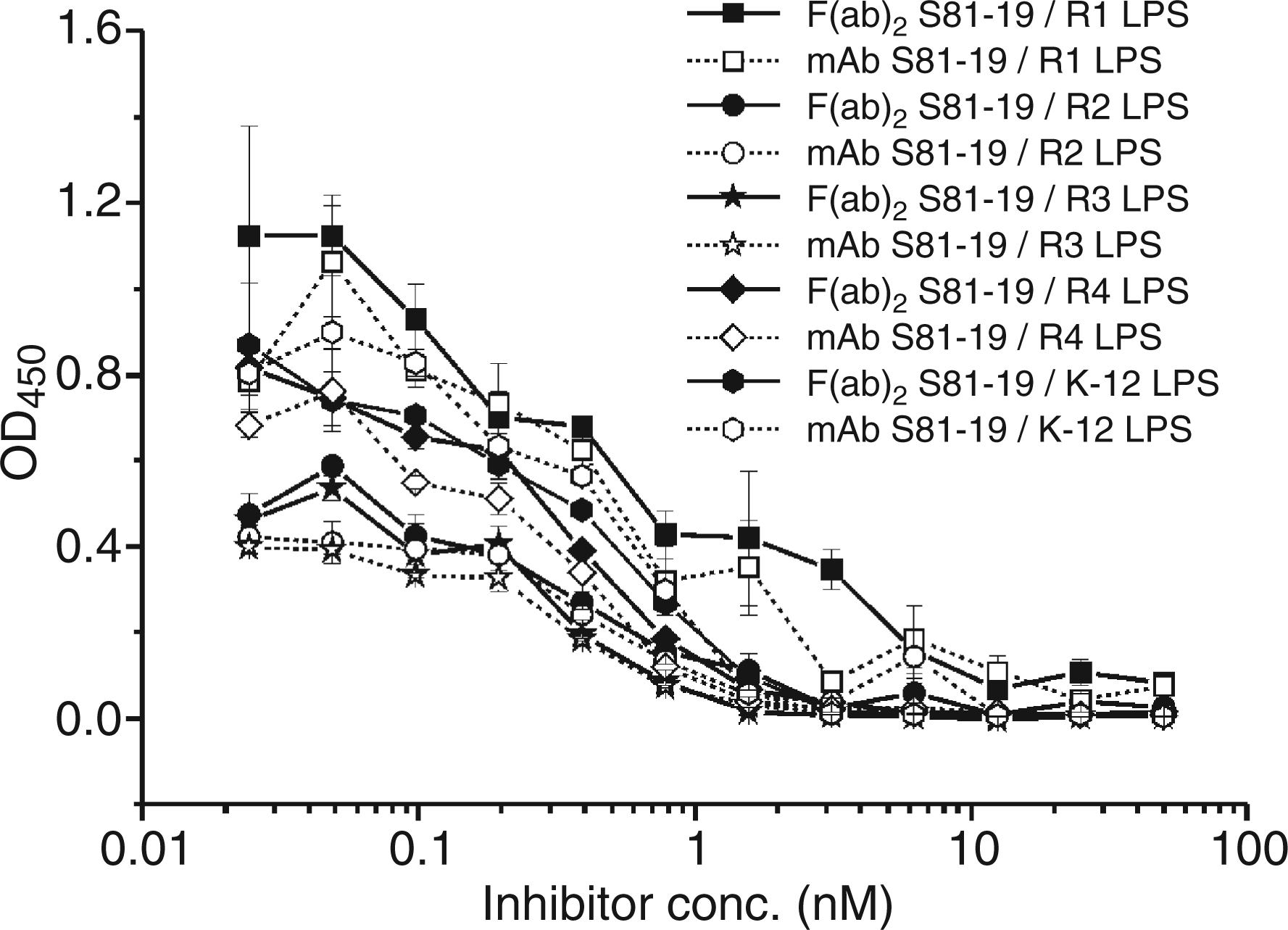

As we were focussing on LPS core-reactive antibodies, we constructed E. coli mutants unable to synthesize ECA (unpublished data); the silver-stained gel after SDS-PAGE (Figure 5A) showed that the LPS of these mutants was unaltered and that they were not stained with the anti-ECA antibody (Figure 5B). Immunostaining with antiserum K437 (day 576) confirmed that among ECA negative strains, a reaction was only observed with the LPS core fraction of E. coli R3 (Figure 5C).
Silver-stained SDS-PAGE (A) and Western Blot after immunostaining with the ECA specific mAb 898 (1 ng/ml, B) or rabbit-antiserum K437 (1:100) taken at day 576 (C). Lanes 1 to 5 were loaded with LPS of Escherichia coli F470 (R1), F576 (R2) F653 (R3), F2513 (R4), and W3110 (K-12), respectively. Lanes 6-10 were loaded with the respective LPS of mutants unable to synthesize ECA. For silver staining and western blots the amount of LPS loaded per lane was 2 µg (A), 10 µg (B) and 20 µg (C).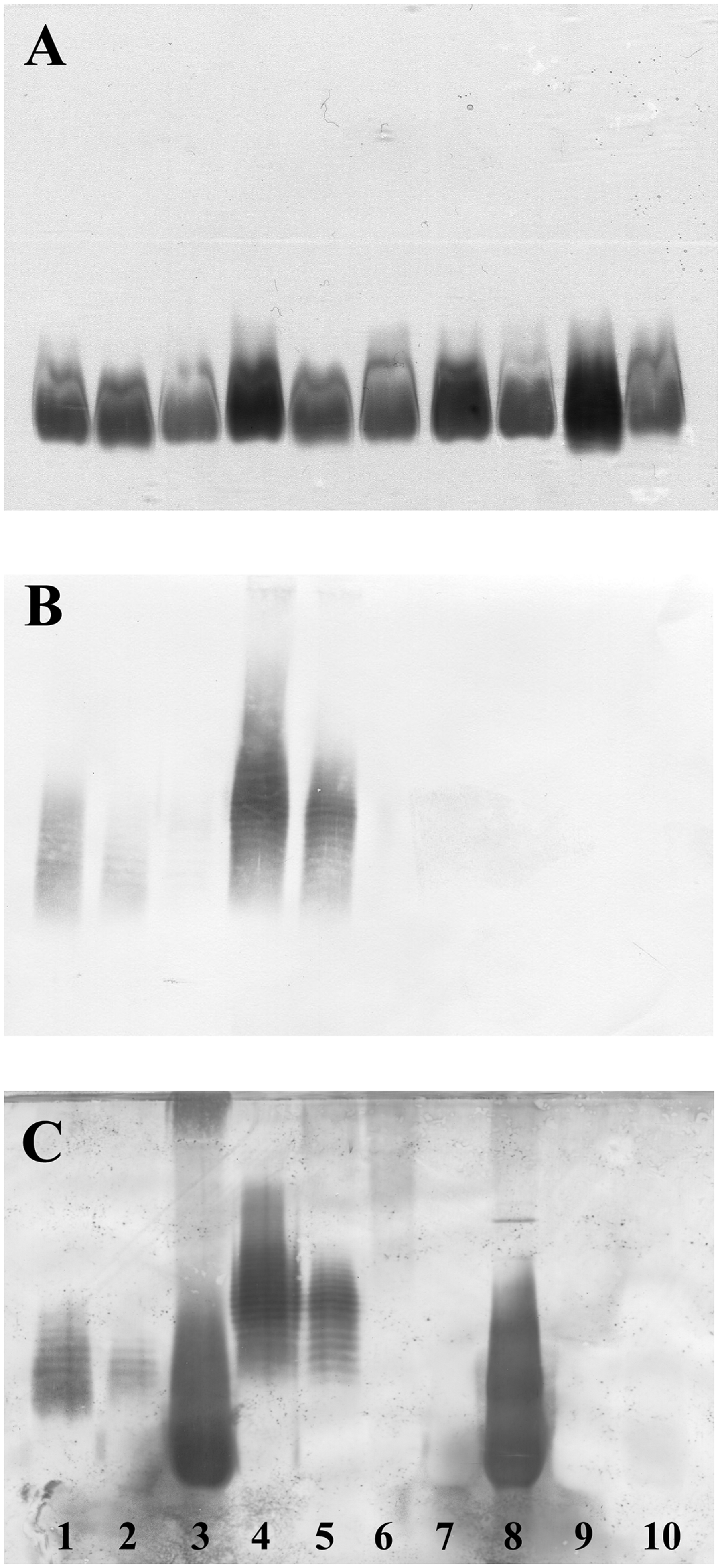
Because of the induction of core-reactive antibodies which bound ECA, we have reinvestigated the binding of WN1 222-5 to LPS of all E. coli core types by ELISA. There was no significant difference between LPS from ECA positive and negative bacteria (Figure 6).
Comparison of mAb WN1 222-5 binding to 20 ng/cup immobilized LPS of ECA+ and ECA-- Escherichia coli strains of core types R1 to R4 and K-12 in ELISA.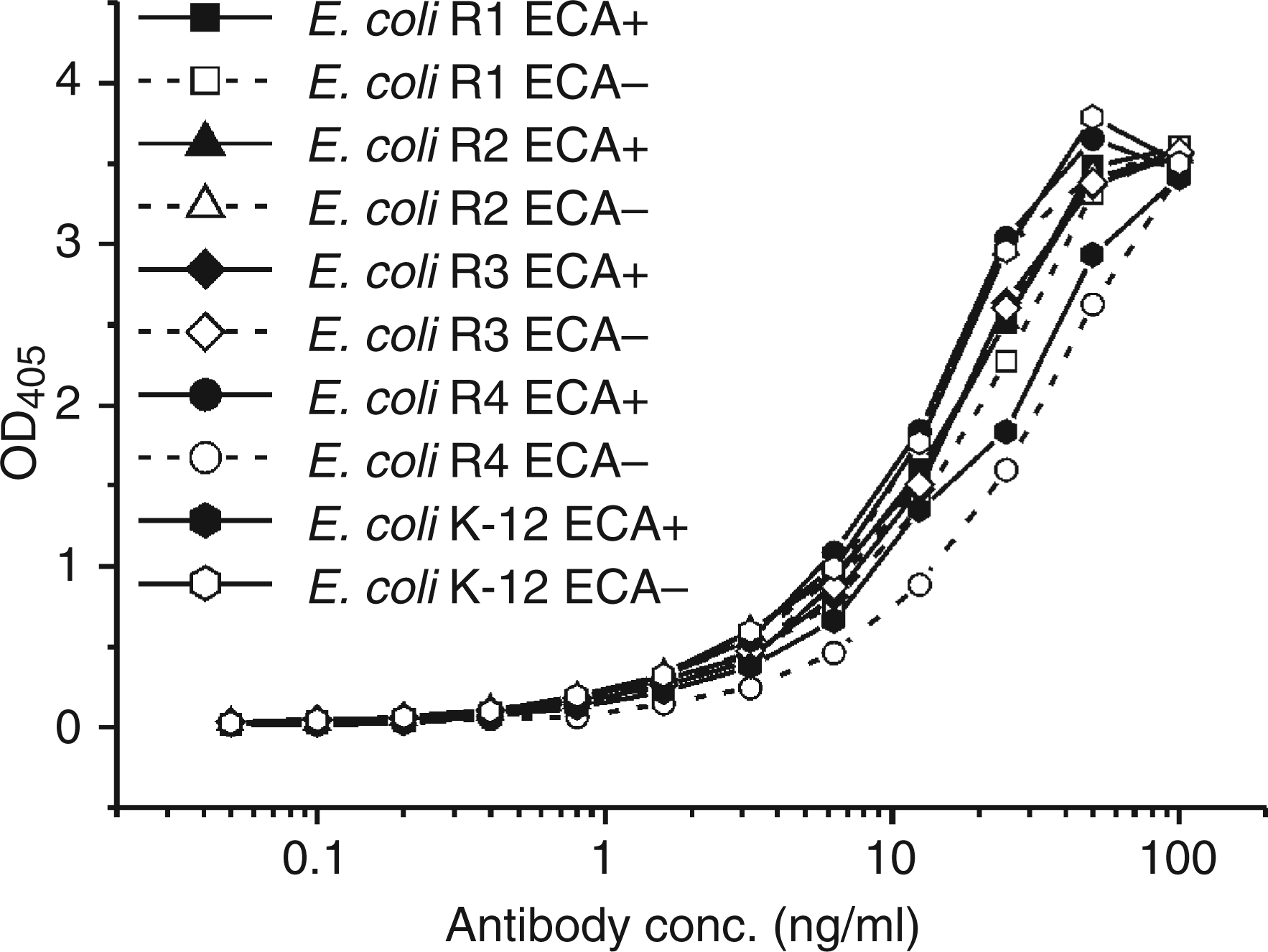
Western-Blots (Figure 7) revealed that only the E. coli F653 rough-type LPS and E. coli smooth LPS of serotypes O15 and O111, that all possess the R3 core type, were stained by the hyperimmune sera collected on days 359 (rabbits 436 and 437) and 371 (rabbits 438 and 439). Each rabbit thus developed an immune response against the E. coli R3 rough LPS. The preimmune sera (day 0) of K437 and 438 already had antibodies against E. coli O111 and O15, respectively. In both cases, however, only the high molecular mass LPS, i.e. molecules with multiple O-chain repeating units, were stained, whereas the low molecular mass LPS without O-antigen was not stained. After immunization all rabbits showed seroconversion with antibodies that bound to the high and low molecular mass fractions equally well. We also tested binding to smooth LPS of E. coli O4, O6 and O18 (R1 core type) and E. coli O12 (R2 core type); none of the animals had raised antibodies against these antigens (data not shown) apart from the reactivity with the ECA. Figure 8 shows the development of antibodies over the observed period of time; 5 out of the 12 sera from two animals with the strongest reactivity are shown. After the first two immunizations (days 0 and 42), neither antibodies against the O-PS nor the core-OS of LPS were detected in the sera collected on day 49. At day 135 (rabbit K437) and day 144 (rabbit K438), antibodies against the rough LPS and the O-PS containing LPS were present, which at this time point stained smooth LPS only weakly. Rabbit K437 received the last booster injection on day 629 and was exsanguinated on day 636. This rabbit received a booster injection on day 326 and the serum sample taken 33 d later had substantially increased antibodies against smooth and rough LPS. The final serum of this rabbit, showed a further strong increase in antibody titers binding to smooth and also rough type LPS. Rabbit K438 received a booster injection on day 487 and serum was collected on day 508. Also in this rabbit, this late serum had a substantially increased activity of antibodies binding smooth and rough LPS. The final serum, which was collected on day 728 after a booster injection on day 718, did not show any further increase compared to that taken on day 508.
Reactivity in Western blot of preimmune (left panel) versus hyperimmune sera (right panel) from four rabbits immunized with S81-19. The LPS used are 1, E. coli J-5; 2, E. coli R1; 3, E. coli R2; 4, E. coli R3; 5, E. coli R4; 6, E. coli K-12; 7, E. coli O111; 8, E. coli O15. Each stripe contained 4 µg of LPS separated by SDS-PAGE and blotted onto PVDF membranes. Sera were diluted 1:100. Time-course of the development of anti-anti-idiotypic antibodies in sera from rabbits immunized with S81-19. A, rabbit K437; B, K438. Lipopolysaccharide of E. coli O111 or E. coli F653 (R3 core type) were separated by SDS-PAGE and blotted onto PVDF membrane; each stripe contained 8 µg of E. coli O111 smooth LPS (left panel) or 4 µg of E. coli F653 rough LPS (right panel). Days after immunization are indicated for each stripe. Sera were diluted 1:100.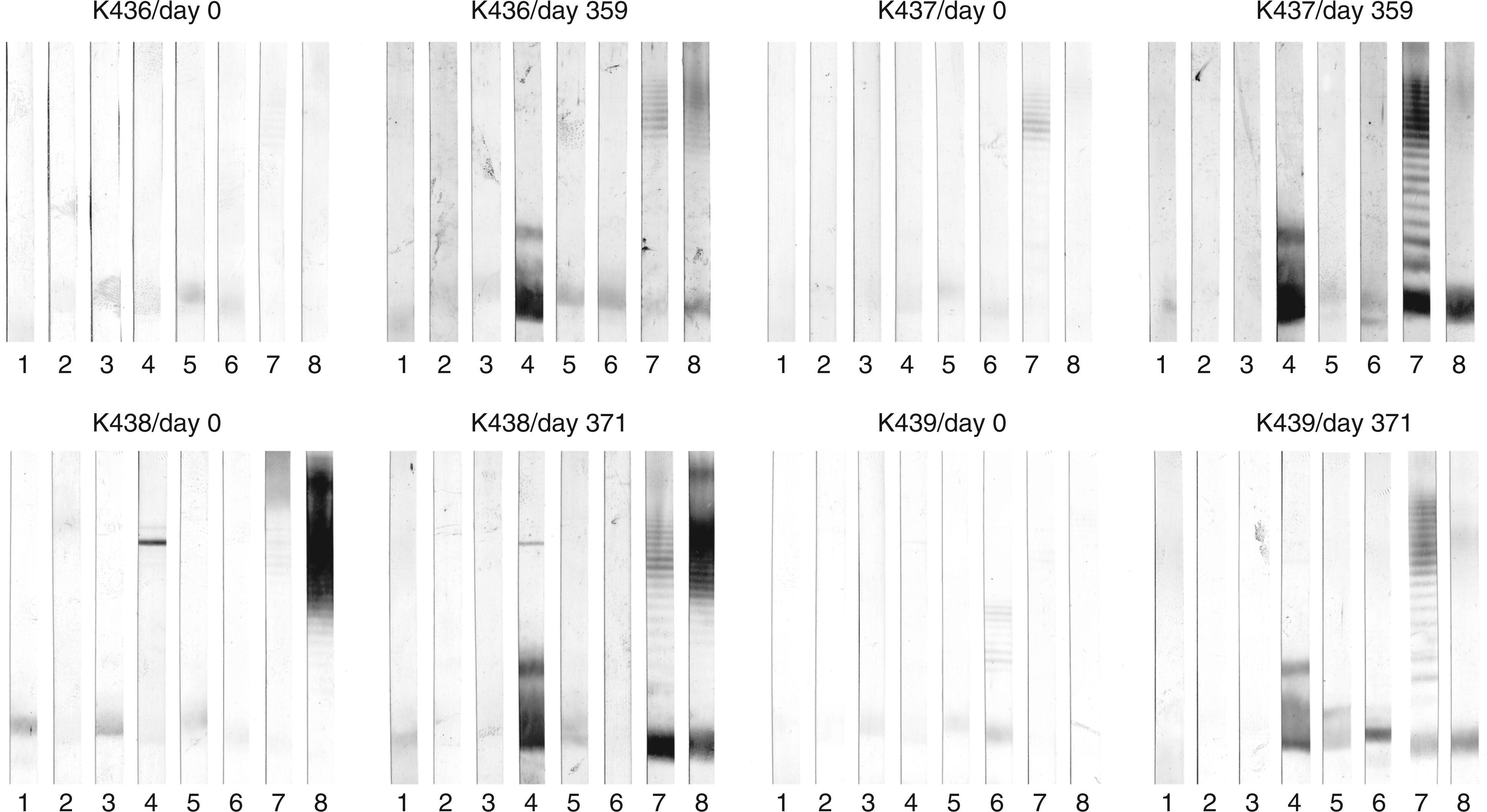
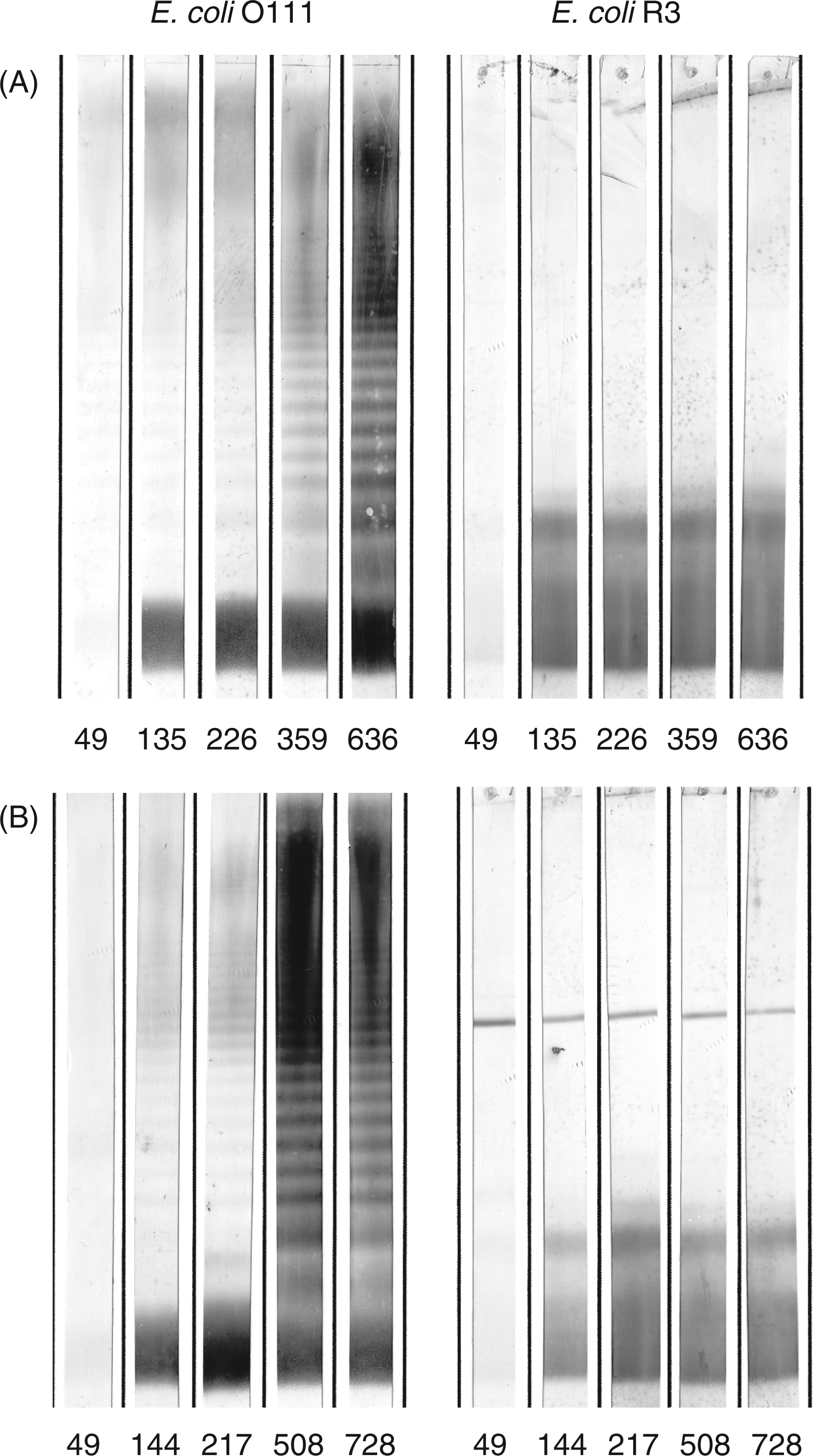
Finally, two sera (K437/day 636 and K438/day 728) were titrated in Western blots using E. coli O111 LPS. As depicted in Figure 9, the sera had an exceptional high activity with titers of more than 1:50,000. This held true for the reactivity with the low (rough) and the high (smooth) molecular mass fractions of the LPS.
Titration of anti-anti-idiotypic antisera from rabbits immunized with mAb S81-19. A, rabbit K437/day 636; B, K438/day 728. Lipopolysaccharide of E. coli O111 was separated by SDS-PAGE and blotted onto PVDF membrane; each stripe contained 8 µg of LPS. Serum dilutions are: 1, 1:200; 2, 1:800; 3, 1:3,200; 4, 1:12,800; 5, 1:51,200, lane 1 to 5, respectively.
The binding observed in Western blots after SDS-PAGE was verified by ELISA where the homologous reaction with mAb S81-19 yielded titers of 1 to 2 millions (Figure 10A and D), whereas the titer to immobilized E. coli R3 (Figure 10B, E) and O111 LPS (Figure 10C, and F) was three orders of magnitude lower.
Titer of anti-anti-idiotypic rabbit immune sera binding to immobilized mAb S81-19 (A and D), immobilized LPS of E. coli F653 (B and E), and E. coli O111 (C and F) determined by ELISA. Shown on the left (A-C) are sera from rabbit 436 (dashed) and 437 (solid) and on the right (D-F) sera from rabbit 438 (dashed) and 439 (solid) taken at indicated days after the first injection. Starting dilutions were 1:500 for the homologous S81-19 reaction and 1:100 in case of LPS as antigen. The immune response was followed over a time course of up to 728 d. Days at which booster injections were applied are indicated by arrows. Note the different y-axis scales for A, D and B, C, E, F, respectively. Measurements were performed as duplicates.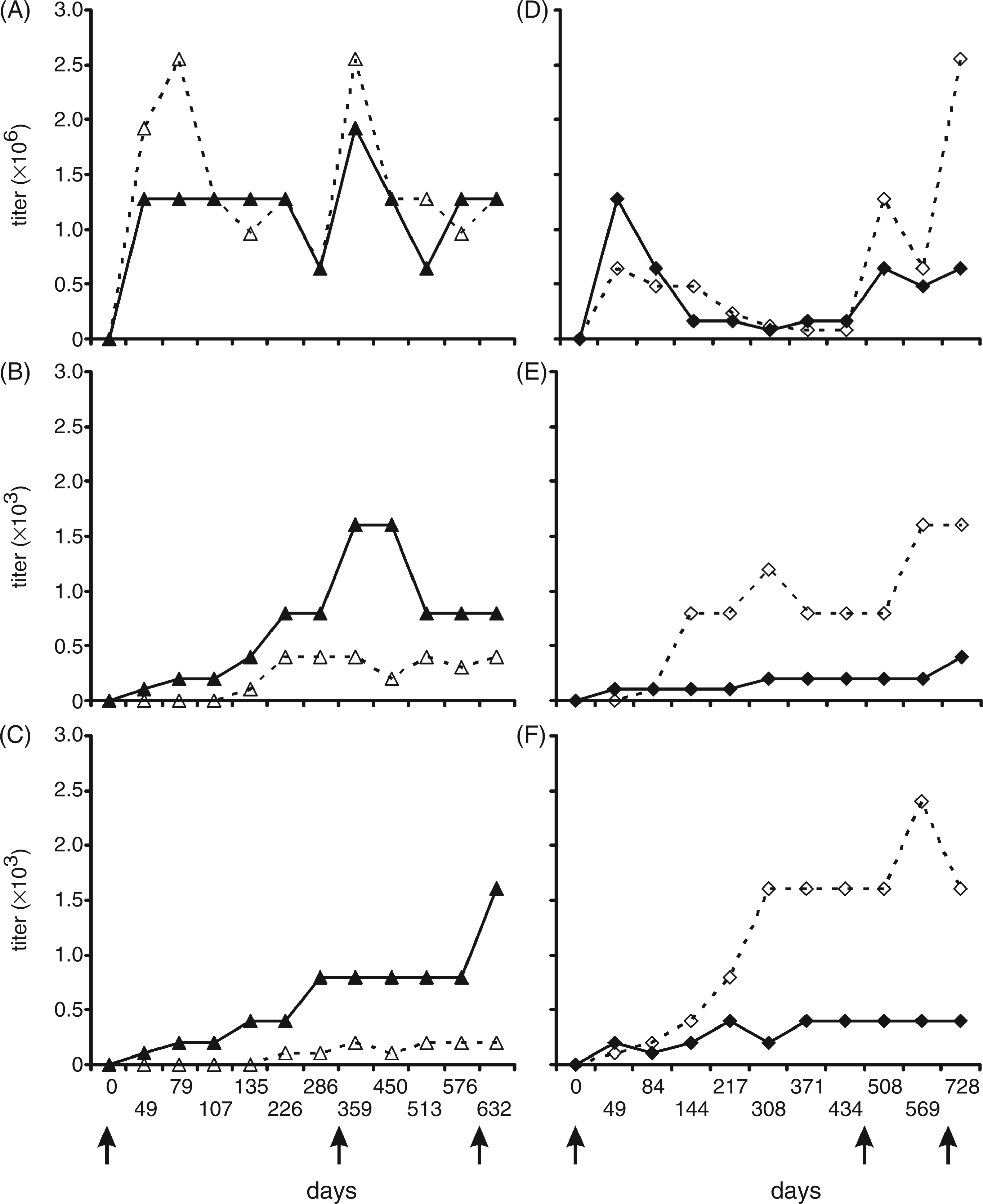
Neutralizing activity of anti-anti-idiotypic antisera for the induction of TNF-α by smooth and rough E. coli LPS
Finally, we determined the endotoxin neutralizing capacity of the anti-anti-idiotypic antibodies. Therefore, human MNC were stimulated with a constant amount of E. coli R3, O111 or O15 LPS in the absence, or presence of, increasing amounts of isolated rabbit IgG after immunization with the anti-idiotypic mAb S81-19. The TNF-α release, induced by incubation with LPS from either E. coli R3 or O111, was strongly inhibited by the isolated rabbit IgG [400 µg/ml] from two different animals (K437, K436), ranging from 71 to 93% (Figure 11A). This inhibition was specific, as neither isolated IgG from a hyperimmune serum (K109/d122) against an unrelated antigen nor the preimmune serum of rabbit K437 (K437/d0) showed any substantial inhibition when used at the same concentration. The capacity of the rabbit IgG to neutralize LPS developed over a long period of time. It was already above 50% after 286 d; however, a further increase of 13-17% could be detected with both sera from day 632 (Figure 11B).
Inhibition of TNF-α release from peripheral human mononuclear cells stimulated with LPS of E. coli F653 or O111 by isolated IgG of rabbit sera obtained after immunization with anti-idiotypic mAb S81-19. The upper panel (A) shows the specificity of the neutralization; the hyperimmune rabbit serum K109 against an unrelated antigen (LPS of E. coli Re mutant strain F515) and the preimmune serum (day 0) from rabbit K437 have no neutralizing effect whereas the late sera (day 632) from rabbits K437 and K436 show up to 90% inhibition. The lower panel (B) shows the development of neutralizing activity against TNF induction by LPS of E. coli O15 over the observed period of time. Inhibitory capacity is calculated from the TNF-inducing activity of 1 ng/l LPS mixed with 400 µg/ml IgG vs. 1 ng/ml LPS alone.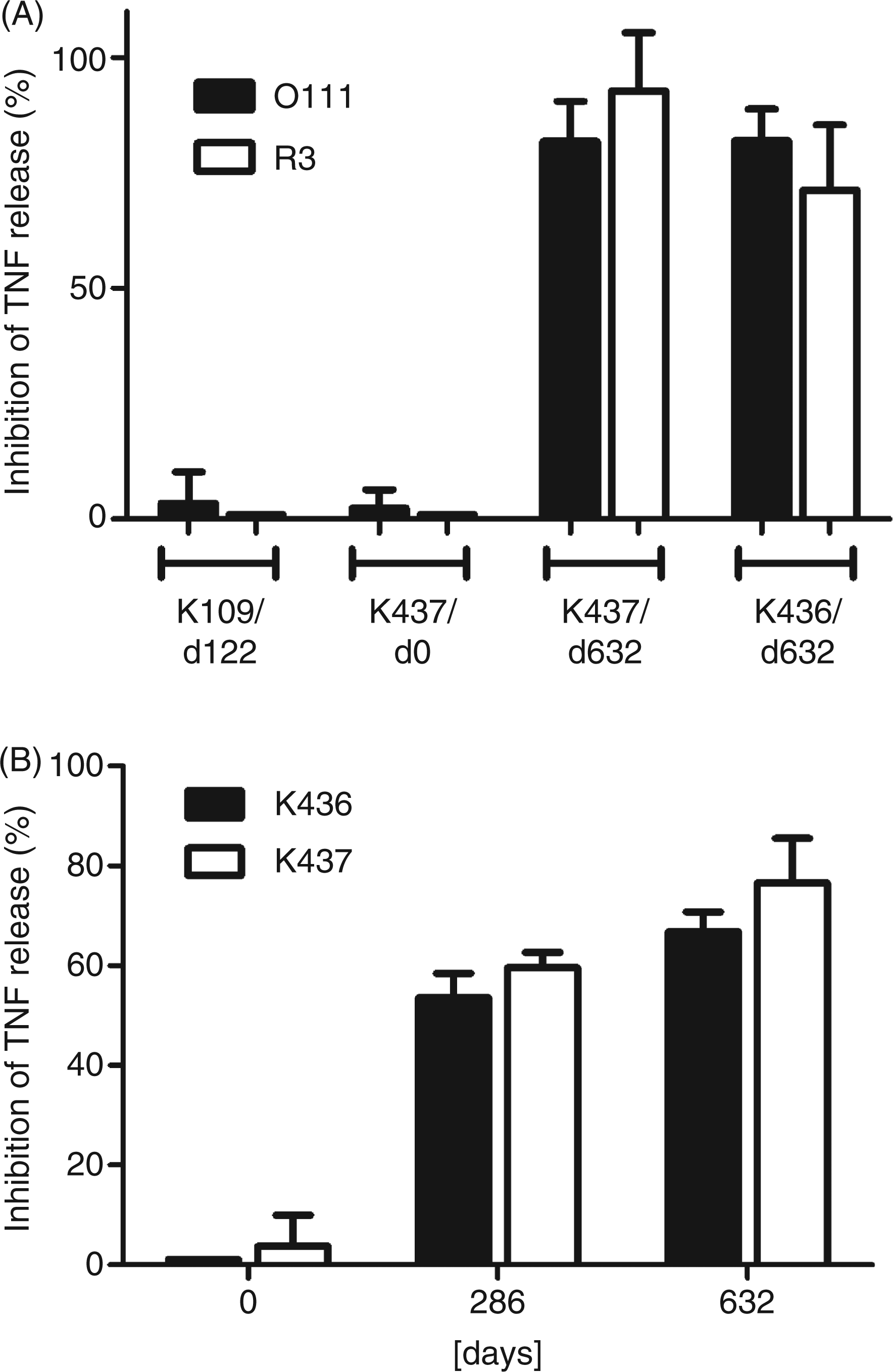
Discussion
Anti-idiotypic antibodies are regarded as potentially useful vaccines for the prevention of a variety of infectious and non-infectious human diseases.26,39 The attraction of anti-idiotypic antibodies arises from the observation that they can serve as carbohydrate surrogates for immunization and offer a principal way to replace T-cell independent carbohydrate immunogens by T-cell dependant protein or peptide antigens. As yet, little is known about this phenomenon at the structural level; however, it can only be understood if a complete three-dimensional shape complementarity of the interacting surfaces is not a necessity for this concept to be successful. Indeed, a structural comparison of antibody SYA/J6 complexed with a carbohydrate oligosaccharide antigen of Shigella flexneri Y or a peptide mimetic, showed that despite substantial differences in the mode of recognition, a partial structural mimicry may exist which is sufficient to obtain a functional mimicry.31,32 A similar observation has been made for a 21-mer peptide binding to the HIV-1 neutralizing antibody 2G12, also binding with high affinity to clustered high mannose structures on gp120. 62 However, it is clear that general conclusions for the understanding of molecular mimicry cannot yet be drawn, and from a functional point of view every antibody-antigen pair will certainly display its own characteristics leading to high affinity binding.
In our study, we investigated whether it would be possible to obtain anti-idiotypic antibodies (Ab2) against the broadly cross-reactive and LPS-neutralizing mAb WN1 222-5 (Ab1) and evaluated whether the main beneficial biological properties of mAb WN1 222-5, i.e. recognition of the core oligosaccharide in the presence of the O-polysaccharide, serotype cross-reactivity and neutralization of LPS can be transferred to a polyclonal immune response (Ab3) by immunization of a heterologous host with an anti-idiotypic antibody (Ab2).
We showed that it is possible to obtain anti-idiotypic antibodies against the mouse mAb WN1 222-5 in principle. Despite the fact that high titers were achieved upon immunization with the native mAb WN1 222-5, only immunization after chemical modification by glutardialdehyde cross-linking gave rise to an anti-idiotypic antibody, mAb S81-19, which was able to inhibit the binding of mAb WN1 222-5 to several carbohydrate ligands derived from E. coli and Salmonella LPS (Table 3). As could be expected, there was no difference if the antigen was presented as LPS or neoglycoconjugate, and the binding to all core types was inhibited by similar concentrations of mAb S81-19.
Immunization of rabbits with IgG and F(ab)2 or F(ab)2 only of S81-19 then yielded polyclonal antisera that contained antibodies against LPS. While all rabbits responded qualitatively in a similar fashion, there were differences in the strength of the antibody immune response measured as the ability of the serum to stain LPS on blots or bind LPS in ELISA which may have been as a result of the generation of antibodies differing in affinities or quantitative differences in antibody serum concentrations. Titers of E. coli R3 LPS-reactive antibodies were in the range of 1:400 to 1:1600 (Figure 10). Unlike the broadly cross-reactive properties of the mAb WN1 222-5, antibodies in the sera were predominantly directed against LPS of the R3 core type and, thus, strongly stained LPS from the E. coli rough mutant F653 and smooth E. coli O111 and O15 (Figures 5C and 7), but not against the mutant E. coli J-5 which contains a truncated R3 core-OS. 63 Despite the presence of a weak J-5 reactivity in the preimmune serum of rabbit K438, immunization with S81-19 led to a strong induction of the R3 specific immune response only, without enhancement of the J-5 reactivity. While this excluded a polyclonal stimulation by immunization with the anti-idiotypic antibody or the adjuvants, it also showed a somewhat different specificity of the induced antibodies as compared to mAb WN1 222-5. 25 Our data (Figure 6) and previous studies 25 have shown that the affinity of mAb WN1 222-5 for the R3 LPS is not higher as compared to other core types and affinity differences, thus not explaining the induction of specificity for the R3 core-type only.
In the anti-anti-idiotypic antisera we have observed antibodies with a specificity so far not understood. These antibodies bind to core-attached ECA as present in E. coli, but not to ECA bound to phosphatidylglycerol as present in K. pneumoniae (Figure 4). The only available explanation for this observation is the recognition of a conformational epitope within the LPS core-OS which is formed or exposed upon attachment of either the ECA polysaccharide or the R3 outer core. It is so far unknown how the ECA polysaccharide is attached to the different E. coli core types and, therefore, this result cannot yet be fully interpreted at the structural level.
The observations may be explained in three principal ways: first, it may be that anti-R3 LPS specific antibodies that are somehow related to the cross-reactive mAb WN1 222-5 by sharing idiotypic markers have been predominantly induced by S81-19 immunization; second, the induced R3-specific antibodies bind to an epitope formed by parts of the inner and outer core oligosaccharides, like WN1 222-5 does, but differ from the latter in being more dependent on the outer core structure, possibly as a result of different variable domain of antibody light chain (VL) or variable domain of antibody heavy chain (VH) usage; and third, the induced antibodies bind the same epitope, like WN1 222-5, but they may be of lower affinity because of an incomplete mimicry by S81-19, therefore leading to a higher sensitivity towards a certain conformation imposed on the inner core by the outer core oligosaccharide. The latter assumption would also provide an explanation for the binding of the anti-anti-idiotypic antisera to ECALPS of E. coli. Whereas mAb WN1 222-5 binding is less dependent on the outer core structure, the anti-anti-idiotypic antibodies recognize a conformation only adopted when the R3 core is present or when the ECA polysaccharide is attached to any of the other outer core types. We have previously observed an increase in affinity of mAb WN1 222-5 for oligosaccharides that contain an outer core in comparison to core-OS devoid of outer core sugars, 25 indicating some conformational dependence.
In accordance with the observed binding specificities, TNF-α release assays using human LPS-stimulated MNC showed that the rabbit immune sera neutralized E. coli R3 and O111 LPS in a specific manner and that the neutralizing capacity increased over time therefore correlating with the antibody titer (Figure 11). It is important to note that rabbits 436 and 437 received their first injection five months earlier than rabbits 438 and 439. Nevertheless, all rabbits showed a significant increase of LPS-specific anti-anti-idiotypic antibodies after approximately 250 d, albeit with different absolute titers.
Our results showed that mAb S81-19 was, in part, able to mimic the structure of the natural carbohydrate ligand of mAb WN1 222-5. Anti-anti-idiotypic antisera could be obtained which bound LPS in the presence of an O-PS and importantly reduced the release of TNF-α, one of the key proinflammatory cytokines in the development of septic shock initiated by endotoxin. The observation that the anti-anti-idiotypic antibodies developed over such an unusual long period of time has two implications: first, anti-anti-idiotypic antibodies may, in general, develop with a different time course than the normal immune response and it may therefore be necessary to wait longer to see whether such antibodies are formed; and second, while such a long time is clearly impractical for clinical use, changing adjuvants or the immunization regimen may allow this problem to be overcome in the future. It is important to note, however, that WN1-222-5-type antibodies have never been obtained by immunization with a single carbohydrate immunogen and that mAb WN1 222-5 was obtained after immunization with a mixture of LPS containing several E. coli core types. 22 While the cross-reactive properties of the WN1 222-5 antibody could not be retained in this approach, the mere observation of neutralizing LPS core-specific antibodies represents a promising starting point for further development. In cooperation with Stephen Evans (University of Victoria, BC, Canada) we have obtained cocrystals of mAb WN1 222-5 in complex with LPS core-OS ligands (manuscript in preparation) to understand the unique cross-reactive properties of WN1 222-5 at the structural level and have begun cocrystallization studies with Fab S81-19 aimed at elucidating the reason for incomplete structural mimicry. This approach will provide a basis for a rational development of antigens able to induce WN1 222-5 type antibodies for use in the fight against Gram-negative sepsis.
Footnotes
Abbreviations
Acknowledgements
This work was financially supported by the Deutsche Forschungsgemeinschaft [grant number SFB470, C1]. We thank U. Agge, A. Baier, I. v. Cube, I. Goroncy, D. Meyer, C. Schneider and V. Susott for technical assistance.
